Abstract
Cardiovascular disease is the major cause of death and disability in the world, with ischemic heart disease accounting for the vast majority of this health problem. Current treatments for ischemic heart disease are primarily aimed at either increasing blood and oxygen supply to the heart or decreasing the heart’s oxygen demand. A novel treatment strategy involves increasing the efficiency of oxygen use by the heart. During and following ischemia, the heart can become inefficient in using oxygen, due in part to an excessive use of fatty acids as a source of fuel. One potential strategy to increase cardiac efficiency is to inhibit this use of fatty acid oxidation as a fuel source, while stimulating the use of glucose oxidation as a fuel source, which allows the heart to produce energy more efficiently and reduces the acidosis associated with ischemia/reperfusion, both of which are beneficial to the heart. Malonyl CoA is a potent endogenous inhibitor of cardiac fatty acid oxidation, secondary to inhibition of carnitine palmitoyl transferase-I, the gatekeeper of mitochondrial fatty acid uptake. Malonyl CoA is synthesized in the heart by acetyl CoA carboxylase and degraded by malonyl CoA decarboxylase (MCD). Strategies aimed at increasing cardiac malonyl CoA levels, such as via inhibition of MCD, are associated with a decrease in fatty acid oxidation rates, and a parallel increase in glucose oxidation rates. This is associated with a decrease in acidosis and an improvement in cardiac function and efficiency during and following ischemia. Therefore, targeting malonyl CoA is a novel exciting approach for the treatment of cardiac ischemia/reperfusion.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cardiovascular disease (CVD) is a major health problem worldwide, and is now the number one killer of people in the world [67]. The underlying cause for the majority of people living with CVD is an imbalance between the oxygen supply and the oxygen demand of the heart, a condition coined “ischemic heart disease.”
In the last two decades, enormous advances have been made in the treatment of ischemic heart disease, including better risk prevention strategies, aggressive lipid lowering strategies and timely re-introduction of blood flow to the ischemic myocardium [2]. Current treatment strategies that increase coronary blood flow and oxygen supply (such as percutaneous and surgical techniques or the use of vasodilators, antiplatelet agents, anticoagulants or thrombolytic therapy), or to decrease myocardial oxygen demand (such as β-blockers or nitrates) have greatly improved the overall prognosis of ischemic heart disease patients. However, there are still a number of patients who prove to be ineligible or unresponsive to conventional treatment. Thus, new approaches to treat such patients are necessary, one of these approaches being the optimization of cardiac energy metabolism.
In the setting of ischemic heart disease, one approach to optimize cardiac energy metabolism is to either stimulate glucose oxidation or inhibit fatty acid oxidation [59]. The oxidation of one glucose molecule produces more ATP per oxygen molecule consumed than that of a fatty acid molecule, which allows the heart to produce energy more efficiently. Furthermore, stimulating glucose oxidation either directly, or secondarily due to an inhibition of fatty acid oxidation, results in improved coupling between glycolysis and glucose oxidation, which decreases proton production and alleviates myocardial acidosis, improving cardiac efficiency [9, 32–34].
Cardiac fatty acid oxidation can be inhibited in a number of ways, such as blocking the transport of fatty acids into the cardiac myocyte, inhibiting mitochondrial fatty acid uptake, or directly inhibiting the enzymatic machinery of the mitochondrial β-oxidative pathway [54, 60]. While there are existing agents that target all these approaches, this review will focus on inhibiting mitochondrial fatty acid uptake. A potent endogenous inhibitor of mitochondrial fatty acid uptake is malonyl CoA, which inhibits carnitine palmitoyltranferase 1 (CPT 1), the rate-limiting enzyme for mitochondrial fatty acid uptake. Agents which either mimic malonyl CoA’s actions on CPT 1, or that increase the myocardial levels of malonyl CoA (by either stimulating malonyl CoA synthesis or inhibiting malonyl CoA degradation) are two approaches that have been used to inhibit mitochondrial fatty acid uptake [21]. This paper reviews the literature on the regulation of malonyl CoA via both its synthesis and its degradation, and discusses in detail how the malonyl CoA axis has been manipulated in animal models to implicate it as a novel target to treat cardiac ischemia/reperfusion.
