Abstract
Affective and non-affective psychoses are severe and frequent psychiatric disorders. Amongst others, they not only have a profound impact on affected individuals through their symptomatology, but also regarding cognition, brain structure and function. Cognitive impairment influences patients’ quality of life as well as their ability to work and being employed. While exercise therapy has been implemented in the treatment of psychiatric conditions since the days of Kraepelin and Bleuler, the underlying mechanisms have never been systematically studied. Since the early 1990s, studies emerged examining the effect of physical exercise in animal models, revealing stimulation of neurogenesis, synaptogenesis and neurotransmission. Based on that body of work, clinical studies have been carried out in both healthy humans and in patient populations. These studies differ with regard to homogenous study samples, sample size, type and duration of exercise, outcome variables and measurement techniques. Based on their review, we draw conclusions regarding recommendations for future research strategies showing that modern therapeutic approaches should include physical exercise as part of a multimodal intervention programme to improve psychopathology and cognitive symptoms in schizophrenia and affective disorders.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Prospective epidemiological studies show that together with obesity, low diet quality and smoking, reduced physical activity belongs to the combination of lifestyle factors being responsible for up to 55 % of deaths [140]. However, only little is known regarding the effects of physical exercise on symptoms of mental disorders. In a longitudinal epidemiological study, subjects with regular physical activity had lower incidence of mental diseases in general, anxiety, somatoform and dysthymic disorder, while the incidence of bipolar disorder was increased [130]. Accordingly, adolescents who engaged regularly in physical activity have been characterised by lower depression and anxiety symptoms [83]. Vice versa, little or no physical activity has been linked to the development of depressive symptoms [37]. In this review, we intend to summarise effects of physical exercise in severe mental disorders and to recommend future research strategies.
Schizophrenia
Schizophrenia is a severe mental disorder with prevalence in the range of 0.7–1 % [75, 98], which in the majority of cases already starts at a young age [80]. Negative symptoms like alogia, blunted affect, anhedonia, avolition and apathy occur early in prodromal states and remain persistent during the course of the illness, if at all responding poorly to antipsychotic treatment [57, 74, 84]. Patients’ cognitive deficits, which also occur at an early stage and mainly affect memory, attention, executive functioning and perceptual processes, remain stable over time, represent a core feature of the disease, and are the main predictor for poor social-functioning outcome [24, 46, 61, 65, 66]. As a consequence of persisting negative symptoms and cognitive impairment, the disease often leads to lifelong disabilities thus constituting an enormous burden for patients and their families as well as health care systems [105]. These impairments have a distinct negative influence on patients’ ability to function in all facets of daily life and therefore represent an important therapeutic target [49, 50, 78] which cannot be treated satisfactorily with currently available antipsychotics [47, 57]. Thus, new therapeutic approaches are needed which can fundamentally alter mechanisms in neuronal plasticity [157]. This, for instance, can be achieved via aerobic exercise, which has been proven to ameliorate cognition deficits and increase hippocampal blood flow in both mice [35, 141, 143] and healthy humans [110]. Furthermore, aerobic exercise was shown to have beneficial effects on hippocampal volume and metabolism in schizophrenia patients [109, 121].
Additionally, it should be kept in mind that patients with schizophrenia tend to lead a less healthy lifestyle [14, 20, 92, 128]. Due to medication [45] and genetic liability [145], they also run a high risk for developing a metabolic syndrome, diabetes and cardiovascular diseases [13, 14, 40, 56]. This risk might be increased in combination with reduced physical activity [18].
Affective disorders
Affective disorders, including unipolar or major depressive disorder (MDD) and bipolar disorder (BD), are amongst the most prevalent psychiatric diseases [2, 101, 156].
Both types of affective disorders display cognitive deficits. Impaired cognitive performance mainly relating to attention, memory and executive tasks has indeed been found in both types of affective disorders (meta-analysis by Torres et al. [136]). These cognitive symptoms are thought to contribute to deterioration of social skills, to loss of autonomy and job-related problems. However, little attention has been paid to the development of specific treatment options for cognitive symptoms apart from the established pharmacological and psychotherapeutic therapies in MDD and BD. Considering current pathophysiological concepts in affective disorders, which emphasise the relevance of adult neurogenesis, hippocampal dysfunction and atrophy and neurotrophic signalling [122], there is good reason to hypothesise beneficial effects from physical exercise interventions.
Effect of physical exercise on neurobiological mechanisms
Several biological mechanisms have been proposed to be influenced by physical exercise. Colcombe et al. [19] for instance reported that exercise not only has a positive influence on the number of synaptic connections through developing new neurons but also increases the cortical capillary blood supply. In mice, voluntary exercise is known to stimulate adult hippocampal neurogenesis, learning and memory [141–143], and this stimulation also increases exploratory behaviour [119]. Additionally, vigilance and memory are improved [42]. Physical exercise may exert antidepressant effects via the peptide precursor of neurotrophic factors VGF [73] or serotonergic 5-HT1A receptors [51] and via neurogenesis [41]. Furthermore, positive effects of physical exercise on mood symptoms have been associated with increased production of brain neurotrophic factors, like insulin-like growth factor 1 (IGF1), brain-derived neurotrophic factor (BDNF) and vascular endothelial growth factor (VEGF), and increased activity and production of neurotransmitters (serotonin, norepinephrine and dopamine) related to psychiatric disorders [26, 103, 108]. Moreover, Floel et al. [38] linked cognitive functioning and physical activity through the effects of neurotrophines which are up-regulated through physical exercise and induce neuronal growth and synaptogenesis in animal models [8, 79, 104]. To the best of our knowledge, there is merely one animal study examining the effects of exercise in a schizophrenia risk mouse model [158] and only very few on the neuroprotective effect of exercise to decrease prenatal stress, a known risk factor for psychiatric diseases, including schizophrenia [4, 93]. In humans, one main focus of interventional trials on physical exercise is to evaluate effects on cognitive performance, individual psychopathology as well as brain function and structure. In light of the obvious limitations of current pharmacotherapy with respect to residual symptoms and cognitive deficits, unravelling the neurobiological effects of physical exercise in severe psychiatric diseases may contribute to finding novel treatment strategies—presumably acting by neuroplastic processes in specific brain regions. In this review, we intend to focus on effects of physical exercise in clinical studies of schizophrenia and affective disorders and to discuss critically methodological aspects leading to variable results.
Search algorithms
To achieve a comprehensive overview of the available studies dealing with our topic, we performed a systematic literature search and study evaluation according to the Cochrane criteria, using the computerised search databases PubMed (1966–2012) and Web of Knowledge. Only studies published in English were considered for this review. The keywords used for this search were ‘‘schizophrenia’’ or ‘‘psychosis’’, “depression” or “depressive disorder”, “bipolar disorder” or “mania”, “physical exercise” or “exercise” or “aerobic exercise”, ‘‘sMRI’’ or ‘‘structural imaging’’, “symptoms” or ‘‘symptomatology’’ and ‘‘cognition’’ or ‘‘cognitive function” or ‘‘cognitive dysfunction”. Although our search was limited to studies in humans’, relevant animal studies were also included in this review. The obtained literature was then carefully screened based on title and abstract. After selecting all pertinent articles, their full text was reviewed followed by a critical evaluation of study outcomes. Finally, we carefully checked the reference lists of these articles for other relevant studies on the field that might have been overlooked in our initial search.
Methodology of exercise interventions in schizophrenia
There are a number of different approaches for designing the adequate physical training intervention and selecting primary outcome parameters. Tables 1 and 2 display various exemplary solutions for schizophrenia and affective disorders.
Exercise therapy studies reveal a great variety of interventions ranging from team sports like basketball [133] over aerobic exercise [109, 120] to yoga [7, 31], which incidentally is not easily classifiable into a strict category because it contains physical as well as mental aspects and is connected to religious or spiritual teachings [7]. While there are also a number of studies focussing on different relaxation techniques [147], a clear preference for aerobic exercise seems to emerge [1, 6, 27, 82, 109, 129, 153]. Some articles also describe resistance training as an add-on to aerobic endurance training [96, 115, 120]. Several interventions also contain educational aspects concerning healthy lifestyle and diet [102, 133, 153]. Exercise intensity ranges from a daily activity level [102] to 85 % or even 95 % of the highest heart rate resulting from the pre-test [59]. Likewise, the duration of these interventions also varies: some lasting only 8 weeks [59], while others went on for 18 months [115]. The duration of the training sessions varies similarly from less than 30 or 30 min maximally [5, 27, 153] to 60 min [7, 31, 96, 115, 120].
