Abstract
Synaptic loss is the major neurobiological substrate of cognitive dysfunction in Alzheimer’s disease (AD). Synaptic failure is an early event in the pathogenesis that is clearly detectable already in patients with mild cognitive impairment (MCI), a prodromal state of AD. It progresses during the course of AD and in most early stages involves mechanisms of compensation before reaching a stage of decompensated function. This dynamic process from an initially reversible functionally responsive stage of down-regulation of synaptic function to stages irreversibly associated with degeneration might be related to a disturbance of structural brain self-organization and involves morphoregulatory molecules such as the amyloid precursor protein. Further, recent evidence suggests a role for diffusible oligomers of amyloid β in synaptic dysfunction. To form synaptic connections and to continuously re-shape them in a process of ongoing structural adaptation, neurons must permanently withdraw from the cell cycle. Previously, we formulated the hypothesis that differentiated neurons after having withdrawn from the cell cycle are able to use molecular mechanisms primarily developed to control proliferation alternatively to control synaptic plasticity. The existence of these alternative effector pathways within neurons might put them at risk of erroneously converting signals derived from plastic synaptic changes into the program of cell cycle activation, which subsequently leads to cell death. The molecular mechanisms involved in cell cycle activation might, thus, link aberrant synaptic changes to cell death.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
“Error is modern while truth is ancient” (Ramón y Cajal, 1928 [168])
When Ramón y Cajal pointed out “One also might imagine that amnesia, a paucity of thought associations, retardation, and dementia could result when synapses between neurons are weakened as a result of a more or less pathological condition, that is, when processes atrophy and no longer form contacts, when cortical mnemonic or association areas suffer partial disorganization” [167], he was probably the first to realize that dementia results from a dysfunction of synaptic contacts.
In the late nineteenth century, some scientists began to speculate about how neuronal extension and retraction, even in the adult brain, could relate to behavior [45, 49, 50, 108, 118–121, 165, 166, 210, 215, 220]. One particularly attractive idea was that axons and dendrites of different cells could grow closer to each other at frequently used junctions to enhance communication. This sort of action, some scientists thought, could account for learning and perhaps even the association of higher ideas. Rabl-Rückhard [165] and others [49] advanced the idea that certain conscious activities may be interpreted in terms of neuronal "ameboid" movements that could account for learning and memory. Forgetting, on the contrary, was believed to involve a “spreading of the gap.” Disuse, diseases of the nervous system, and aging were believed to be the factors that could cause processes to pull away from each other at the intercellular junction. The idea that intercellular junctional growth could account for memory was first anticipated in 1872 by Alexander Bain [14]. With this idea, however, Bain was much ahead of his time, and contemporaries did not give his theory serious consideration. In the 1890s, Ramón y Cajal similarly believed it probable that information could be stored by modifying interneuronal connections and that mental excercise leads to greater growth of neuronal collaterals in the stimulated regions of the brain [166, 167]. He also proposed that the proliferation of neural connections in the cerebral cortex may correlate with intelligence. In the Croonian Lecture given in England in 1894, he surmised that “brain jogging” (“gymnastique cérébrale”) may increase the growth of certain dendrites and the branching of axon collaterals [166]. Intelligence, he argued, can be achieved economically, without adding more neurons and without demanding more space, by increasing dendrite and axon branching. Only a few years later, in 1897, Charles Scott Sherrington coined the term "synapse" to describe the junction between nerve cells.
Synapse loss is the major neurobiological substrate of cognitive dysfunction in Alzheimer’s disease
More than 100 years after Ramón y Cajal’s original suggestion, his ideas on the neurobiological substrate of amnesia and dementia have basically been proven [212]. Data obtained by electron microscopy [40, 42–44, 182–184], immunocytochemical and biochemical analyses on synaptic marker proteins in AD biopsies and autopsies [47, 59, 83, 140, 211] indicate that synaptic loss in the hippocampus and neocortex is an early event [81, 135] and the major structural correlate of cognitive dysfunction [19, 27, 42–44, 63, 66, 72, 80, 83, 106, 130, 134, 158, 228].
Quantitative ultrastructural studies performed on temporal and frontal cortical biopsies within 2 to 4 years after the clinical onset of the disease have revealed a 25% to 35% decrease in the numerical density of synapses and a 15% to 35% loss in the number of synapses per neuron [40]. Synaptic loss might even be more pronounced in the hippocampus where it amounts to 44% to 55% [183].
From all cortical areas analyzed, the hippocampus appears to be the most severely affected by the loss of synaptic proteins, while the occipital cortex is affected least [22, 25, 26, 33, 38, 39, 56, 59, 79–81, 83, 106, 113, 117, 135, 139, 140, 157, 196, 206, 207, 211]. In other words, there appear to be regional differences within the cerebral cortex with respect to the severity of synaptic marker loss that basically match the pattern of neurofibrillary degeneration as outlined in the Braaks’ staging [24]. Further, the highly correlative relationship between loss of synaptic markers and neurofibrillary tangle counts in the same brain region [79, 83, 117, 132, 180, 221] also supports this link between tangle formation and synaptic dysfunction. More detailed studies on the relationship between synaptophysin mRNA and tangle formation at the level of individual neurons directly proved a significant reduction of synaptophysin mRNA in neurons affected by tangle formation [29, 30].
On the contrary, results of studies on plaques are less consistent. While a few studies [106, 180, 207] described a link between higher plaques counts and lower synaptic protein measures, other studies failed to establish such a relationship [79, 80, 83, 117, 141].
Most studies analyzing the link between premortem severity of cognitive impairment and synaptic pathology in AD agree on the correlation between lower synaptic protein levels and some aspect of cognitive dysfunction [22, 79, 106, 131, 132, 133, 149, 180, 181, 206, 207, 211, 221], although in several studies, correlations between synaptic proteins and cognitive functions are region-specific.
The reduction in synaptic vesicle proteins in AD is likely related to the clinical symptoms of dementia, given their function in vesicle trafficking, docking and fusion to the synaptic membrane and neurotranmitter exocytosis. Synaptic pathology in AD is reflected by a loss of all major components of small synaptic vesicles that accommodate classical neurotransmitters and large dense core vesicles that store most peptides, together with a loss of molecular components of pre- and postsynaptic compartments accompanied by extensive pathological changes of the synapse [38, 84, 95, 105–107, 109, 128, 170, 194, 195, 206, 207, 221, 233] (Table 1).
Although degeneration of subcortical input might contribute to cortical synapse loss [7], most of the synaptic loss in the neocortex might derive from loss of cortico-cortical associational fibers [86, 110, 136, 137, 150].
Synaptic failure is an early event and accelerates slowly in a process of dynamic reorganization
Synapse formation and stabilization in the nervous system are dynamic processes and so is the synaptic degeneration in AD. AD is a slowly progressing disorder apparently preceded by a clinically silent period of several years or even decades. Similarly, synaptic degeneration might be a slow process progressing from an initially reversible functionally responsive stage of down-regulation of synaptic function to stages irreversibly associated by marked synapse loss [169].
Disturbances of synaptic integrity can be detected already in patients with mild cognitive impairment (MCI), a prodromal state of AD [133, 184], suggesting that synaptic degeneration occurs very early on in the process of AD. Ultrastructural stereological investigations on rapid postmortem autopsy samples revealed an 18% synapse loss in the hippocampal CA1 region of MCI patients that progressed to a 55% synapse loss in mild AD [183]. Although with progression of the disease there is a steady decline in synaptic population, eventually accompanied by the loss of about 10-20% of cortical neurons [94, 130], recent studies suggest a biphasic process with an initial rise of synaptic markers in early stages of the disease, suggesting synaptic reorganization. An initial transient rise, for example, was observed for synaptophysin and other presynaptic proteins in cortical association areas at Braak stage III, i.e., prior to neurofibrillary pathology [153]. Other studies indicated an increase in drebrin in the frontal cortex of patients with MCI followed by a 40% to 60% decrease in severe AD [36], an increase in the expression of postsynaptic density protein PSD-95 in AD brain [109], as well as an elevation in glutamatergic presynaptic bouton density in midfrontal gyrus of MCI patients [16]. Functional magnetic resonance imaging (fMRI) on MCI patients similarly suggests that there might be a phase of paradoxically increased activation early in the course of prodromal AD [200].
Dynamic synaptic reorganization during the process of degeneration is further supported by observations of an increase in synaptic size that accompanies synaptic loss in various cortical regions in AD and might reflect the attempt of a functional compensatory increase of synaptic efficacy [18, 44, 182]. As a result, the total synaptic area per unit tissue volume is initially preserved, but decreases thereafter, paralleled by cognitive decline [44, 138, 211]. Synaptic compensatory mechanisms that in normal aging [15, 19, 20, 28, 55] succeed in preserving considerable cognitive function are, thus, disrupted in AD. At least in early stages of the disease, processes of compensation and decompensation might be present at the time in the same region as well as in different brain regions, which might to some extent obscure direct linear relationships between brain pathology and functional measures.
Findings of a deregulation of proteins involved in structural plasticity of axons and dendrites [1, 51, 77, 93, 116, 146] as well as results of computational studies [76, 85] indicate a failure of local neuronal regulatory mechanisms of synaptic plasticity and make a primary disturbance of synapse turnover very likely. This assumption is further supported by alterations in the composition [20, 67, 102, 126, 205] and fluidity of membranes [53, 238] as well as by direct morphological evidence of a disturbed axonal and dendritic remodeling (for review, see [4, 5]).
Recent studies applying functional magnetic resonance imaging (fMRI) provide further evidence of disrupted organization of functional brain connectivity in AD. AD patients have significantly lower regional connectivity and show disrupted global functional brain network organization when compared to healthy controls [199, 224]. Reduced functional connectivity of the hippocampus can already be obtained in MCI patients compared to normal aging control subjects [13, 92]. In AD patients, cognitive decline is associated with disrupted functional connectivity in the entire brain [204].