Cardiac energy metabolism in the normal heart and during ischemia/reperfusion
In the normal healthy heart, almost all (>95%) ATP generated in the heart arises from mitochondrial oxidative phosphorylation (primarily carbohydrate and fatty acid oxidation), with the remainder derived from glycolysis [35]. Despite generating more ATP per molecule than carbohydrates, fatty acids are not as oxygen-efficient, requiring approximately 10% more oxygen to generate an equivalent amount of ATP [54]. In addition, fatty acids directly inhibit the oxidation of carbohydrates [43], due to a build up of fatty acid oxidative by-products inhibiting mitochondrial pyruvate dehydrogenase, the rate limiting enzyme in glucose oxidation. This uncouples glycolysis from glucose oxidation, resulting in an increased proton production [9, 32–34]. In the aerobic heart, this production of protons does not present a problem as the heart has a sufficient energy supply to deal with it.
During ischemia cardiac energy metabolism is dramatically altered, which can include an increase in glycolysis rates, which attempts to provide an anaerobic source of ATP to make up for the reduction in oxidative ATP production. Of importance, is that even though overall mitochondrial oxidative metabolism decreases in proportion to the severity of ischemia, fatty acid oxidation dominates as the main source of residual oxidative metabolism [62], which occurs at the expense of glucose oxidation. This result in an uncoupling between glycolysis and glucose oxidation, contributing to the acidosis observed in the ischemic heart, which reduces cardiac efficiency [9, 32–34].
During reperfusion of the heart following ischemia, glycolytic rates remain elevated, while fatty acids dominate as a source of oxidative energy production [32]. These high rates of fatty acid oxidation can account for >90% of energy production in the reperfused heart, which inhibits glucose oxidation via the Randle Cycle effect [43]. Thus, similar to ischemia, reperfusion of the ischemic heart is accompanied by an increased production of protons from glycolysis uncoupled to glucose oxidation that lowers cardiac efficiency. In this scenario, the accumulation of protons can be detrimental to the heart, as ATP is being diverted away from contractile function, in order to restore ion homeostasis during the initial critical stages of the heart’s recovery from the ischemic insult [33, 34, 55, 56].
The reason for the excessive use of fatty acids as a fuel source during and following ischemia can be primarily attributed to two factors: (1) plasma levels of fatty acids rapidly increase during and following ischemia, resulting in the heart being exposed to high levels of fatty acids [30, 36, 40], and (2) subcellular changes occur in the cardiac myocyte itself, resulting in a decreased control of fatty acid oxidation [28, 29]. With regard to the second point, cardiac malonyl CoA levels decrease in the heart due to the rapid activation of 5′AMP activated protein kinase (AMPK) during ischemia [as a result of an increase in AMP levels and activation of the upstream AMPK kinase (AMPKK)], which persists into reperfusion, resulting in the phosphorylation induced inhibition of acetyl CoA carboxylase (ACC) [12, 16, 28, 29]. Lower malonyl CoA levels relieves the inhibition of CPT 1, resulting in an accelerated mitochondrial fatty acid uptake for subsequent β-oxidation.
As will be discussed, one approach to optimize cardiac energy metabolism is to increase cardiac malonyl CoA levels. This inhibits mitochondrial fatty acid uptake, leading to a subsequent inhibition of fatty acid β-oxidation and secondary increase in glucose oxidation [16, 32]. This approach increases the efficiency of oxygen utilization and cardiac energy production, while preventing the production of protons and development of acidosis.
Regulation of malonyl CoA in the heart
As discussed, malonyl CoA is a potent endogenous inhibitor of CPT 1, the rate limiting enzyme for mitochondrial fatty acid uptake. Thus, an increase in myocardial malonyl CoA levels result in a decrease in mitochondrial fatty acid uptake and subsequent fatty acid β-oxidation. As malonyl CoA has quite a rapid turnover, with a half life of about 1.25 min [44], both the production and the degradation of malonyl CoA are important determinants of cardiac malonyl CoA levels, and therefore fatty acid oxidation.