A number of studies assess physical performance [6, 27, 59, 96, 109, 120, 129], while others reveal methodological deficits in this area [1, 5, 7, 31, 39, 82, 102, 115, 132, 133, 151, 153]. Without a standardised assessment of physical performance, its correlations with symptoms, cognitive performance and neuroimaging cannot be generated. This also applies to the individual exercise intensity which otherwise is not determinable. Furthermore, the applied methods of assessment of physical performance are not comparable and fundamentally different from each other. While the 6-min walking test [6, 27, 96] measures the distance that a person can walk in 6 min and is practically simple [90], exercise tests on treadmills or cycle ergometers can be more complex and may therefore provide data in higher quality for more parameters. The 6-min walking test is suitable for patients with low cardiorespiratory fitness, reflects the intensity of daily life activities and allows a rough estimation of physical performance. Other exercise tests can be carried out with a maximal [59, 109] or submaximal [27, 129] intensity. Maximal exercise tests have a higher cardiovascular demand and thus are not recommendable for all patients. Contraindications need to be respected [137]. In addition, maximum values depend on the patient’s motivation, which can be disturbed in mental illness. Maximal test protocols, however, can be used and result in data for the highest oxygen uptake [59, 109, 121, 129], but maximum values for oxygen uptake should be interpreted carefully because it is discussible whether mentally ill patients are able to reach exhaustion as defined for healthy individuals.
Guaranteeing the predictability, measurability and interpretability of training effects should not only be the basic design principle for clinical trials, but should always also be included and discussed in the respective publications. In detail, we recommend that authors report (a) exercising parameters (period of time, frequency, duration of single exercise sessions, intensity, equipment and type of exercise—in order to intentionally create an efficient training stimulus), (b) the selection of measured variables for the documentation and control of the training progress (e.g. heart rate, blood lactate concentration and written logbook) and (c) the consideration of the reasonableness and feasibility of the intervention in light of the reduced capability of the patients.
Physiological adaptations to regular physical exercise afford a certain amount of time. Investigations in sport science led to the development of a model, which proposes that after approximately 6 weeks of training, a new level of adaptation is reached [33, 72, 107]. Subsequently, training intensity should be modified accordingly. Physical exercise according to the continuous method with no changes in intensity needs neither warming up nor cooling down and is feasible even with cognitively impaired patients [64, 144]. High-intensity interval training seems to be very promising as well, especially concerning the improvement of peak oxygen uptake levels [62]. Training durations of at least 30 min provide stimuli for metabolic as well as neurologic, muscular and cardiovascular systems [106]. In addition, 30 min are well tolerated by the patients [109, 129]. As experiences from current studies show even patients only accustomed to shorter continuous physical activity are able to exercise for 30 min after a short habituation phase. There are indications, however, that longer training sessions show an even greater positive effect on positive and depressive symptoms in schizophrenia [27, 96, 120]. Unless a deliberate decision in favour of high-intensity training is made, exercise intensity should be equivalent to 10–13 points (fairly light to moderately difficult) on the scale of perceived exertion (BORG scale) [12]. Although experiences in using the BORG scale for mentally ill patients [96, 133] exist, it is still uncertain whether it can be used with these patients in the same way as with healthy controls. If the training load corresponds to both the mental and physical precondition of the patient, the exercise sessions themselves should not be too stressful for the patients [72]. Thus, training intensity should always be chosen according to patients’ individual results on the assessments of their physical performance. Blood lactate concentrations of approximately 2.0 mmol/l are recommended to ensure a predominantly aerobic energy metabolism and prevent stressful experiences with increasing anaerobic metabolism and overload [69, 70]. We propose exercise tests both before and after exercise intervention since changes in heart rate (Physical Working Capacity 130) and blood lactate concentration (performance with 2 mmol/l) within stages similar to training intensities are to be expected, while the highest oxygen uptake will not necessarily change. Exercise testing allows generating different training intensities by applying concepts of ventilatory or lactate thresholds. Further research is needed to ensure that these parameters can be used for patients. Control of patients’ individual exercise intensity through blood lactate concentrations can be used to back up training in an aerobic zone. The heart rate can be influenced by a number of pharmacological treatments, which is why an electrocardiogram (ECG) is also essential. Regarding the higher cardiovascular mortality rate in schizophrenia [126, 134, 161], some safety precautions should be considered, e.g. a doctor on call, emergency equipment and as mentioned an exercise testing with stress ECG before the intervention [137]. The use of bicycle ergometers in the intervention assures an exact measurement of training intensity and provides the option to standardise. Cycling is, above all, also a type of exercise that is feasible with patients who are obese or have orthopaedic problems because it is joint-friendly [89, 135]. Its circular movement is easy to practice and seems to also have a relaxing influence because of the rhythmic repetitions [87]. During a long-term exercise therapy regimen, other cardiorespiratory exercises like running on a treadmill might also be appealing to patients and will help to avoid monotonous training stimuli.
Methodology of exercise interventions in affective disorders
A majority of studies in affective disorders do not fulfil adequate methodological requirements. In their review, Wolff et al. [159] excluded all studies not meeting minimum methodological criteria and suggested only moderate effect sizes in studies comparing physical exercise with pharmacological treatment. A review of existing studies on BD and physical exercise also proposes that most do not meet methodological standards [160]. Furthermore, doses of training might again influence results. Dunn et al. [30] systematically investigated the effects of low versus high doses of aerobic exercise finding high doses to be more effective in reducing depressive symptoms in MDD than low doses and that after 12 weeks of intervention exercise, frequency had not shown influence on the severity of depressive symptoms. Compared with a control condition (social visits), for instance, an individualised physical activity programme for older patients with depressive symptoms was not more effective in ameliorating the severity of their symptoms [81]. It must be kept in mind, however, that participants of the study by Kerse et al. had engaged in only moderate levels of physical activity. In addition, findings of sport intervention studies might also be influenced by the point in time of assessing main outcome measures: Weinstein et al., for example, [154] reported that in a group of depressive patients, depressive symptoms had decreased directly after exercise, while 30 min after exercise, depressive symptoms and fatigue had again increased in the patient group.
Effects of physical exercise on schizophrenia symptomatology
After having been neglected as a therapeutic intervention despite its cost-effectiveness for a long time, exercise has been in the focus as therapeutic add-on in various psychiatric disorders since the late 1990s [16, 118]. In the beginning, the effects of exercise on schizophrenia were seen mainly with regard to physiological changes like obesity or metabolic syndrome while psychological alterations, not to mention neurobiological changes, were being ignored. Additionally, there were many methodological limitations in these early studies [16]. Major concerns, e.g., are missing healthy and/or patient control groups, too small sample sizes and non-randomized or non-controlled design [1, 27, 148]. Until now, many exercise studies still only focus on body weight as sole outcome parameter without paying attention to psychopathological changes [5, 32, 102, 152] or measure only unspecific global outcomes with scales not tailored to the specific schizophrenia psychopathology [96, 129].
