Abstract
Background
Reverse total shoulder arthroplasty (RSA) can improve function in cuff tear arthropathy (CTA) shoulders, but limited exact data are available about the maximum values in 3D motion analysis, and as to how improvements translate into the normal range of motion (ROM) in activities of daily living (ADLs).
Methods
This study included nine consecutive patients (n = 9) who received RSA for CTA without muscle transfers. We measured shoulder movement by a novel 3D motion analysis using the Heidelberg upper extremity model (HUX) which can eliminate compensatory movements of the scapula, and the trunk. The measurement included active maximum values, and four ADLs.
Results
Comparing the pre- to the 1-year postoperative status, RSA was associated with a significant increase in the mean maximum values for active flexion of about 43° (SD ± 31) from 66° to 109° (p = 0.001), for active abduction of about 37° (SD ± 26) from 57° to 94° (p = 0.001), and for the active adduction of about 28° (SD ± 10) from 5° to 33° (p = 0.002). Comparing the preoperative to the postoperative ROM in the ADLs in flexion/extension, ROM improved significantly in all ADLs, in abduction/adduction in three of four ADLs. No significant changes were observed in internal/external rotation in any ADLs.
Conclusion
RSA improves the active maximum ROM for flexion, abduction, and adduction. The patients are able to take advantage of this ROM increase in ADLs in flexion and in most ADL in abduction, but only in trend in internal and external rotation.
Level of evidence
Level IV, Case Series with no comparison group.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
A massive defect of the rotator cuff in combination with secondary glenohumeral osteoarthritis are the characteristics of a cuff tear arthropathy (CTA) [1, 2]. Patients with advanced CTA suffer from pain and a reduced range of motion (ROM) and they have trouble to perform activities of daily living (ADL). Reverse total shoulder arthroplasty (RSA) is widely used in these patients to regain and improve function. As described by Grammont [3], a RSA medializes and lowers the glenohumeral center of rotation. This increases the lever arm of the deltoid muscle [4] and increases muscle fiber recruitment of the anterior and posterior deltoid to compensate for a deficient rotator cuff in CTA shoulders. Postoperatively, in addition to pain relief, RSA can improve CTA shoulder function [5–10], but it is unclear if the ability to process ADL also enhances. The constant score (CS) includes a common basic subjective assessment of ADL [11, 12] by use of questions. To date there is very limited data about objective 3D motion analysis of ADLs after RSA. Therefore, the purpose of this study was three fold: (1) to objectively examine how RSA changes the maximum active glenohumeral ROM; (2) how this translates into the ability to perform ADL and (3) in which plane the ROM in ADL changes using a prospective 3D motion analysis study [13]. The hypothesis was that RSA significantly improved the ability to perform ADL, and the ROM in ADL. The focus was on the question which plane displays the biggest improvement.
Materials and methods
Patients
The study included nine consecutive patients (seven women, two men), mean age 73.4 years (SD ± 4.5), who received an Aequalis reverse shoulder prosthesis (Tornier Inc, USA) for CTA. The dominant side was involved in all nine cases, and the right side was affected in all patients, too. In all nine patients the same surgeon (FZ) at our department had performed the surgical procedure. The CS, the American shoulder and elbow scores (ASES), and the disability of arm, shoulder and hand (DASH) scores [12, 14, 15] were rated pre- and post-operatively. Patients were included in the study in cases of CTA with superior humeral head migration and subsequent concave deformity of the acromion, and entire lack of the supraspinatus and infraspinatus tendons with retraction and fatty infiltration of the muscles. Patients were excluded, in cases of a stiff shoulder, CTA after humeral fractures, and neurological and muscular diseases or comorbidity which made the movement examination impossible, a lack of verbal communication, and additionally fracture. The preoperative X-ray imaging in true AP view showed an acetabularization (concave deformity of the acromion undersurface [16]) in all cases and, correspondingly, a neoglenoid cavity. We used the classification of Sirveaux et al. [9] to assess the glenoid erosion preoperatively. The preoperative status of the rotator cuff was rated in accordance with Fuchs [17] which is an adaptation of Goutallier et al. [18] for MRI. All patients had severe atrophy (Fuchs stage 3 or 4) or complete loss of the teres minor muscle [19].