Mechanisms of synaptic failure in Alzheimer’s disease, potential roles of Aβ, APP and tau
Synaptic loss is currently the best neurobiological correlate of cognitive deficits in AD. In addition to the synapse loss due to the death of neurons, there is evidence that still living neurons lose their synapses in AD [34]. Further, synaptic function is impaired in living neurons as demonstrated by decrements in transcripts related to synaptic vesicle trafficking [34]. The question, thus, has been addressed to what extent the established amyloid and tau pathology might contribute to synaptic dysfunction in AD.
Disruption of synaptic function by Aβ oligomers
Although Aβ might show some deleterious cellular effects, brain amyloid load does not correlate well with synaptic loss, neuronal death or cognitive dysfunction ([211], see above). Defects in synaptic transmission, furthermore, occur in AD well before the formation of amyloid plaques and neurofibrillary tangles [4, 5, 223]. It might, thus, be argued that soluble molecular species that are generated at very early stages of the disease and that only at more advanced stages are deposited in an aggregated form could be involved in synaptic failure. It has, thus, been suggested that soluble assembly states of Aβ peptides can cause cognitive problems by disrupting synaptic function in the absence of significant neurodegeneration. There is evidence that soluble oligomers of Aβ can selectively impair synaptic plasticity mechanisms necessary for memory processing [35, 71, 176]. Both cell-derived and synthetic soluble Aβ oligomers can disrupt hippocampal long-term potentiation in slices and in vivo, and can also impair the memory of complex learned behavior in rodents and decrease dendritic spine density in organotypic hippocampal slice cultures [48, 98, 99, 104, 156, 190, 192, 222]. Intracerebroventricular injections of soluble synthetic Aβ1-40-dimers rapidly inhibit the plasticity of excitatory synaptic transmission at doses of 10-42 pmol comparable to natural Aβ [87]. Other studies, however, have suggested opposite effects with picomolar levels of Aβ1-40 playing a neurotrophic role in cell cultures [161, 231]. Also, low picomolar concentrations of preparations containing both Aβ1-42 monomers and oligomers have been shown to be able to induce a marked increase in hippocampal long-term potentiation and produce a pronounced enhancement of both reference and contextual fear memory [164]. Current findings on the effects of soluble Aβ oligomers on synaptic function are, thus, not entirely conclusive. Still, a recent study applying array tomography, a technique that combines ultrathin sectioning of tissue with immunofluorescence, to a mouse model of AD provides compelling evidence that senile plaques are a potential reservoir of synaptotoxic oligomeric Aβ [100].
Synaptotrophic function of human wild-type APP and its failure in FAD-mutated APP
Disturbances of synaptic function and plasticity have also been observed in transgenic mice overexpressing FAD-mutated APP [32, 54, 62, 65]. APP is a type I transmembrane protein that belongs to a conserved family including Apl-1 in Caenorhabditis elegans [37], APPL in Drosophila [129, 173] and APP [209], APP-like protein 1 (APLP1) [226] and APLP2 [197, 227] in mammals. Within the brain, APP can be detected in synaptic membranes [97] and has been shown to localize to postsynaptic densities, axons and dendrites [187, 193]. APP undergoes fast axonal transport [101, 178, 229] and is targeted to synaptic sites [187, 193]. Studies on overexpression or knockout of APP and its homologues indicate a critical role in neuronal survival, neurite outgrowth, synaptogenesis and synaptic plasticity [41, 115, 148, 152, 191].
The proteolytic processing of APP has been investigated extensively, and numerous studies have focused on elevations in the Aβ-peptide, which is generated during normal metabolism of APP [70] and forms the major component of plaques, a major hallmark of the disease [142]. While mechanisms of APP processing giving rise to Aβ have received the most attention, the physiological function of APP and potential sequelae of impaired function are less well understood.
Several genetic knockout models for APP and its homologues have been generated to gain insight into its function [78, 219, 237]. Triple knockouts for APP and its homologues are lethal and show cortical dysplasia [82]. Studies on the Drosophila APP homologue APPL support a potential role in axonal arborization, axonal transport and synapse formation both during development and in the mature nervous system [69, 111, 122, 214]. APPL does not contain an Aβ-peptide sequence, suggesting that the conserved physiological function of APP does not involve Aβ. Mice deficient in APP show abnormalities in expression of synaptic markers and in axonal and dendritic arborization, together with neurological and behavioral dysfunction and impaired long-term potentiation [41, 124, 159, 188, 237]. During rat brain development, expression of APP peaks in the second postnatal week, the time of synapse formation. After development is completed, high expression levels of APP persist in the adult olfactory bulb, where continuous synaptogenesis occurs in the adult animal [114, 179], and its expression level increases in animals reared in enriched environments [89]. This all suggests an involvement of APP in the process of cell differentiation and the establishment and plastic maintenance of synaptic contacts [68].
Evidence for synaptotrophic effects of APP were, in addition, obtained by a variety of transgenic approaches generating different lines of mice overexpressing different hAPP isoforms under control of different promotors [131, 151, 171, 214]. It had not been analyzed previously, however, whether this synaptotrophic effect is maintained in mutated forms of APP that show linkage to familial forms of AD (FAD) and thus have been identified as one potential cause of the disease [64].
In a recent study, we analyzed whether familiar AD (FAD)-linked mutations of APP might impair synaptotrophic function, potentially contributing to synaptic deficiencies seen in AD. We therefore performed a quantitative electron microscopy study on synapse number in the cerebral cortex of well-characterized expression-matched transgenic mouse lines expressing either wild-type human (h)APP or FAD-mutated hAPP [152], using unbiased stereological methods. We could obtain clear evidence for a synaptotrophic effect in mice overexpressing wild-type hAPP demonstrated by an increase in synaptic number [189]. This effect was abolished when FAD-mutated APPsw,Ind was expressed instead of wild-type APP. In agreement with previous reports on reduced synaptic vesicle number in APP/APLP2 knockout mice [225, 230], we further observed increased density of synaptic vesicles in mice overexpressing wild-type hAPP. Again, this effect was abolished when FAD-mutated hAPP was expressed instead of wild-type hAPP.
Taken together, this strongly indicates a failure in synaptotrophic function of FAD-mutated forms of APP, which likely contributes to synaptic pathology in AD. Thus, at least in familiar cases of AD, not only “too much Aβ” but also “too little functional intact APP” might be of potential pathogenetic significance for synaptic loss. It remains to be determined whether a similar mechanism might be relevant also for sporadic cases of AD.
Studies on APP fragment function have demonstrated that both sAPPalpha and the APP intracellular domain may have neurotrophic properties and enhance synaptic plasticity and memory [2, 57, 91, 111, 123, 143–145, 151, 171, 198, 213]. Intracerebral administration of sAPP significantly increases synaptogenesis, reduces neuronal injury and improves functional motor outcome following brain injury in rats [17, 213], an effect that likely depends on a conserved motif in the C-terminus [111]. On the other hand, activity-dependent cleavage of APP by BACE enhances short-term and long-term synaptic plasticity. This effect correlates with elevated levels of the APP intracellular domain, which has been implicated in the regulation of gene transcription and calcium signaling [123].
Recent evidence suggests that APP and neurotrophic factors such as NGF and BDNF use similar intracellular pathways to control neuronal plasticity [3]. The low-affinity neurotrophin receptor p75 shares similarities in processing with APP. After initial cleavage by alpha secretase, p75 is cleaved by gamma secretase [234] in an event identical to the cleavage of APP. Both NGF and BDNF enhance APP promoter activity in a process that involves activation of the ras-MAP-kinase pathway [21, 174, 175, 177, 216]. As we showed previously, the ras-MAP-kinase pathway is induced already at very early stages of AD prior to any noticeable pathological alterations, such as plaque or tangle formation [60, 61]. It might, thus, be suggested to regard the activation of this signaling pathway as an, apparently ineffective, attempt to compensate for the strongly attenuated synaptotrophic effects of APP. As the ras-MAP-kinase pathway also mediates mitogenic effects eventually resulting in activation of the cell cycle (see below), a scenario can be envisaged where a disturbed synaptotrophic action of APP triggers an aberrant activation of intracellular signaling cascades that eventually induce cell cycle activation and subsequent cell death [12, 155, 217].
Synaptic disconnection in a model of cerebral hypometabolism is associated with PHF-like phosphorylation of tau
Recently, we have demonstrated that hypometabolism during topor in hibernating animals is associated with a PHF-like pattern of phosphorylation of the microtubule-asssociated protein tau, a process believed to be critically involved in the mechanism of neurofibrillary degeneration in AD [11, 74, 201, 202]. Furthermore, the stage of torpor in hibernating animals shows significant analogies to the pathophysiological condition of AD with respect to an altered synaptic connectivity [11, 125, 162, 163, 203, 218] and the impairment of cognitive function [147, 201].
Depression of the metabolic state of neurons during torpor in hibernating mammals leads to greatly reduced electroencephalographic activity [58, 103]. As activity is a measure of use, and neuronal connections remain functional through regular use, this decrease negatively affects the maintenance of neuronal connections [96]. The hibernation cycle thus represents a physiological model that allows the study of sequelae of reduced neuronal connectivity. Synaptic regression during torpor and subsequent reinnervation in phases of arousal have been particularly well characterized for mossy fibers terminating on CA3 hippocampal pyramidal neurons [11, 90, 162, 163]. Stages of synaptic disconnection are associated with the formation of PHF-like phosphorylated tau in CA3 pyramidal cells, which lose their afferentation. This PHF-like tau phosphorylation is quickly and fully reversible during arousal when mossy fibers re-connect to pyramidal neurons. These findings implicate a critical link between a dysfunction and/or loss of synaptic afferentation and PHF-like phosphorylation of tau.
A direct link between hyperphosphorylated tau and synaptic pathology is also supported by recent reports on an accumulation of abnormally phosphorylated tau species within synaptic terminals in AD brains and APP Swedish mutant transgenic mice [154, 208].
Synaptic plasticity and cell cycle activation in neurons are alternative effector pathways
To form synaptic connections and to continuously re-shape them in a process of ongoing structural adaptation, neurons must permanently withdraw from the cell cycle. That means synaptic plasticity can only occur at the expense of the ability to proliferate. In the “Dr. Jekyll and Mr. Hyde concept,” we have formulated the hypothesis that differentiated neurons after having withdrawn from the cell cycle are able to use molecular mechanisms primarily developed to control proliferation alternatively to control synaptic plasticity [5]. The existence of these alternative effector pathways within a neuron might put it at risk of erroneously converting signals derived from plastic synaptic changes into the program of cell cycle activation that subsequently leads to cell death. The molecular mechanisms involved in cell cycle activation might, thus, link synaptic plasticity to cell death [5, 6].