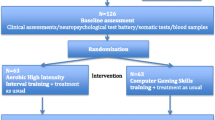
As for other psychiatric disorders [159], a number of studies point to a significant improvement of schizophrenia symptoms via aerobic exercise, which has been confirmed by a first meta-analysis [48]. Some authors measured quality of life as additional outcome parameter and unanimously report respective improvement as well [1, 6, 7, 31]. Scheewe et al. used the Camberwell Assessment of Need (CAN) [114] also finding significant improvement [120]. In a randomized controlled trial (RCT), Beebe et al. [6] were able to show reductions of positive and negative symptoms in chronically ill schizophrenia patients after aerobic exercise, though the differences did not reach statistical significance. Another RCT in chronically ill schizophrenia patients with aerobic exercise (indoor cycling) compared to a patient control group and healthy controls found a reduction in the severity of total symptoms of up to 9 % in the cycling group, while non-exercising patients presented a total symptom increase of 13 % measured by the positive and negative symptom scale (PANSS); again the findings did not reach statistical significance most likely due to small sample sizes [109]. This is in accordance with the results of Takahashi et al. [133] who found an improvement of the general psychopathology scale but not in the positive and negative symptom scale of the PANSS after exercising. This finding is further verified by an RCT with a large sample of schizophrenia patients reporting a significant reduction in the PANSS total score, especially by decreasing positive symptoms, disorganisation, excitement, emotional distress and a trend level significance in the decrease in negative symptoms. Also, exercise therapy had a positive effect on depressive symptoms [120]. Only one study with a relatively small sample of chronically ill schizophrenia patients undergoing aerobic high-intensity training compared to patient controls playing computer games did not find an improvement of positive, negative or depressive symptoms in either group via using PANSS and the Calgary Depression Scale for Schizophrenia (CDSS) [59]. The noted missing effects in this study could be due to the relatively small sample sizes and overall duration of the intervention, but also an effect of the different exercise intensities. After combining aerobic exercise (walking) and training of muscular strength in a small sample, persons with schizophrenia or schizoaffective disorders showed significantly reduced depressive symptoms plus significant improvement in their total score on the Mental Health Inventory, using a structured self-report questionnaire, as compared with controls [96].
Additionally, there are three RCTs pointing to a good if not even superior positive influence on schizophrenia symptoms through yoga compared to aerobic exercise: while one only compared patients with a yoga intervention to patients in a waiting list condition [151], both Behere et al. [7] and Duraiswamy et al. [31] investigated different outcomes of yoga therapy compared to a therapeutic exercise add-on. Each of the studies reported a significantly better total, positive and negative PANSS score plus a significantly improved socio-occupational status. This better outcome might be due to the yoga-intrinsic dual focus on physical and mental conditions, specifically including relaxation exercises. Nevertheless, the reported findings should be interpreted with caution since both studies present methodological weaknesses with regard to adherence, compliance rate, monitoring the therapeutic add-on under outpatient conditions and by not including healthy controls. Furthermore, the study designs were based on different yoga techniques, and the two latter were conducted in India where yoga might be better accepted than in Western countries [149].
Effects of physical exercise on cognitive performance in schizophrenia
Although cognitive deficits are regarded as a core feature of schizophrenia (e.g. [24, 46, 61, 65]) and despite some evidence derived from studies in healthy humans (e.g. [19, 34, 71, 110]), macaque monkeys [116] and rodents [4, 35, 110, 141] that exercise enhances cognition, this aspect still is widely neglected in schizophrenia research. To our knowledge, only one RCT specifically focussed on neurocognitive effects induced by aerobic exercise in chronically ill schizophrenia patients. Here, aerobic exercise led to a 34 % improvement in the short-term memory of schizophrenia patients score of the German version of the Rey Auditory Verbal Learning Test. Somehow surprising, no improvement of memory scores were found in healthy controls. This may be due to the fact that controls already had a very high pre-test short-term memory level. Likewise, the long-term memory and Corsi direct block span score were investigated, but no significant effects were detected [109]. While we originally speculated these somewhat unexpected findings might to be due to the small sample size of our trial, Scheewe et al. also failed to find any changes in cognition in their larger sample of schizophrenia patients (personal communication).
Neuroimaging studies on aerobic exercise in schizophrenia
At present, there are only three randomized structural imaging trials investigating the effects of aerobic exercise on brain volumes in schizophrenia patients. In a first pilot study, Pajonk et al. could demonstrate a mean hippocampal volume increase of 12 % in the schizophrenia exercise group and 16 % in the healthy control group positively correlated with fitness in comparison with the non-exercising group. Additionally, exercising patients presented a statistically significant increase of 35 % in the NAA/Cr ratio in magnetic resonance spectroscopy, whereas the patient control group showed a decline of 16 %. The authors did not find changes in total brain volume or total grey matter volumes [109]. In a far larger sample again, neither hippocampal volume changes nor changes in global brain volume or cortical thickness could be found. Nevertheless, fitness improvement was related to an increase in total cerebral matter volume (or attenuated volume decrease) and a decrease in lateral and third ventricle volume (or less increase) in patients with schizophrenia, but not in healthy controls. Additionally, an association of cardiorespiratory fitness improvement with cortical thickening in most left frontal, temporal and cingulate cortical areas was found in both schizophrenia patients and healthy controls, which may point to less volume decrease over time in the schizophrenia exercise group [121]. In contrast, using another method, Falkai et al. [36] found no difference in cortical thickness and surface expansion in schizophrenia patients after exercising, while healthy controls showed these changes in right frontal and occipital pole regions. According to a trial in healthy probands, increase in hippocampal volume seems to take place only in the anterior section and is related to greater serum levels of BDNF, which is known as a mediator of synaptic plasticity and neurogenesis [34].
In two recent studies focusing on the extrastriate body area, located in the posterior temporal-occipital cortex and associated with understanding others’ behaviours, Takahashi et al. [132, 133] used functional magnetic resonance imaging and found increased activation of the extrastriate body area in the exercising patient group in comparison with non-exercising schizophrenia patients. This increase was associated with an improved general psychopathology measured by PANSS. However, both studies are no RCTs, and hence, general conclusions are limited.
Effects of physical exercise on affective symptoms
The first study describing effects of physical exercise on symptoms of depression has been published in 1985 [95]. Levels of physical activity have been inversely associated with the severity of depressive symptoms and with prevalence rates of depression in the general population [43, 55, 99, 131]. Interestingly, one trial resulted in a significant association between more steps/day and lower prevalence of depression in women, but not in men [99]. Other studies observed the effect of regular physical exercise on the remission rate and onset of depression.
In their recent study, Jerstad et al. reported that physical exercise has a protective effect for the onset of major/minor depression in adolescents. They also suggested that the occurrence of depressive symptoms reduces physical activity in adolescents [77]. Assumptions of physical exercise being effective in preventing depression [91] were confirmed by the finding that an aerobic training programme was superior to pharmacological treatment in preventing a relapse in patients with MDD (6-month observation). This effect had been stronger in patients who had continued to exercise on their own [3].
To investigate systematically whether there is a causal relationship between physical activity and psychopathological symptoms, intervention studies with 2–3 weekly exercise sessions have been carried out [9, 63]. Interventional sports trials in MDD patients indicated a beneficial effect of moderate physical activity on depressive symptoms, as measured with well-established depression scales (e.g. Hamilton depression scale [54]; Beck depression scale [58]) [100, 159]. Moreover, an exercise support programme, lasting 3 months, reduced depression scores of mothers suffering from postpartum depression [60].
However, a meta-analysis including 25 interventional trials revealed only small-to-moderate effect sizes on clinical symptoms in MDD [100]. Similarly, in a new Cochrane review and meta-analyses including 28 studies, physical exercise showed moderate clinical effects [117]. Another meta-analyses of randomized controlled trials showed only modest effects of exercise and little evidence of long-term beneficial effect of exercise in patient with depression [86]. In many of these studies, the sample sizes were small and other methodological shortcomings need to be considered. Therefore, to improve comparability between studies, there is a need for large controlled clinical trials meeting standard criteria for intervention and rater-based evaluation of symptoms. For example, using only self-reported ratings of depressive symptoms, Chalder et al. [17] found no improvement in the intervention group. Several studies reveal a reduction of depressive symptoms in MDD via a controlled physical exercise programme [10, 23, 25, 29, 85, 97, 123, 125, 127]. It has been suggested that the effects were particularly strong for positive mood and energy, and less strong for improving distress, confusion, fatigue, tension or anger [11].
Other studies comparing physical exercise with other treatments, e.g. pharmacological treatment, indicated that physical exercise was similarly effective as established therapies [159]. Hoffman et al. compared initial treatment assignment, antidepressant medication and physical exercise in a 1 year follow-up and found that physical exercise was the best predictor of depression scores compared with other conditions [67]. Comparing a 12-months lasting aerobic exercise group with a group receiving antidepressant medication showed that the exercise group had a significant decrease in depressive symptoms compared to controls [23]. One work group found antidepressant therapy to show better effects than physical exercise at the beginning of treatment. However, 4 months later, both treatment options were regarded as similarly helpful in reducing depressive symptoms [9]. Several studies have shown that after physical activity, decreased depressive symptoms and increased quality of life are not merely due to improvement of physical fitness [28, 94, 124]. Thus, neurobiological mechanisms may be altered before the physical fitness level rises.
