Abstract
Generalized anxiety disorder (GAD) is characterized by excessive and persistent worrying. Neural substrates of this disorder are insufficiently understood, which relates to functional as well as to structural brain abnormalities. Especially, findings on the neuroanatomy of GAD have been inconsistent and were predominantly derived from pediatric samples. Therefore, we studied adult patients. Thirty-one women (16 patients with GAD and 15 healthy control participants) underwent structural MRI scanning. Gray matter volumes for specific brain regions involved in worrying, anticipatory anxiety, and emotion regulation were analyzed by means of voxel-based morphometry. Relative to controls, patients with GAD had larger volumes of the amygdala and the dorsomedial prefrontal cortex (DMPFC). Moreover, patients’ self-reports on symptom severity were positively correlated with volumes of the DMPFC and the anterior cingulate cortex. Patients with GAD show localized gray matter volume differences in brain regions associated with anticipatory anxiety and emotion regulation. This abnormality may represent either a predisposition for GAD or a consequence of disorder-specific behavior, such as chronic worrying. This issue should be addressed in future MRI studies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Generalized anxiety disorder (GAD) is characterized by persistent, excessive worries about everyday life circumstances (e.g. finances, work) and additional somatic symptoms (e.g. muscle tension, sleep disturbance). Although the clinical manifestation may begin in childhood, GAD is more common in adulthood [1, 28]. Different psychological models have been proposed to explain GAD. Borkovec et al. [2] underline the cognitive avoidance function of worrying. Worries are primarily verbal-semantic (as opposed to visual) in order to disengage from threatening imagery. It is the abstract nature of worrying that helps to control anticipatory arousal. Other psychological approaches refer to emotion processing and emotion regulation deficits in GAD [3]. This patient group has a poor understanding of their own emotions, a negative cognitive reactivity to affective experiences as well as difficulties in effectively dampening negative feelings by means of cognitive strategies [3].
Functional neuroimaging has provided evidence for an altered amygdala function in GAD. Three studies showed that afflicted patients displayed greater amygdala activity than healthy individuals, when they anticipated the presentation of an affective scene [4] and when they were exposed to pictures with angry or fearful faces [5, 6]. However, Blair et al. [7] identified amygdalar hyporesponsiveness to fearful faces in adult patients with GAD, whereas Whalen et al. [8] observed similar amygdalar reactivity in patients with GAD and in healthy controls. These conflicting results might be a consequence of an intra-amygdalar perturbation as demonstrated by Etkin et al. [9]. The authors examined patients with GAD in a resting condition and showed that they were characterized by a reduced connectivity between basolateral and centromedial amygdalar regions relative to healthy controls. Moreover, the clinical group showed increased amygdala connectivity with different prefrontal cortex (PFC) regions, such as the dorsomedial and ventromedial PFC.
There are several other brain imaging findings pointing to an altered PFC function in GAD. Hoehn-Saric et al. [10] presented patients with GAD with worry and neutral sentences. After pharmacological treatment, worry-induced activation decreased in the medial PFC, in the ACC, and in the insula. In a picture perception paradigm with affective facial expressions, adolescents with GAD displayed enhanced ventrolateral PFC activation and an attentional bias away from angry faces [11]. In a study by Paulesu et al. [12], dorsomedial PFC and ACC activity was positively correlated with symptom severity in patients with GAD while they read worry-eliciting sentences. Etkin et al. [13] identified a deficit in ACC recruitment in patients with GAD during emotional conflict. The patients had been presented with emotional facial expressions together with either congruent or incongruent affective labels. The ACC hyporesponsiveness to incongruence was interpreted to reflect a dysfunction in automatic emotion regulation.
A few structural imaging studies focused on GAD in children and adolescents [14–16]. In a first study [14], the pediatric GAD sample had larger amygdala volumes than the healthy control sample. A subsequent investigation [15] demonstrated that the superior temporal gyrus (white and gray matter) was significantly larger in patients with GAD compared with controls. Finally, Milham et al. [16] studied a group of children with different anxiety disorders (social phobia, GAD, separation anxiety). Relative to healthy controls, the patients were characterized by reduced amygdala volumes. This reduction was more pronounced in patients with a GAD diagnosis as opposed to those without GAD. The heterogeneous findings may be due to difficulties in reliably classifying pediatric GAD. This disorder may be overdiagnosed in children presenting with anxiety [1, 28]. Moreover, neurodevelopmental aspects (e.g. individual differences in brain development) may contribute to increased variance and sample-dependent findings.
To our knowledge, there is only one published morphometric study on GAD in adults [9]. The authors reported increased amygdala volume for the clinical group. Of the 16 studied patients, ten suffered from at least one comorbid disorder (e.g. major depression, social anxiety disorder, panic disorder) and four patients were taking antidepressant medication. This constellation is typical for GAD, where additional diagnoses are common [1]. However, the comorbidity makes a direct attribution of structural abnormalities to the GAD diagnosis difficult.
