Abstract
Astrocytes regulate central nervous system development, maintain its homeostasis and orchestrate repair upon injury. Emerging evidence support functional specialization of astroglia, both between and within brain regions. Different subtypes of gray matter astrocytes have been identified, yet molecular and functional diversity of white matter astrocytes remains largely unexplored. Nonetheless, their important and diverse roles in maintaining white matter integrity and function are well recognized. Compelling evidence indicate that impairment of normal astrocytic function and their response to injury contribute to a wide variety of diseases, including white matter disorders. In this review, we highlight our current understanding of astrocyte heterogeneity in the white matter of the mammalian brain and how an interplay between developmental origins and local environmental cues contribute to astroglial diversification. In addition, we discuss whether, and if so, how, heterogeneous astrocytes could contribute to white matter function in health and disease and focus on the sparse human research data available. We highlight four leukodystrophies primarily due to astrocytic dysfunction, the so-called astrocytopathies. Insight into the role of astroglial heterogeneity in both healthy and diseased white matter may provide new avenues for therapies aimed at promoting repair and restoring normal white matter function.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Astrocytes are the most abundant glial cell type in the central nervous system (CNS). They have long been considered as passive cells providing structural support to neurons, but are now recognized to exert numerous functions essential to support and maintain a healthy CNS [10, 127]. It remains debatable whether all astrocytes exert a variety of complex functions or functional specialization exists among distinct subtypes. Originally, two main subtypes were characterized: protoplasmic and fibrous astrocytes, residing in the gray and white matter (GM and WM), respectively [88]. Protoplasmic astrocytes show extensive branching of bushy processes oriented in a globoid distribution, which contact numerous synapses and envelop blood vessels with their endfeet. Contrastingly, fibrous astrocytes have smaller, elongated cell somas and long, fiber-like processes that align with myelinated axons and contact blood vessels and axons at the nodes of Ranvier [127]. In addition to this, diversity can be appreciated at the molecular level regarding expression of glutamate transporters [50, 113], ion channels [108], gap junction proteins [94], and receptors [63]. These differences indicate that astrocytes may be specialized to their location in the CNS [28, 100].
The concept of astroglia heterogeneity only gained attention in the last 2 decades, resulting in the build-up of evidence on diversity in morphology, gene expression profiles, and physiology, both between and within brain regions. This led to the identification of additional astrocyte subtypes residing in GM regions [28, 42, 45, 60, 100]. Contrastingly, there still remains a lack of understanding on the molecular and functional diversity of astrocytes residing in the WM. WM astrocytes are crucial for maintaining healthy WM structure and function [9, 35, 82]. Accordingly, impairment of normal astrocytic function and its response to injury contribute to a wide variety of CNS diseases, including WM disorders [106, 145, 146]. Here, we discuss our current understanding on astrocyte heterogeneity in the WM of the mammalian brain. We review how astrocytic diversity may arise and discuss what the implications are for WM structure and function in health and disease by focusing on the sparse human research data available. We also discuss how heterogeneity of astrocytes may contribute to selective vulnerability of white matter regions in the scope of several genetic WM disorders.
The rise of heterogeneous astrocytes
Mature astrocytes can generally be recognized by their star-like appearance with radially oriented processes and expression of major intermediate filament glial fibrillary acidic protein (GFAP) [127]. Its expression, however, is not detectable in all healthy astrocytes and varies greatly according to CNS region [50, 65] and activation state [127]. Identification of new astrocyte markers over the past few years enabled the characterization of different astroglial subtypes, providing clues for their functions. These include S100 calcium-binding protein β, glutamate transporter 1 (GLT-1), glutamate aspartate transporter (GLAST), aldehyde dehydrogenase 1 family member L1, aldolase C, aquaporin-4 (AQP4), and Sox9 [24, 90, 113]. Nevertheless, studying astroglial diversity remains complicated due to the lack of markers that clearly distinguish between subtypes and developmental stages [90]. Here, we discuss our current understanding of the rise of heterogeneous astroglia and how an interplay between cell-intrinsic and cell-extrinsic factors determine their cell identity.
Cell-intrinsic factors
Emerging evidence indicate morphological and molecular astroglia heterogeneity across CNS regions [6, 13, 24, 39, 42, 113], but the mechanisms driving this diversification remain poorly understood. For neurons, it is well established that distinct subtypes are generated during early development by patterning of the neuroepithelium into discrete progenitor domains by morphogens. As a result, these progenitors express a unique combination of transcription factors, giving rise to diverse cell types in a region-dependent manner [12, 28, 90]. Regional diversity of astrocytes implies derivation from distinct progenitor pools, suggesting that the mechanisms driving neuronal diversity may similarly contribute to astrocyte diversification.
A major source of astrocytes during late embryonic development are radial glia in the ventricular zone, which are GFAP-positive cells derived from the neuroepithelium. Radial glia have multipotent neural stem cell (NSC)-like properties and give rise to neurons and, after a gliogenic switch, to oligodendrocytes and astrocytes (reviewed in [12]). In addition, progenitors in the subventricular zone (SVZ) that persist into adulthood can differentiate into interneurons, but also give rise to oligodendrocytes and astrocytes in GM or WM [131]. Radial glia and SVZ progenitors are organized into distinct spatial domains, suggesting that spatial patterning may indeed contribute to heterogeneity of astrocytes [12, 28, 60, 90, 131, 154]. In support of this, clonal analysis of the adult cortex revealed that protoplasmic and fibrous astrocytes are derived from separate cell lineages [48]. In addition, progenitors expressing proteoglycan neural/glial-antigen 2 (NG2), termed as NG2-glia or oligodendrocyte precursor cells (OPCs), give rise to myelinating oligodendrocytes in the GM and WM throughout life. They were also shown to generate a subpopulation of protoplasmic, but not fibrous, astrocytes in the ventral forebrain [158]. By contrast, basic helix–loop–helix oligodendrocyte transcription factor 2 (Olig2), which is expressed in a subset of progenitors in the neonatal and adult forebrain, is involved in the differentiation of oligodendrocytes [83, 104] and WM, but not GM, astrocytes [25]. Similarly, in glial progenitor pools in the dorsal mouse spinal cord, differential expression of transcription factor Ascl1 was shown to regulate the numbers of protoplasmic versus fibrous astrocytes [148]. These findings suggest that regionally distinct astrocytes, including GM and WM astrocytes, may derive from distinct cell lineages as a result of spatial patterning.
Importantly, spatial patterning of progenitor domains could also contribute to diversification of astroglia within the WM. A homeodomain code was shown to specify three distinct progenitor domains in the ventral spinal ventricular zone termed p1–p3, which give rise to three distinct astrocyte subpopulations in the WM termed VA1–VA3 [62]. Interestingly, the spatial organization of these WM astrocytes along the dorso-ventral axis mirrors the progenitors in the p1–3 domains [62], revealing that positional identity of progenitors may contribute to astroglial heterogeneity within the spinal cord WM. Taken together, increasing evidence indicate that developmental patterning followed by regional specialization could underlie astrocyte diversity throughout the CNS [48, 92, 134]. Given that spatial allocation is retained into adulthood and during disease [134], the regional specification of astrocytes appears to be an intrinsic property contributing to regional variation in form and function.
Cell-extrinsic factors
Astroglia also display diversity at a local level, with distinct subtypes co-existing within the same CNS region [42, 45, 60, 100]. In the cerebral cortex, astrocytes are diverse in morphological, molecular and transcriptional properties across its layers [64, 72, 93, 101]. Distinct astrocyte subtypes are also found in hippocampal subregions [42]. In addition, the cerebellar cortex reveals intraregional heterogeneity, where velate astrocytes and Bergmann glia are located in the granular and Purkinje cell layer, respectively. Despite deriving from the same progenitor pool, these subtypes harbor distinct morphological features and gene expression profiles in mice [67]. This suggests that local (micro-)environmental cues may induce functional adaptations in astrocytes tailored to the requirements of their local cellular environment. In support of this, signaling from adjacent neurons is crucial in determining the mature astrocyte phenotype in the adult mouse brain [72]. Neuron-derived morphogen Shh drives the distinct molecular and physiological profile of velate astrocytes and Bergmann glia in the mouse cerebellar cortex [44]. Similarly, Shh regulates the molecular diversity of spatially distinct astrocyte subpopulations within the mature mouse forebrain [49]. While human data on this subject are sparse, the extent of astrocyte diversity in form and function in the human brain, even within the same brain region [16, 100, 101, 130], suggests that both developmental origin and local (micro-)environmental cues underlie their heterogeneity.
The concept that GM astrocytes adapt their molecular properties in response to cell-extrinsic factors raises the question whether WM astrocytes might have the same potential. So far, two types of GFAP-positive WM astrocyte populations have been identified in cultures of the developing rat optic nerve based on shape, antigenic properties, and growth characteristics, which were termed type 1 and type 2 astrocytes [111]. These astrocyte subtypes remain to be confirmed in vivo. In conclusion, astroglial cell-fate specification depends on a combination of complex cell-intrinsic and cell-extrinsic mechanisms, including transcription factor expression, neuronal feedback, as well as epigenetic mechanisms (reviewed elsewhere [132]). These mechanisms are highly regulated in time and space to ensure proper neural network formation and drive astroglial diversity in the mammalian brain.
Astrocyte diversity in healthy WM
Diversity in morphological and molecular features between distinct astrocyte subtypes implies functional specialization tailored to their surrounding tissue. Protoplasmic astrocytes tile the healthy GM in non-overlapping spatial domains [127], wherein they facilitate information processing by regulating blood flow and shaping synaptic transmission [107]. The functions of GM astrocytes are thought to be regionally specialized to support their surrounding neuronal subtype and neural circuit [12, 29, 55, 65]. In the WM, astrocytes show extensive intermingling of processes with neighboring astrocytes [101] and they support healthy WM development and function, including myelination [9, 35, 82]. But how do diverse astrocyte subtypes contribute to the maintenance of healthy WM?