To our knowledge, there are only few studies on physical exercise in BD patients [15, 76, 138, 139]. Wright et al. [160] assumed that physical exercise may reduce stress as well as depressive and anxious symptoms in BD. In a recent review, it has been shown that medical co-morbidity, lower education status and social isolation have been associated with decreased physical activity in bipolar patients [146]. Therefore, in prospective studies, these variables should be evaluated.
Effects of physical exercise on cognitive performance in affective disorders
Studies investigating the effects of physical exercise on cognitive performance in affective disorders are rare [68, 87, 88, 150, 159]. For MDD, two studies investigated cognitive skills immediately after an exercise session and showed improved attention and inhibitory control function [87], as well as increased selective attention [150]. When compared with cognitive therapy or antidepressants in MDD, exercise was found to be as effective as either of these treatments [159]. In contrast, one trial compared four groups of patients with MDD: one group participated in supervised exercise; a second group engaged in home-based exercises; a third group received an antidepressive drug (sertraline) and a fourth group was given a placebo. There were no differences across all neuropsychological tests when comparing the effects of placebo and exercise, but exercising participants performed better than participants receiving sertraline on tests of executive functioning [68]. While two studies had assessed cognitive function immediately after the exercise sessions [87, 150], participants in the study by Hoffman et al. [68] were measured later and were of older age.
A recent review of BD revealed that the results in BD mirror those from studies in MDD, suggesting that exercise is a viable treatment option for neurocognitive dysfunction in BD [88]. Other reviews on physical exercise in BD reported exercise-induced neurobiological changes and effects on BDNF levels [2].
While there are a number of studies assessing the effect sizes of sports interventions in patients with cognitive decline and dementia, such analyses do not yet exist for trials in affective disorders. To sum it up, there is substantial evidence for significant effects of physical exercise interventions on individual psychopathology in MDD, while effects on cognitive performance in these patients are less well established.
Neurophysiology and physical exercise effects in affective disorders
Relatively few studies have examined the influence of regular physical exercise on brain functioning in affective disorders, and none of them assessed BD patients. The existing studies mainly focused on electroencephalography (EEG), which has also been used to assess electrophysiological correlates of brain function in clinical studies of depression [21, 22]. Recent findings indicate that mood symptoms are associated with cortical asymmetry mainly in the alpha band (8–13 Hz) [52, 53, 111–113]. However, to the best of our knowledge, only two studies systematically examined the effects of exercise treatment on EEG activity patterns in patients with MDD [23, 123]. One study investigating a group of patients with MDD, receiving a combination of aerobic exercise and pharmacological treatment for 1 year, revealed no changes in EEG pattern after the treatment period. In contrast, a control group with only pharmacological treatment showed an increase in alpha band power in the right hemisphere. After 1 year, however, the authors reported a reduction in depressive symptoms after 1 year in the exercise group but not in the control group [23]. The second study showed increased EEG mean frequency for both, an aerobic exercise group (2 times a week, 6 months) and a control group of MDD patients, but no significant group differences [123]. In sum, the existing number of studies using imaging approaches in affective disorders is too small to fit these findings into a pathophysiological model. Assessing brain function with fMRI and brain morphological changes with volumetric methods may be better suited for the assessment of training effects. The findings in schizophrenia are of interest for studies in affective disorders, which mirror some of the negative symptoms of schizophrenia (depressive symptoms). Based on the recent findings, it might be of interest to systematically investigate brain volumes, especially hippocampus volumes, in trials on MDD patients.
Standardised physical activity in psychiatric research: a recommendation
In summary, exercise therapy has shown effects on both cognition and symptoms in schizophrenia and affective disorders. However, exercise therapy interventions in psychiatric research should also be standardised in order to be comparable. Based on our experiences, we recommend the following standard programme for endurance training in clinical studies: a minimum of 12 weeks, 3 sessions per week, general aerobic endurance training (continuous training), a minimum of 30 min per training session, with blood lactate concentrations of ca. 2.0 mmol/l, intensities equivalent to 10–13 on the BORG scale, attendance rates of more than 75 % and finally assessment of physical performance by incremental bicycle ergometry (with measurements of gas exchange, blood lactate concentration and heart rate). This design corresponds to recent research findings in sports medicine and the most recent consort statement by the American College of Sports Medicine [44, 107, 155] and ensures compliance of mentally ill patients [109]. However, to date, it is uncertain whether this scale is applicable in the same way to psychiatric patients, and future studies should address this. In spite of encouraging first results, further exercise therapy studies should comprise larger patient samples to provide more conclusive findings. Finally, it must be kept in mind that long-term exercise therapies call for different training methods to avoid monotonous and thus inefficient training stimuli [72].
References
Acil AA, Dogan S, Dogan O (2008) The effects of physical exercises to mental state and quality of life in patients with schizophrenia. J Psychiatr Ment Health Nurs 15:808–815
Alsuwaidan MT, Kucyi A, Law CW, McIntyre RS (2009) Exercise and bipolar disorder: a review of neurobiological mediators. Neuromol Med 11:328–336
Babyak M, Blumenthal JA, Herman S, Khatri P, Doraiswamy M, Moore K, Craighead WE, Baldewicz TT, Krishnan KR (2000) Exercise treatment for major depression: maintenance of therapeutic benefit at 10 months. Psychosom Med 62:633–638
Baek SS, Jun TW, Kim KJ, Shin MS, Kang SY, Kim CJ (2012) Effects of postnatal treadmill exercise on apoptotic neuronal cell death and cell proliferation of maternal-separated rat pups. Brain Dev 34:45–56
Ball MP, Coons VB, Buchanan RW (2001) A program for treating olanzapine-related weight gain. Psychiatr Serv 52:967–969
Beebe LH, Tian L, Morris N, Goodwin A, Allen SS, Kuldau J (2005) Effects of exercise on mental and physical health parameters of persons with schizophrenia. Issues Mental Health Nurs 26:661–676
Behere RV, Arasappa R, Jagannathan A, Varambally S, Venkatasubramanian G, Thirthalli J, Subbakrishna DK, Nagendra HR, Gangadhar BN (2011) Effect of yoga therapy on facial emotion recognition deficits, symptoms and functioning in patients with schizophrenia. Acta Psychiatr Scand 123:147–153