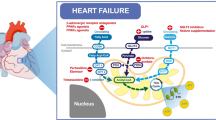
Malonyl CoA is synthesized via carboxylation of acetyl CoA to malonyl CoA by acetyl CoA carboxylase (ACC) (Fig. 1) [5, 57]. There are two isoforms of ACC in the heart, α and β, with a predominance of ACCβ [57]. This has led to the suggestion that the malonyl CoA produced by this isoform is more involved in the regulation of fatty acid oxidation, as opposed to the high abundance of ACCα in the liver, where the malonyl CoA produced by this isoform may be primarily involved in the regulation of fatty acid biosynthesis [38]. Studies from our laboratory have confirmed the key role of ACCβ in regulating cardiac fatty acid oxidation [47]. The regulation of ACC is under phosphorylation/dephosphorylation control, with 5′AMP-activated protein kinase (AMPK) playing a major role in its phosphorylation-induced inactivation in the heart (Fig. 1) [15, 28]. As a result, AMPK has a key signaling role in the control of cardiac energy metabolism during reperfusion following ischemia.
Malonyl CoA regulation in the aerobic heart and during ischemia/reperfusion. Malonyl CoA is synthesized via carboxylation of acetyl CoA by ACC, whereas it is degraded via decarboxylation back into acetyl CoA by MCD. In addition, ACC is negatively regulated by phosphorylation via AMPK. Increased production of malonyl CoA inhibits mitochondrial uptake of fatty acids through CPT-I, thereby reducing rates of fatty acid β-oxidation. During ischemia, decreased ATP production and a subsequent increase in AMP, as well as activation of AMPKK, result in the rapid activation of AMPK, which phosphorylates and inhibits ACC, resulting in a dramatic drop in malonyl CoA levels. Following aerobic reperfusion of the ischemic heart, AMPK activity is sustained, while MCD activity remains high. This keeps malonyl CoA levels low, allowing fatty acids to dominate as the main source of oxidative ATP production
Malonyl CoA is degraded via decarboxylation of malonyl CoA to acetyl CoA by malonyl CoA decarboxylase (MCD) (Fig. 1) [12]. Studies in both rat and mouse have demonstrated that MCD is very important in regulating cardiac malonyl CoA levels, and that inhibition of MCD can increase malonyl CoA levels and limit rates of fatty acid oxidation, leading to a secondary increase in glucose oxidation. This decrease in fatty acid oxidation and increase in glucose oxidation is also associated with an improvement in the functional recovery of the heart during ischemia/reperfusion [8, 13, 14, 53]. In addition, peroxisome proliferator activated receptor alpha (PPARα), which is a major transcription factor involved in the regulation of fatty acid oxidation, has been shown to regulate expression of MCD [31, 63]. An increase in MCD expression by PPARα activation is accompanied by an increase fatty acid oxidation [19, 31].
Previous work in our laboratory has shown that the high rates of cardiac fatty acid oxidation observed during reperfusion of ischemic hearts are due, in part, to a rapid reduction in malonyl CoA levels, as opposed to direct alterations in CPT 1 [28]. Furthermore, we have shown that this reduction in malonyl CoA levels can be explained by the rapid activation of AMPK during ischemia, which persists into reperfusion, causing the phosphorylation-induced inactivation of ACC and decrease in malonyl CoA levels (Fig. 1) [28]. It has also been suggested that MCD is a direct target of AMPK, but our laboratory has been unable to reproduce those findings [48]. In summary, a decrease in ACC activity via AMPK phosphorylation, together with a maintained MCD activity, are two major factors responsible for the rapid decline in cardiac malonyl CoA levels observed during ischemia/reperfusion.
Targeting malonyl CoA to treat cardiac ischemia/reperfusion
One approach to increasing malonyl CoA levels in the heart to inhibit fatty acid oxidation and treat ischemic heart disease is to target one or more of the enzymes involved in regulating its production (i.e. ACC, MCD, and/or AMPK). The section will examine the targeting of each of these enzymes individually as potential treatments for ischemia/reperfusion.
Targeting ACC to treat ischemia/reperfusion injury in the heart
The literature examining the effects of ACC inhibition on ischemia/reperfusion injury is limited, as the majority of literature on ACC manipulation has focused on inhibiting ACC in the liver and adipose tissue. In lipogenic tissues such as the liver it has been postulated that inhibiting ACC to decrease malonyl CoA levels will decrease fatty acid biosynthesis, thereby preventing the development of hepatosteatosis and obesity [7, 38, 41]. To date, there are no selective pharmacological inhibitors of ACC available, and if there were, they should be specific for inhibition of the β isoform of ACC, which predominates in the heart and is more tightly linked to the regulation of fatty acid oxidation [47].