The WM is largely composed of myelinated axons. Myelin sheaths are crucial for fast saltatory conduction of action potentials along the axons and support axonal integrity and trophism [98]. WM astrocytes play important roles in both developmental myelination and remyelination following injury [82] (Fig. 1). They contribute to each step of the myelinating process (reviewed in [35, 82, 91]). In brief, astrocytes secrete growth and trophic factors, including brain-derived neurotrophic factor, ciliary neurotrophic factor (CNTF), insulin-like growth factor 1, fibroblast growth factor-2, leukemia inhibitory factor, and tissue inhibitor of metalloproteinase-1, that enhance myelination by promoting survival or differentiation of oligodendrocytes [9, 66, 78, 91]. In addition, they provide metabolic support to oligodendrocytes by supplying energy substrates via gap junctions [78, 82]. Conversely, astrocytes can also inhibit myelination. Secretion of platelet-derived growth factor-α (PDGFα) prevents premature differentiation of OPCs, thereby regulating the timing of myelination in the developing CNS [84]. Astrocytes play dual roles in regulating oligodendrocyte survival and differentiation, and also myelination. In support of this, it has been shown that the astrocytic phenotype determines their ability to support myelination. Plating rat astrocytes on extracellular matrix (ECM) glycoprotein tenascin C induces a quiescent phenotype that inhibits myelination of spinal cord cultures in vitro, whereas treating astrocytes with cytokine CNTF induces an activated phenotype that promotes myelination [96]. This is in line with other studies reporting a pro-regenerative phenotype of CNTF-treated astrocytes (reviewed in [66]). These findings suggest that astrocytes can change their phenotype in response to environmental factors and, depending on their activation state, may hamper or support myelination.
Supporting and detrimental functions of astrocytes on oligodendrocyte survival, differentiation and myelination. (Left) Astrocytes support survival and differentiation of oligodendrocyte precursor cells (OPC) into myelinating oligodendrocytes via several secreted factors, including brain-derived neurotrophic factor (BDNF), ciliary neurotrophic factor (CNTF), insulin-like growth factor 1 (IGF-1), fibroblast growth factor-2 (FGF-2), leukemia inhibitory factor (LIF), and tissue inhibitor of metalloproteinase-1 (TIMP-1). They also metabolically support oligodendrocytes by taking up glucose from the bloodstream through glucose transporters (GLUT1) and storing it in the form of glycogen. When demanded, glycogen can be transformed into pyruvate by astrocytes. This can subsequently be broken down into lactate, which is transported to oligodendrocytes. Astrocytes also support the maintenance of myelin and axonal integrity. (Right) Astrocytes can also have detrimental effects on oligodendrocytes and/or myelin, compromising the integrity of myelin and, consequently, the axons. Such astrocyte-derived factors include platelet-derived growth factor-α (PDGFα), tumor necrosis factor α (TNFα), interferon-gamma (IFNγ), C–X–C motif chemokine 10 (CXCL10), bone morphogenetic proteins (BMP), and extracellular matrix (ECM) components such as hyaluronan. They also upregulate GFAP in response to damage and disease
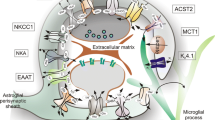
Regulation of myelination only represents part of the essential roles of astrocytes in maintaining WM homeostasis (Fig. 2). Through bidirectional crosstalk with other cell types, including neurons, pericytes, endothelial cells, and other non-neural cells, astrocytes affect nearly all aspects of healthy WM functioning. Astrocytes regulate neurite outgrowth via secreted factors and ECM components [109, 112] and interact with microglia during development (e.g., synaptogenesis and synapse pruning) [112]. In addition, they contribute to the induction and maintenance of the blood–brain barrier (BBB) [102] and regulation of blood flow [5]. Astrocytes are also involved in the production and recycling of several neurotransmitters, including glutamate via GLAST and GLT-1 transporters [151]. Moreover, they release gliotransmitters to signal to neurons [57] and shape neurotransmission and plasticity [151]. They play important roles in ion–water homeostasis, including spatial potassium buffering via Kir4.1 channels, which is important for healthy WM function [78, 82, 122]. Astrocytes also serve as an energy supplier via uptake of glucose from the bloodstream and storage of glycogen, a function exclusive to astrocytes in the CNS [151]. Astrocyte-derived glycogen is released in the form of lactate and delivered to axons to support their high metabolic demands [8, 151] and to oligodendrocytes to use as substrate for myelin production [116]. They also provide many growth and trophic factors, including iron, to support surrounding cells in the WM [66, 78]. These complex functions of WM astrocytes illustrate their important and diverse roles in supporting healthy WM structure and function.
Essential astrocytic functions in the CNS WM. Astrocytes play essential roles in the CNS WM through proliferation, support of oligodendrocytes and myelination of axons via secreted factors or direct astrocyte–oligodendrocyte and astrocyte–axonal interactions, glutamate transport and glutamine synthesis, regulation of synaptic neurotransmission and plasticity, support of ion and water homeostasis, regulation of neurite outgrowth, maintenance of the BBB, interaction with microglia to induce innate immune responses, and responses to injury and protection of healthy tissue, including formation of glial scar tissue surrounding the damaged area
Notably, the CNS WM is not a homogeneous structure, but consists of many different fiber tracts with their own unique organization, density and diameter of nerve fibers, myelin thickness, and functions. Different fiber tracts may require tailored and fine-tuned support from local astrocytes. In support of this, astroglial density varies substantially across WM tracts, including relatively low density in the stria terminalis and high density in the optic tract [42]. In addition, astroglial calcium signaling properties may vary throughout the WM. Neighboring astrocytes are coupled in large complex networks via gap junction channels that allow intercellular passage of ions and small molecules. This includes rapid propagation of intracellular calcium waves, thereby enabling intercellular communication in the astrocyte network [119]. In mice, cortical astrocytes are highly coupled by gap junctions, which is required for calcium wave propagation. By contrast, gap junctions are rarely found in the corpus callosum and calcium wave propagation here relies on ATP release [53]. Another study reported the strongest and highest degree of coupling in the rat optic nerve tracts compared to other CNS regions, including the spinal cord [73]. This argues that gap junction coupling of astrocytes may be varied throughout the WM. Although the precise functional implications of these differences remain unclear, they support the view that WM astrocytes are tailored to the functional requirements of local WM tracts. In-depth characterization of their cellular diversity in the human brain will contribute to our understanding of the complex and multifaceted role of astrocytes in WM structure and function.
Astrocyte diversity in diseased WM
Diversity in reactive astrogliosis
Astrocytes react upon all types of CNS injury. During this response, also termed reactive astrogliosis, astrocytes undergo proliferation, cell hypertrophy, process extension and interdigitation, and profound changes in gene expression [127]. Astrogliosis is induced by various signaling factors released from inflamed and damaged areas and is a hallmark of diseased CNS tissue. Together with other glia, neural cells, and non-neural cells entering from the bloodstream, reactive astrocytes orchestrate a multicellular response aimed at restoring homeostasis by minimizing tissue damage and supporting repair [23, 125]. The functional implications of astrogliosis remain, however, debatable [106, 126, 127]. Astrogliosis has been considered a major limiting factor in CNS repair due to glial scar formation. It has been reported to hamper axonal regeneration [121] and acts as a physical barrier for OPCs entering the lesion, resulting in remyelination failure [95]. In response to injury, astrocytes may also inhibit maturation of OPCs through overproduction of ECM components, including hyaluronan, which accumulates in demyelinated lesions and hinders myelin regeneration [7, 124]. Several other astrocyte-derived factors, including interferon-gamma, tumor necrosis factor α (TNFα), C–X–C motif chemokine 10, and bone morphogenetic proteins (BMP) hamper remyelination by inhibiting OPC survival and/or preventing differentiation into mature oligodendrocytes [9, 66, 78, 91, 149] (Fig. 1). In addition, reactive astrocytes promote an inflammatory response mediated by activated microglial cells that could exacerbate tissue damage and BBB disruption [102]. On the other hand, reactive astrogliosis can be beneficial and neuroprotective. Glial scar tissue restricts the spread of inflammatory cells into the surrounding healthy parenchyma and helps restore BBB integrity [126, 150]. Contrary to previous findings, astrogliosis has also been reported to aid, rather than prevent, axonal regeneration after spinal cord injury [4]. Furthermore, reactive astrocytes promote survival and repair of damaged tissue by providing trophic support to cells surrounding the lesion [95] and facilitate remyelination by promoting OPC survival and differentiation into myelinating oligodendrocytes [91, 95]. They recruit and instruct microglia to clear myelin-debris, which is essential for remyelination [99, 123]. Moreover, ablation of reactive astrogliosis following CNS injury causes increased inflammation, more severe demyelination and oligodendrocyte death, and worse clinical outcome [46, 147].
What drives these heterogeneous astrocyte responses to injury and disease? Astrogliosis is not a uniform response, but involves a spectrum of changes tailored to context, insult type, and severity [3, 127]. Two complementing studies described that astrocytes acquire distinct molecular states depending on the nature of the insult. Reactive astrocytes induced in a mouse model of ischemic injury upregulated neurotrophic factors and may thus promote CNS recovery and repair [80, 157]. By contrast, LPS-induced neuroinflammation activated microglia that subsequently induced reactive astrocytes via secretion of specific cytokines. These latter astrocytes displayed a pro-inflammatory gene expression profile and caused loss of neurons and mature oligodendrocytes [80], consequently contributing to failure of repair and restoration of CNS homeostasis. Evidence on the existence of dichotomous reactive astrocytes, divided into ‘A1’ neurotoxic and ‘A2’ neuroprotective phenotypes in the human brain, are, however, largely absent, especially in vivo. Recent data on the diversity of reactive astrocyte states advocates that we should move beyond the concept of a mere binary division of (reactive) astrocytes (reviewed by [43]). Reactive astrogliosis likely involves a continuous spectrum of changes and encompasses a wide phenotypic diversity across diseases [2, 64, 153]. Moreover, signaling cascades inducing reactive gliosis may be beneficial in one disease context and detrimental in another. Signaling of STAT3, a transcription factor regulating astrogliosis and glial scar formation, was shown to restrict inflammation and lesion size in a mouse model of spinal cord injury [59]. In an Alzheimer’s disease (AD) mouse model, on the other hand, deletion rather than activation of STAT3 was shown to ameliorate pathological hallmarks, decrease pro-inflammatory cytokine levels and attenuate spatial learning and memory decline [114]. Astrocytes may also be heterogeneous within one disease context. Different transcriptomic states of astrocytes have been identified in a mouse model of AD, including a shift from GFAP-low to GFAP-high astrocyte states with disease progression and a subset of disease-associated astrocytes specific to AD [54]. In addition, different morphologies and staining patterns of GFAP-positive astrocytes were found to be associated with different types of plaques in AD patient tissue [14].