Biedermann S, Fuss J, Zheng L, Sartorius A, Falfan-Melgoza C, Demirakca T, Gass P, Ende G, Weber-Fahr W (2012) In vivo voxel based morphometry: detection of increased hippocampal volume and decreased glutamate levels in exercising mice. Neuroimage 61:1206–1212
Blumenthal JA, Babyak MA, Moore KA, Craighead WE, Herman S, Khatri P, Waugh R, Napolitano MA, Forman LM, Appelbaum M, Doraiswamy PM, Krishnan KR (1999) Effects of exercise training on older patients with major depression. Arch Intern Med 159:2349–2356
Blumenthal JA, Sherwood A, Rogers SD, Babyak MA, Doraiswamy PM, Watkins L, Hoffman BM, O’Connell C, Johnson JJ, Patidar SM, Waugh R, Hinderliter A (2007) Understanding prognostic benefits of exercise and antidepressant therapy for persons with depression and heart disease: the upbeat study—rationale, design, and methodological issues. Clin Trials (London, England) 4:548–559
Bodin T, Martinsen EW (2004) Mood and self-efficacy during acute exercise in clinical depression. A randomized, controlled study. J Sport Exercise Psychol 26:623–633
Borg G (1970) Perceived exertion as an indicator of somatic stress. Scand J Rehabil Med 2:92–98
Brown S, Birtwistle J, Roe L, Thompson C (1999) The unhealthy lifestyle of people with schizophrenia. Psychol Med 29:697–701
Brown S, Inskip H, Barraclough B (2000) Causes of the excess mortality of schizophrenia. Br J Psychiatry 177:212–217
Cairney J, Veldhuizen S, Faulkner G, Schaffer A, Rodriguez C (2009) Bipolar disorder and leisure-time physical activity: results from a national survey of Canadians. Mental Health Phys Activity 2:65–70
Callaghan P (2004) Exercise: a neglected intervention in mental health care? J Psychiatr Ment Health Nurs 11:476–483
Chalder M, Wiles NJ, Campbell J, Hollinghurst SP, Haase AM, Taylor AH, Fox KR, Costelloe C, Searle A, Baxter H, Winder R, Wright C, Turner KM, Calnan M, Lawlor DA, Peters TJ, Sharp DJ, Montgomery AA, Lewis G (2012) Facilitated physical activity as a treatment for depressed adults: randomised controlled trial. BMJ 344:e2758
Chamove AS (1986) Positive short-term effects of activity on behaviour in chronic schizophrenic patients. British J Clin Psychol/British Psychol Soc 25(Pt 2):125–133
Colcombe SJ, Kramer AF, Erickson KI, Scalf P, McAuley E, Cohen NJ, Webb A, Jerome GJ, Marquez DX, Elavsky S (2004) Cardiovascular fitness, cortical plasticity, and aging. Proc Natl Acad Sci USA 101:3316–3321
de Leon J, Diaz FJ (2005) A meta-analysis of worldwide studies demonstrates an association between schizophrenia and tobacco smoking behaviors. Schizophr Res 76:135–157
Deslandes A, Veiga H, Cagy M, Fiszman A, Piedade R, Ribeiro P (2004) Quantitative electroencephalography (qeeg) to discriminate primary degenerative dementia from major depressive disorder (depression). Arq Neuropsiquiatr 62:44–50
Deslandes AC, de Moraes H, Pompeu FA, Ribeiro P, Cagy M, Capitao C et al (2008) Electroencephalographic frontal asymmetry and depressive symptoms in the elderly. Biol Psychol 79:317–322
Deslandes AC, Moraes H, Alves H, Pompeu FA, Silveira H, Mouta R, Arcoverde C, Ribeiro P, Cagy M, Piedade RA, Laks J, Coutinho ES (2010) Effect of aerobic training on eeg alpha asymmetry and depressive symptoms in the elderly: a 1-year follow-up study. Braz J Med Biol Res 43:585–592
Dickinson D, Ramsey ME, Gold JM (2007) Overlooking the obvious: a meta-analytic comparison of digit symbol coding tasks and other cognitive measures in schizophrenia. Arch Gen Psychiatry 64:532–542
Dimeo F, Bauer M, Varahram I, Proest G, Halter U (2001) Benefits from aerobic exercise in patients with major depression: a pilot study. Br J Sports Med 35:114–117
Dishman RK, Berthoud HR, Booth FW, Cotman CW, Edgerton VR, Fleshner MR et al (2006) Neurobiology of exercise. Obesity 14:345–356
Dodd KJ, Duffy S, Stewart JA, Impey J, Taylor N (2011) A small group aerobic exercise programme that reduces body weight is feasible in adults with severe chronic schizophrenia: a pilot study. Disabil Rehabil 33:1222–1229
Doyne EJ, Ossip-Klein DJ, Bowman ED, Osborn KM, McDougall-Wilson IB, Neimeyer RA (1987) Running versus weight lifting in the treatment of depression. J Consult Clin Psychol 55:748–754
Dunn A, Trivedi M, O’Neal H (2001) Physical activity dose-response effects on outcomes of depression and anxiety. Med Sci Sports Exerc 33:587–597
Dunn AL, Trivedi MH, Kampert JB, Clark CG, Chambliss HO (2005) Exercise treatment for depression: efficacy and dose response. Am J Prev Med 28:1–8
Duraiswamy G, Thirthalli J, Nagendra HR, Gangadhar BN (2007) Yoga therapy as an add-on treatment in the management of patients with schizophrenia–a randomized controlled trial. Acta Psychiatr Scand 116:226–232
Ellis N, Crone D, Davey R, Grogan S (2007) Exercise interventions as an adjunct therapy for psychosis: a critical review. British J Clin Psychol/British Psychol Soc 46:95–111
Engelhardt M, Neumann G (1994) Sports medicine. BLV Verlagsgesellschaft, Munich
Erickson KI, Voss MW, Prakash RS, Basak C, Szabo A, Chaddock L, Kim JS, Heo S, Alves H, White SM, Wojcicki TR, Mailey E, Vieira VJ, Martin SA, Pence BD, Woods JA, McAuley E, Kramer AF (2011) Exercise training increases size of hippocampus and improves memory. Proc Natl Acad Sci USA 108:3017–3022
Fabel K, Kempermann G (2008) Physical activity and the regulation of neurogenesis in the adult and aging brain. Neuromolecular Med 10:59–66
Falkai P, Malchow B, Wobrock T, Gruber O, Schmitt A, Honer WG, Pajonk FG, Sun F, Cannon TD (2012) The effect of aerobic exercise on cortical architecture in patients with chronic schizophrenia: a randomized controlled MRI study. Eur Arch Psychiatry Clin Neurosci [Epub ahead of print]. PubMed PMID:23161338
Farmer ME, Locke BZ, Moscicki EK, Dannenberg AL, Larson DB, Radloff LS (1988) Physical activity and depressive symptoms: the NHANES I epidemiologic follow-up study. Am J Epidemiol 128:1340–1351
Floel A, Ruscheweyh R, Kruger K, Willemer C, Winter B, Volker K, Lohmann H, Zitzmann M, Mooren F, Breitenstein C, Knecht S (2010) Physical activity and memory functions: are neurotrophins and cerebral gray matter volume the missing link? Neuroimage 49:2756–2763
Fogarty M, Happell B (2005) Exploring the benefits of an exercise program for people with schizophrenia: a qualitative study. Issues Mental Health Nurs 26:341–351
Ford ES, Giles WH, Dietz WH (2002) Prevalence of the metabolic syndrome among us adults: findings from the third national health and nutrition examination survey. JAMA 287:356–359
Fuss J, Ben Abdallah NM, Hensley FW, Weber KJ, Hellweg R, Gass P (2010) Deletion of running-induced hippocampal neurogenesis by irradiation prevents development of an anxious phenotype in mice. PloS One 5(9)
Fuss J, Ben Abdallah NM, Vogt MA, Touma C, Pacifici PG, Palme R, Witzemann V, Hellweg R, Gass P (2010) Voluntary exercise induces anxiety-like behavior in adult c57bl/6j mice correlating with hippocampal neurogenesis. Hippocampus 20:364–376
Galper DI, Trivedi MH, Barlow CE, Dunn AL, Kampert JB (2006) Inverse association between physical inactivity and mental health in men and women. Med Sci Sports Exerc 38:173–178
Garber CE, Blissmer B, Deschenes MR, Franklin BA, Lamonte MJ, Lee IM, Nieman DC, Swain DP (2011) American college of sports medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: guidance for prescribing exercise. Med Sci Sports Exerc 43:1334–1359
Goff DC, Sullivan LM, McEvoy JP, Meyer JM, Nasrallah HA, Daumit GL, Lamberti S, D’Agostino RB, Stroup TS, Davis S, Lieberman JA (2005) A comparison of ten-year cardiac risk estimates in schizophrenia patients from the catie study and matched controls. Schizophr Res 80:45–53
