Abstract
Introduction
Percutaneous radiofrequency ablation (RFA) has been considered, in recent years, the standard treatment for osteoid osteoma (OO) of the appendicular skeleton. The variable clinical presentations in the foot and ankle pose problems in diagnosis, localization and thus treatment. The aim of this study was to assess the efficacy of RFA for patients with osteoid osteoma of the foot and ankle.
Materials and methods
A total of 29 patients (22 males, 7 females; mean age 16.7 years; range 8–44 years) with OO of the foot and ankle (distal tibia, n = 17; distal fibula, n = 6; talus, n = 3; calcaneus, n = 3) were enrolled in the study. A CT-guided RFA was performed, using a cool-tip electrode without the cooling system, heating the lesion up to 90 °C for 4–5 min. Clinical success, assessed at a minimum follow-up of 1 year, was defined as complete or partial pain relief after RFA. Pain and clinical outcomes were scored pre-operatively and at the follow-up with a visual analogue scale (VAS) and with the American Orthopaedic Foot and Ankle Society (AOFAS) score. Complications and local recurrences were also recorded.
Results
Clinical success was achieved in 26 patients (89.6 %). After RFA, mean VAS and AOFAS score significantly improved from 8 ± 1 to 2 ± 1 (p < 0.05) and from 60.7 ± 12.7 to 89.6 ± 7.1 (p < 0.05), respectively. Two patients experienced partial relief of pain and underwent a second successful ablation. Local recurrences were found in three patients, always associated with pain. These underwent conventional excision through open surgery. No early or late complications were detected after RFA.
Conclusion
CT-guided RFA of foot and ankle osteoid osteoma is a safe and effective procedure, showing similar results for the rest of the appendicular skeleton.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Percutaneous radiofrequency ablation (RFA) can be considered a safe and effective minimally invasive treatment for osteoid osteoma of the appendicular skeleton [1]. Several studies reported high success rate comparable to open surgery, but with fewer complications and faster recovery time [2, 3].
Some authors have suggested that percutaneous treatments of osteoid osteoma (OO) of the hands and feet may carry a greater risk of injury to nerves, or that the small size of the bones at these sites may pre-dispose to osteonecrosis [2]. Previous studies reported no major complications in patients treated with RFA for OO of the foot [4, 5]. However, these case series reported the clinical results in a low number of patients.
The purpose of this study was to evaluate the safety and effectiveness of RFA in a large series of patients with OO of the foot and ankle.
Materials and methods
Between May 1999 and March 2009, 29 patients (22 male and 7 female patients; age range, 8–44 years; mean age, 16.7 years) underwent a CT-guided RFA treatment for OO in the foot and ankle. The lesions were located in the distal tibia (n = 17), distal fibula (n = 6), talus (n = 3), and calcaneus (n = 3) (Fig. 1). All the procedures were performed for initial tumour treatment.
Written consent was obtained from all the patients or their parents of legal guardian. The study was performed in accordance with the declaration of Helsinki and Local Ethics Committee guidelines.
The diagnosis of OO was made with both clinical criteria (pain, worse at night and rest, and relief after administration of nonsteroidal anti-inflammatory drugs) and suggestive imaging findings [1]. Furthermore all patients received a dynamic-contrast CT which shows a characteristic arterial enhancement within the nidus [6].
Procedure
All the procedures were carried out in the CT radiology room on an in-patient basis, after the administration of general or spinal anaesthesia and CT-guidance by a team consisting of an orthopaedic oncological surgeon, a musculoskeletal radiologist and an anaesthetist.
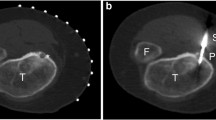
Dynamic CT scans with a section thickness of 1–3 mm (Tomoscan LX; Philips, Eindhoven, The Netherlands) were obtained for the localization of the nidus (Fig. 2). Along with that you obtain under contrast agent a typically parallel increase of the houndsfield units in the measuring range of the nidus (Fig. 3). Under CT-guidance a crossed laser beam was adjusted according to the desired angle and location. A 20-gauge needle was then inserted for periosteal anaesthesia with 2–5 ml of 0.5 % bupivacaine hydrochloride. With the needle in place a single CT cut confirmed the correct approach (Fig. 4). A skin incision was made at the puncture site and access to the nidus was established either using a 11-gauge Jamshidi hollow biopsy needle or a 2 mm coaxial drill system (Richards Surgical Products, Kalamazoo, Michigan), depending on the hardness of the adjacent bone. The position of the inserted instrument was imaged by additional CT.