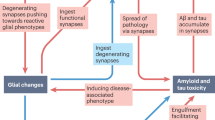
Up-regulation of a variety of molecules critically involved in the activation and progression of the cell cycle, indicating a cell cycle re-entry of neurons, occurs at early phases of neurodegeneration in AD [12, 155, 217] (Fig. 1). This cell-cycle re-activation most likely is a down-stream effect of aberrantly activated mitogenic signalling pathways [10, 60, 61]. The p21ras-MAP-kinase pathway, a mitogenic pathway that in cycling cells controls proliferation, in the adult nervous system regulates neuronal plasticity of differentiated neurons [9]. These observations provide direct evidence that depending on the cellular context, cell cycle activation and plasticity might involve identical molecular pathways. In AD, these pathways are upregulated very early during the course of the disease. This activation can be found, for example, in the frontal isocortex, as early as Braak stage I-II, i.e., prior to any other noticeable sign of pathology [10, 60, 61].
According to a hypothesis we proposed several years ago [5], neurons might have evolutionarily acquired the ability to use molecular mechanisms primarily developed to control proliferation alternatively to control synaptic plasticity. Cell cycle regulation and control of synaptic plasticity might, thus, be alternative effector pathways of “canonical” cell cycle regulating molecules. During neuronal differentiation a switch might take place from the control of the cell cycle to the control of synaptic plasticity. This switch might be reversed during degeneration, an event that might be critical for cell death
Cell cycle regulators might serve non-canonical functions in differentiated neurons: linking synaptic plasticity to cell cycle
Recent studies indicate that, contrary to classical beliefs, molecules known to be involved in activation and progression of the cell cycle are not entirely repressed in differentiated neurons in the adult nervous system where they might serve in alternative “non-canonical functions” such as regulation of neuronal and synaptic plasticity [185, 186]. A functional link between cell cycle regulation and synaptic changes potentially requires a signaling mechanism between synaptic terminals and gene regulation. Such a mechanism has indeed been identified in Drosophila, where Latheo might serve as an information shuttle between the nerve terminal and the nucleus participating in synapse-to-nucleus sigaling [52]. Latheo is present in the cytoplasm of postmitotic neurons and is also abundant in boutons of presynaptic terminals at the Drosophila neuromuscular junction, far from the nuclei [172]. It regulates both evoked transmission amplitude and activity-dependent forms of synaptic facilitation and potentiation [172] and has been implicated in learning [23, 160, 172]. Latheo is a homologue of ORC3, a component of the origin recognition complex (ORC), a critical “guard” of DNA replication that controls initiation of DNA replication and prevents re-replication during the cell cycle [88]. These data show that Latheo might play a dual neuronal role: a nuclear role in DNA replication/transcription and a role in synaptic plasticity. A similar role was shown for ORC subunits in the mammalian brain. ORC3 and ORC5 loss of function phenotypes in hippocampal pyramidal neurons induced by siRNA in mice neuronal cultures revealed a regulation of dendrite and spine development [88]. These data directly support our hypothesis that neurons have evolutionarily acquired the ability to use molecular mechanisms primarily developed to control proliferation alternatively to control synaptic plasticity [5]. More recently, we could provide evidence for an involvement of ORC units in AD pathology [8]. In AD, ORC units show pathological alterations of their subcellular compartmentation, basically reflected by a close association with neurofibrillar tau pathology in the form of neurofibrillary tangles, neuropil threads and plaque-associated dystrophic neurites. This abnormal compartmentation might segregate these regulatory elements from their physiological function in regulating plasticity and gene silencing and will, thus, potentially result in de-repression of genes triggering an apopototic phenotype.
Taken together, synaptic degeneration is the major neurobiological substrate of cognitive dysfunction in AD. Synaptic failure occurs very early in the course of the disease and progresses slowly in a process of dynamic reorganisation. Although the cause for this failure is still unknown, recent evidence indicates a link between plastic synaptic changes and control of differentiation and cell-cycle-repression within a neuron. We have thus put forward the hypothesis [5] that molecular mechanisms are shared between control of synaptic plasticity and control of the cell cycle, and as a consequence of this link, attempts to compensate for synapse loss may activate the cell cycle, which finally may lead to cell death. It will thus be the challenge for future therapeutic approaches to lock the neurons in a differentiated stage but still in a highly plastic phenotype.
References
Aoki C, Mahadomrongkul V, Fujisawa S, Habersat R, Shirao T (2007) Chemical and morphological alterations of spines within the hippocampus and entorhinal cortex precede the onset of Alzheimer’s disease pathology in double knock-in mice. J Comp Neurol 505(4):352–362
Araki W, Kitaguchi N, Tokushima Y, Ishii K, Aratake H, Shimohama S, Nakamura S, Kimura J (1991) Trophic effect of beta-amyloid precursor protein on cerebral cortical neurons in culture. Biochem Biophys Res Commun 181:265–271
Aranchio O, Chao MV (2007) Neurotrophins, synaptic plasticity and dementia. Curr Opin Neurobiol 17:1–6
Arendt T (2001) Alzheimer’s disease as a disorder of mechanisms underlaying structural brain self-organization. Commentary. Neuroscience 102:723–765
Arendt T (2003) Synaptic plasticity and cell cycle activation in neurons are alternative effector pathways. The Dr. Jekyll and Mr. Hyde theory of Alzheimer’s disease or The yin and yang of neuroplasticity. Progr Neurobiol 71:83–248
Arendt T (2008) Differentiation and de-differentiation—Neuronal cell cycle regulation during development and age-related neurodegenerative disorders. In: Lajtha A et al (eds) Handbook of Neurochemistry and Molecular Neurobiology. Springer, New York, pp 160–197
Arendt T, Bigl V, Tennstedt A, Arendt A (1985) Neuronal loss in different parts of the nucleus basalis is related to neuritic plaque formation in cortical target areas in Alzheimer’s disease. Neuroscience 14:1–14
Arendt T, Brückner MK (2007) Linking cell-cycle dysfunction in Alzheimer’s disease to a failure of synaptic plasticity. Biochim Biophys Acta 1772(4):413–421
Arendt T, Gärtner U, Seeger G, Barmashenko G, Palm K, Mittmann T, Yan L, Hümmeke M, Behrbohm J, Brückner MK, Holzer M, Wahle P, Heumann R (2004) Neuronal activation of Ras regulates synaptic connectivity. Eur J Neurosci 19:2953–2966
Arendt T, Holzer M, Großmann A, Zedlick D, Brückner MK (1995) Increased expression and subcellular translocation of the mitogen-activated protein kinase kinase and mitogen-activated protein kinase in Alzheimer’s disease. Neuroscience 68:5–18
Arendt T, Stieler J, Strijkstra AM, Hut RA, Rüdiger J, Van der Zee EA, Harkany T, Holzer M, Härtig W (2003) Reversible PHF-like phosphorylation of tau is an adaptive process associated with neuronal plasticity in hibernating animals. J Neurosci 18:6972–6981
Arendt T, Rödel L, Gärtner U, Holzer M (1996) Expression of the cyclin-dependent kinase inhibitor p16 in Alzheimer’s disease. Neuroreport 7:3047–3049
Bai F, Zhang Z, Watson DR, Yu H, Shi Y, Yuan Y, Zang Y, Zhu C, Qian Y (2008) Abnormal functional connectivity of hippocampus during episodic memory retrieval processing network in amnestic mild cognitive impairment. Biol Psychiatry. 2008 Nov 22. [Epub ahead of print]
Bain A (1872) Mind and body. The theories of their relation. D. Appleton & Company, New York
Barnes CA (1994) Normal aging: regionally specific changes in hippocampal synaptic transmission. Trends Neurosci 17:13–18
Bell KF, Bennett DA, Cuello AC (2007) Paradoxical upregulation of glutamatergic presynaptic boutons during mild cognitive impairment. J Neurosci 27:10810–10817
Bell KF, Zheng L, Fahrenholz F, Cuello AC (2008) ADAM-10 over-expression increases cortical synaptogenesis. Neurobiol Aging 29:554–565
Bertoni-Freddari C, Fattoretti P, Casoli T, Meier-Ruge W, Ulrich J (1990) Morphological adaptive response of the synaptic junctional zones in the human dentate gyrus during aging and Alzheimer’s disease. Brain Res 517:69–75
Bertoni-Freddari C, Fattoretti P, Meier-Ruge W, Ulrich J (1989) Computer-assisted morphometry of synaptic plasticity during aging and dementia. Pathol Res Pract 185:799–802