Astrogliosis is also heterogeneous with respect to distance from the site of injury, with variation in proliferation rates, morphology and gene expression levels [127, 150]. In areas close to the lesion site, severe astrogliosis is observed, whereas this is mild or moderate in areas distant from the lesion site (Fig. 3). In addition, the astrocytic response depends on the affected area. Reactive astrocytes appear at a faster rate in the subcortical WM tracts compared to GM areas after diffuse traumatic brain injury of the rat brain in vivo and differ regionally in morphology and vimentin expression levels [61]. In addition, astrocytes across brain regions respond differently to scratch wound injury and neurotrophins, with more rapid and beneficial responses in the striatum than in the hippocampus and cortex in vitro [32]. Astrocytes across the WM may also show region-specific responses. After focal demyelinating injury, GFAP expression levels are more elevated in the spinal cord WM than in the corpus callosum in mice in vivo [156]. Interestingly, heterogeneous astrogliosis is also observed at the local level. Intermingling reactive astrocytes show varying expression levels of cytokines when stimulated with inflammatory mediators in vivo [56]. In addition, expression levels of pSTAT3, which regulates astrogliosis and glial scar formation, may vary at the single cell level after spinal cord injury in vivo [59]. Taken together, the astrocytic response to injury is diverse at multiple levels and can both hinder and support CNS repair, depending on the context.
Morphologic heterogeneity of astrocytes in CARASAL brain tissue with respect to distance from the lesion site. a Cathepsin A-related arteriopathy with strokes and leukoencephalopathy (CARASAL) is a recently identified adult-onset cerebral leukodystrophy due to mutations in the CTSA gene, encoding cathepsin A. The disease is characterized by tissue rarefaction and abnormal morphology of astrocytes with enlarged nuclei. GFAP staining of CARASAL brain tissue shows reactive astrocytes with hypertrophied cell bodies forming a glial scar at the border of the lesion site in the frontal subcortical WM. b Close to the lesion site, reactive, but non-gliotic, astrocytes can be observed. c Away from the lesion site, only few astrocytes are present in the WM that have a non-reactive morphology. d In the corresponding cortex, gliotic astrocytes can be observed in areas close to the lesion site. Scale bar 20 µm
Due to the heterogeneous nature of reactive astrogliosis and its dual effects on CNS repair, a tightly regulated response of astrocytes to injury is crucial for successful regeneration. It is, therefore, not surprising that dysfunction of astrocytes plays a pivotal role in a wide variety of CNS disorders [106, 145, 146]. Astrocytes contribute to CNS disease through loss of beneficial functions and/or gain of detrimental functions. As such, astrogliopathologies can manifest as (1) sustained reactive astrogliosis, with or without glial scar formation, (2) glial asthenia or atrophy with loss of function, or (3) pathological remodeling [106, 146]. Compelling evidence suggest that glial asthenia with loss of function, rather than sustained reactive astrogliosis, is associated with normal aging of the brain, as well as the development of age-dependent neurodegenerative diseases [143, 144]. Importantly, the diversity of astrocytes across CNS regions [100] suggests that they could respond differently to injury and disease. This implies that some subtypes might be more susceptible to become chronically activated and others functionally asthenic. This could consequently account for selective vulnerability of specific CNS regions to disease. Accordingly, WM astrocytes are supposedly more sensitive to ischemic injury than GM astrocytes [120]. Moreover, frontal WM astrocytes appear to be more vulnerable to ischemic damage than temporal and parietal astrocytes [30], which might relate to frontal predominance of cerebrovascular disease [152]. Even in normal aging, astrocytic transcriptional profiles change in a region-dependent manner, with striking differences in reactive astrocyte phenotypes across CNS regions [13, 31]. While future studies should focus on further characterizing the heterogeneity of astrocytes and its physiological implications, the data discussed here clearly illustrate the diversity of (reactive) astrocytes and how this heterogeneity could drive the pathogenesis of several CNS disorders. Importantly, selective vulnerability of distinct astrocyte subtypes to pathological changes could at least in part explain regional variation in disease severity. Similarly, astrocyte heterogeneity could differentially impact on the process of reactive gliosis. This possibility, however, still awaits investigation.
Astrocyte diversity in leukodystrophies
Leukodystrophies are a group of genetically determined disorders with predominant involvement of the CNS WM [138]. They were originally considered to primarily involve myelin and oligodendrocytes. The emergence of more sensitive genetic tools combined with imaging pattern recognition, however, revealed that surprisingly only few leukodystrophies are caused by mutations in myelin- or oligodendrocyte-specific genes. Many are rather caused by defects in other WM structural components, including axons, microglia, oligodendrocytes, and blood vessels. A subset of leukodystrophies, termed astrocytopathies, is due to primary astrocytic dysfunction [138]. By acting in cell-autonomous or non-cell-autonomous ways, astrocytes have widespread effects on the structural integrity of the WM and might contribute to or even drive its degeneration [35, 78, 138]. Remarkably, many astrocytopathies present with frontal predominance [138], suggesting selective vulnerability of frontal WM astrocytes to pathological changes relative to those of other WM areas. In other words, astrocyte heterogeneity could perhaps in part explain the regional variation in disease severity in genetic leukodystrophies that affect all astrocytes. In light of this, we discuss four common astrocytopathies and highlight current insights on how heterogeneity of astrocytes could play a role in its pathogenesis.
Aicardi–Goutières syndrome (AGS) is an early-onset leukoencephalopathy characterized by neuropathological features that mimic a congenital viral infection [51]. These include progressive microcephaly, WM and basal ganglia calcifications, microangiopathy, cerebral and brain stem atrophy, inflammatory responses, and on brain imaging WM abnormalities primarily in the frontotemporal regions [11, 118] (Fig. 4). WM abnormalities include cavitation, lack of myelin with relative sparing of U-fibers, and reactive astrogliosis [68, 135]. AGS is a genetically heterogeneous disorder caused by autosomal recessive mutations in any of the following genes: TREX1, RNASEH2A, RNASEH2B, RNASEH2C, SAMHD1, ADAR1, and IFIH1/MDA5 [118], which encode proteins involved in nucleic acid metabolism. Dysfunction of these proteins may lead to nucleic acid accumulation, triggering an autoimmune response [105, 155]. AGS is characterized by overproduction of the cytokine Interferon-α (IFNα) and its downstream effectors [118]. How IFNα drives AGS-associated brain pathology remains poorly understood. It is considered that increased IFNα levels induce a cascade of events that trigger an innate immune response and subsequent WM damage [118]. Emerging evidence suggest that astrocytes, the major source of IFNα in the brain, play a central role as they have been shown to overproduce IFNα in AGS [142]. In agreement with this, transgenic mice with astrocytes chronically producing IFNα develop clinical and neuropathological phenotypes resembling human AGS [1, 26]. In addition, chronic treatment of cultured human NSC-derived astrocytes with IFNα reduces proliferation, increases reactivity, and dysregulates expression of astrocyte-specific proteins crucial for WM maintenance [33]. Neuropathological examination of AGS brain tissue revealed apoptotic oligodendrocytes and markedly increased numbers of cells expressing OPC marker platelet-derived growth factor receptor alpha (PDGFRα) in the non-infarcted WM [68], providing a clue for the WM abnormalities in AGS. Taken together, overproduction of IFNα by astrocytes triggers neuroinflammatory events that could induce WM damage in AGS. Importantly, IFNα signaling also seems to drive phenotypic changes in astrocytes themselves that negatively affect their WM maintenance potential. An important question is whether astrocyte subpopulations across the CNS are differently affected in AGS. Given the frontal WM predominance and relative sparing of the cerebellum (Fig. 4), astrocytes in the cerebral WM might be more susceptible to AGS-associated pathological changes, but this has yet to be confirmed.
Radiological and neuropathological features of AGS. a T1-weighted sagittal MRI image of a 16-month-old patient with AGS shows a cyst in the anterior temporal region. The anterior temporal WM is swollen and rarefied. The lateral ventricles are enlarged and the cerebellar hemispheres are small. b On the T2-weighted image, the cerebral WM is reduced in volume and diffusely abnormal in signal intensity. c Susceptibility-weighted imaging (SWI) shows spots of low signal intensity in the cerebral WM, basal nuclei and cerebellar dentate nuclei, reflecting calcium deposits. d The calcifying microangiopathy of the brain can be recognized on macro- and e micro-scale. f AGS has frontotemporal predominance: in the frontal WM, conglomerates of IFNα- and g GFAP-positive astrocytes can be observed. h The cerebellar WM shows less affected GFAP-positive astrocytes and calcification of blood vessels. Scale bar 20 µm
Megalencephalic leukoencephalopathy with subcortical cysts (MLC) is a genetic infantile-onset disease characterized by macrocephaly, frontotemporal subcortical fluid cyst-like formation, and chronic WM edema with cerebral swelling followed by WM atrophy [136, 137] (Fig. 5). It is clinically characterized by delayed-onset neurological deterioration, cerebellar ataxia, motor degeneration, spasticity, and epileptic seizures [136]. Histology shows numerous fluid-filled vacuoles within the outer layers of the myelin sheaths. Vacuoles are also apparent in astrocytic endfeet contacting capillaries [137] (Fig. 5). MLC is caused by mutations in the MLC1 or GLIALCAM gene, leading to loss of normal MLC1 protein function [74, 81]. MLC1 is a membrane protein involved in astrocytic volume regulation [41, 115] and is mainly expressed in astrocyte endfeet contacting the BBB and the pial surface [15, 133]. GlialCAM is a glial cell adhesion-like molecule that traffics MLC1 and ClC-2, a chloride channel also involved in cell volume regulation, to cell–cell junctions [27, 41, 81]. Under normal conditions, astrocytes clear extracellular potassium ions released upon neuronal activity through spatial potassium buffering [70]. This is accompanied by water uptake and leads to astrocyte swelling, which can be counteracted by activation of volume-regulated anion channels that decrease cell volume. In MLC, astrocytic volume regulation in response to osmotic changes is hampered [20, 27, 41, 115], causing defects in ion–water homeostasis. The defect in potassium buffering could contribute to hyperexcitability of neuronal networks and cause early-onset seizures in patients [40, 89]. Importantly, astrocyte abnormalities with swelling of processes are the earliest pathological change in Mlc1-null and Glialcam-null mice. This is followed by intramyelinic edema and progressive WM vacuolization [20, 41], indicating that WM pathology is secondary to astrocyte dysfunction in MLC. Interestingly, the degree in severity of WM edema varies regionally, with anterior cerebral regions being more severely affected than posterior regions, followed by the cerebellum, and relative sparing of the corpus callosum and brainstem [136]. The regional variation in WM vulnerability might correspond to regional heterogeneity of astrocytes. Since astrocytes differ in expression of ion channels, gap junction coupling, and electrophysiological properties across brain regions [100], ion–water homeostasis may be variably affected by loss of MLC1 function. In support of this notion, MLC1 expression in mice is the highest in the cerebellum [133]. This is also the most affected region in MLC mouse models, as mice virtually have no cerebral WM [20, 41]. This shows that regional astrocyte heterogeneity may underlie selective vulnerability of WM regions in MLC.