Operative technique and implants
Standard surgical technique through a deltopectoral approach was employed according to Neer in all patients [20]. A tenotomy of the subscapularis tendon (if still present) and the long biceps tendon was realized. The Aequalis reverse shoulder prosthesis (Tornier Inc, USA) was used in all patients. No revision surgery was performed for implant failure. All the humeral stems were cemented after a cement restrictor was inserted. Cemented stems were used in all cases because a cementless stem in RSA was not recommended by the company at that time. Furthermore, to our knowledge there are no data that cementless stems in RSA provide the same excellent long-term outcome as the cemented ones [2]. Nonconstrained humeral polyethylene liners of 6 mm were then impacted on the humeral components. In all nine cases subscapularis reconstruction was possible, and the tendon was repaired using three nonabsorbable tendon-to-tendon sutures. Postoperative rehabilitation was also standardized with a 45° abduction pillow for 4 weeks. During the first 6 weeks, only passive mobilization was allowed as a combined elevation and abduction of 60° and 0° of external rotation in an outpatient facility. To exercise muscle strength and ROM, 6 weeks after surgery, the patients started with a 21-day program of free passive and active movement at a specialized rehabilitation center.
Motion analysis
The local ethics committee authorized the study protocol (S-305/2007), and informed consent was gathered from all patients. The patients were examined the day before RSA and 1 year after surgery. We used a 3D motion analysis and the HUX model as described before [21] with a 12-camera motion analysis system (Vicon 612; Vicon, Lake Forest, USA) operating at 120 Hz to monitor the patients’ movements. The HUX model as described by Rettig [13] uses seven segments for 3D motion analysis of the upper extremities. The characteristic of the HUX is that it calculates the shoulder joint center from the motion data and is able to capture the motion of this calculated shoulder joint center in relation to the torso. The centers of the joints are not deterministically defined via anthropometric averages based on markers that are fixed on bony prominences, which is the case, for example, in the model of Rab [22], but rather the functional center of the joints and the axes of the joints are determined across the motion of the markers. In this way, a more exact exposure of the glenohumeral ROM is possible and the success of surgery, especially for the glenohumeral joint, can be evaluated better. For the motion analysis, four markers were placed on the trunk of the patients as recommended by the International Society of Biomechanics [23] for this measurement, and supplementary four markers were placed on each forearm: two markers, connected with a wand, on the ulna close to the elbow joint, one at the ulnar and one at the radial styloid process of the wrist (Fig. 1).
After placing the markers, the patient performed isolated movements of elbow flexion/extension, shoulder flexion/extension, and shoulder abduction/adduction to determine the position of the center of rotation of the glenohumeral joint. In these shoulder calibration trials, the sternoclavicular joint was treated as a Cardan joint [13]. We did not deduce the technical coordinate systems for the ulna/forearm, humerus, clavicle, and thorax by optimization methods as has been done for marker clusters [24], in fact they were based directly on marker trajectories, i.e., the direction vectors between them, using cross-products as reviewed by Chiari et al. [25]. After these calibration trials, the actual measurement started: first, the maximum values were recorded for flexion (anteversion), extension (retroversion), abduction, and adduction. For this, the subjects sat on a stool and were instructed to move their arms to the respective maximum position without moving their torso. To calculate the results for flexion/extension together with abduction/adduction, the corresponding angles between the long axis of the body and the humerus were recorded. The body’s long axis is fixed to the thorax; hence, compensatory motion of the thorax can be monitored and separated from shoulder motion. Angles of flexion/extension and abduction/adduction were expressed as projection angles relative to the proximal anatomical coordinate system. The maximum rotation, defined by the globe convention [26], was measured at 90° of arm abduction. After determining the maximum values, we recorded ADL, including the following movements: “combing the hair (cmb)”, “washing the opposite armpit (wsh)”, “tying an apron (aprn)” and “taking a book from a shelf (shlf)”. Starting from the sitting position mentioned above, the subject was asked to implement these movements without moving the torso. Original position was the static calibration recording and each movement was carried out three times in a row. For “cmb” the subject hat a comb in his/her hand and placed it at the forehead for combing from there to the back of the head and back to the original position. For “wsh” the subject was given a washcloth and asked to move it to the opposite armpit and make a typical motion of washing there and then to go back to the original position. For the movement “aprn” the subject was also given a washcloth and asked to move his/her hand to their bottom and make a typical motion of wiping and then to return to the original position. For “shlf” a self-constructed, height-adjustable shelf was employed. The height of the shelf was set at the forehead and the book positioned at the subjects’ respective arm length. Then, the test person took the book in his/her hand, moved back to the original position with the book, put it back on the shelf, and finally returned to the original position without the book.