Gold JM, Goldberg RW, McNary SW, Dixon LB, Lehman AF (2002) Cognitive correlates of job tenure among patients with severe mental illness. Am J Psychiatry 159:1395–1402
Goldberg TE, Goldman RS, Burdick KE, Malhotra AK, Lencz T, Patel RC, Woerner MG, Schooler NR, Kane JM, Robinson DG (2007) Cognitive improvement after treatment with second-generation antipsychotic medications in first-episode schizophrenia: is it a practice effect? Arch Gen Psychiatry 64:1115–1122
Gorczynski P, Faulkner G (2010) Exercise therapy for schizophrenia. Schizophr Bull 36:665–666
Green MF (1996) What are the functional consequences of neurocognitive deficits in schizophrenia? Am J Psychiatry 153:321–330
Green MF, Kern RS, Braff DL, Mintz J (2000) Neurocognitive deficits and functional outcome in schizophrenia: are we measuring the “right stuff”? Schizophr Bull 26:119–136
Greenwood BN, Foley TE, Day HE, Campisi J, Hammack SH, Campeau S, Maier SF, Fleshner M (2003) Freewheel running prevents learned helplessness/behavioral depression: role of dorsal raphe serotonergic neurons. J Neurosci 23:2889–2898
Hall EE, Ekkekakis P, Petruzzello SJ (2007) Regional brain activity and strenuous exercise: predicting affective responses using eeg asymmetry. Biol Psychol 75:194–200
Hall EE, Petruzzello SJ (1999) Frontal asymmetry, dispositional affect and physical activity in older adults. J Aging Phys Activ 7:76–90
Hamilton M (1960) A rating scale for depression. J Neurol Neurosurg Psychiatry 23:56–62
Harris AH, Cronkite R, Moos R (2006) Physical activity, exercise coping, and depression in a 10-year cohort study of depressed patients. J Affect Disord 93:79–85
Harris EC, Barraclough B (1998) Excess mortality of mental disorder. Br J Psychiatry 173:11–53
Hasan A, Falkai P, Wobrock T, Lieberman J, Glenthoj B, Gattaz WF, Thibaut F, Moller HJ (2012) World federation of societies of biological psychiatry (wfsbp) guidelines for biological treatment of schizophrenia, part 1: update 2012 on the acute treatment of schizophrenia and the management of treatment resistance. World J Biol Psychiatry 13:318–378
Hautzinger M, Keller F, Kühner C (2006) Das beck depressionsinventar ii. Deutsche bearbeitung und handbuch zum bdi ii. Harcourt Test Services, Frankfurt a. M
Heggelund J, Nilsberg GE, Hoff J, Morken G, Helgerud J (2011) Effects of high aerobic intensity training in patients with schizophrenia: a controlled trial. Nord J Psychiatry 65:269–275
Heh SS, Huang LH, Ho SM, Fu YY, Wang LL (2008) Effectiveness of an exercise support program in reducing the severity of postnatal depression in Taiwanese women. Birth (Berkeley, Calif) 35:60–65
Heinrichs RW, Zakzanis KK (1998) Neurocognitive deficit in schizophrenia: a quantitative review of the evidence. Neuropsychology 12:426–445
Helgerud J, Hoydal K, Wang E, Karlsen T, Berg P, Bjerkaas M, Simonsen T, Helgesen C, Hjorth N, Bach R, Hoff J (2007) Aerobic high-intensity intervals improve vo2max more than moderate training. Med Sci Sports Exerc 39:665–671
Heyn P, Abreu BC, Ottenbacher KJ (2004) The effects of exercise training on elderly persons with cognitive impairment and dementia: a meta-analysis. Arch Phys Med Rehabil 85:1694–1704
Heyn PC, Johnson KE, Kramer AF (2008) Endurance and strength training outcomes on cognitively impaired and cognitively intact older adults: a meta-analysis. J Nutr Health Aging 12:401–409
Hoff AL, Sakuma M, Wieneke M, Horon R, Kushner M, DeLisi LE (1999) Longitudinal neuropsychological follow-up study of patients with first-episode schizophrenia. Am J Psychiatry 156:1336–1341
Hoff AL, Svetina C, Shields G, Stewart J, DeLisi LE (2005) Ten year longitudinal study of neuropsychological functioning subsequent to a first episode of schizophrenia. Schizophr Res 78:27–34
Hoffman BM, Babyak MA, Craighead WE, Sherwood A, Doraiswamy PM, Coons MJ, Blumenthal JA (2011) Exercise and pharmacotherapy in patients with major depression: one-year follow-up of the smile study. Psychosom Med 73:127–133
Hoffman BM, Blumenthal JA, Babyak MA, Smith PJ, Rogers SD, Doraiswamy PM, Sherwood A (2008) Exercise fails to improve neurocognition in depressed middle-aged and older adults. Med Sci Sports Exerc 40:1344–1352
Hollmann W (1963) Höchst- und dauerleistungsfähigkeit des sportlers. Barth, München
Hollmann W, Hettinger T (2000) Sportmedizin. Grundlagen für arbeit, training und präventivmedizin Schattauer, Stuttgart
Hopkins ME, Davis FC, Vantieghem MR, Whalen PJ, Bucci DJ (2012) Differential effects of acute and regular physical exercise on cognition and affect. Neuroscience 215:59–68
Hottenrott K, Neumann G (2010) Methods of endurance training. Verlag Karl Hofmann, Schorndorf
Hunsberger JG, Newton SS, Bennett AH, Duman CH, Russell DS, Salton SR, Duman RS (2007) Antidepressant actions of the exercise-regulated gene vgf. Nat Med 13:1476–1482
Jablensky A (2000) Epidemiology of schizophrenia: the global burden of disease and disability. Eur Arch Psychiatry Clin Neurosci 250:274–285
Jablensky A (1995) Schizophrenia: recent epidemiologic issues. Epidemiol Rev 17:10–20
Jerome J, Young DR, Dalcin A et al (2009) Physical activity levels of persons with mental illness attending psychiatric rehabilitation programs. Schizophr Res 108:252–257
Jerstad SJ, Boutelle KN, Ness KK, Stice E (2010) Prospective reciprocal relations between physical activity and depression in female adolescents. J Consult Clin Psychol 78:268–272
Keefe RS (2007) Cognitive deficits in patients with schizophrenia: effects and treatment. J Clin Psychiatry 68(Suppl 14):8–13
Kempermann G, Fabel K, Ehninger D, Babu H, Leal-Galicia P, Garthe A, Wolf SA (2010) Why and how physical activity promotes experience-induced brain plasticity. Frontiers Neurosci 4:189
Kendler KS, Gallagher TJ, Abelson JM, Kessler RC (1996) Lifetime prevalence, demographic risk factors, and diagnostic validity of nonaffective psychosis as assessed in a us community sample. The national comorbidity survey. Arch Gen Psychiatry 53:1022–1031
Kerse N, Hayman KJ, Moyes SA, Peri K, Robinson E, Dowell A, Kolt GS, Elley CR, Hatcher S, Kiata L, Wiles J, Keeling S, Parsons J, Arroll B (2010) Home-based activity program for older people with depressive symptoms: dellite–a randomized controlled trial. Ann Family Med 8:214–223
Killackey E, Anda AL, Gibbs M, Alvarez-Jimenez M, Thompson A, Sun P, Baksheev GN (2011) Using internet enabled mobile devices and social networking technologies to promote exercise as an intervention for young first episode psychosis patients. BMC Psychiatry 11:80
Kirkcaldy BD, Shephard RJ, Siefen RG (2002) The relationship between physical activity and self-image and problem behaviour among adolescents. Soc Psychiatry Psychiatr Epidemiol 37:544–550
Kirkpatrick B, Buchanan RW, McKenney PD, Alphs LD, Carpenter WT Jr (1989) The schedule for the deficit syndrome: an instrument for research in schizophrenia. Psychiatry Res 30:119–123
Knubben K, Reischies FM, Adli M, Schlattmann P, Bauer M, Dimeo F (2007) A randomised, controlled study on the effects of a short-term endurance training programme in patients with major depression. Br J Sports Med 41:29–33
Krogh J, Nordentoft M, Sterne JA, Lawlor DA (2011) The effect of exercise in clinically depressed adults: systematic review and meta-analysis of randomized controlled trials. J Clin Psychiatry 72:529–538