Radiological and neuropathological features of MLC. a, b T1-weighted sagittal images of a 22-month-old patient with MLC show the swollen WM and a cyst in the anterior temporal region. The U-fibers of the occipital lobe, corpus callosum, brainstem and cerebellum are relatively normal in signal intensity. c, d T2-weighted images show diffusely swollen cerebral WM. The posterior limb of the internal capsule is medially and laterally affected. The corpus callosum is largely spared. e, f FLAIR images show the diffuse swelling and involvement of the cerebral WM with tissue rarefaction in the subcortical areas of the frontal and parietal lobe, and bilateral pseudo-cysts in the anterior temporal lobes. g T2-weighted image shows severe signal abnormalities in the anterior temporal WM. In the cerebellum, the deep WM is mildly abnormal in signal, but not swollen. h In MLC, GFAP-positive astrocytes with swollen endfeet surrounding a blood vessel are often found, as in this section from the frontal subcortical WM. Scale bar 20 µm
Alexander disease (AXD) is a degenerative disorder, of which the manifestation depends on the age of onset. Early-onset (type I) disease has a progressive course with macrocephaly, bulbar dysfunction, psychomotor regression, seizures, and usually leads to death within the first 2 decades [117]. Type I disease shows widespread cerebral WM abnormalities with frontal predominance and, over time, cystic degeneration of frontal WM and cerebellar and brainstem atrophy on MRI [140] (Fig. 6). Contrastingly, late-onset (type II) disease has a more heterogeneous disease course with generally less severe symptoms. MRI features of late-onset patients include WM lesions and atrophy predominantly in the medulla oblongata [138], demonstrating regional heterogeneity in disease severity depending on the age of onset. AxD is caused by gain-of-function mutations in the GFAP gene [17]. The pathogenic mutations result in misfolding of GFAP and its accumulation in cytoplasmic Rosenthal fibers together with vimentin, plectin, ubiquitin, and small heat shock proteins (e.g., Hsp27 and αB-crystallin) [58, 110]. Rosenthal fibers are the pathological hallmark of the disease found in astrocytes adjacent to damaged areas [52, 85] (Fig. 6). Additional pathological features include loss of oligodendrocytes and myelin, as well as neurodegeneration [128]. How GFAP gene mutations lead to AxD-associated pathology remains to be elucidated. A toxic gain-of-function mechanism has been suggested, in which AxD astrocytes acquire abnormal morphology and function due to the disrupted intermediate filament network [87]. In AxD mouse models, GFAP accumulation induces a phenotypic change of protoplasmic astrocytes to severely reactive astrocytes that are multi-nucleated and hypertrophic [129]. In addition, it causes cellular stress in astrocytes that leads to release of inflammatory molecules and microglia activation, thus triggering an immune response detrimental to neighboring cells [103, 128]. AxD astrocytes may also lose their normal supportive functions, as demonstrated by loss of GLT-1 [128, 129]. This could result in diminished extracellular glutamate clearance and eventually glutamate-mediated excitotoxicity, hence explaining the loss of oligodendrocytes and neurons in AxD. AxD patient induced pluripotent stem cell (iPSC)-derived astrocytes inhibit proliferation of iPSC-derived OPC and reduce their myelinating potential in vitro [79], indicating that AxD astrocytes lose their ability to support and maintain myelination. Furthermore, failure of other supporting functions of astrocytes, including extracellular potassium buffering via Kir4.1 and Na+, K+-ATPase activity, has been proposed to contribute to myelin degeneration [86]. Selective involvement of the CNS WM in AxD is in line with earlier studies showing higher GFAP expression in WM astrocytes compared to GM astrocytes [24, 50], making the WM more susceptible to gain-of-function GFAP mutations. It remains unclear if astrocyte subtypes across WM regions are equally affected by the mutations. Nevertheless, it is conceivable that differential expression of GFAP in astrocytes across WM areas contributes to regional vulnerability in AxD.
Radiological and neuropathological features of AXD. a T1-weighted sagittal MRI image of a 10-year-old patient with infantile-onset A × D shows marked hypointensity of the frontal WM with thinning of the corpus callosum and enlargement of the ventricular system. In the posterior frontal subcortical WM, an area of cavitation can be appreciated (arrow). b Axial T2-weighted image shows diffuse signal hyperintensity of the cerebral WM with a frontal predominance and involvement of the capsules, and relative sparing of the parietal and occipital U-fibers. c T1-weighted axial image after gadolinium administration shows punctate areas of contrast enhancement anterior to the anterior horns of the lateral ventricles and along the ventricular surface of the caudate nuclei. d H&E staining of the medulla oblongata shows rarefaction of the white matter surrounding the inferior olivary nucleus. e H&E staining of the cerebellar WM shows characteristic protein inclusions called Rosenthal fibers. f Ubiquitin staining of the subventricular zone shows accumulation in the Rosenthal fibers at this site. g GFAP-positive astrocytes are dysmorphic and multi-nucleated in AxD. Scale bar 300 (d) and 20 (e–g) µm
Vanishing white matter (VWM) is one of the most prevalent leukodystrophies. It mainly affects young children, but may present at any age [47, 141]. Clinical characteristics include cerebellar ataxia with slow progressive neurological deterioration, spasticity, and seizures. Stresses as fever or minor trauma trigger episodes of rapid clinical decline, which may result in coma and death [141]. MRI shows diffuse loss of cerebral WM, which is replaced by fluid over time, and relative sparing of the GM (Fig. 7). This is consistent with neuropathological findings describing progressive WM rarefaction and cystic degeneration of tissue, mainly in the frontoparietal regions [18, 141]. VWM is caused by recessive mutations in any of the genes encoding the five subunits of the eukaryotic translation initiation factor 2B (eIF2B), which is essential for the translation of mRNA into proteins, and regulates the mRNA translation rate, also under cellular stress conditions [139]. It remains unclear how a genetic defect in eIF2B, a ubiquitously expressed factor with central housekeeping functions, leads to selective involvement of the CNS WM [22]. Accumulating evidence identify oligodendrocytes and astrocytes as predominantly affected cell types (reviewed in [22]). Around cavitating lesions, paucity of myelin is paradoxically accompanied by increased numbers of OPCs. They, however, fail to mature into myelinating oligodendrocytes [19], hence explaining the lack of myelin. In addition, the degree of reactive astrogliosis is disproportionally scarce considering the severity of WM degeneration [18]. This may reflect functional impairment of the astrocytes, as they remain immature and are dysmorphic, with coarse, blunt cell processes [19]. They overexpress the GFAP δ-isoform relative to the α-isoform [19], indicating that a disrupted intermediate filament network might contribute to their abnormal morphology and function. Recent findings implicate astrocytes as the primary drivers of VWM pathophysiology. Astrocytes of mice expressing VWM mutations were shown to inhibit WT mouse OPC maturation via secreted factors in vitro [36]. These findings suggest that VWM astrocytes fail in their mature functions to maintain WM integrity, with secondary effects on both oligodendroglia [36] and axons [69]. Notably, cerebellar Bergmann glia, retinal Müller glia, and spinal cord WM astrocytes are also affected in VWM [36, 75]. The disease severity shows regional dependency. The frontal WM is consistently severely affected, whereas the cerebellar WM is relatively spared [21]. Interestingly, the regional variation in disease severity co-varies with the degree in astrocyte abnormality in the corresponding areas (Fig. 7). In addition, accumulation of high molecular weight hyaluronan is more pronounced in the frontal WM compared to cerebellar WM in VWM [21]. Hyaluronan is mainly produced by astrocytes and inhibits OPC maturation [7, 124], which could in part explain the lack of mature oligodendroglia and WM repair in VWM [21]. These findings illustrate the diverse astrocyte phenotypes across WM regions in VWM, indicating that astrocyte heterogeneity may underlie the regional variability in disease severity.
Radiological and neuropathological features of VWM. a Sagittal T1-weighted image of a 6-year-old patient with VWM shows rarefaction of the frontal and parietal WM. b–d Axial FLAIR images confirm the diffuse rarefaction of the hemispheric WM with frontal predominance. The U-fibers show signal hyperintensity, but are not rarefied. Infratentorially, a signal hyperintensity without tissue rarefaction can be appreciate in the cerebellar hemispheric WM surrounding the dentate nuclei and in the pontine tectum. e, f Axial T2-weighted images also show a diffuse signal hyperintensity in the cerebral hemispheric WM, with relative sparing of the U-fibers, and in the tegmentum and tectum of the pons and medulla oblongata. g H&E staining of the frontal WM reveals WM tissue rarefaction and dysmorphic astrocytes in VWM with few, short and blunt cell processes. h The morphology of astrocytes is also abnormal in the relatively preserved WM in VWM, together with increased OPC numbers, i compared to those of the cerebellar WM. Scale bars 20 (g, i) and 50 (h) µm
Astrocytes as targets for therapy
For many years, oligodendrocyte and myelin dysfunction were recognized as primary drivers of WM diseases. Therapies aiming at promoting remyelination by targeting oligodendrocytes, however, have yielded little success. This demonstrates that the deficits in these diseases are not necessarily intrinsic to the myelinating cells. In fact, failure of WM repair may be the result of an unfavorable microenvironment. Disturbed crosstalk between cell types and factors present in the diseased microenvironment, including ECM components, may fail to support regeneration [35, 77, 78]. Since astrocytes dynamically regulate the microenvironment and functionally interact with all other cell types, they are crucial in determining the outcome of the regenerative response [9, 35, 82] and represent an interesting target for therapies aimed at restoring WM integrity.
Currently, there are no curative treatments for astrocytopathies. Since they are caused by genetic defects [138], a possible therapeutic strategy is glial cell replacement therapy [37]. In this, healthy glial precursor cells (GPCs) are transplanted into the CNS, where they differentiate into astrocytes and/or myelinating oligodendrocytes to replace their dysfunctional counterparts. Recent insights into the heterogeneous and dynamic nature of astrocytes [28, 45, 100] have implications for the design of these treatment strategies. To promote successful regeneration, progenitors might require pre-differentiation into a specific astrocyte subtype before transplantation. This aspect remains challenging because it remains debatable which factors drive astrocyte diversification. So far, a few attempts have been made to generate distinct astrocyte subtypes in vitro. In one study, astroglial subtypes were generated from human iPSCs by patterning GPCs to regional identities with morphogens used for neuronal subtype specification. These subtypes differentially express homeodomain transcription factors and are functionally diverse [71]. Astrocytes generated without addition of morphogens display a dorsal telencephalic phenotype, whereas retinoic acid and Shh induce astrocytes with spinal cord and ventral forebrain phenotypes, respectively. Notably, these astrocytes retained a stable and functional phenotype after transplantation into the mouse brain [71]. In another approach, GM and WM-like astrocytes were generated by stimulating iPSC-derived GPCs with fetal bovine serum or CNTF, respectively [76]. These astrocytes differed in size, morphological complexity, and expression profiles, resembling primary GM and WM astrocytes. Using this strategy, in vitro models for VWM were developed. Both human and mouse iPSC-derived WM-like astrocytes showed intrinsic subtype-specific responses to VWM mutations, including inhibition of OPC maturation into oligodendrocytes [76]. Another study reported functional differences between distinct astrocyte populations in the ability to promote CNS repair after transplantation. Human GPC-derived astrocytes pre-differentiated with BMP promoted behavioral recovery, neuronal survival, and axonal growth in the injured adult rat spinal cord, whereas astrocytes pre-differentiated with CNTF failed to do so [34]. These findings suggest that diversity of astrocytes may impact on their regenerative capacities, implying that optimized pre-transplantation differentiation protocols should be implemented to increase the therapeutic benefit of glial replacement therapies. Finally, the influence of the diseased microenvironment should be taken into consideration. Since (reactive) astrocytes adapt their phenotype in response to environmental cues after CNS injury [97], cell replacement strategies could be combined with treatments that alter the microenvironment into a niche more permissive for recovery [38, 77].