Statistics
The statistical analysis was performed using SPSS Version 16.0 (SPSS Inc., Chicago, IL, USA). Group mean values (MV) and standard deviations (SD) were calculated. P values <0.05 were considered as significant. A paired t test was used to compare the preoperative and postoperative CS and subscores. The distribution of the data was assessed by using the Shapiro–Wilk test and the homogeneity of variance using the Levene test. The angle between the long axis of the humerus and the trunk position was determined. Deviations in shoulder joint angles between target and reproduced positions were compared between the pre- and post-operative examination by using the Wilcoxon test.
Results
Clinical outcome and patient satisfaction
The mean raw CS for the entire cohort improved from 26.3 points [±7.1 (17–40 points)] preoperatively to 40.9 points [±7.6 (26–51 points)] postoperatively (p < 0.0001) (Table 1). Significant differences were also found in terms of pain relief, power, activity, mobility, shoulder flexion, abduction, and external and internal rotation (p < 0.05). The ASES and DASH score improved significantly pre- to post-operatively (Table 1).
Maximum values
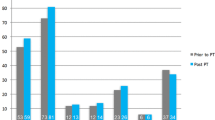
Comparing the pre- to the 1-year post-operative status, RSA resulted in a significant increase in the average maximum values for active flexion of about 43° (SD ± 31) from 66° to 109° (p = 0.001), for active abduction of about 37° (SD ± 26) from 57° to 94° (p = 0.001), and for active adduction of about 28° (SD ± 10) from 5° to 33° (p = 0.002) (Fig. 2). For the internal and external rotation there were no significant changes. By trend for the active external rotation, the ROM improved by about 5° (SD ± 16) from 13° to 18° (p = 0.582); by trend for the active internal rotation, the ROM improved by about 10° (SD ± 17) from 7° to 17° (p = 0.132) (Fig. 2). By trend retroversion (extension) deteriorated by about 7° (SD ± 15) from 36° to 29° (p = 0.398) (Fig. 2).
ROM in ADLs
One year after surgery, almost all of the RSA patients were able to perform the four ADLs, whereas before surgery, only about half of the patients were able to do so (Table 2). Comparing the pre- to the post-operative status, RSA resulted in a significant increase in the mean ROM values for flexion/extension for four ADLs and abduction/adduction for three ADLs. In flexion/extension, a significant improvement was observed for the Cmb task, from 35°–8°–0° preoperatively to 95°–30°–0° postoperative (p < 0.001), from 42°–11°–0° to 63°–22°–0° (p = 0.012) for Wsh, from 18°–0°–27° preoperatively to 37°–0°–28° postoperatively (p = 0.022) for Aprn, and from 19°–1°–0° to 87°–10°–0° (p = 0.002) for the Shlf task. In abduction/adduction, ROM improved significantly for Cmb from 29°–5°–0° preoperatively to 98°–24°–0° postoperatively (p < 0.001), for Aprn from 27°–11°–0° to 43°–19°–0° (p = 0.013), and for Shlf from 16°–4°–0° to 99°–15°–0° (p = 0.002) (Fig. 3). There were no significant changes in internal/external rotation for any of the ADLs (Fig. 3).