Bertoni-Freddari C, Meier-Ruge W, Ulrich J (1988) Quantitative morphology of synaptic plasticity in the aging brain. Scanning Microsc 2:1027–1034
Binnington JC, Kalisch BE (2007) Nitric oxide synthase inhibitors modulate nerve growth factor-mediated regulation of amyloid precursor protein expression in PC12 cells. J Neurochem 101:422–433
Blennow K, Bogdanovic N, Alafuzoff I, Ekman R, Davidsson P (1996) Synaptic pathology in Alzheimer’s disease: relation to severity of dementia, but not to senile plaques, neurofibrillary tangles, or the ApoE4 allele. J Neural Transm 103(5):603–618
Boynton S, Tully T (1992) latheo, a new gene involved in associative learning and memory in Drosophila melanogaster, identified from P element mutagenesis. Genetics 131:655–672
Braak H, Braak E (1991) Neuropathological staging of Alzheimer related changes. Acta Neuropathol 82:239–259
Brown DF, Risser RC, Bigio EH, Tripp P, Stiegler A, Welch E, Eagan KP, Hladik CL, White CL 3rd (1998) Neocortical synapse density and Braak stage in the Lewy body variant of Alzheimer disease: a comparison with classic Alzheimer disease and normal aging. J Neuropathol Exp Neurol 57(10):955–960
Brun A, Liu X, Erikson C (1995) Synapse loss and gliosis in the molecular layer of the cerebral cortex in Alzheimer’s disease and in frontal lobe degeneration. Neurodegeneration 4(2):171–177
Brunelli MP, Kowall NW, Lee JM, McKee AC (1991) Synaptophysin immunoreactivity is depleted in cortical laminae with dense dystrophic neurites and neurofibrillary tangles. J Neuropathol Exp Neurol 50:315
Buell SJ, Coleman PD (1979) Dendritic growth in the aged human brain and failure of growth in senile dementia. Science 206:854–856
Callahan LM, Vaules WA, Coleman PD (2002) Progressive reduction of synaptophysin message in single neurons in Alzheimer disease. J Neuropathol Exp Neurol 61(5):384–395
Callahan LM, Vaules WA, Coleman PD (1999) Quantitative decrease in synaptophysin message expression and increase in cathepsin D message expression in Alzheimer disease neurons containing neurofibrillary tangles. J Neuropathol Exp Neurol 58(3):275–287
Chang JW, Schumacher E, Coulter PM 2nd, Vinters HV, Watson JB (1997) Dendritic translocation of RC3/neurogranin mRNA in normal aging, Alzheimer disease and fronto-temporal dementia. J Neuropathol Exp Neurol 56:1105–1118
Chapman PF, White GL, Jones MW, Cooper-Blacketer D, Marshall VJ, Irizarry M, Younkin L, Good MA, Bliss TV, Hyman BT, Younkin SG, Hsiao KK (1999) Impaired synaptic plasticity and learning in aged amyloid precursor protein transgenic mice. Nat Neurosci 2:271–276
Clinton J, Blackman SE, Royston MC, Roberts GW (1994) Differential synaptic loss in the cortex in Alzheimer’s disease: a study using archival material. Neuroreport 12; 5(4):497–500
Coleman PD, Yao PJ (2003) Synaptic slaughter in Alzheimer’s disease. Neurobiol Aging 24:1023–1027
Cooke SF, Bliss TV (2006) Plasticity in the human central nervous system. Brain 129:1659–1673
Counts SE, Nadeem M, Lad SP, Wuu J, Mufson EJ (2006) Differential expression of synaptic proteins in the frontal and temporal cortex of elderly subjects with mild cognitive impairment. J Neuropathol Exp Neurol 65(6):592–601
Daigle I, Li C (1993) apl-1, a Caenorhabditis elegans gene encoding a protein related to the human beta-amyloid protein precursor. PNAS 90:12045–12049
Davidsson P, Blennow K (1998) Neurochemical dissection of synaptic pathology in Alzheimer’s disease. Int Psychogeriatr 10(1):11–23
Davidsson P, Jahn R, Bergquist J, Ekman R, Blennow K (1996) Synaptotagmin, a synaptic vesicle protein, is present in human cerebrospinal fluid: a new biochemical marker for synaptic pathology in Alzheimer disease? Mol Chem Neuropathol 27(2):195–210
Davies CA, Mann DM, Sumpter PQ, Yates PO (1987) A quantitative morphometric analysis of the neuronal and synaptic content of the frontal and temporal cortex in patients with Alzheimer’s disease. J Neurol Sci 78:151–164
Dawson GR, Seabrook GR, Zheng H, Smith DW, Graham S, O’Dowd G, Bowery BJ, Boyce S, Trumbauer ME, Chen HY, Van der Ploeg LH, Sirinathsinghji DJ (1999) Age-related cognitive deficits, impaired long-term potention and reduction in synaptic marker density in mice lacking the beta-amyloid precursor protein. Neurosci 90:1–13
DeKosky ST, Harbaugh RE, Schmitt FA, Bakay RA, Chui HC, Knopman DS, Reeder TM, Shetter AG, Senter HJ, Markesbery WR (1992) Cortical biopsy in Alzheimer’s disease: diagnostic accuracy and neurochemical, neuropathological, and cognitive correlations. Intraventricular Bethanecol Study Group. Ann Neurol 32(5):625–632
DeKosky ST, Scheff SW, Styren SD (1996) Structural correlates of cognition in dementia: quantification and assessment of synapse change. Neurodegeneration 5(4):417–421 Review
DeKosky ST, Scheff SW (1990) Synapse loss in frontal cortex biopsies in Alzheimer’s disease: correlation with cognitive severity. Ann Neurol 27(5):457–464
Demoor J (1896) La plasticité morphologique des neurones cérébraux. Archives de Biologie 14:723–749
Dessi F, Colle MA, Hauw JJ, Duyckaerts C (1997) Accumulation of SNAP-25 immunoreactive material in axons of Alzheimer’s disease. Neuroreport 8:3685–3689
Dickson DW, Crystal HA, Bevona C, Honer W, Vincent I, Davies P (1995) Correlations of synaptic and pathological markers with cognition of the elderly. Neurobiol Aging 16(3):285–298 discussion 298-304
Dinamarca MC, Colombres M, Cerpa W, Bonansco C, Inestrosa NC (2008) Beta-amyloid oligomers affect the structure and function of the postsynaptic region: role of the Wnt signaling pathway. Neurodegener Dis 5:149–152
Duval M (1895) Hypothèses sur la physiologie des centres nerveux; théorie histologique du sommeil. Comptes Rendus Hebdomadaires des Séances et Mémoires de la Société de Biologie 47:74–77
Duval M (1900) Les neurones. L’amiboïsme nerveux la théorie histologique du sommeil. Revue de l’Ècole d’Anthropologie de Paris 10:37–71
Espinosa B, Zenteno R, Mena R, Robitaille Y, Zenteno E, Guevara J (2001) O-Glycosylation in sprouting neurons in Alzheimer disease, indicating reactive plasticity. J Neuropathol Exp Neurol 60:441–448
Featherstone DE, Broadie K (2000) Surprises from Drosophila: genetic mechanisms of synaptic development and plasticity. Brain Res Bull 53(5):501–511
Fernandes MA, Proenca MT, Nogueira AJ, Oliveira LM, Santiago B, Santana I, Oliveira CR (1999) Effects of apolipoprotein E genotype on blood lipid composition and membrane platelet fluidity in Alzheimer’s disease. Biochem Biophys Acta 1454:89–96
Fitzjohn SM, Morton RA, Kuenzi F, Rosahl TW, Shearman M, Lewis H, Smith D, Reynolds DS, Davies CH, Collingridge GL, Seabrook GR (2001) Age-related impairment of synaptic transmission but normal long-term potentiation in transgenic mice that overexpress the human APP695SWE mutant form of amyloid precursor protein. J Neurosci 21:4691–4698
Flood DG, Coleman PD (1986) Failed compensatory dendritic growth as a pathophysiological process in Alzheimer’s disease. Can J Neurol Sci 13(Suppl. 4):475–479
Fukumoto H, Cheung BS, Hyman BT, Irizarry MC (2002) Beta-secretase protein and activity are increased in the neocortex in Alzheimer disease. Arch Neurol 59(9):1381–1389
Furukawa K, Sopher BL, Rydel RE, Begley JG, Pham DG, Martin GM, Fox M, Mattson MP (1996) Increased activity-regulating and neuroprotective efficacy of alpha-secretase-derived secreted amyloid precursor protein conferred by a C-terminal heparin-binding domain. J Neurochem 67:1882–1896
Gabriel A, Klussmann FW, Igelmund P (1998) Rapid temperature changes induce adenosine-mediated depression of synaptic transmission in hippocampal slices from rats (non-hibernators). Neuroscience 86:67–77
Gabriel SM, Haroutunian V, Powchik P, Honer WG, Davidson M, Davies P, Davis KL (1997) Increased concentrations of presynaptic proteins in the cingulate cortex of subjects with schizophrenia. Arch Gen Psychiatry 54(6):559–66. Erratum in: Arch Gen Psychiatry 1997 54(10):912
Gärtner U, Holzer M, Arendt T (1999) Elevated expression of p21ras is an early event in Alzheimer’s disease and precedes neurofibrillary degeneration. Neuroscience 91:1–5
Gärtner U, Holzer M, Heumann R, Arendt T (1995) Induction of p21ras in Alzheimer pathology. Neuroreport 6:1441–1444
Giacchino J, Criado JR, Games D, Henriksen S (2000) In vivo synaptic transmission in young and aged amyloid precursor protein transgenic mice. Brain Res 876:185–190
Gibson PH (1983) EM study of the numbers of cortical synapses in the brains of ageing people and people with Alzheimer-type dementia. Acta Neuropathol (Berl) 62:127–133
Goate A, Chartier-Harlin MC, Mullan M, Brown J, Crawford F, Fidani L, Giuffra L, Haynes A, Irving N, James L, Mant R, Newton P, Rooke K, Roques P, Tabot C, Williamson R, Rossor M, Hardy J (1991) Segregation of a missense mutation in the amyloid precursor protein gene with familial Alzheimer’s disease. Nature 349:704–706