Outcome
Short-term outcome was evaluated to detect procedure-related problems. Each patient was examined at the procedure site for evidence of bleeding, swelling, and burns the day after the procedure. Neurovascular integrity was assessed, and the patient was asked about pain. If necessary, a short course of pain killers was prescribed. Patients were discharged 24 h after treatment from the orthopaedic ward, when free of pain or when pain was controlled by simple oral analgesics. Patients were instructed to avoid sport activities for at least 6 weeks; all other daily activities were allowed immediately after discharge. Patients were also evaluated 2 weeks after the procedure in order to detect late complications (burns, infections, wound problems) and resolution of procedure-related pain. The follow-up period has a maximum of 132 months (range 12–132 months; mean 67.2 months). No needle biopsy was necessary to obtain histological confirmation of the diagnosis. The America Foot and Ankle Society (AOFAS) score and a Visual Analogue Scale (VAS) for pain (0 for least and 10 for worst pain) was assessed pre- and post-operatively at the last follow-up in order to evaluate the results of the procedure. If the patient was free of pain without the use of pain killers, the clinical outcome was defined as successful. In case of recurrence of symptoms, CT and MR imaging were performed systematically. We determined any differences in the outcome scores (AOFAS score and VAS for pain) between pre-operative and latest follow-up using a Student’s t test or a nonparametric Wilcoxon test after checking for normal distribution of the samples with a Kolmogorov-Smirnoff Z test. The significance level was set at p < 0.05. Analyses were performed using SPSS 13.0 for Windows (SPSS, Chicago, IL).
Results
Clinical long-term success was achieved in 26 of the 29 patients, regarding pain relief was obtained at an average follow-up time of 67.2 months (ranging from 12–132 months). Two patients experienced partial relief of pain and underwent a second successful ablation whereby a second RFA was performed 4 weeks later. However, in three patients (of 29) a recurrence of the symptoms similar to that experienced on initial clinical presentation developed after 36 months. A CT-scan revealed a residual nidus despite a retrospective analysis showing that the needle tip was properly centered in the nidus. These patients underwent conventional excision through open surgery, with complete resolution of symptoms at 6 months after the index procedure. No procedure-related complications were observed.
According to the AOFAS and VAS score, clinical results revealed an improvement from 60.7 ± 12.7 to 89.6 ± 7.1 (p < 0.05) and from 8 ± 1 to 2 ± 1 (p < 0.05), respectively. All patients fully resumed their previous sport activities at the last follow-up. No fractures or other late complications occurred in any patient (Fig. 5).
Discussion
In the past years, open surgical excision was considered the gold standard treatment for OO of the foot and ankle. However, the central nidus can be difficult to identify intraoperatively, requiring an excessive amount of bone resection, which may result in weakening of the bony structure and an increased risk of fracture [7]. Furthermore, during an en bloc resection, part of the articular surface could be compromised to completely remove the lesion leading to an increased risk of osteoarthritis.
Minimally invasive techniques have been further developed in order to decrease the risk of the aforementioned complications. Percutaneous resection [8], ethanol injection [9], laser photocoagulation [7, 10], and RFA [2, 3] have showed high success rates with low complications for the treatment of OO. Because of the potentional advantages of the noninvasive treatments, surgery is now reserved for cases that cannot be treated percutaneously.
Different methods of treatment when these lesions occur in the ankle and foot have been described [11–13]. Only few cases involving the foot being treated with RFA have been previously reported, with outcomes and risks comparable to operative excision [4, 5, 7, 14, 15]. RFA of OO requires only small surgical access to allow insertion of the electrode. Therefore, bone loss is minimal and this is particularly useful in ankle and foot lesions that are often juxta or intra-articular, which may otherwise require an open arthrotomy or arthroscopic removal [16].
In this cohort of patients, no histological confirmation was required since a dynamic-contrast CT was obtained in all patients. In the foot, OO is commonly located in cancellous bone or within a joint, where the periosteal reaction is minimal or often absent. A dynamic-contrast CT has been showed to distinguish OO from subacute osteomyelitis [17]. As 26 of our 29 patients had a successful clinical outcome, we conclude that bone biopsy should not be performed routinely in-patient with suspected OO of the foot and ankle [18].
In conclusion, percutaneous RFA is a safe and efficient treatment for OO involving foot and ankle, because it can be performed with a short hospitalization and it is associated with a short period of convalescence and high success rate. Further studies are needed, in order to clarify the role of percutaneous RFA for the treatment of iuxta-articular OO of the foot and ankle.