Discussion
The current study for the first time used a prospective 3D motion analysis study and the HUX model [13] in patients with CTA with deficient teres minor and no muscle transfers. The purpose of this study was three fold: (1) to objectively examine how RSA changes the maximum active glenohumeral ROM; (2) how this translates into the ability to perform ADL and (3) in which plane the ROM in ADL changes. The study demonstrates that RSA improves the active glenohumeral ROM for flexion by about 43°, abduction by about 37°, and adduction by about 28°. The patients are able to take advantage of the latter ROM increase in ADLs, but there was no significant change in internal and external rotation. By trend, there was an improvement in active external rotation by about 5° and for active internal rotation by about 10°. By trend, extension deteriorated by about 7°. In comparison to the average postoperative active flexion of 123° reported in other studies (Table 3), our RSA group showed less postoperative glenohumeral ROM at 109°. This might be explained by the different measurement methods. Flexion in the clinical papers is a combination of glenohumeral and scapulothoracic motion and flexion was measured using a conventional manual goniometer or an electric goniometer, whereby it is hardly possible to achieve sufficient separation of the glenohumeral joint movement from compensatory motion of the shoulder girdle or the trunk. It can be assumed that the scapulothoracic motion and maybe compensatory movements of the trunk were included in the values of the clinical papers. Comparable to the present study is the electromagnetic study of Alta et al. [27]: they measured actively performed ROM tasks and found a minor active glenohumeral ROM for forward flexion (71°) in contrast to the average reported in other studies after 23 months. We assume that these lower values better describe the true benefit of RSA, although we are aware that a comparison is difficult. Follow-up in the present study varies from that in other published studies: the follow-up is shorter and ROM can further improve over time. Another fact influencing the postoperative ROM is the shift of the center of rotation of the glenohumeral joint, classically combined with a medialization to improve efficiency of the deltoid [3, 28–30]. Lateralizing the center of rotation in RSA has been suggested as a potential means of limiting the degree of scapular notching and of improving external rotation [31]. As we described previously [32], the location of the functional center of rotation can be located using the HUX model with an accuracy of 0.1 mm. In the aforementioned study, a comparable patient group showed a medialization of 8.3 mm. It has also been shown that the HUX model better represents glenohumeral joint motion than manual goniometer measurements [13, 33]; however, this is not the only advantage in contrast to electromagnetic tracking device studies [27]. Indeed, the benefit of marker-based 3D motion analysis using the HUX model is that it provides an objective, precise analysis of complex ADLs, such as taking a book from a shelf, as performed in the present study. Thus, our study provides valuable insight into the movement of a reverse, replaced shoulder while performing ADLs and shows that patients were able to use the postoperative ROM increase in flexion, abduction, and adduction while performing ADLs. In the present study, we tried to answer the question, how RSA patients use the ROM changes in performing ADLs: the results show that RSA improves flexion ROM in ADLs but when performing ADLs, RSA patients do not use their maximum available flexion ROM of 109°. In ADLs they use a maximum of 95° in performing the Cmb task. In the literature, the study group published a comparable control group, performing the same ADLs [34]. These healthy controls used 139°–0°–69° in flexion/extension and 118°–0°–37° in abduction/adduction. Thus, postoperatively, RSA permits the patient of this study to perform nearly all required ADLs painlessly (Table 2). Considering the maximum ROM, RSA patients were not able using their maximum available ROM to perform the ADLs in the early follow-up. This might be related to limitations in active ROM, and changed shoulder geometry and motion patterns after RSA. Further studies with evaluation of the muscular motion patterns should focus on that.
3D motion analysis using the HUX model seems to be a valuable tool to exactly detect surgery-related changes in CTA shoulders after RSA. Patients included in this study had a deficient teres minor. There is some data that this can cause only minor improvement of external rotation after RSA. There are some promising results of transfer of the latissimus dorsi and the teres major in a one-stage procedure with RSA [35]. Future studies could exactly measure the improvement of ARO in ADL in these patients.
There are some limitations of the current study: there are very few studies which exactly measure glenohumeral motion, but clinically flexion includes a combined motion of the scapula on the trunk and the glenohumeral joint. Therefore, a comparison of the current data to the clinical values is difficult. Future studies will have to address the issue of the exact measurement of the scapulathoracic motion. Only nine patients performing four ADL could be included in the current “pilot study” which is a relatively small study population. This however, can be explained by the complex and elaborate study setup that prevents the examination of larger study populations.