Concluding remarks and future perspectives
Astrocytes are a heterogeneous cell population with distinct subtypes. Their diversity originates from spatial patterning of progenitor domains during brain development (i.e., cell-intrinsic factors), but is also acquired in response to local, micro-environmental cues (i.e., cell-extrinsic factors), emphasizing the dynamic nature of their cell identity. Importantly, their diverse morphological and molecular properties likely serve to fulfill specialized local functions to maintain a healthy CNS. In the WM, astrocytes play crucial roles in maintaining its integrity and function in health and disease. Although limited, compelling data suggest that astrocytes might be heterogeneous throughout the WM. Astrocytopathies, a group of leukodystrophies caused by primary astrocyte dysfunction, provide a window for assessing how loss of physiological astrocytic functions or gain of detrimental functions affect CNS WM in a region-dependent manner. Importantly, astrocyte heterogeneity may contribute to selective vulnerability of WM regions in astrocytopathies and, probably, in all leukodystrophies. The underlying mechanisms driving these regional variations in disease severity remain, however, unclear. Insight into the role of astroglial heterogeneity in both healthy and diseased WM may provide new avenues for therapies aimed at promoting repair and restoring normal WM function.
References
Akwa Y, Hassett DE, Eloranta ML, Sandberg K, Masliah E, Powell H et al (1998) Transgenic expression of IFN-alpha in the central nervous system of mice protects against lethal neurotropic viral infection but induces inflammation and neurodegeneration. J Immunol 161:5016–5026
Al-Dalahmah O, Sosunov AA, Shaik A, Ofori K, Liu Y, Vonsattel JP et al (2020) Single-nucleus RNA-seq identifies Huntington disease astrocyte states. Acta Neuropathol Commun 8:19. https://doi.org/10.1186/s40478-020-0880-6
Anderson MA, Ao Y, Sofroniew MV (2014) Heterogeneity of reactive astrocytes. Neurosci Lett 565:23–29. https://doi.org/10.1016/j.neulet.2013.12.030
Anderson MA, Burda JE, Ren Y, Ao Y, O’Shea TM, Kawaguchi R et al (2016) Astrocyte scar formation aids central nervous system axon regeneration. Nature 532:195–200. https://doi.org/10.1038/nature17623
Attwell D, Buchan AM, Charpak S, Lauritzen M, Macvicar BA, Newman EA (2010) Glial and neuronal control of brain blood flow. Nature 468:232–243. https://doi.org/10.1038/nature09613
Bachoo RM, Kim RS, Ligon KL, Maher EA, Brennan C, Billings N et al (2004) Molecular diversity of astrocytes with implications for neurological disorders. Proc Natl Acad Sci U S A 101:8384–8389. https://doi.org/10.1073/pnas.0402140101
Back SA, Tuohy TM, Chen H, Wallingford N, Craig A, Struve J et al (2005) Hyaluronan accumulates in demyelinated lesions and inhibits oligodendrocyte progenitor maturation. Nat Med 11:966–972. https://doi.org/10.1038/nm1279
Baltan S (2015) Can lactate serve as an energy substrate for axons in good times and in bad, in sickness and in health? Metab Brain Dis 30:25–30. https://doi.org/10.1007/s11011-014-9595-3
Barnett SC, Linington C (2013) Myelination: do astrocytes play a role? Neuroscientist 19:442–450. https://doi.org/10.1177/1073858412465655
Barres BA (2008) The mystery and magic of glia: a perspective on their roles in health and disease. Neuron 60:430–440. https://doi.org/10.1016/j.neuron.2008.10.013
Barth PG (2002) The neuropathology of Aicardi–Goutieres syndrome. Eur J Paediatr Neurol 6(Suppl A):A27-31 (discussion A37–29, A77–86)
Bayraktar OA, Fuentealba LC, Alvarez-Buylla A, Rowitch DH (2014) Astrocyte development and heterogeneity. Cold Spring Harb Perspect Biol 7:a020362. https://doi.org/10.1101/cshperspect.a020362
Boisvert MM, Erikson GA, Shokhirev MN, Allen NJ (2018) The aging astrocyte transcriptome from multiple regions of the mouse brain. Cell Rep 22:269–285. https://doi.org/10.1016/j.celrep.2017.12.039
Boon BDC, Bulk M, Jonker AJ, Morrema THJ, van den Berg E, Popovic M et al (2020) The coarse-grained plaque: a divergent Abeta plaque-type in early-onset Alzheimer’s disease. Acta Neuropathol 140:811–830. https://doi.org/10.1007/s00401-020-02198-8
Boor PK, de Groot K, Waisfisz Q, Kamphorst W, Oudejans CB, Powers JM et al (2005) MLC1: a novel protein in distal astroglial processes. J Neuropathol Exp Neurol 64:412–419
Bradley RA, Shireman J, McFalls C, Choi J, Canfield SG, Dong Y et al (2019) Regionally specified human pluripotent stem cell-derived astrocytes exhibit different molecular signatures and functional properties. Development. https://doi.org/10.1242/dev.170910
Brenner M, Johnson AB, Boespflug-Tanguy O, Rodriguez D, Goldman JE, Messing A (2001) Mutations in GFAP, encoding glial fibrillary acidic protein, are associated with Alexander disease. Nat Genet 27:117. https://doi.org/10.1038/83679
Bugiani M, Boor I, Powers JM, Scheper GC, van der Knaap MS (2010) Leukoencephalopathy with vanishing white matter: a review. J Neuropathol Exp Neurol 69:987–996. https://doi.org/10.1097/NEN.0b013e3181f2eafa
Bugiani M, Boor I, van Kollenburg B, Postma N, Polder E, van Berkel C et al (2011) Defective glial maturation in vanishing white matter disease. J Neuropathol Exp Neurol 70:69–82. https://doi.org/10.1097/NEN.0b013e318203ae74
Bugiani M, Dubey M, Breur M, Postma NL, Dekker MP, Ter Braak T et al (2017) Megalencephalic leukoencephalopathy with cysts: the Glialcam-null mouse model. Ann Clin Transl Neurol 4:450–465. https://doi.org/10.1002/acn3.405
Bugiani M, Postma N, Polder E, Dieleman N, Scheffer PG, Sim FJ et al (2013) Hyaluronan accumulation and arrested oligodendrocyte progenitor maturation in vanishing white matter disease. Brain 136:209–222. https://doi.org/10.1093/brain/aws320
Bugiani M, Vuong C, Breur M, van der Knaap MS (2018) Vanishing white matter: a leukodystrophy due to astrocytic dysfunction. Brain Pathol 28:408–421. https://doi.org/10.1111/bpa.12606
Burda JE, Sofroniew MV (2014) Reactive gliosis and the multicellular response to CNS damage and disease. Neuron 81:229–248. https://doi.org/10.1016/j.neuron.2013.12.034
Cahoy JD, Emery B, Kaushal A, Foo LC, Zamanian JL, Christopherson KS et al (2008) A transcriptome database for astrocytes, neurons, and oligodendrocytes: a new resource for understanding brain development and function. J Neurosci 28:264–278. https://doi.org/10.1523/JNEUROSCI.4178-07.2008
Cai J, Chen Y, Cai WH, Hurlock EC, Wu H, Kernie SG et al (2007) A crucial role for Olig2 in white matter astrocyte development. Development 134:1887–1899. https://doi.org/10.1242/dev.02847
Campbell IL, Krucker T, Steffensen S, Akwa Y, Powell HC, Lane T et al (1999) Structural and functional neuropathology in transgenic mice with CNS expression of IFN-alpha. Brain Res 835:46–61
Capdevila-Nortes X, Lopez-Hernandez T, Apaja PM, Lopez de Heredia M, Sirisi S, Callejo G et al (2013) Insights into MLC pathogenesis: GlialCAM is an MLC1 chaperone required for proper activation of volume-regulated anion currents. Hum Mol Genet 22:4405–4416. https://doi.org/10.1093/hmg/ddt290
Chaboub LS, Deneen B (2012) Developmental origins of astrocyte heterogeneity: the final frontier of CNS development. Dev Neurosci 34:379–388. https://doi.org/10.1159/000343723
Chai H, Diaz-Castro B, Shigetomi E, Monte E, Octeau JC, Yu X et al (2017) Neural circuit-specialized astrocytes: transcriptomic, proteomic, morphological, and functional evidence. Neuron 95(531–549):e539. https://doi.org/10.1016/j.neuron.2017.06.029
Chen A, Akinyemi RO, Hase Y, Firbank MJ, Ndung’u MN, Foster V et al (2016) Frontal white matter hyperintensities, clasmatodendrosis and gliovascular abnormalities in ageing and post-stroke dementia. Brain 139:242–258. https://doi.org/10.1093/brain/awv328
Clarke LE, Liddelow SA, Chakraborty C, Munch AE, Heiman M, Barres BA (2018) Normal aging induces A1-like astrocyte reactivity. Proc Natl Acad Sci U S A 115:E1896–E1905. https://doi.org/10.1073/pnas.1800165115
Cragnolini AB, Montenegro G, Friedman WJ, Masco DH (2018) Brain-region specific responses of astrocytes to an in vitro injury and neurotrophins. Mol Cell Neurosci 88:240–248. https://doi.org/10.1016/j.mcn.2018.02.007
Cuadrado E, Jansen MH, Anink J, De Filippis L, Vescovi AL, Watts C et al (2013) Chronic exposure of astrocytes to interferon-alpha reveals molecular changes related to Aicardi–Goutieres syndrome. Brain 136:245–258. https://doi.org/10.1093/brain/aws321
Davies SJ, Shih CH, Noble M, Mayer-Proschel M, Davies JE, Proschel C (2011) Transplantation of specific human astrocytes promotes functional recovery after spinal cord injury. PLoS ONE 6:e17328. https://doi.org/10.1371/journal.pone.0017328
Domingues HS, Portugal CC, Socodato R, Relvas JB (2016) Oligodendrocyte, astrocyte, and microglia crosstalk in myelin development, damage, and repair. Front Cell Dev Biol 4:71. https://doi.org/10.3389/fcell.2016.00071