References
Motamedi D, Learch TJ, Ishimitsu DN, Motamedi K, Katz MD, Brien EW, Menendez L (2009) Thermal ablation of osteoid osteoma: overview and step-by-step guide. Radiographics 29:2127–2141
Rosenthal DI, Hornicek FJ, Wolfe MW, Jennings LC, Gebhardt MC, Mankin HJ (1998) Percutaneous radiofrequency ablation of osteoid osteoma compared with operative treatment. J Bone Joint Surg Am 80:815–821
Rosenthal DI, Hornicek FJ, Torriani M, Gebhardt MC, Mankin HJ (2003) Osteoid osteoma: percutaneous treatment with radiofrequency energy. Radiology 229:171–175
Le Corroller T, Champsaur P (2011) Osteoid osteoma of the midfoot: percutaneous radiofrequency treatment in 2 cases. J Foot Ankle Surg 50:754–757
Migues A, Velan O, Solari G, Pace G, Slullitel G, Araujo ES (2005) Osteoid osteoma of the calcaneus: percutaneous radiofrequency ablation. J Foot Ankle Surg 44:469–472
Woertler K, Vestring T, Boettner F, Winkelmann W, Heindel W, Lindner N (2001) Osteoid osteoma: CT-guided percutaneous radiofrequency ablation and follow-up in 47 patients. J Vasc Interv Radiol 12:717–722
Zouari L, Bousson V, Hamz_e B, Roulot E, Roqueplan F, Laredo JD (2008) CT-guided percutaneous laser photocoagulation of osteoid osteomas of the hands and feet. Eur Radiol 18:2635–2641
Sans N, Galy-Fourcade D, Assoun J, Jarlaud T, Chiavassa H, Bonnevialle P, Railhac N, Giron J, Morera-Maupome H, Railhac JJ (1999) Osteoid osteoma: CT-guided percutaneous resection and follow-up in 38 patients. Radiology 212:687–692
Adam G, Neuerburg J, Vorwerk D, Forst J, Gunther RW (1997) Percutaneous treatment of osteoid osteomas: combination of drill biopsy and subsequent ethanol injection. Semin Musculoskelet Radiol 1:281–284
Gangi A, Alizadeh H, Wong L, Buy X, Dietemann JL, Roy C (2007) Osteoid osteoma: percutaneous laser ablation and follow-up in 114 patients. Radiology 242:293–301
Barca F, Letti Acciaro A, Spina V (2002) Intra-articular osteoid osteoma of the lower extremity: diagnostic problems. Foot Ankle Int 23:264–267
Donley BG, Philbin T, Rosenberg GA, Schils JP, Recht M (2000) Percutaneous CT guided resection of osteoid osteoma of the tibial plafond. Foot Ankle Int 21:596–598
Ghanem I, Collet LM, Kharrat K, Samaha E, Deramon H, Mertl P, Dagher F (2003) Percutaneous radiofrequency coagulation of osteoid osteoma in children and adolescents. J Pediatr Orthop B 12:244–252
Lindner NJ, Ozaki T, Roedl R, Gosheger G, Wortler K, Winkelmann W (2001) Percutaneous radiofrequency ablation in osteoid osteoma. J Bone Joint Surg Br 83:391–396
Vanderschueren GM, Taminiau AH, Obermann WR, Bloem JL (2002) Osteoid osteoma: clinical results with thermocoagulation. Radiology 224:82–86
Resnick RB, Jarolem KL, Sheskier SC, Desai P, Cisa J (1995) Arthroscopic removal of an osteoid osteoma of the talus: a case report. Foot Ankle Int 16:212–215
McGrath BE, Bush CH, Nelson TE, Scarborough MT (1996) Evaluation of suspected osteoid osteoma. Clin Orthop Relat Res 327:247–252
Henrichs MP, Hardes J, Gosheger G, Vieth V (2011) Monarthritis of the ankle due to osteoid osteoma of the talus. Difficulties in diagnosis and treatment. Orthopade 40:344–348
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Daniilidis, K., Martinelli, N., Gosheger, G. et al. Percutaneous CT-guided radio-frequency ablation of osteoid osteoma of the foot and ankle. Arch Orthop Trauma Surg 132, 1707–1710 (2012). https://doi.org/10.1007/s00402-012-1614-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00402-012-1614-4