Conclusion
RSA improves the active glenohumeral ROM for flexion, abduction, and adduction. The values measured in 3D motion analysis are lower than the ones measured by use of a manual goniometer. The patients who received a RSA could perform at least three out of four ADL. The patients had an improved flexion, abduction, and adduction in the four tested ADLs, but there is no significant change in internal and external rotation. This supports the point of view that muscle transfers have to be added if this issue should be addressed.
References
Neer CS, Craig EV, Fukuda H (1983) Cuff-tear arthropathy. J Bone Jt Surg Am 65(9):1232–1244
Cil A et al (2010) Survivorship of the humeral component in shoulder arthroplasty. J Shoulder Elb Surg 19(1):143–150
Grammont PM, Baulot E (1993) Delta shoulder prosthesis for rotator cuff rupture. Orthopedics 16(1):65–68
Ladermann A et al (2009) Objective evaluation of lengthening in reverse shoulder arthroplasty. J Shoulder Elb Surg 18(4):588–595
Kwon YW et al (2012) Kinematic analysis of dynamic shoulder motion in patients with reverse total shoulder arthroplasty. J Shoulder Elb Surg 21(9):1184–1190
Boileau P et al (2006) Neer award 2005: the Grammont reverse shoulder prosthesis: results in cuff tear arthritis, fracture sequelae, and revision arthroplasty. J Shoulder Elb Surg 15(5):527–540
Frankle M et al (2005) The Reverse shoulder prosthesis for glenohumeral arthritis associated with severe rotator cuff deficiency. A minimum two-year follow-up study of sixty patients. J Bone Jt Surg Am 87(8):1697–1705
Guery J et al (2006) Reverse total shoulder arthroplasty. Survivorship analysis of eighty replacements followed for five to ten years. J Bone Jt Surg Am 88(8):1742–1747
Sirveaux F et al (2004) Grammont inverted total shoulder arthroplasty in the treatment of glenohumeral osteoarthritis with massive rupture of the cuff. Results of a multicentre study of 80 shoulders. J Bone Jt Surg Br 86(3):388–395
Werner CM et al (2005) Treatment of painful pseudoparesis due to irreparable rotator cuff dysfunction with the delta III reverse-ball-and-socket total shoulder prosthesis. J Bone Jt Surg Am 87(7):1476–1486
Constant CR (1997) An evaluation of the Constant-Murley shoulder assessment. J Bone Jt Surg Br 79(4):695–696
Constant CR, Murley AH (1987) A clinical method of functional assessment of the shoulder. Clin Orthop Relat Res 214:160–164
Rettig O et al (2009) A new kinematic model of the upper extremity based on functional joint parameter determination for shoulder and elbow. Gait Posture 30(4):469–476
Germann G, Wind G, Harth A (1999) The DASH (disability of arm-shoulder-hand) questionnaire—a new instrument for evaluating upper extremity treatment outcome. Handchir Mikrochir Plast Chir 31(3):149–152
Offenbacher M et al (2003) Validation of a German version of the ‘Disabilities of Arm, Shoulder and Hand’ questionnaire (DASH-G). Z Rheumatol 62(2):168–177
Hamada K et al (1990) Roentgenographic findings in massive rotator cuff tears. A long-term observation. Clin Orthop Relat Res 254:92–96
Fuchs B et al (2006) Clinical and structural results of open repair of an isolated one-tendon tear of the rotator cuff. J Bone Jt Surg Am 88(2):309–316
Goutallier D et al (1994) Fatty muscle degeneration in cuff ruptures. Pre- and post-operative evaluation by CT scan. Clin Orthop Relat Res 304:78–83
Simovitch RW et al (2007) Impact of fatty infiltration of the teres minor muscle on the outcome of reverse total shoulder arthroplasty. J Bone Jt Surg Am 89(5):934–939
Neer CS, Watson KC, Stanton FJ (1982) Recent experience in total shoulder replacement. J Bone Jt Surg Am 64(3):319–337
Veeger HE et al (2006) A kinematical analysis of the shoulder after arthroplasty during a hair combing task. Clin Biomech (Bristol, Avon) 21(Suppl 1):S39–S44
Rab G, Petuskey K, Bagley A (2002) A method for determination of upper extremity kinematics. Gait Posture 15(2):113–119
Wu G et al (2005) ISB recommendation on definitions of joint coordinate systems of various joints for the reporting of human joint motion—part II: shoulder, elbow, wrist and hand. J Biomech 38(5):981–992