Therefore, in the present study, we included only those patients with GAD who did not currently suffer from another mental disorder and did not take any psychiatric medication. Based on the previous functional and structural MRI studies [4, 9, 13], we hypothesized that GAD would be associated with increased amygdala volume. Further, we related self-reports on symptom severity to gray matter volumes in previously identified brain regions involved in worrying and uncertainty processing, such as the amygdala, the insula, the ACC, and prefrontal regions (DMPFC, VLPFC, VMPFC).
Method
Sample
Sixteen women suffering from GAD according to DSM-IV [1] and 15 healthy women participated in this study. Both groups were matched with regard to their age (GAD: M = 22.9 years, SD = 4.1 years; CG: M = 23.7 years, SD = 3.7 years), their years of education (GAD = 11.1 years, SD = 2.9 years; CG = 11.3 years, SD = 3.3 years), and with regard to their profession (all white collar). The sample had been restricted to women as the GAD prevalence is considerably higher in the female population [1, 28]. The patients suffered on average 3.1 years from GAD (SD = 4.7 years), but reported that they felt anxious and nervous all their lives. The diagnosis of a further mental disorder besides GAD at the time of assessment led to exclusion from the patient sample. All women were nonmedicated, right-handed and had been recruited via announcements in local newspapers. Written informed consent was obtained from each participant prior to entry. The study had been approved by the local ethics committee.
Procedure
The study consisted of two different sessions. In the first session, all participants underwent a standardized clinical interview for DSM-IV diagnoses [17] and answered questionnaires: (a) the Penn State Worry Questionnaire [18] is a 16-item measure most frequently used to assess worrying in both clinical and nonclinical samples. The Cronbach’s α is 0.87. (b) The MetaCognition Questionnaire [19] assesses beliefs about worries with 65 items, e.g. the concept that worrying contributes to problem solving. The Cronbach’s α is 0.93. (c) All participants completed the BDI [20]; α = 0.88). Patients with GAD additionally received a nonstandardized exploration for specific aspects of their disorder (e.g. content of worries, duration of disorder). In the second session, all participants underwent structural scanning.
MRI scanning and data processing
High resolution structural MRI scans (field of view = 256 × 256 mm, TR 1,900 ms, TE: 2,200 ms, TI: 900 ms, flip angle: 90°, voxel size: 1 × 1 × 1 mm) were acquired using a 3D-MPRAGE sequence on a 3T tomograph (Siemens Trio, Erlangen, Germany). Data analysis was carried out with SPM5 (Wellcome Department of Imaging Neuroscience, London, UK) and the VBM toolbox (VBM5.1 version 1.19; http://dbm.neuro.uni-jena.de/vbm/vbm5-for-spm5/). Individual structural scans were segmented using unified segmentation [21]. A hidden Markov random field approach was applied for the segmentation in order to avoid misclassification of noncontiguous voxels. Modulated segments were used in order to assess volume differences. As a final preprocessing step, segments were smoothed by a Gaussian kernel of 12 mm. Two-sample t-tests (GAD <> CG) were computed to assess group differences. In addition, covariations of volumes with self-report measures were computed by means of multiple regressions for each group. In all analyses, total gray matter was considered a covariate. Segments were thresholded prior to all statistical analysis with an absolute value of 0.15. We computed exploratory whole brain analyses as well as region of interest (ROI) analyses. The ROIs were created with WFUPickatlas (version 2.4, [22]) and based on the parcellation of Tzourio-Mazoyer et al. [23]. Statistical parametric maps were initially thresholded by an uncorrected P of 0.005. P-values <0.05 corrected for family-wise error were considered significant for exploratory and ROI analyses. Small volume correction was conducted individually for each ROI analysis for the following masks: amygdala, insula, ACC, DMPFC, VMPFC, and VLPFC.
Results
Self-report data
Relative to the control group, the patients with GAD scored higher on the PSWQ (M GAD = 62.8, SD = 4.7; M CG = 40.1, SD = 6.8; t(29) = 9.24, P < 0.001), on the MCQ (M GAD = 131.0, SD = 20.5; M CG = 113.5, SD = 22.2; t(29) = 2.3, P = 0.03), and on the BDI (M GAD = 11.4, SD = 5.1; M CG = 4.9, SD = 4.4; t(29) = 2.6, P = 0.016).
GMV data
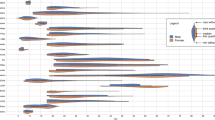
The ROI analysis showed that the patients with GAD had larger gray matter volumes of the bilateral amygdala and the right DMPFC (Table 1; Fig. 1). The reversed contrast (CG > GAD) showed no localized increases in GMV. The exploratory tests were nonsignificant.