Götz J, Ittner LM, Kins S (2006) Do axonal defects in tau and amyloid precursor protein transgenic animals model axonopathy in Alzheimer’s disease? J Neurochem 98:993–1006
Gonatas NK, Anderson W, Evangelista I (1967) The contribution of altered synapses in the senile plaque: an electron microscopic study in Alzheimer’s dementia. J Neuropathol. Exp Neurol 26:25–39
Gottfries CG, Karlsson I, Svennerholm L (1996) Membrane components separate early-onset Alzheimer’s disease from senile dementia of the Alzheimer type. Int Psychogeriatr 8:365–372
Gralle M, Ferreira ST (2007) Structure and functions of the human amyloid precursor protein: the whole is more than the sum of its parts. Progr Neurobiol 82:11–32
Gunawardena S, Goldstein LS (2001) Disruption of axonal transport and neuronal viability by amyloid precursor protein mutations in Drosophila. Neuron 32:389–401
Haass C, Schlossmacher MG, Hung AY, Vigo-Pelfrey C, Mellon A, Ostaszewski BL, Lieberburg I, Koo EH, Schenk D, Teplow DB, Selkoe DJ (1992) Amyloid beta-peptide is produced by cultured cells during normal metabolism. Nature 359:322–325
Haass C, Selkoe DJ (2007) Soluble protein oligomers in neurodegeneration: lessons from the Alzheimer’s amyloid beta-peptide. Nat Rev Mol Cell Biol 8:101–112
Hamos JE, DeGennaro LJ, Drachman DA (1989) Synaptic loss in Alzheimer’s disease and other dementias. Neurol 39:355–361
Hansen LA, Daniel SE, Wilcock GK, Love S (1998) Frontal cortical synaptophysin in Lewy body diseases: relation to Alzheimer’s disease and dementia. J Neurol Neurosurg Psychiatry 64:653–656
Härtig W, Stieler J, Boerema AS, Wolf J, Schmidt U, Weißfuß J, Bullmann T, Strijkstra AM, Arendt T (2007) Hamster model of tau hyperphosphorylation: selective vulnerability of cholinergic forebrain neurons during hibernation—implications for Alzheimer’s disease. Eur J Neurosci 25:69–80
Harigaya Y, Shoji M, Shirao T, Hirai S (1996) Disappearance of actin-binding protein, drebrin, from hippocampal synapses in Alzheimer’s disease. J Neurosci Res 43:87–92
Hasselmo ME (1997) A computational model of the progression of Alzheimer’s disease. MD Comput 14:181–191
Hatanpää K, Isaacs KR, Shirao T, Brady DR, Rapoport SI (1999) Loss of proteins regulating synaptic plasticity in normal aging of the human brain and in Alzheimer disease. J Neuropathol Exp Neurol 58:637–643
Heber S, Herms J, Gajic V, Hainfellner J, Aguzzi A, Rülicke T, von Kretzschmar H, von Koch C, Sisodia S, Tremml P, Lipp HP, Wolfer DP, Müller U (2000) Mice with combined gene knock-outs reveal essential and partially redundant functions of amyloid precursor protein family members. J Neurosci 20:7951–7963
Heffernan JM, Eastwood SL, Nagy Z, Sanders MW, McDonald B, Harrison PJ (1998) Temporal cortex synaptophysin mRNA is reduced in Alzheimer’s disease and is negatively correlated with the severity of dementia. Exp Neurol 150(2):235–239
Heinonen O, Lehtovirta M, Soininen H, Helisalmi S, Mannermaa A, Sorvari H, Kosunen O, Paljärvi L, Ryynänen M, Riekkinen PJ Sr (1995) Alzheimer pathology of patients carrying apolipoprotein E epsilon 4 allele. Neurobiol Aging 16(4):505–513
Heinonen O, Soininen H, Sorvari H, Kosunen O, Paljärvi L, Koivisto E, Riekkinen PJ Sr (1995) Loss of synaptophysin-like immunoreactivity in the hippocampal formation is an early phenomenon in Alzheimer’s disease. Neuroscience 64(2):375–384
Herms J, Anliker B, Heber S, Ring S, Fuhrmann M, Kretzschmar H, Sisodia S, Müller U (2004) Cortical dysplasia resembling human type 2 lissencephaly in mice lacking all three APP family members. EMBO J 23:4106–4115
Honer WG, Dickson DW, Gleeson J, Davies P (1992) Regional synaptic pathology in Alzheimer’s disease. Neurobiol Aging 13(3):375–382
Honer WG (2003) Pathology of presynaptic proteins in Alzheimer’s disease: more than simple loss of terminals. Neurobiol Aging 24:1047–1062
Horn D, Levy N, Ruppin E (1996) Neuronal-based synaptic compensation: a computational study in Alzheimer’s disease. Neural Comput 8:1227–1243
Horwitz B, Grady CL, Schlageter NL, Duara R, Rapoport SI (1987) Intercorrelations of regional cerebral glucose metabolic rates in Alzheimer’s disease. Brain Res 407:294–306
Hu NW, Smith IM, Walsh DM, Rowan MJ (2008) Soluble amyloid-beta peptides potently disrupt hippocampal synaptic plasticity in the absence of cerebrovascular dysfunction in vivo. Brain 131:2414–2424
Huang Z, Zang K, Reichardt LF (2005) The origin recognition core complex regulates dendrite and spine development in postmitotic neurons. J Cell Biol 170:527–535
Huber G, Bailly Y, Martin JR, Mariani J, Brugg B (1997) Synaptic beta-amyloid precursor proteins increase with learning capacity in rats. Neurosci 80:313–320
Hut RA, de Wilde MC, Strijkstra AM, Van der Zee EA, Daan S (2001) Neuronal changes in the hippocampus and SCN of hibernating ground squirrels. Soc Neurosci Abstr 27:535.11
Ishida A, Furukawa K, Keller JN, Mattson MP (1997) Secreted form of beta-amyloid precursor protein shifts the frequency dependency for induction of LTD, and enhances LTP in hippocampal slices. Neuroreport 8:2133–2137
Johnson SC, Schmitz TW, Moritz CH, Meyerand ME, Rowley HA, Alexander AL, Hansen KW, Gleason CE, Carlsson CM, Ries ML, Asthana S, Chen K, Reiman EM, Alexander GE (2006) Activation of brain regions vulnerable to Alzheimer’s disease: the effect of mild cognitive impairment. Neurobiol Aging 27:1604–1612
Jorgensen OS, Balázs R (1993) Plastic neuronal changes in Alzheimer’s disease associated with activation of astrocytes and enhanced neurotrophic activity. In: Corain B, Iqbal K, Nicolini M, Winblad B, Wisniewski H, Zatta P (eds) Alzheimer’s disease: advances in clinical and basic research. Wiley, Chichester, pp 189–198
Katzman R (1986) Alzheimer’s disease. N Engl J Med 314:964–973
Kaufmann WA, Barnas U, Humpel C, Nowakowski K, DeCol C, Gurka P, Ransmayr G, Hinterhuber H, Winkler H, Marksteiner J (1998) Synaptic loss reflected by secretoneurin-like immunoreactivity in the human hippocampus in Alzheimer’s disease. Eur J Neurosci 10(3):1084–1094
Kavanau JL (1997) Memory, sleep and the evolution of mechanisms of synaptic efficacy maintenance. Neuroscience 79:7–44
Kirazov E, Kirazov L, Bigl V, Schliebs R (2001) Ontogenetic changes in protein level of amyloid precursor protein (APP) in growth cones and synaptosomes from rat brain and prenatal expression pattern of APP mRNA isoforms in developing rat embryo. Int J Dev Neurosci 19:287–296
Klyubin I, Walsh DM, Cullen WK, Fadeeva JV, Anwyl R, Selkoe DJ, Rowan MJ (2004) Soluble Arctic amyloid beta protein inhibits hippocampal long-term potentiation in vivo. Eur J Neurosci 19:2839–2846
Klyubin I, Walsh DM, Lemere CA, Cullen WK, Shankar GM, Betts V, Spooner ET, Jiang L, Anwyl R, Selkoe DJ, Rowan MJ (2005) Amyloid beta protein immunotherapy neutralizes Abeta oligomers that disrupt synaptic plasticity in vivo. Nat Med 11:556–561
Koffie RM, Meyer-Luehmann M, Hashimoto T, Adams KW, Mielke ML, Garcia-Alloza M, Micheva KD, Smith SJ, Kim ML, Lee VM, Hyman BT, Spires-Jones TL (2009) Oligomeric amyloid beta associates with postsynaptic densities and correlates with excitatory synapse loss near senile plaques. Proc Natl Acad Sci USA 106(10):4012–4017
Koo EH, Sisodia SS, Archer DR, Martin LJ, Weidemann A, Beyreuther K, Fischer P, Masters CL, Price DL (1990) Precursor of amyloid protein in Alzheimer disease undergoes fast anterograde axonal transport. PNAS 87:1561–1565
Korade Z, Kenworthy AK (2008) Lipid rafts, cholesterol, and the brain. Neuropharmacology 55(8):1265–1273
Krilowicz BL, Glotzbach SF, Heller HC (1988) Neuronal activity during sleep and complete bouts of hibernation. Am J Physiol 255:R1008–R1019
Lambert MP, Barlow AK, Chromy BA, Edwards C, Freed R, Liosatos M, Morgan TE, Rozovsky I, Trommer B, Viola KL, Wals P, Zhang C, Finch CE, Krafft GA, Klein WL (1998) Diffusible, nonfibrillar ligands derived from Abeta1–42 are potent central nervous system neurotoxins. Proc Natl Acad Sci USA 95:6448–6453
Lassmann H, Fischer P, Jellinger K (1993) Synaptic pathology of Alzheimer’s disease. Ann N Y Acad Sci 695:59–64
Lassmann H, Weiler R, Fischer P, Bancher C, Jellinger K, Floor E, Danielczyk W, Seitelberger F, Winkler H (1992) Synaptic pathology in Alzheimer’s disease: immunological data for markers of synaptic and large dense-core vesicles. Neuroscience 46:1–8
Lechner T, Adlassnig C, Humpel C, Kaufmann WA, Maier H, Reinstadler-Kramer K, Hinterhölzl J, Mahata SK, Jellinger KA, Marksteiner J (2004) Chromogranin peptides in Alzheimer’s disease. Exp Gerontol 39(1):101–113
Lépine MR (1895) Théorie mécanique de la paralysie hystérique, du somnambulisme, du sommeil naturel et de la distraction. Soc Biol 5:85–86
Leuba G, Savioz A, Vernay A, Carnal B, Kraftsik R, Tardif E, Riederer I, Riederer BM (2008) Differential changes in synaptic proteins in the Alzheimer frontal cortex with marked increase in PSD-95 postsynaptic protein. J Alzheimers Dis 15:139–151