Kubesch S, Bretschneider V, Freudenmann R, Weidenhammer N, Lehmann M, Spitzer M, Gron G (2003) Aerobic endurance exercise improves executive functions in depressed patients. J Clin Psychiatry 64:1005–1012
Kucyi A, Alsuwaidan MT, Liauw SS, McIntyre RS (2010) Aerobic physical exercise as a possible treatment for neurocognitive dysfunction in bipolar disorder. Postgrad Med 122:107–116
Kutzner I, Heinlein B, Graichen F, Rohlmann A, Halder AM, Beier A, Bergmann G (2012) Loading of the knee joint during ergometer cycling: telemetric in vivo data. J Orthop Sports Phys Ther 42:1032–1038
Laboratories ACoPSfCPF (2002) Ats statement: guidelines for the six-minute walk test. Am J Respir Crit Care Med 166:111–117
Lampinen P, Heikkinen E (2003) Reduced mobility and physical activity as predictors of depressive symtposm among community-dwelling older adults: an eight-year follow-up study. Aging Clin Exp Res 15:205–211
Lindamer LA, McKibbin C, Norman GJ, Jordan L, Harrison K, Abeyesinhe S, Patrick K (2008) Assessment of physical activity in middle-aged and older adults with schizophrenia. Schizophr Res 104:294–301
Mabandla MV, Kellaway LA, Daniels WM, Russell VA (2009) Effect of exercise on dopamine neuron survival in prenatally stressed rats. Metab Brain Dis 24:525–539
Martinsen EW, Hoffart A, Solberg O (1989) Comparing aerobic with nonaerobic forms of exercise in the treatment of clinical depression: a randomized trial. Compr Psychiatry 30:324–331
Martinsen EW, Medhus A, Sandvik L (1985) Effects of aerobic exercise on depression: a controlled study. Br Med J (Clin Res Ed) 291:109
Marzolini S, Jensen B, Melville P (2009) Feasibility and effects of a group-based resistance and aerobic exercise program for individuals with severe schizophrenia: a multidisciplinary approach. Mental Health Phys Activity 2:29–36
Mather AS, Rodriguez C, Guthrie MF, McHarg AM, Reid IC, McMurdo ME (2002) Effects of exercise on depressive symptoms in older adults with poorly responsive depressive disorder: randomised controlled trial. Br J Psychiatry 180:411–415
McGrath J, Saha S, Chant D, Welham J (2008) Schizophrenia: a concise overview of incidence, prevalence, and mortality. Epidemiol Rev 30:67–76
McKercher CM, Schmidt MD, Sanderson KA, Patton GC, Dwyer T, Venn AJ (2009) Physical activity and depression in young adults. Am J Prev Med 36:161–164
Mead GE, Morley W, Campbell P, Greig CA, McMurdo M, Lawlor DA (2009) Exercise for depression. Cochrane Database Syst Rev (3):CD004366. doi:10.1002/14651858.CD004366.pub4
Merikangas KR, Akiskal HS, Angst J, Greenberg PE, Hirschfeld RM, Petukhova M, Kessler RC (2007) Lifetime and 12-month prevalence of bipolar spectrum disorder in the national comorbidity survey replication. Arch Gen Psychiatry 64:543–552
Methapatara W, Srisurapanont M (2011) Pedometer walking plus motivational interviewing program for thai schizophrenic patients with obesity or overweight: a 12-week, randomized, controlled trial. Psychiatry Clin Neurosci 65:374–380
Moraes H, Deslandes A, Ferreira C, Pompeu FAMS, Ribeiro P, Laks J (2007) Physical exercise in the treatment of depression in the elderly: a systematic review. Rev Psiquiatr R Gd Sul 29:79
Morroni F (2011) Repeated physical training and environmental enrichment induce neurogenesis and synaptogenesis following neuronal injury in an inducible mouse model. J Behav Brain Sci 01:199–209
Murray CJ, Lopez AD (1996) Evidence-based health policy–lessons from the global burden of disease study. Science 274:740–743
Neumann G, Schüler KP (1994) Diagnostics in sports medicine. Johann Ambrosius Barth Verlag, Leipzig Berlin Heidelberg
Neumann NU, Frasch K (2008) New aspects regarding physical exercise in psychiatry-clinical and scientific basics. Dtsch Z Sportmed 59:28–33
Oliff HS, Berchtold NC, Isackson P, Cotman CW (1998) Exercise-induced regulation of brain-derived neurotrophic factor (bdnf) transcripts in the rat hippocampus. Brain Res Mol Brain Res 61:147–153
Pajonk FG, Wobrock T, Gruber O, Scherk H, Berner D, Kaizl I, Kierer A, Muller S, Oest M, Meyer T, Backens M, Schneider-Axmann T, Thornton AE, Honer WG, Falkai P (2010) Hippocampal plasticity in response to exercise in schizophrenia. Arch Gen Psychiatry 67:133–143
Pereira AC, Huddleston DE, Brickman AM, Sosunov AA, Hen R, McKhann GM, Sloan R, Gage FH, Brown TR, Small SA (2007) An in vivo correlate of exercise-induced neurogenesis in the adult dentate gyrus. Proc Natl Acad Sci USA 104:5638–5643
Petruzzello SJ, Hall EE, Ekkekakis P (2001) Regional brain activation as a biological marker of affective responsivity to acute exercise: influence of fitness. Psychophysiology 38:99–106
Petruzzello SJ, Landers DM (1994) State anxiety reduction and exercise: does hemispheric activation reflect such changes? Med Sci Sports Exerc 26:1028–1035
Petruzzello SJ, Tate AK (1997) Brain activation, affect, and aerobic exercise: an examination of both state-independent and state-dependent relationships. Psychophysiology 34:527–533
Phelan M, Slade M, Thornicroft G, Dunn G, Holloway F, Wykes T, Strathdee G, Loftus L, McCrone P, Hayward P (1995) The Camberwell assessment of need: the validity and reliability of an instrument to assess the needs of people with severe mental illness. Br J Psychiatry 167:589–595
Poulin MJ, Chaput JP, Simard V, Vincent P, Bernier J, Gauthier Y, Lanctot G, Saindon J, Vincent A, Gagnon S, Tremblay A (2007) Management of antipsychotic-induced weight gain: prospective naturalistic study of the effectiveness of a supervised exercise programme. Aust NZ J Psychiatry 41:980–989
Rhyu IJ, Bytheway JA, Kohler SJ, Lange H, Lee KJ, Boklewski J, McCormick K, Williams NI, Stanton GB, Greenough WT, Cameron JL (2010) Effects of aerobic exercise training on cognitive function and cortical vascularity in monkeys. Neuroscience 167:1239–1248
Rimer J, Dwan K, Lawlor DA, Greig CA, McMurdo M, Morley W, Mead GE (2012) Exercise for depression. Cochrane Database Syst Rev 7:CD004366
Roine E, Roine RP, Rasanen P, Vuori I, Sintonen H, Saarto T (2009) Cost-effectiveness of interventions based on physical exercise in the treatment of various diseases: a systematic literature review. Int J Technol Assess Health Care 25:427–454
Sahay A, Scobie KN, Hill AS, O’Carroll CM, Kheirbek MA, Burghardt NS, Fenton AA, Dranovsky A, Hen R (2011) Increasing adult hippocampal neurogenesis is sufficient to improve pattern separation. Nature 472:466–470
Scheewe TW, Backx FJ, Takken T, Jörg F, van Strater AC, Kroes AG, Kahn RS, Cahn W (2013) Exercise therapy improves mental and physical health in schizophrenia: a randomised controlled trial. Acta Psychiatr Scand 127(6):464–473
Scheewe TW, van Haren NE, Sarkisyan G, Schnack HG, Brouwer RM, de Glint M, Hulshoff Pol HE, Backx FJ, Kahn RS, Cahn W (2013) Exercise therapy, cardiorespiratory fitness and their effect on brain volumes: a randomised controlled trial in patients with schizophrenia and healthy controls. Eur Neuropsychopharmacol 23(7):675–685
Schneider B, Prvulovic D, Oertel-Knochel V, Knochel C, Reinke B, Grexa M, Weber B, Hampel H (2011) Biomarkers for major depression and its delineation from neurodegenerative disorders. Prog Neurobiol 95:703–717
Silveira H, Deslandes AC, de Moraes H, Mouta R, Ribeiro P, Piedade R, Laks J (2010) Effects of exercise on electroencephalographic mean frequency in depressed elderly subjects. Neuropsychobiology 61:141–147
Singh NA, Clements KM, Fiatarone MA (1997) A randomized controlled trial of the effect of exercise on sleep. Sleep 20:95–101