Within the patient sample, PSWQ scores were positively correlated with bilateral gray matter volumes of the ACC and the DMPFC. MCQ scores were positively correlated with the right DMPFC volume. Such correlations were not present in the control group.
Discussion
The present VBM investigation on GAD identified enlarged volumes of the amygdala and the dorsomedial prefrontal cortex (DMPFC) in the clinical group relative to the healthy comparison group. Thus, we were able to replicate and extend findings of a previous morphometric study on adult patients with GAD [9]. The observed difference in amygdalar gray matter might be linked to a disorder-related hyperresponsiveness of this structure. Nitschke et al. [4] demonstrated that the patients with GAD were characterized by enhanced amygdala responses during aversive anticipation. As patients with GAD consistently experience apprehensive expectations (over many years), this might lead to a volume enlargement of the amygdala. Several structural imaging studies have identified activity-dependent selective changes in gray matter before [24, 25].
The second enlarged brain area, the DMPFC, has been implicated in worry states [10, 12] and in anticipatory anxiety [26, 29]. Moreover, Etkin et al. [9] identified increased amygdalar connectivity with the DMPFC in their studied GAD sample. This altered coupling may be due to the habitual engagement in worrying that can be understood as a dysfunctional control mechanism to dampen negative affective states. Further, patients with GAD were unable to decrease DMPFC activation after the occurrence of an emotional conflict [13]. Thus, they continued to use cognitive coping despite changed situational requirements.
In line with these previous reports, the present study showed that patients’ DMPFC volumes were positively associated with their self-report on the degree of worrying (PSWQ) and with their belief about the possible usefulness of worries (MCQ). Finally, the ACC volume in patients with GAD was positively correlated with their PSWQ scores. The ACC is concerned with many emotion-relevant processes including anticipatory arousal [27]. In a study by Paulesu et al. [12], DMPFC and ACC activity had been positively correlated with symptom severity in patients with GAD while worrying. Altogether, the localized GMV differences identified in the present VBM study fit nicely to functional data for this patient group as well as to psychological GAD models.
Some limitations of the present study need to be mentioned. We only studied female patients with GAD Therefore, the results cannot be generalized to men. The sample size was modest and should be extended in the future. However, the clinical group can be considered homogenous as the patients did not suffer from comorbid disorders at the time of the testing and they were nonmedicated. Previous heterogeneous findings (e.g. with regard to increased vs. decreased amygdala volume) might be a result of different comorbidity constellations. Moreover, the functional meaning of the observed GMV differences needs to be elaborated. Are the observed increased volumes indicators of an elevated GAD proneness or a consequence of persistent worrying? The latter interpretation implies that the localized GMV alteration might be reversible by pharmacotherapy or psychotherapy. This issue should be addressed in a future study.
References
American Psychiatric Association (2000) Diagnostic and statistical manual of mental disorders, 4th edn. American Psychiatric Press, Washington, DC
Borkovec TD, Alcaine O, Behar E (2004) Avoidance theory of worry and generalized anxiety disorder. In: Heimberg RG, Turk C, Mennin DS (eds) General anxiety disorder: advances in research and practice. Guilford Press, New York, pp 77–108
Mennin DS, Heimberg RG, Turk CL, Fresco DM (2005) Preliminary evidence for an emotion dysregulation model of generalized anxiety disorder. Behav Res Ther 43:1281–1310
Nitschke JB, Sarinopoulos I, Oathes DJ, Johnstone T, Whalen PJ, Davidson RJ, Kalin NH (2009) Anticipatory activation in the amygdala and anterior cingulate in generalized anxiety disorder and prediction of treatment reponse. Am J Psychiatry 166:302–310
Monk CS, Telzer EH, Mogg K, Bradley BP, Mai X, Louro HM, Chen G, McClure-Tone EB, Ernst M, Pine DS (2008) Amygdala and ventrolateral prefrontal cortex activation to masked angry faces in children and adolescents with generalized anxiety disorder. Arch Gen Psychiatry 65:568–576
McClure EB, Monk CS, Nelson EE, Parrish JM, Adler A, Blair RJ, Fromm S, Charney DS, Leibenluft E, Ernst M, Pine DS (2007) Abnormal attention modulation of fear circuit function in pediatric generalized anxiety disorder. Arch Gen Psychiatry 64:97–106