Lewis DA, Campbell MJ, Terry RD, Morrison JH (1987) Laminar and regional distributions of neurofibrillary tangles and neuritic plaques in Alzheimer’s disease: a quantitative study of visual and auditory cortices. J Neurosci 7:1799–1808
Leyssen M, Ayaz D, Hébert SS, Reeve S, De Strooper B, Hassan BA (2005) Amyloid precursor protein promotes post-developmental neurite arborization in the Drosophila brain. EMBO J 24:2944–2955
Lippa CF, Hamos JE, Pulaski-Salo D, DeGennaro LJ, Drachman DA (1992) Alzheimer’s disease and aging: effects on perforant pathway perikarya and synapses. Neurobiol Aging 13:405–411
Liu X, Erikson C, Brun A (1996) Cortical synaptic changes and gliosis in normal aging, Alzheimer’s disease and frontal lobe degeneration. Dementia 7(3):128–134
Löffler J, Huber G (1992) β-amyloid precirsor protein isoforms in various rat brain regions and during development. J. Neurochem 59:1316–1324
López-Sánchez N, Müller U, Frade JM (2005) Lengthening of G2/mitosis in cortical precursors from mice lacking β-amyloid precursor protein. Neurosci 130:51–60
Lubec G, Nonaka M, Krapfenbauer K, Gratzer M, Cairns N, Fountoulakis M (1999) Expression of the dihydropyrimidinase related protein 2 (DRP-2) in Down syndrome and Alzheimer’s disease brain is downregulated at the mRNA and dysregulated at the protein level. J Neural Transm, Suppl. 57:161–177
Lue LF, Brachova L, Civin WH, Rogers J (1996) Inflammation, A beta deposition, and neurofibrillary tangle formation as correlates of Alzheimer’s disease neurodegeneration. J Neuropathol Exp Neurol 55(10):1083–1088
Lugaro E (1895) Sulle Modificazioni delle cellule nervose. Lo Sperimentale 49:159–193
Lugaro E (1898) Sulle modificazioni morfologiche funzionali die dendriti delle cellule nervose. Rivista di Patologia Nervosa e Mentale 3:337–359
Lugaro E (1899) I recenti progressi dell’anatomia del sistema nervoso in rapporto alla psicologia ed alla psichiatria. Rivista di Patologia Nervosa e Mentale 4:481–514
Lugaro E (1900) I recenti progressi dell’anatomia del sistema nervoso in rapporto alla psicologia ed alla psichiatria. Rivista Sperimentale di Freniatria e Medicina Legale delle Alienazioni Mentali 26:831–894
Luo LQ, Martin-Morris LE, White K (1990) Identification, secretion, and neural expression of APPL, a Drosophila protein similar to human amyloid protein precursor. J Neurosci 10:3849–3861
Ma H, Lesné S, Kotilinek L, Steidl-Nichols JV, Sherman M, Younkin L, Younkin S, Forster C, Sergeant N, Delacourte A, Vassar R, Citron M, Kofuji P, Boland LM, Ashe KH (2007) Involvement of beta-site APP cleaving enzyme 1 (BACE1) in amyloid precursor protein-mediated enhancement of memory and activity-dependent synaptic plasticity. PNAS 104:8167–8172
Magara F, Müller U, Li ZW, Lipp HP, Weissmann C, Stagljar M, Wolfer DP (1999) Genetic background changes the pattern of forebrain commissure defects in transgenic mice underexpressing the beta-amyloid-precursor protein. PNAS 96:4656–4661
Magariños AM, McEwen BS, Saboureau M, Pevet P (2006) Rapid and reversible changes in intrahippocampal connectivity during the course of hibernation in European hamsters. Proc Natl Acad Sci USA 103:18775–18780
Majocha RE, Jungalwala FB, Rodenrys A, Marotta CA (1989) Monoclonal antibody to embryonic CNS antigen A2B5 provides evidence for the involvement of membrane components at sites of Alzheimer degeneration and detects sulfatides as well as gangliosides. J Neurochem 53:953–961
Marksteiner J, Kaufmann WA, Gurka P, Humpel C (2002) Synaptic proteins in Alzheimer’s disease. J Mol Neurosci 18(1–2):53–63
Marksteiner J, Lechner T, Kaufmann WA, Gurka P, Humpel C, Nowakowski C, Maier H, Jellinger KA (2000) Distribution of chromogranin B-like immunoreactivity in the human hippocampus and its changes in Alzheimer’s disease. Acta Neuropathol 100(2):205–212
Martin-Morris LE, White K (1990) The Drosophila transcript encoded by the beta-amyloid protein precursor-like gene is restricted to the nervous system. Development 110:185–195
Masliah E (1995) Mechanisms of synaptic dysfunction in Alzheimer’s disease. Histol Histopathol 10:509–519
Masliah E, Alford M, DeTeresa R, Mallory M, Hansen L (1996) Deficient glutamate transport is associated with neurodegeneration in Alzheimer’s disease. Ann Neurol 40(5):759–766
Masliah E, Ellisman M, Carragher B, Mallory M, Young S, Hansen L, DeTeresa R, Terry RD (1992) Three-dimensional analysis of the relationship between synaptic pathology and neuropil threads in Alzheimer disease. J Neuropathol Exp Neurol 51(4):404–414
Masliah E, Mallory M, Alford M, DeTeresa R, Hansen LA, McKeel DW Jr, Morris JC (2001) Altered expression of synaptic proteins occurs early during progression of Alzheimer’s disease. Neurol 56:127–129
Masliah E, Mallory M, Ge N, Alford M, Veinbergs I, Roses AD (1995) Neurodegeneration in the central nervous system of apoE-deficient mice. Exp Neurol 136(2):107–122
Masliah E, Mallory M, Hansen L, DeTeresa R, Alford M, Terry R (1994) Synaptic and neuritic alterations during the progression of Alzheimer’s disease. Neurosci Lett 174(1):67–72
Masliah E, Mallory M, Hansen L, DeTeresa R, Terry RD (1993) Quantitative synaptic alterations in the human neocortex during normal aging. Neurology 43:192–197
Masliah E, Terry R (1993) The role of synaptic proteins in the pathogenesis of disorders of the central nervous system. Brain Pathol 3(1):77–85
Masliah E, Terry R (1994) The role of synaptic pathology in the mechanisms of dementia in Alzheimer’s disease. Clin Neurosci 1:192–198
Masliah E, Terry RD, Alford M, DeTeresa R, Hansen LA (1991) Cortical and subcortical patterns of synaptophysinlike immunoreactivity in Alzheimer’s disease. Am J Pathol 138(1):235–246
Masliah E, Terry RD, DeTeresa RM, Hansen LA (1989) Immunohistochemical quantification of the synapse-related protein synaptophysin in Alzheimer disease. Neurosci Lett 103(2):234–239
Masliah E, Terry RD, Mallory M, Alford M, Hansen LA (1990) Diffuse plaques do not accentuate synapse loss in Alzheimer’s disease. Am J Pathol 137(6):1293–1297
Masters CL, Simms G, Weinman NA, Multhaup G, McDonald BL, Beyreuther K (1985) Amyloid plaque core protein in Alzheimer disease and Down syndrome. PNAS 82:4245–4249
Mattson MP, Cheng B, Culwell AR, Esch FS, Lieberburg I, Rydel RE (1993) Evidence for excitoprotective and intraneuronal calcium-regulating roles for secreted forms of the beta-amyloid precursor protein. Neuron 10:243–254
Mattson MP (1994) Secreted forms of beta-amyloid precursor protein modulate dendrite outgrowth and calcium responses to glutamate in cultured embryonic hippocampal neurons. J Neurobiol 25:439–450
Meziane H, Dodart JC, Mathis C, Little S, Clemens J, Paul SM, Ungerer A (1998) Memory-enhancing effects of secreted forms of the beta-amyloid precursor protein in normal and amnestic mice. Proc Natl Acad Sci USA 95:12683–12688
Mikkonen M, Soininen H, Tapiola T, Alafuzoff I, Miettinen R (1999) Hippocampal plasticity in Alzheimer’s disease: changes in highly polysialylated NCAM immunoreactivity in the hippocampal formation. Eur J Neurosci 11:1754–1764
Millesi E, Prossinger H, Dittami JP, Fieder M (2001) Hibernation effects on memory in European ground squirrels (Spermophilus citellus). J Biol Rhythm 16:264–271
Milward EA, Papadopoulos R, Fuller SJ, Moir RD, Small D, Beyreuther K, Masters CL (1992) The amyloid protein precursor of Alzheimer’s disease is a mediator of the effects of nerve growth factor on neurite outgrowth. Neuron 9:129–137
Minger SL, Honer WG, Esiri MM, McDonald B, Keene J, Nicoll JA, Carter J, Hope T, Francis PT (2001) Synaptic pathology in prefrontal cortex is present only with severe dementia in Alzheimer disease. J Neuropathol Exp Neurol 60(10):929–936
Morrison JH, Hof PR, Campell MJ, DeLima AD, Voigt T, Bouras C, Cox K, Young WG (1990) Cellular pathology in Alzheimer’s disease: implications for corticocortical disconnection and differential vulnerability. In: Rapoport SI, Petit H, Leys D, Christen Y (eds) Imaging, Cerebral Topography and Alzheimer’s disease. Research and Perspectives in Alzheimer’s disease. Springer, Berlin, pp 19–40
Mucke L, Masliah E, Johnson WB, Ruppe MD, Alford M, Rockenstein EM, Forss-Petter S, Pietropaolo M, Mallory M, Abraham CR (1994) Synaptotrophic effects of human amyloid beta protein precursors in the cortex of transgenic mice. Brain Res 666:151–167
Mucke L, Masliah E, Yu GQ, Mallory M, Rockenstein EM, Tatsuno G, Hu K, Kholodenko D, Johnson-Wood K, McConlogue L (2000) High-level neuronal expression of Aβ1–42 in wild-type human amyloid protein precursor transgenic mice: synaptotoxicity without plaque formation. J Neurosci 20:4050–4058