Sjosten N, Vaapio S, Kivela SL (2008) The effects of fall prevention trials on depressive symptoms and fear of falling among the aged: a systematic review. Aging Mental Health 12:30–46
Srihari VH, Phutane VH, Ozkan B, Chwastiak L, Ratliff JC, Woods SW, Tek C (2013) Cardiovascular mortality in schizophrenia: defining a critical period for prevention. Schizophr Res 146:64–68
Stathopoulou G, Powers M, Berry A et al (2006) Exercise interventions for mental health: a quantitative and qualitative review. Clin PSychol Sci Pract 13:180–193
Strassnig M, Brar JS, Ganguli R (2006) Increased caffeine and nicotine consumption in community-dwelling patients with schizophrenia. Schizophr Res 86:269–275
Strassnig MT, Newcomer JW, Harvey PD (2012) Exercise improves physical capacity in obese patients with schizophrenia: pilot study. Schizophr Res 141:284–285
Strohle A, Hofler M, Pfister H, Muller AG, Hoyer J, Wittchen HU, Lieb R (2007) Physical activity and prevalence and incidence of mental disorders in adolescents and young adults. Psychol Med 37:1657–1666
Sui X, Laditka JN, Church TS, Hardin JW, Chase N, Davis K, Blair SN (2009) Prospective study of cardiorespiratory fitness and depressive symptoms in women and men. J Psychiatr Res 43:546–552
Takahashi H, Kato M, Sassa T, Shibuya T, Koeda M, Yahata N, Matsuura M, Asai K, Suhara T, Okubo Y (2010) Functional deficits in the extrastriate body area during observation of sports-related actions in schizophrenia. Schizophr Bull 36:642–647
Takahashi H, Sassa T, Shibuya T, Kato M, Koeda M, Murai T, Matsuura M, Asai K, Suhara T, Okubo Y (2012) Effects of sports participation on psychiatric symptoms and brain activations during sports observation in schizophrenia. Transl Psychiatry 2:e96
Tay YH, Nurjono M, Lee J (2013) Increased Framingham 10-year cvd risk in Chinese patients with schizophrenia. Schizophr Res 147:187–192
Taylor ED, Theim KR, Mirch MC, Ghorbani S, Tanofsky-Kraff M, Adler-Wailes DC, Brady S, Reynolds JC, Calis KA, Yanovski JA (2006) Orthopedic complications of overweight in children and adolescents. Pediatrics 117:2167–2174
Torres IJ, Boudreau VG, Yatham LN (2007) Neuropsychological functioning in euthymic bipolar disorder: a meta-analysis. Acta Psychiatr Scand 116(Suppl 434):17–26
Trappe HJ, Lollgen H (2000) guidelines for ergometry. German society of cardiology—heart and cardiovascular research. Z Kardiol 89:821–831
Ussher M, Stanbury L, Cheeseman V, Faulkner G (2007) Physical activity preferences and perceived barriers to activity among those with severe mental illness in the United Kingdom. Psychiatr Serv 58:405–408
Van Citters AD, Pratt SI, Jue K et al (2010) A pilot evaluation of the in shape individualized health promotion intervention for adults with mental illness. Community Ment Health J 46:540–552
van Dam RM, Li T, Spiegelman D, Franco OH, Hu FB (2008) Combined impact of lifestyle factors on mortality: prospective cohort study in us women. BMJ 337:a1440
van Praag H, Christie BR, Sejnowski TJ, Gage FH (1999) Running enhances neurogenesis, learning, and long-term potentiation in mice. Proc Natl Acad Sci USA 96:13427–13431
van Praag H, Kempermann G, Gage FH (1999) Running increases cell proliferation and neurogenesis in the adult mouse dentate gyrus. Nat Neurosci 2:266–270
van Praag H, Shubert T, Zhao C, Gage FH (2005) Exercise enhances learning and hippocampal neurogenesis in aged mice. J Neurosci 25:8680–8685
van Uffelen JG, Chinapaw MJ, Hopman-Rock M, van Mechelen W (2009) Feasibility and effectiveness of a walking program for community-dwelling older adults with mild cognitive impairment. J Aging Phys Activity 17:398–415
van Welie H, Derks EM, Verweij KH, de Valk HW, Kahn RS, Cahn W (2013) The prevalence of diabetes mellitus is increased in relatives of patients with a non-affective psychotic disorder. Schizophr Res 143:354–357
Vancampfort D, Correll CU, Probst M, Sienaert P, Wyckaert S, De Herdt A, Knapen J, De Wachter D, De Hert M (2013) A review of physical activity correlates in patients with bipolar disorder. J Affect Disord 145(3):285–291
Vancampfort D, Correll CU, Scheewe TW, Probst M, De Herdt A, Knapen J, De Hert M (2013) Progressive muscle relaxation in persons with schizophrenia: a systematic review of randomized controlled trials. Clin Rehabil 27(4):291–298
Vancampfort D, Probst M, Scheewe T, Knapen J, De Herdt A, De Hert M (2012) The functional exercise capacity is correlated with global functioning in patients with schizophrenia. Acta Psychiatr Scand 125:382–387
Vancampfort D, Vansteelandt K, Scheewe T, Probst M, Knapen J, De Herdt A, De Hert M (2012) Yoga in schizophrenia: a systematic review of randomised controlled trials. Acta Psychiatr Scand 126:12–20
Vasques PE, Moraes H, Silveira H, Deslandes AC, Laks J (2011) Acute exercise improves cognition in the depressed elderly: the effect of dual-tasks. Clinics (Sao Paulo, Brazil) 66:1553–1557
Visceglia E, Lewis S (2011) Yoga therapy as an adjunctive treatment for schizophrenia: a randomized, controlled pilot study. J Altern Complement Med 17:601–607
Wang J, Fan X, Liu D, Yi Z, Freudenreich O, Goff D, Henderson DC (2012) Both physical activity and food intake are associated with metabolic risks in patients with schizophrenia. Schizophr Res 140:260–261
Warren KR, Ball MP, Feldman S, Liu F, McMahon RP, Kelly DL (2011) Exercise program adherence using a 5-kilometer (5 k) event as an achievable goal in people with schizophrenia. Biol Res Nurs 13:383–390
Weinstein AA, Deuster PA, Francis JL, Beadling C, Kop WJ (2010) The role of depression in short-term mood and fatigue responses to acute exercise. Int J Behav Med 17:51–57
Wen CP, Wai JP, Tsai MK, Yang YC, Cheng TY, Lee MC, Chan HT, Tsao CK, Tsai SP, Wu X (2011) Minimum amount of physical activity for reduced mortality and extended life expectancy: a prospective cohort study. Lancet 378:1244–1253
Williams DR, Gonzalez HM, Neighbors H, Nesse R, Abelson JM, Sweetman J, Jackson JS (2007) Prevalence and distribution of major depressive disorder in african americans, caribbean blacks, and non-hispanic whites: results from the national survey of american life. Arch Gen Psychiatry 64:305–315
Wobrock T, Hasan A, Falkai P (2012) Innovative treatment approaches in schizophrenia enhancing neuroplasticity: aerobic exercise, erythropoietin and repetitive transcranial magnetic stimulation. Curr Pharm Biotechnol 13:1595–1605
Wolf SA, Melnik A, Kempermann G (2011) Physical exercise increases adult neurogenesis and telomerase activity, and improves behavioral deficits in a mouse model of schizophrenia. Brain Behav Immun 25:971–980
Wolff E, Gaudlitz K, von Lindenberger BL, Plag J, Heinz A, Strohle A (2011) Exercise and physical activity in mental disorders. Eur Arch Psychiatry Clin Neurosci 261(Suppl 2):S186–S191
Wright KA, Everson-Hock ES, Taylor AH (2009) The effects of physical activity on physical and mental health among individuals with bipolar disorder: a systematic review. Mental Health Phys Activity 2:86–94
Yogaratnam J, Biswas N, Vadivel R, Jacob R (2013) Metabolic complications of schizophrenia and antipsychotic medications–an updated review. East Asian Arch Psychiatry 23(1):21–28
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Malchow, B., Reich-Erkelenz, D., Oertel-Knöchel, V. et al. The effects of physical exercise in schizophrenia and affective disorders. Eur Arch Psychiatry Clin Neurosci 263, 451–467 (2013). https://doi.org/10.1007/s00406-013-0423-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00406-013-0423-2