Carman AB, Milburn PD (2006) Determining rigid body transformation parameters from ill-conditioned spatial marker co-ordinates. J Biomech 39(10):1778–1786
Chiari L et al (2005) Human movement analysis using stereophotogrammetry. Part 2: instrumental errors. Gait Posture 21(2):197–211
Doorenbosch CA, Harlaar J, Veeger DH (2003) The globe system: an unambiguous description of shoulder positions in daily life movements. J Rehabil Res Dev 40(2):147–155
Alta TD et al (2011) Kinematic and clinical evaluation of shoulder function after primary and revision reverse shoulder prostheses. J Shoulder Elb Surg 20(4):564–570
Henninger HB et al (2012) Effect of deltoid tension and humeral version in reverse total shoulder arthroplasty: a biomechanical study. J Shoulder Elb Surg 21(4):483–490
Boileau P et al (2005) Grammont reverse prosthesis: design, rationale, and biomechanics. J Shoulder Elb Surg 14(1 Suppl S):147S–161S
Henninger HB et al (2012) Effect of lateral offset center of rotation in reverse total shoulder arthroplasty: a biomechanical study. J Shoulder Elb Surg 21(9):1128–1135
Cuff D et al (2008) Reverse shoulder arthroplasty for the treatment of rotator cuff deficiency. J Bone Jt Surg Am 90(6):1244–1251
Rettig O et al (2013) Does the reverse shoulder prosthesis medialize the center of rotation in the glenohumeral joint? Gait Posture 37(1):29–31
Maier MW et al (2012) Proprioception 3 years after shoulder arthroplasty in 3D motion analysis: a prospective study. Arch Orthop Trauma Surg 132(7):1003–1010
Maier MW et al (2014) 3D motion capture using the HUX model for monitoring functional changes with arthroplasty in patients with degenerative osteoarthritis. Gait Posture 39(1):7–11
Boileau P et al (2008) Reverse shoulder arthroplasty combined with a modified latissimus dorsi and teres major tendon transfer for shoulder pseudoparalysis associated with dropping arm. Clin Orthop Relat Res 466(3):584–593
Jobin CM et al (2012) Reverse total shoulder arthroplasty for cuff tear arthropathy: the clinical effect of deltoid lengthening and center of rotation medialization. J Shoulder Elb Surg 21(10):1269–1277
Lawrence TM et al (2012) Patient reported activities after reverse shoulder arthroplasty: part II. J Shoulder Elb Surg 21(11):1464–1469
Seebauer L, Walter W, Keyl W (2005) Reverse total shoulder arthroplasty for the treatment of defect arthropathy. Oper Orthop Traumatol 17:1–24
Favard L, Le Du C, Bicknell R, Sirveaux F, Levigne C, Boileau P, Walch G (2006) Reverse prostheses for cuff tear arthritis (Hamada IV and V) without previous surgery. Nice shoulder course: reverse shoulder arthroplasty. Sauramps Medical, Montpellier, pp 113–123
Acknowledgements
We thank the research fund of the Department of Orthopaedic and Trauma Surgery of the University of Heidelberg for the financial support of the study. Furthermore, we would like to thank the motion analysis team of the University of Heidelberg, especially Oliver Rettig and Waltraud Schuster, for the practical support during the study. This study was funded by the Research fund of the Department of Orthopaedic and Trauma Surgery of the Hospital of the University of Heidelberg.
Conflict of interest
All authors, their immediate family, and any research foundation with which they are affiliated did not receive any financial payments or other benefits from any commercial entity related to the subject of this article.
Ethical standards
The local ethics committee approved the study (S-305/2007) and all patients consented to the study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Maier, M.W., Caspers, M., Zeifang, F. et al. How does reverse shoulder replacement change the range of motion in activities of daily living in patients with cuff tear arthropathy? A prospective optical 3D motion analysis study. Arch Orthop Trauma Surg 134, 1065–1071 (2014). https://doi.org/10.1007/s00402-014-2015-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00402-014-2015-7