Dooves S, Bugiani M, Postma NL, Polder E, Land N, Horan ST et al (2016) Astrocytes are central in the pathomechanisms of vanishing white matter. J Clin Invest 126:1512–1524. https://doi.org/10.1172/JCI83908
Dooves S, Leferink PS, Krabbenborg S, Breeuwsma N, Bots S, Hillen AEJ et al (2019) Cell replacement therapy improves pathological hallmarks in a mouse model of leukodystrophy vanishing white matter. Stem Cell Rep 12:441–450. https://doi.org/10.1016/j.stemcr.2019.01.018
Dooves S, van der Knaap MS, Heine VM (2016) Stem cell therapy for white matter disorders: don’t forget the microenvironment! J Inherit Metab Dis 39:513–518. https://doi.org/10.1007/s10545-016-9925-1
Doyle JP, Dougherty JD, Heiman M, Schmidt EF, Stevens TR, Ma G et al (2008) Application of a translational profiling approach for the comparative analysis of CNS cell types. Cell 135:749–762. https://doi.org/10.1016/j.cell.2008.10.029
Dubey M, Brouwers E, Hamilton EMC, Stiedl O, Bugiani M, Koch H et al (2018) Seizures and disturbed brain potassium dynamics in the leukodystrophy megalencephalic leukoencephalopathy with subcortical cysts. Ann Neurol 83:636–649. https://doi.org/10.1002/ana.25190
Dubey M, Bugiani M, Ridder MC, Postma NL, Brouwers E, Polder E et al (2015) Mice with megalencephalic leukoencephalopathy with cysts: a developmental angle. Ann Neurol 77:114–131. https://doi.org/10.1002/ana.24307
Emsley JG, Macklis JD (2006) Astroglial heterogeneity closely reflects the neuronal-defined anatomy of the adult murine CNS. Neuron Glia Biol 2:175–186. https://doi.org/10.1017/S1740925X06000202
Escartin C, Galea E, Lakatos A, O’Callaghan JP, Petzold GC, Serrano-Pozo A et al (2021) Reactive astrocyte nomenclature, definitions, and future directions. Nat Neurosci 24:312–325. https://doi.org/10.1038/s41593-020-00783-4
Farmer WT, Abrahamsson T, Chierzi S, Lui C, Zaelzer C, Jones EV et al (2016) Neurons diversify astrocytes in the adult brain through sonic hedgehog signaling. Science 351:849–854. https://doi.org/10.1126/science.aab3103
Farmer WT, Murai K (2017) Resolving astrocyte heterogeneity in the CNS. Front Cell Neurosci 11:300. https://doi.org/10.3389/fncel.2017.00300
Faulkner JR, Herrmann JE, Woo MJ, Tansey KE, Doan NB, Sofroniew MV (2004) Reactive astrocytes protect tissue and preserve function after spinal cord injury. J Neurosci 24:2143–2155. https://doi.org/10.1523/JNEUROSCI.3547-03.2004
Fogli A, Schiffmann R, Bertini E, Ughetto S, Combes P, Eymard-Pierre E et al (2004) The effect of genotype on the natural history of eIF2B-related leukodystrophies. Neurology 62:1509–1517
Garcia-Marques J, Lopez-Mascaraque L (2013) Clonal identity determines astrocyte cortical heterogeneity. Cereb Cortex 23:1463–1472. https://doi.org/10.1093/cercor/bhs134
Garcia AD, Petrova R, Eng L, Joyner AL (2010) Sonic hedgehog regulates discrete populations of astrocytes in the adult mouse forebrain. J Neurosci 30:13597–13608. https://doi.org/10.1523/JNEUROSCI.0830-10.2010
Goursaud S, Kozlova EN, Maloteaux JM, Hermans E (2009) Cultured astrocytes derived from corpus callosum or cortical grey matter show distinct glutamate handling properties. J Neurochem 108:1442–1452. https://doi.org/10.1111/j.1471-4159.2009.05889.x
Goutieres F, Boulloche J, Bourgeois M, Aicardi J (1996) Leukoencephalopathy, megalencephaly, and mild clinical course. A recently individualized familial leukodystrophy. Report on five new cases. J Child Neurol 11:439–444. https://doi.org/10.1177/088307389601100604
Goyal M, Mehndiratta S, Faruq M, Dwivedi MK, Kapoor S (2014) Infantile onset alexander disease with normal head circumference: a genetically proven case report. J Clin Diagn Res 8:PD03-04. https://doi.org/10.7860/JCDR/2014/10211.5200
Haas B, Schipke CG, Peters O, Sohl G, Willecke K, Kettenmann H (2006) Activity-dependent ATP-waves in the mouse neocortex are independent from astrocytic calcium waves. Cereb Cortex 16:237–246. https://doi.org/10.1093/cercor/bhi101
Habib N, McCabe C, Medina S, Varshavsky M, Kitsberg D, Dvir-Szternfeld R et al (2020) Disease-associated astrocytes in Alzheimer’s disease and aging. Nat Neurosci 23:701–706. https://doi.org/10.1038/s41593-020-0624-8
Haim BL, Rowitch DH (2017) Functional diversity of astrocytes in neural circuit regulation. Nat Rev Neurosci 18:31–41. https://doi.org/10.1038/nrn.2016.159
Hamby ME, Coppola G, Ao Y, Geschwind DH, Khakh BS, Sofroniew MV (2012) Inflammatory mediators alter the astrocyte transcriptome and calcium signaling elicited by multiple G-protein-coupled receptors. J Neurosci 32:14489–14510. https://doi.org/10.1523/jneurosci.1256-12.2012
Harada K, Kamiya T, Tsuboi T (2015) Gliotransmitter release from astrocytes: functional, developmental, and pathological implications in the brain. Front Neurosci 9:499. https://doi.org/10.3389/fnins.2015.00499
Heaven MR, Flint D, Randall SM, Sosunov AA, Wilson L, Barnes S et al (2016) Composition of Rosenthal fibers, the protein aggregate hallmark of Alexander disease. J Proteome Res 15:2265–2282. https://doi.org/10.1021/acs.jproteome.6b00316
Herrmann JE, Imura T, Song B, Qi J, Ao Y, Nguyen TK et al (2008) STAT3 is a critical regulator of astrogliosis and scar formation after spinal cord injury. J Neurosci 28:7231–7243. https://doi.org/10.1523/JNEUROSCI.1709-08.2008
Hewett JA (2009) Determinants of regional and local diversity within the astroglial lineage of the normal central nervous system. J Neurochem 110:1717–1736. https://doi.org/10.1111/j.1471-4159.2009.06288.x
Hill SJ, Barbarese E, McIntosh TK (1996) Regional heterogeneity in the response of astrocytes following traumatic brain injury in the adult rat. J Neuropathol Exp Neurol 55:1221–1229
Hochstim C, Deneen B, Lukaszewicz A, Zhou Q, Anderson DJ (2008) Identification of positionally distinct astrocyte subtypes whose identities are specified by a homeodomain code. Cell 133:510–522. https://doi.org/10.1016/j.cell.2008.02.046
Hoft S, Griemsmann S, Seifert G, Steinhauser C (2014) Heterogeneity in expression of functional ionotropic glutamate and GABA receptors in astrocytes across brain regions: insights from the thalamus. Philos Trans R Soc Lond B Biol Sci 369:20130602. https://doi.org/10.1098/rstb.2013.0602
John Lin CC, Yu K, Hatcher A, Huang TW, Lee HK, Carlson J et al (2017) Identification of diverse astrocyte populations and their malignant analogs. Nat Neurosci 20:396–405. https://doi.org/10.1038/nn.4493
Khakh BS, Sofroniew MV (2015) Diversity of astrocyte functions and phenotypes in neural circuits. Nat Neurosci 18:942–952. https://doi.org/10.1038/nn.4043
Kiray H, Lindsay SL, Hosseinzadeh S, Barnett SC (2016) The multifaceted role of astrocytes in regulating myelination. Exp Neurol 283:541–549. https://doi.org/10.1016/j.expneurol.2016.03.009
Kita Y, Kawakami K, Takahashi Y, Murakami F (2013) Development of cerebellar neurons and glias revealed by in utero electroporation: Golgi-like labeling of cerebellar neurons and glias. PLoS ONE 8:e70091. https://doi.org/10.1371/journal.pone.0070091
Klok MD, Bakels HS, Postma NL, van Spaendonk RM, van der Knaap MS, Bugiani M (2015) Interferon-alpha and the calcifying microangiopathy in Aicardi–Goutieres syndrome. Ann Clin Transl Neurol 2:774–779. https://doi.org/10.1002/acn3.213
Klok MD, Bugiani M, de Vries SI, Gerritsen W, Breur M, van der Sluis S et al (2018) Axonal abnormalities in vanishing white matter. Ann Clin Transl Neurol 5:429–444. https://doi.org/10.1002/acn3.540
Kofuji P, Newman EA (2004) Potassium buffering in the central nervous system. Neuroscience 129:1045–1056. https://doi.org/10.1016/j.neuroscience.2004.06.008
Krencik R, Weick JP, Liu Y, Zhang ZJ, Zhang SC (2011) Specification of transplantable astroglial subtypes from human pluripotent stem cells. Nat Biotechnol 29:528–534. https://doi.org/10.1038/nbt.1877
Lanjakornsiripan D, Pior BJ, Kawaguchi D, Furutachi S, Tahara T, Katsuyama Y et al (2018) Layer-specific morphological and molecular differences in neocortical astrocytes and their dependence on neuronal layers. Nat Commun 9:1623. https://doi.org/10.1038/s41467-018-03940-3
Lee SH, Kim WT, Cornell-Bell AH, Sontheimer H (1994) Astrocytes exhibit regional specificity in gap-junction coupling. Glia 11:315–325. https://doi.org/10.1002/glia.440110404
Leegwater PA, Yuan BQ, van der Steen J, Mulders J, Konst AA, Boor PK et al (2001) Mutations of MLC1 (KIAA0027), encoding a putative membrane protein, cause megalencephalic leukoencephalopathy with subcortical cysts. Am J Hum Genet 68:831–838. https://doi.org/10.1086/319519
Leferink PS, Breeuwsma N, Bugiani M, van der Knaap MS, Heine VM (2018) Affected astrocytes in the spinal cord of the leukodystrophy vanishing white matter. Glia 66:862–873. https://doi.org/10.1002/glia.23289
Leferink PS, Dooves S, Hillen AEJ, Watanabe K, Jacobs G, Gasparotto L et al (2019) Astrocyte subtype vulnerability in stem cell models of vanishing white matter. Ann Neurol. https://doi.org/10.1002/ana.25585