Mukaetova-Ladinska EB, Garcia-Siera F, Hurt J, Gertz HJ, Xuereb JH, Hills R, Brayne C, Huppert FA, Paykel ES, McGee M, Jakes R, Honer WG, Harrington CR, Wischik CM (2000) Staging of cytoskeletal and beta-amyloid changes in human isocortex reveals biphasic synaptic protein response during progression of Alzheimer’s disease. Am J Pathol 157(2):623–636
Muntané G, Dalfó E, Martinez A, Ferrer I (2008) Phosphorylation of tau and alpha-synuclein in synaptic-enriched fractions of the frontal cortex in Alzheimer’s disease, and in Parkinson’s disease and related alpha-synucleinopathies. Neuroscience 152:913–923
Nagy Z, Esiri MM, Smith AD (1997) Expression of cell division markers in the hippocampus in Alzheimer’s disease and other neurodegenerative conditions. Acta Neuropathol 93:294–300
Origlia N, Righi M, Capsoni S, Cattaneo A, Fang F, Stern DM, Chen JX, Schmidt AM, Arancio O, Yan SD, Domenici L (2008) Receptor for advanced glycation end product-dependent activation of p38 mitogen-activated protein kinase contributes to amyloid-beta-mediated cortical synaptic dysfunction. J Neurosci 28:3521–3530
Perdahl E, Adolfsson R, Alafuzoff I, Albert KA, Nestler EJ, Greengard P, Winblad B (1984) Synapsin I (protein I) in different brain regions in senile dementia of Alzheimer type and in multi-infarct dementia. J Neural Transm 60(2):133–141
Perry EK, Tomlinson BE, Blessed G, Bergmann K, Gibson PH, Perry RH (1978) Correlation of cholinergic abnormalities with senile plaques and mental test scores in senile dementia. Br Med J 25;2(6150):1457–1459
Phinney AL, Calhoun ME, Wolfer DP, Lipp HP, Zheng H, Jucker M (1999) No hippocampal neuron or synaptic bouton loss in learning-impaired aged beta-amyloid precursor protein-null mice. Neurosci 90:1207–1216
Pinto S, Quintana DG, Smith P, Mihalek RM, Hou ZH, Boynton S, Jones CJ, Hendricks M, Velinzon K, Wohlschlegel JA, Austin RJ, Lane WS, Tully T, Dutta A (1999) latheo encodes a subunit of the origin recognition complex and disrupts neuronal proliferation and adult olfactory memory when mutant. Neuron 23:45–54
Plant LD, Boyle JP, Smith IF, Peers C, Pearson HA (2003) The production of amyloid beta peptide is a critical requirement for the viability of central neurons. J Neurosci 23:5531–5535
Popov VI, Bocharova LS (1992) Hibernation-induced structural changes in synaptic contracts between mossy fibres and hippocampal neurons. Neuroscience 48:53–62
Popov VL, Bocharova LS, Bragin AG (1992) Repeated changes of dendritic morphology in the hippocampus of ground squirrels in the course of hibernation. Neuroscience 48:45–51
Puzzo D, Privitera L, Leznik E, Fà M, Staniszewski A, Palmeri A, Arancio O (2008) Picomolar amyloid-beta positively modulates synaptic plasticity and memory in hippocampus. J Neurosci 28:14537–14545
Rabl-Rückhard (1890) Sind die Ganglienzellen amöboid? Eine Hypothese zur Mechanik psychischer Vorgänge. Neurologisches Centralblatt 9:199–200
Ramón y Cajal S (1894) The Croonian lecture. La fine structure des centres nerveux. Proceedings of the Royal Society of London. vol. LV. Harrison and Sons, London, pp 444–468
Ramón y Cajal S (1911) Histologie du systeme nerveux. A. Maloine. Paris. (This is the second edition, in French, of the Textura del sistema nervioso del hombre y los vertebrados of 1899. Quotations here are from the English translation by N. and L. Swanson, vol 1. Oxford University Press 1995, New York
Ramón y Cajal S (1928) Degeneration and regeneration of the nervous system. Oxford University Press, London
Rapoport SI (1999) In vivo PET imaging and postmortem studies suggest potentially reversible and irreversible stages of brain metabolic failure in Alzheimer’s disease. Eur Arch Psychiatry Clin Neurosci 249(Suppl 3):46–55
Reddy PH, Mani G, Park BS, Jacques J, Murdoch G, Whetsell W Jr, Kaye J, Manczak M (2005) Differential loss of synaptic proteins in Alzheimer’s disease: implications for synaptic dysfunction. J Alzheimers Dis 7:103–117
Roch JM, Masliah E, Roch-Levecq AC, Sundsmo MP, Otero DA, Veinbergs I, Saitoh T (1994) Increase of synaptic density and memory retention by a peptide representing the trophic domain of the amyloid beta/A4 protein precursor. PNAS 91:7450–7454
Rohrbough J, Pinto S, Mihalek RM, Tully T, Broadie K (1999) latheo, a Drosophila gene involved in learning, regulates functional synaptic plasticity. Neuron 23:55–70
Rosen DR, Martin-Morris L, Luo LQ, White K (1989) A Drosophila gene encoding a protein resembling the human beta-amyloid protein precursor. PNAS 86:2478–2482
Rossner S, Ueberham U, Schliebs R, Perez-Polo JR, Bigl V (1998) p75 and TrkA receptor signaling independently regulate amyloid precursor protein mRNA expression, isoform composition, and protein secretion in PC12 cells. J.Neurochem 71:757–766
Rossner S, Ueberham U, Schliebs R, Perez-Polo JR, Bigl V (1998) The regulation of amyloid precursor protein metabolism by cholinergic mechanisms and neurotrophin receptor signaling. Progr Neurobiol 56:541–569
Rowan MJ, Klyubin I, Wang Q, Hu NW, Anwyl R (2007) Synaptic memory mechanisms: Alzheimer’s disease amyloid beta-peptide-induced dysfunction. Biochem Soc Trans 35:1219–1223
Ruiz-León Y, Pascual A (2004) Regulation of beta-amyloid precursor protein expression by brain-derived neurotrophic factor involves activation of both the Ras and phosphatidylinositide 3-kinase signalling pathways. J Neurochem 88:1010–1018
Sabo SL, Ikin AF, Buxbaum JD, Greengard P (2003) The amyloid precursor protein and its regulatory protein, FE65, in growth cones and synapses in vitro and in vivo. J Neurosci 23:5407–5415
Salbaum JM, Ruddle FH (1994) Embryonic expression pattern of amyloid protein precursor suggests a role in differentiation of specific subsets of neurons. J Exp Zool 269:116–127
Samuel W, Alford M, Hofstetter CR, Hansen L (1997) Dementia with Lewy bodies versus pure Alzheimer disease: differences in cognition, neuropathology, cholinergic dysfunction, and synapse density. J Neuropathol Exp Neurol 56(5):499–508
Samuel W, Terry RD, DeTeresa R, Butters N, Masliah E (1994) Clinical correlates of cortical and nucleus basalis pathology in Alzheimer dementia. Arch Neurol 51(8):772–778
Scheff SW, DeKosky ST, Price DA (1990) Quantitative assessment of cortical synaptic density in Alzheimer’s disease. Neurobiol Aging 11(1):29–37
Scheff SW, Price DA, Schmitt FA, DeKosky ST, Mufson EJ (2007) Synaptic alterations in CA1 in mild Alzheimer disease and mild cognitive impairment. Neurology 68:1501–1508
Scheff SW, Price DA, Schmitt FA, Mufson EJ (2006) Hippocampal synaptic loss in early Alzheimer’s disease and mild cognitive impairment. Neurobiol Aging 27(10):1372–1384
Schmetsdorf S, Gärtner U, Arendt T (2005) Expression of cell cycle-related proteins in developing and adult mouse hippocampus. Int J Dev Neurosci 23:101–112
Schmetsdorf S, Gärtner U, Arendt T (2007) Constitutive expression of functionally active cyclin-dependent kinases and their binding partners suggests noncanonical functions of cell cycle regulators in differentiated neurons. Cereb. Cortex 17(8):1821–1829
Schubert W, Prior R, Weidemann A, Dircksen H, Multhaup G, Masters CL, Beyreuther K (1991) Localization of Alzheimer beta A4 amyloid precursor protein at central and peripheral synaptic sites. Brain Res 563:184–194
Seabrook GR, Smith DW, Bowery BJ, Easter A, Reynolds T, Fitzjohn SM, Morton RA, Zheng H, Dawson GR, Sirinathsinghji DJ, Davies CH, Collingridge GL, Hill RG (1999) Mechanisms contributing to the deficits in hippocampal synaptic plasticity in mice lacking amyloid precursor protein. Neuropharmacology 38:349–359
Seeger G, Gärtner U, Ueberham U, Rohn S, Arendt T (2009) FAD-mutation of APP is associated with a loss of its synaptotrophic activity. Neurobiology of Disease (under revision)
Selkoe DJ (2008) Soluble oligomers of the amyloid beta-protein impair synaptic plasticity and behavior. Behav Brain Res 192:106–113
Selkoe DJ (1994) Normal and abnormal biology of the beta-amyloid precursor protein. Annu Rev Neurosci 17:489–517
Shankar GM, Li S, Mehta TH, Garcia-Munoz A, Shepardson NE, Smith I, Brett FM, Farrell MA, Rowan MJ, Lemere CA, Regan CM, Walsh DM, Sabatini BL, Selkoe DJ (2008) Amyloid-beta protein dimers isolated directly from Alzheimer’s brains impair synaptic plasticity and memory. Nat Med 14:837–842
Shigematsu K, McGeer PL, McGeer EG (1992) Localization of amyloid precursor protein in selective postsynaptic densities of rat cortical neurons. Brain Res 592:353–357
Shimohama S, Fujimoto S, Sumida Y, Akagawa K, Shirao T, Matsuoka Y, Taniguchi T (1998) Differential expression of rat brain synaptic proteins in development and aging. Biochem Biophys Res Commun 251(1):394–398
Shimohama S, Kamiya S, Taniguchi T, Akagawa K, Kimura J (1997) Differential involvement of synaptic vesicle and presynaptic plasma membrane proteins in Alzheimer’s disease. Biochem Biophys Res Commun 236(2):239–242