Blair K, Shaywitz J, Smith BW, Rhodes R, Geraci M, Jones M, McCaffrey D, Vythilingam M, Finger E, Mondillo K, Jacobs M, Charney DS, Blair RJ, Drevets WC, Pine DS (2008) Response to emotional expressions in generalized social phobia and generalized anxiety disorder: evidence for separate disorders. Am J Psychiatry 165:1193–1202
Whalen PJ, Johnstone T, Somerville LH, Nitschke JB, Polis S, Alexander AL, Davidson RJ, Kalin NH (2007) A functional magnetic resonance imaging predictor of treatment response to venlafaxine in generalized anxiety disorder. Biol Psychiatry 63:858–863
Etkin A, Prater KE, Schatzberg AF, Menon V, Greicius MD (2009) Disrupted amygdalar subregion functional connectivity and evidence for a compensatory network in generalized anxiety disorder. Arch Gen Psychiatry 66:1361–1372
Hoehn-Saric R, Schlund MW, Wong SSY (2004) Effects of citalopram on worry and brain activation in patients with generalized anxiety disorder. Psychiat Res Neuroim 13:111–121
Monk CS, Nelson EE, McClure EB, Mogg K, Bradley BP, Leibenluft E (2006) Ventrolateral prefrontal cortex activation and attentional bias in response to angry faces in adolescents with generalized anxiety disorder. Am J Psychiatry 163:1091–1097
Paulesu E, Sambugaro E, Torti T, Danelli L, Ferri F, Scialfa G, Sberna M, Ruggiero GM, Bottini G, Sassaroli S (2009) Neural correlates of worry in generalized anxiety disorder and in normal controls: a functional MRI study. Psychol Med 7:1–8
Etkin A, Prater KE, Hoeft F, Menon V, Schatzberg AF (2010) Failure of anterior cingulate activation and connectivity with the amygdala during implicit regulation of emotional processing in Generalized Anxiety Disorder. Am J Psychiatry 167:545–554
De Bellis MD, Casey BJ, Dahl R, Birmaher B, Williamson D, Thomas KM, Axelson DA, Frustaci K, Boring AM, Hall J, Ryan ND (2000) A pilot study of amygdala volumes in pediatric generalized anxiety disorder. Biol Psychiatry 48:51–57
De Bellis MD, Keshavan MS, Shifflett H, Iyengar S, Dahl RE, Axelson DA (2002) Superior temporal gyrus volumes in pediatric generalized anxiety disorder. Biol Psychiatry 51:553–562
Milham MP, Nugent AC, Drevets WC, Dickstein DP, Leibenluft E, Ernst M, Charney D, Pine DS (2005) Selective reduction in amygdala volume in pediatric anxiety disorders: a voxel-based morphometry investigation. Biol Psychiatry 57:961–966
Margraf J (1994) Diagnostisches kurz-interview bei psychischen Störungen (Mini-DIPS). Springer, Berlin
Stoeber J (1995) Penn state worry questionnaire–German version. Z Diff Diagn Psychol 16:50–63
Möbius J, Hoyer J (2003) Metacogntion questionnaire (MKF). In: Hoyer J, Margraf J (eds) Angst-diagnostik. Grundlagen und Testverfahren. Berlin, Springer, pp 220–223
Hautzinger M, Bailer M, Worall H, Keller F (1993) Beck-depressions-inventar (BDI). Bern, Huber
Ashburner J, Friston K (2006) Unified segmentation. NeuroImage 26:839–851
Maldjian JA, Laurienti PJ, Burdette JB, Kraft RA (2003) An automated method for neuroanatomic and cytoarchitectonic atlas-based interrogation of fMRI data sets. NeuroImage 19:1233–1239
Tzourio-Mazoyer N, Landeau B, Papathanassiou D, Crivello F, Etard O, Delcroix N (2002) Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. NeuroImage 15:273–289
Draganski B, Gaser C, Busch V, Schuierer G, Bogdahn U (2004) Neuroplasticity: changes in gray matter induced by training. Nature 427:311–312
Maguire EA, Gadian DG, Johnsrude IS, Good CD, Ashburner J (2000) Navigation-related structural change in the hippocampi of taxi drivers. Proc Natl Acad Sci USA 97:4398–4403
Nitschke JB, Sarinopoulos I, Mackiewicz KL, Schaefer HS, Davidson RJ (2006) Functional neuroanatomy of aversion and its anticipation. NeuroImage 29:106–116
Critchely H, Mathias C, Dolan R (2001) Neural activity in the human brain relating to uncertainty and arousal during anticipation. Neuron 29:537–545
Angst J, Gamma A, Baldwin DS, Ajdacic-Gross V, Rösler W (2009) The generalized anxiety spectrum: prevalence, onset, course and outcome. Eur Arch Psychiatry Clin Neurosci 259:37–45
Schienle A, Schäfer A, Hermann A, Rohrmann S, Vaitl D (2007) Symptom provocation and symptom reduction in spider phobia. Eur Arch Psychiatry Clin Neurosci 257:486–493
Conflict of interest
The authors have no conflict of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schienle, A., Ebner, F. & Schäfer, A. Localized gray matter volume abnormalities in generalized anxiety disorder. Eur Arch Psychiatry Clin Neurosci 261, 303–307 (2011). https://doi.org/10.1007/s00406-010-0147-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00406-010-0147-5