Leferink PS, Heine VM (2018) The healthy and diseased microenvironments regulate oligodendrocyte properties: implications for regenerative medicine. Am J Pathol 188:39–52. https://doi.org/10.1016/j.ajpath.2017.08.030
Li J, Zhang L, Chu Y, Namaka M, Deng B, Kong J et al (2016) Astrocytes in oligodendrocyte lineage development and white matter pathology. Front Cell Neurosci 10:119. https://doi.org/10.3389/fncel.2016.00119
Li L, Tian E, Chen X, Chao J, Klein J, Qu Q et al (2018) GFAP mutations in astrocytes impair oligodendrocyte progenitor proliferation and myelination in an hiPSC model of alexander disease. Cell Stem Cell 23(239–251):e236. https://doi.org/10.1016/j.stem.2018.07.009
Liddelow SA, Guttenplan KA, Clarke LE, Bennett FC, Bohlen CJ, Schirmer L et al (2017) Neurotoxic reactive astrocytes are induced by activated microglia. Nature 541:481–487. https://doi.org/10.1038/nature21029
Lopez-Hernandez T, Ridder MC, Montolio M, Capdevila-Nortes X, Polder E, Sirisi S et al (2011) Mutant GlialCAM causes megalencephalic leukoencephalopathy with subcortical cysts, benign familial macrocephaly, and macrocephaly with retardation and autism. Am J Hum Genet 88:422–432. https://doi.org/10.1016/j.ajhg.2011.02.009
Lundgaard I, Osorio MJ, Kress BT, Sanggaard S, Nedergaard M (2014) White matter astrocytes in health and disease. Neuroscience 276:161–173. https://doi.org/10.1016/j.neuroscience.2013.10.050
Marshall CA, Novitch BG, Goldman JE (2005) Olig2 directs astrocyte and oligodendrocyte formation in postnatal subventricular zone cells. J Neurosci 25:7289–7298. https://doi.org/10.1523/JNEUROSCI.1924-05.2005
McKinnon RD, Waldron S, Kiel ME (2005) PDGF alpha-receptor signal strength controls an RTK rheostat that integrates phosphoinositol 3’-kinase and phospholipase Cgamma pathways during oligodendrocyte maturation. J Neurosci 25:3499–3508. https://doi.org/10.1523/JNEUROSCI.5049-04.2005
Messing A, Brenner M, Feany MB, Nedergaard M, Goldman JE (2012) Alexander disease. J Neurosci 32:5017–5023. https://doi.org/10.1523/JNEUROSCI.5384-11.2012
Messing A, Head MW, Galles K, Galbreath EJ, Goldman JE, Brenner M (1998) Fatal encephalopathy with astrocyte inclusions in GFAP transgenic mice. Am J Pathol 152:391–398
Mignot C, Boespflug-Tanguy O, Gelot A, Dautigny A, Pham-Dinh D, Rodriguez D (2004) Alexander disease: putative mechanisms of an astrocytic encephalopathy. Cell Mol Life Sci 61:369–385. https://doi.org/10.1007/s00018-003-3143-3
Miller RH, Raff MC (1984) Fibrous and protoplasmic astrocytes are biochemically and developmentally distinct. J Neurosci 4:585–592
Min R, van der Knaap MS (2018) Genetic defects disrupting glial ion and water homeostasis in the brain. Brain Pathol 28:372–387. https://doi.org/10.1111/bpa.12602
Molofsky AV, Krencik R, Ullian EM, Tsai HH, Deneen B, Richardson WD et al (2012) Astrocytes and disease: a neurodevelopmental perspective. Genes Dev 26:891–907. https://doi.org/10.1101/gad.188326.112
Moore CS, Abdullah SL, Brown A, Arulpragasam A, Crocker SJ (2011) How factors secreted from astrocytes impact myelin repair. J Neurosci Res 89:13–21. https://doi.org/10.1002/jnr.22482
Morel L, Chiang MSR, Higashimori H, Shoneye T, Iyer LK, Yelick J et al (2017) Molecular and functional properties of regional astrocytes in the adult brain. J Neurosci 37:8706–8717. https://doi.org/10.1523/JNEUROSCI.3956-16.2017
Morel L, Men Y, Chiang MSR, Tian Y, Jin S, Yelick J et al (2019) Intracortical astrocyte subpopulations defined by astrocyte reporter Mice in the adult brain. Glia 67:171–181. https://doi.org/10.1002/glia.23545
Nagy JI, Patel D, Ochalski PA, Stelmack GL (1999) Connexin30 in rodent, cat and human brain: selective expression in gray matter astrocytes, co-localization with connexin43 at gap junctions and late developmental appearance. Neuroscience 88:447–468
Nair A, Frederick TJ, Miller SD (2008) Astrocytes in multiple sclerosis: a product of their environment. Cell Mol Life Sci 65:2702–2720. https://doi.org/10.1007/s00018-008-8059-5
Nash B, Thomson CE, Linington C, Arthur AT, McClure JD, McBride MW et al (2011) Functional duality of astrocytes in myelination. J Neurosci 31:13028–13038. https://doi.org/10.1523/JNEUROSCI.1449-11.2011
Nathan FM, Li S (2017) Environmental cues determine the fate of astrocytes after spinal cord injury. Neural Regen Res 12:1964–1970. https://doi.org/10.4103/1673-5374.221144
Nave KA (2010) Myelination and support of axonal integrity by glia. Nature 468:244–252. https://doi.org/10.1038/nature09614
Neumann H, Kotter MR, Franklin RJ (2009) Debris clearance by microglia: an essential link between degeneration and regeneration. Brain 132:288–295. https://doi.org/10.1093/brain/awn109
Oberheim NA, Goldman SA, Nedergaard M (2012) Heterogeneity of astrocytic form and function. Methods Mol Biol 814:23–45. https://doi.org/10.1007/978-1-61779-452-0_3
Oberheim NA, Takano T, Han X, He W, Lin JH, Wang F et al (2009) Uniquely hominid features of adult human astrocytes. J Neurosci 29:3276–3287. https://doi.org/10.1523/JNEUROSCI.4707-08.2009
Obermeier B, Daneman R, Ransohoff RM (2013) Development, maintenance and disruption of the blood-brain barrier. Nat Med 19:1584–1596. https://doi.org/10.1038/nm.3407
Olabarria M, Putilina M, Riemer EC, Goldman JE (2015) Astrocyte pathology in Alexander disease causes a marked inflammatory environment. Acta Neuropathol 130:469–486. https://doi.org/10.1007/s00401-015-1469-1
Ono K, Takebayashi H, Ikeda K, Furusho M, Nishizawa T, Watanabe K et al (2008) Regional- and temporal-dependent changes in the differentiation of Olig2 progenitors in the forebrain, and the impact on astrocyte development in the dorsal pallium. Dev Biol 320:456–468. https://doi.org/10.1016/j.ydbio.2008.06.001
Orcesi S, La Piana R, Fazzi E (2009) Aicardi–Goutieres syndrome. Br Med Bull 89:183–201. https://doi.org/10.1093/bmb/ldn049
Pekny M, Pekna M, Messing A, Steinhauser C, Lee JM, Parpura V et al (2016) Astrocytes: a central element in neurological diseases. Acta Neuropathol 131:323–345. https://doi.org/10.1007/s00401-015-1513-1
Perea G, Navarrete M, Araque A (2009) Tripartite synapses: astrocytes process and control synaptic information. Trends Neurosci 32:421–431. https://doi.org/10.1016/j.tins.2009.05.001
Poopalasundaram S, Knott C, Shamotienko OG, Foran PG, Dolly JO, Ghiani CA et al (2000) Glial heterogeneity in expression of the inwardly rectifying K(+) channel, Kir4.1, in adult rat CNS. Glia 30:362–372
Qian JA, Bull MS, Levitt P (1992) Target-derived astroglia regulate axonal outgrowth in a region-specific manner. Dev Biol 149:278–294
Quinlan RA, Brenner M, Goldman JE, Messing A (2007) GFAP and its role in Alexander disease. Exp Cell Res 313:2077–2087. https://doi.org/10.1016/j.yexcr.2007.04.004
Raff MC, Abney ER, Cohen J, Lindsay R, Noble M (1983) Two types of astrocytes in cultures of developing rat white matter: differences in morphology, surface gangliosides, and growth characteristics. J Neurosci 3:1289–1300
Reemst K, Noctor SC, Lucassen PJ, Hol EM (2016) The indispensable roles of microglia and astrocytes during brain development. Front Hum Neurosci 10:566. https://doi.org/10.3389/fnhum.2016.00566
Regan MR, Huang YH, Kim YS, Dykes-Hoberg MI, Jin L, Watkins AM et al (2007) Variations in promoter activity reveal a differential expression and physiology of glutamate transporters by glia in the developing and mature CNS. J Neurosci 27:6607–6619. https://doi.org/10.1523/JNEUROSCI.0790-07.2007
Reichenbach N, Delekate A, Plescher M, Schmitt F, Krauss S, Blank N et al (2019) Inhibition of Stat3-mediated astrogliosis ameliorates pathology in an Alzheimer’s disease model. EMBO Mol Med. https://doi.org/10.15252/emmm.201809665
Ridder MC, Boor I, Lodder JC, Postma NL, Capdevila-Nortes X, Duarri A et al (2011) Megalencephalic leucoencephalopathy with cysts: defect in chloride currents and cell volume regulation. Brain 134:3342–3354. https://doi.org/10.1093/brain/awr255
Rinholm JE, Hamilton NB, Kessaris N, Richardson WD, Bergersen LH, Attwell D (2011) Regulation of oligodendrocyte development and myelination by glucose and lactate. J Neurosci 31:538–548. https://doi.org/10.1523/JNEUROSCI.3516-10.2011
Salvi F, Aoki Y, Della Nave R, Vella A, Pastorelli F, Scaglione C et al (2005) Adult Alexander’s disease without leukoencephalopathy. Ann Neurol 58:813–814. https://doi.org/10.1002/ana.20634
Sase S, Takanohashi A, Vanderver A, Almad A (2018) Astrocytes, an active player in Aicardi–Goutieres syndrome. Brain Pathol 28:399–407. https://doi.org/10.1111/bpa.12600
Scemes E, Giaume C (2006) Astrocyte calcium waves: what they are and what they do. Glia 54:716–725. https://doi.org/10.1002/glia.20374
Shannon C, Salter M, Fern R (2007) GFP imaging of live astrocytes: regional differences in the effects of ischaemia upon astrocytes. J Anat 210:684–692. https://doi.org/10.1111/j.1469-7580.2007.00731.x