Simpson IA, Chundu KR, Davies-Hill T, Honer WG, Davies P (1994) Decreased concentrations of GLUT1 and GLUT3 glucose transporters in the brains of patients with Alzheimer’s disease. Ann Neurol 35(5):546–551
Slunt HH, Thinakaran G, Von Koch C, Lo AC, Tanzi RE, Sisodia SS (1994) Expression of a ubiquitous, cross-reactive homologue of the mouse beta-amyloid precursor protein (APP). J Biol Chem 269:2637–2644
Smith-Swintosky VL, Pettigrew LC, Craddock SD, Culwell AR, Rydel RE, Mattson MP (1994) Secreted forms of beta-amyloid precursor protein protect against ischemic brain injury. J Neurochem 63:781–784
Sorg C, Riedl V, Mühlau M, Calhoun VD, Eichele T, Läer L, Drzezga A, Förstl H, Kurz A, Zimmer C, Wohlschläger AM (2007) Selective changes of resting-state networks in individuals at risk for Alzheimer’s disease. Proc Natl Acad Sci USA 104:18760–18765
Sperling R (2007) Functional MRI studies of associative encoding in normal aging, mild cognitive impairment, and Alzheimer’s disease. Ann N Y Acad Sci 1097:146–155
Stieler JT, Boerema AS, Bullmann T, Kohl F, Strijkstra AM, Barnes BM, Arendt T (2008) Activity-state profile of tau kinases in hibernating animals. In: Lovegrove BG, McKechnie AE (eds) Hypometabolism in animals: hibernation, torpor and cryobiology. University of KwaZulu-Nata, Pietermaritzburg, pp 133–142
Stieler JT, Bullmann T, Kohl F, Barnes BM, Arendt T (2009) PHF-like tau phosphorylation in mammalian hibernation is not associated with p25-formation. J Neural Transm 116(3):345–350
Strijkstra AM, Hut RA, de Wilde MC, Stieler J, Van der Zee EA (2003) Hippocampal synaptophysin immunoreactivity is reduced during natural hypothermia in ground squirrels. Neurosci Lett 344:29–32
Supekar K, Menon V, Rubin D, Musen M, Greicius MD (2008) Network analysis of intrinsic functional brain connectivity in Alzheimer’s disease. PLoS Comput Biol 4(6):e1000100
Svennerholm L, Gottfries CG (1994) Membrane lipids, selectively diminished in Alzheimer brains, suggest synapse loss as a primary event in early-onset form (type I) and demyelination in late-onset form (type II). J Neurochem 62:1039–1047
Sze CI, Bi H, Kleinschmidt-DeMasters BK, Filley CM, Martin LJ (2000) Selective regional loss of exocytotic presynaptic vesicle proteins in Alzheimer’s disease brains. J Neurol Sci 175(2):81–90
Sze CI, Troncoso JC, Kawas C, Mouton P, Price DL, Martin LJ (1997) Loss of the presynaptic vesicle protein synaptophysin in hippocampus correlates with cognitive decline in Alzheimer disease. J Neuropathol Exp Neurol 56(8):933–944
Takahashi RH, Capetillo-Zarate E, Lin MT, Milner TA, Gouras GK (2008) Co-occurrence of Alzheimer’s disease beta-amyloid and tau pathologies at synapses. Neurobiol Aging 2008 Sep 2. [Epub ahead of print]
Tanzi RE, Gusella JF, Watkins PC, Bruns GA, St George-Hyslop P, Van Keuren ML, Patterson D, Pagan S, Kurnit DM, Neve RL (1987) Amyloid beta protein gene: cDNA, mRNA distribution, and genetic linkage near the Alzheimer locus. Science 235:880–884
Tanzi E (1893) I fatti e le induzioni nell’odierna istologia del sistema nervoso. Rivista Sperimentale di Frenetria e Medicina Legale della Alienzioni mentali 19:419–472
Terry RD, Masliah E, Salmon DP, Butters N, DeTeresa R, Hill R, Hansen LA, Katzman R (1991) Physical basis of cognitive alterations in Alzheimer’s disease: synapse loss is the major correlate of cognitive impairment. Ann Neurol 30(4):572–580
Terry RD (1996) The pathogenesis of Alzheimer disease: an alternative to the amyloid hypothesis. J Neuropathol Exp Neurol 55(10):1023–1025
Thornton E, Vink R, Blumbergs PC, Van Den Heuvel C (2006) Soluble amyloid precursor protein alpha reduces neuronal injury and improves functional outcome following diffuse traumatic brain injury in rats. Brain Res 1094:38–46
Torroja L, Packard M, Gorczyca M, White K, Budnik V (1999) The Drosophila beta-amyloid precursor protein homolog promotes synapse differentiation at the neuromuscular junction. J Neurosci 19:7793–7803
van Gehuchten A (1900) Anatomie du systéme nerveux de l’homme. 3rd edn. Louvain
Villa A, Latasa MJ, Pascual A (2001) Nerve growth factor modulates the expression and secretion of beta-amyloid precursor protein through different mechanisms in PC12 cells. J Neurochem 77:1077–1084
Vincent I, Rosado M, Davies P (1996) Mitotic mechanisms in Alzheimer’s disease? J Cell Biol 132:413–425
von der Ohe CG, Garner CC, Darian-Smith C, Heller HC (2007) Synaptic protein dynamics in hibernation. J Neurosci 27:84–92
von Koch CS, Zheng H, Chen H, Trumbauer M, Thinakaran G, van der Ploeg LH, Price DL, Sisodia SS (1997) Generation of APLP2 KO mice and early postnatal lethality in APLP2/APP double KO mice. Neurobiol Aging 18:661–669
von Kölliker (1895) Kritik der Hypothesen von Rabl-Rückhard und Duval über amoeboide Bewegungen der Neurodendren. Sitzungsberichte der physik.-med. Gesellschaft: 38-42
Wakabayashi K, Honer WG, Masliah E (1994) Synapse alterations in the hippocampal-entorhinal formation in Alzheimer’s disease with and without Lewy body disease. Brain Res 667(1):24–32
Walsh DM, Klyubin I, Fadeeva JV, Cullen WK, Anwyl R, Wolfe MS, Rowan MJ, Selkoe DJ (2002) Naturally secreted oligomers of amyloid beta protein potently inhibit hippocampal long-term potentiation in vivo. Nature 416:535–539
Walsh DM, Selkoe DJ (2004) Deciphering the molecular basis of memory failure in Alzheimer’s disease. Neuron 44:181–193
Wang L, Zang Y, He Y, Liang M, Zhang X, Tian L, Wu T, Jiang T, Li K (2006) Changes in hippocampal connectivity in the early stages of Alzheimer’s disease: evidence from resting state fMRI. Neuroimage 31:496–504
Wang P, Yang G, Mosier DR, Chang P, Zaidi T, Gong YD, Zhao NM, Dominguez B, Lee KF, Gan WB, Zheng H (2005) Defective neuromuscular synapses in mice lacking amyloid precursor protein (APP) and APP-like protein 2. J Neurosci 25:1219–1225
Wasco W, Bupp K, Magendantz M, Gusella JF, Tanzi RE, Solomon F (1992) Identification of a mouse brain cDNA that encodes a protein related to the Alzheimer disease-associated amyloid beta protein precursor. PNAS 89:10758–10762
Wasco W, Gurubhagavatula S, Paradis MD, Romano DM, Sisodia SS, Hyman BT, Neve RL, Tanzi RE (1993) Isolation and characterization of APLP2 encoding a homologue of the Alzheimer’s associated amyloid beta protein precursor. Nat Genet 5:95–100
Weiler R, Lassmann H, Fischer P, Jellinger K, Winkler H (1990) A high ratio of chromogranin A to synaptin/synaptophysin is a common feature of brains in Alzheimer and Pick disease. FEBS Lett 263:337–339
Yamazaki T, Selkoe DJ, Koo EH (1995) Trafficking of cell surface beta-amyloid precursor protein: retrograde and transcytotic transport in cultured neurons. J Cell Biol 129:431–442
Yang G, Gong YD, Gong K, Jiang WL, Kwon E, Wang P, Zheng H, Zhang XF, Gan WB, Zhao NM (2005) Reduced synaptic vesicle density and active zone size in mice lacking amyloid precursor protein (APP) and APP-like protein 2. Neurosci Lett 384:66–71
Yankner BA, Duffy LK, Kirschner DA (1990) Neurotrophic and neurotoxic effects of amyloid beta protein: reversal by tachykinin neuropeptides. Science 250:279–282
Yao PJ, Morsch R, Callahan LM, Coleman PD (1999) Changes in synaptic expression of clathrin assembly protein AP180 in Alzheimer’s disease analysed by immunohistochemistry. Neuroscience 94:389–394
Yao PJ, Zhu M, Pyun EI, Brooks AI, Therianos S, Meyers VE, Coleman PD (2003) Defects in expression of genes related to synaptic vesicle trafficking in frontal cortex of Alzheimer’s disease. Neurobiol Dis 12(2):97–109
Zampieri N, Xu CF, Neubert TA, Chao MV (2005) Cleavage of p75 neurotrophin receptor by alpha-secretase and gamma-secretase requires specific receptor domains. J Biol Chem 280:14563–14571
Zhan SS, Beyreuther K, Schmitt HP (1993) Quantitative assessment of the synaptophysin immuno-reactivity of the cortical neuropil in various neurodegenerative disorders with dementia. Dementia 4:66–74
Zhan SS, Beyreuther K, Schmitt HP (1994) Synaptophysin immunoreactivity of the cortical neuropil in vascular dementia of Binswanger type compared with the dementia of Alzheimer type and nondemented controls. Dementia 5:79–87
Zheng H, Jiang M, Trumbauer ME, Sirinathsinghji DJ, Hopkins R, Smith DW, Heavens RP, Dawson GR, Boyce S, Conner MW, Stevens KA, Slunt HH, Sisoda SS, Chen HY, Van der Ploeg LH (1995) Beta-amyloid precursor protein-deficient mice show reactive gliosis and decreased locomotor activity. Cell 81:525–531
Zubenko GS, Kopp U, Seto T, Firestone LL (1999) Platelet membrane fluidity individuals at risk for Alzheimer’s disease: a comparison of results from fluorescence spectroscopy and electron spin resonance spectroscopy. Psychopharmacology (Berl.) 145:175–180
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Arendt, T. Synaptic degeneration in Alzheimer’s disease. Acta Neuropathol 118, 167–179 (2009). https://doi.org/10.1007/s00401-009-0536-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-009-0536-x