Silver J, Miller JH (2004) Regeneration beyond the glial scar. Nat Rev Neurosci 5:146–156. https://doi.org/10.1038/nrn1326
Simard M, Nedergaard M (2004) The neurobiology of glia in the context of water and ion homeostasis. Neuroscience 129:877–896. https://doi.org/10.1016/j.neuroscience.2004.09.053
Skripuletz T, Hackstette D, Bauer K, Gudi V, Pul R, Voss E et al (2013) Astrocytes regulate myelin clearance through recruitment of microglia during cuprizone-induced demyelination. Brain 136:147–167. https://doi.org/10.1093/brain/aws262
Sloane JA, Batt C, Ma Y, Harris ZM, Trapp B, Vartanian T (2010) Hyaluronan blocks oligodendrocyte progenitor maturation and remyelination through TLR2. Proc Natl Acad Sci U S A 107:11555–11560. https://doi.org/10.1073/pnas.1006496107
Sofroniew MV (2014) Astrogliosis. Cold Spring Harb Perspect Biol 7:a020420. https://doi.org/10.1101/cshperspect.a020420
Sofroniew MV (2009) Molecular dissection of reactive astrogliosis and glial scar formation. Trends Neurosci 32:638–647. https://doi.org/10.1016/j.tins.2009.08.002
Sofroniew MV, Vinters HV (2010) Astrocytes: biology and pathology. Acta Neuropathol 119:7–35. https://doi.org/10.1007/s00401-009-0619-8
Sosunov A, Olabarria M, Goldman JE (2018) Alexander disease: an astrocytopathy that produces a leukodystrophy. Brain Pathol 28:388–398. https://doi.org/10.1111/bpa.12601
Sosunov AA, Guilfoyle E, Wu X, McKhann GM 2nd, Goldman JE (2013) Phenotypic conversions of “protoplasmic” to “reactive” astrocytes in Alexander disease. J Neurosci 33:7439–7450. https://doi.org/10.1523/JNEUROSCI.4506-12.2013
Sosunov AA, Wu X, Tsankova NM, Guilfoyle E, McKhann GM 2nd, Goldman JE (2014) Phenotypic heterogeneity and plasticity of isocortical and hippocampal astrocytes in the human brain. J Neurosci 34:2285–2298. https://doi.org/10.1523/JNEUROSCI.4037-13.2014
Tabata H (2015) Diverse subtypes of astrocytes and their development during corticogenesis. Front Neurosci 9:114. https://doi.org/10.3389/fnins.2015.00114
Takouda J, Katada S, Nakashima K (2017) Emerging mechanisms underlying astrogenesis in the developing mammalian brain. Proc Jpn Acad Ser B Phys Biol Sci 93:386–398. https://doi.org/10.2183/pjab.93.024
Teijido O, Casaroli-Marano R, Kharkovets T, Aguado F, Zorzano A, Palacin M et al (2007) Expression patterns of MLC1 protein in the central and peripheral nervous systems. Neurobiol Dis 26:532–545. https://doi.org/10.1016/j.nbd.2007.01.016
Tsai HH, Li H, Fuentealba LC, Molofsky AV, Taveira-Marques R, Zhuang H et al (2012) Regional astrocyte allocation regulates CNS synaptogenesis and repair. Science 337:358–362. https://doi.org/10.1126/science.1222381
Uggetti C, La Piana R, Orcesi S, Egitto MG, Crow YJ, Fazzi E (2009) Aicardi-Goutieres syndrome: neuroradiologic findings and follow-up. AJNR Am J Neuroradiol 30:1971–1976. https://doi.org/10.3174/ajnr.A1694
van der Knaap MS, Barth PG, Stroink H, van Nieuwenhuizen O, Arts WF, Hoogenraad F et al (1995) Leukoencephalopathy with swelling and a discrepantly mild clinical course in eight children. Ann Neurol 37:324–334. https://doi.org/10.1002/ana.410370308
van der Knaap MS, Boor I, Estevez R (2012) Megalencephalic leukoencephalopathy with subcortical cysts: chronic white matter oedema due to a defect in brain ion and water homoeostasis. Lancet Neurol 11:973–985. https://doi.org/10.1016/S1474-4422(12)70192-8
van der Knaap MS, Bugiani M (2017) Leukodystrophies: a proposed classification system based on pathological changes and pathogenetic mechanisms. Acta Neuropathol 134:351–382. https://doi.org/10.1007/s00401-017-1739-1
van der Knaap MS, Leegwater PA, Konst AA, Visser A, Naidu S, Oudejans CB et al (2002) Mutations in each of the five subunits of translation initiation factor eIF2B can cause leukoencephalopathy with vanishing white matter. Ann Neurol 51:264–270
van der Knaap MS, Naidu S, Breiter SN, Blaser S, Stroink H, Springer S et al (2001) Alexander disease: diagnosis with MR imaging. AJNR Am J Neuroradiol 22:541–552
van der Knaap MS, Pronk JC, Scheper GC (2006) Vanishing white matter disease. Lancet Neurol 5:413–423. https://doi.org/10.1016/S1474-4422(06)70440-9
van Heteren JT, Rozenberg F, Aronica E, Troost D, Lebon P, Kuijpers TW (2008) Astrocytes produce interferon-alpha and CXCL10, but not IL-6 or CXCL8, in Aicardi–Goutieres syndrome. Glia 56:568–578. https://doi.org/10.1002/glia.20639
Verkhratsky A, Augusto-Oliveira M, Pivoriunas A, Popov A, Brazhe A, Semyanov A (2021) Astroglial asthenia and loss of function, rather than reactivity, contribute to the ageing of the brain. Pflugers Arch 473:753–774. https://doi.org/10.1007/s00424-020-02465-3
Verkhratsky A, Marutle A, Rodriguez-Arellano JJ, Nordberg A (2015) Glial asthenia and functional paralysis: a new perspective on neurodegeneration and Alzheimer’s disease. Neuroscientist 21:552–568. https://doi.org/10.1177/1073858414547132
Verkhratsky A, Parpura V (2016) Astrogliopathology in neurological, neurodevelopmental and psychiatric disorders. Neurobiol Dis 85:254–261. https://doi.org/10.1016/j.nbd.2015.03.025
Verkhratsky A, Rodriguez JJ, Steardo L (2014) Astrogliopathology: a central element of neuropsychiatric diseases? Neuroscientist 20:576–588. https://doi.org/10.1177/1073858413510208
Voskuhl RR, Peterson RS, Song B, Ao Y, Morales LB, Tiwari-Woodruff S et al (2009) Reactive astrocytes form scar-like perivascular barriers to leukocytes during adaptive immune inflammation of the CNS. J Neurosci 29:11511–11522. https://doi.org/10.1523/JNEUROSCI.1514-09.2009
Vue TY, Kim EJ, Parras CM, Guillemot F, Johnson JE (2014) Ascl1 controls the number and distribution of astrocytes and oligodendrocytes in the gray matter and white matter of the spinal cord. Development 141:3721–3731. https://doi.org/10.1242/dev.105270
Wang Y, Cheng X, He Q, Zheng Y, Kim DH, Whittemore SR et al (2011) Astrocytes from the contused spinal cord inhibit oligodendrocyte differentiation of adult oligodendrocyte precursor cells by increasing the expression of bone morphogenetic proteins. J Neurosci 31:6053–6058. https://doi.org/10.1523/JNEUROSCI.5524-09.2011
Wanner IB, Anderson MA, Song B, Levine J, Fernandez A, Gray-Thompson Z et al (2013) Glial scar borders are formed by newly proliferated, elongated astrocytes that interact to corral inflammatory and fibrotic cells via STAT3-dependent mechanisms after spinal cord injury. J Neurosci 33:12870–12886. https://doi.org/10.1523/JNEUROSCI.2121-13.2013
Weber B, Barros LF (2015) The astrocyte: powerhouse and recycling center. Cold Spring Harb Perspect Biol. https://doi.org/10.1101/cshperspect.a020396
Wen W, Sachdev P (2004) The topography of white matter hyperintensities on brain MRI in healthy 60- to 64-year-old individuals. Neuroimage 22:144–154. https://doi.org/10.1016/j.neuroimage.2003.12.027
Wheeler MA, Clark IC, Tjon EC, Li Z, Zandee SEJ, Couturier CP et al (2020) MAFG-driven astrocytes promote CNS inflammation. Nature 578:593–599. https://doi.org/10.1038/s41586-020-1999-0
Yang Y, Jackson R (2019) Astrocyte identity: evolutionary perspectives on astrocyte functions and heterogeneity. Curr Opin Neurobiol 56:40–46. https://doi.org/10.1016/j.conb.2018.11.006
Yang YG, Lindahl T, Barnes DE (2007) Trex1 exonuclease degrades ssDNA to prevent chronic checkpoint activation and autoimmune disease. Cell 131:873–886. https://doi.org/10.1016/j.cell.2007.10.017
Yoon H, Walters G, Paulsen AR, Scarisbrick IA (2017) Astrocyte heterogeneity across the brain and spinal cord occurs developmentally, in adulthood and in response to demyelination. PLoS ONE 12:e0180697. https://doi.org/10.1371/journal.pone.0180697
Zamanian JL, Xu L, Foo LC, Nouri N, Zhou L, Giffard RG et al (2012) Genomic analysis of reactive astrogliosis. J Neurosci 32:6391–6410. https://doi.org/10.1523/JNEUROSCI.6221-11.2012
Zhu X, Bergles DE, Nishiyama A (2008) NG2 cells generate both oligodendrocytes and gray matter astrocytes. Development 135:145–157. https://doi.org/10.1242/dev.004895
Acknowledgements
The study was supported by a grant from ZonMw (TOP 91217006). M.S.v.d.K. is a member of the European Reference Network for Rare Neurological Disorders (ERN-RND), project ID 739510.
Author information
Authors and Affiliations
Contributions
BCP researched data for the article. MBu, BCP and JHKM discussed the content and wrote the manuscript. MBr designed schematic figures and commented on the manuscript. MSvdK provided the MRI images and MBu the histopathological images. MSvdK and MBu reviewed and edited the manuscript before submission. All the authors agreed on the content of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bugiani, M., Plug, B.C., Man, J.H.K. et al. Heterogeneity of white matter astrocytes in the human brain. Acta Neuropathol 143, 159–177 (2022). https://doi.org/10.1007/s00401-021-02391-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-021-02391-3