Although pharmacological inhibitors of ACC in the heart have not been studied, mice deficient for ACCβ (ACCβ−/−) are available, and have been shown to have significantly elevated rates of fatty acid oxidation [1]. This would be expected if malonyl CoA levels were decreased. A recent study in these mice showed the expected reduction in cardiac malonyl CoA levels and triglyceride content, but curiously showed an unexpected elevation in cardiac fatty acid oxidation and glucose oxidation rates, as well as an improved insulin-stimulated glucose uptake [17]. Coupled with its beneficial effects on enhancing insulin sensitivity in muscle and the liver, the authors concluded that ACC inhibition represents viable treatment strategy for obesity and its associated conditions. However, it should be noted in this study that the reported glucose oxidation rates are 10–20 fold lower than what is normally reported in the literature [26, 33, 37, 47], and the reported change in fatty acid oxidation rates were minimal. Moreover, it is surprising to not observe an increase in oxygen consumption in hearts from ACCβ−/− mice if both glucose and fatty acid oxidation were increased simultaneously. Because, obesity itself is a risk factor for the development of ischemic heart disease [23–25], and because elevated rates of fatty acid oxidation may have detrimental effects on cardiac recovery during reperfusion [28, 33, 34, 49, 59], it would be of interest in future studies to examine the effects of ACCβ deletion on ischemia/reperfusion injury in hearts from ACCβ−/− mice.
Targeting MCD to treat ischemia/reperfusion injury in the heart
Recent studies from our laboratory have shown that MCD is a key regulator of cardiac fatty acid oxidation, and that inhibition of this enzyme is a viable target for the treatment of ischemia/reperfusion injury [13, 14]. Employing novel inhibitors of MCD, we have shown that inhibition of MCD in isolated working rat hearts significantly increases malonyl CoA levels [13]. This is associated with a significant decrease in fatty acid oxidation rates, resulting in a significant increase in glucose oxidation rates. The increase in glucose oxidation was accompanied by a significant decrease in proton production during ischemia. These metabolic effects induced via inhibition of MCD result in a significant improvement in cardiac functional recovery of aerobically reperfused ischemic rat hearts. In an in vivo pig model of demand-induced ischemia, MCD inhibition also results in an increase in cardiac malonyl CoA levels and glucose oxidation rates [13, 53]. Moreover, this is accompanied by a significant reduction in myocardial lactate production and a restoration of left ventricular regional wall function. Last, a third study from our laboratory investigated the effects of chronic MCD inhibition using a whole body MCD deficient mouse model [14]. Although hearts from these animals had a significant increase in malonyl CoA levels, isolated aerobic working heart perfusions demonstrated no changes in glucose and fatty acid metabolism compared to wild type control hearts. This may have resulted from a significant up-regulation in the mRNA of a number of different PPARα target gene transcripts, such as pyruvate dehydrogenase kinase 4 and CPT 1. Nevertheless, when hearts from the MCD deficient mice were subjected to an ischemia/reperfusion protocol, a significant improvement in the recovery of cardiac power and function was observed. This improved recovery was accompanied by a significant increase in glucose oxidation rates, such that glucose oxidation became the major source of cardiac ATP production. In an in vivo ischemia model, involving temporary occlusion of the left anterior descending artery, we have also shown that hearts from MCD deficient mice also have a marked reduction in infarct size compared to wild-type mice [61].
While the inhibition of fatty acid oxidation in the heart improves function in the setting of ischemic heart disease, others have postulated that the inhibition of fatty acid oxidation in peripheral tissues, such as the muscle and liver, will exacerbate insulin resistance and type 2 diabetes [7, 42, 66]. This is of potential importance, as a significant number of patients with ischemic heart disease are also obese/type 2 diabetic. From a clinical viewpoint, oral delivery of MCD inhibitors for the treatment of ischemic heart disease would be most practical. However, this would also affect peripheral tissues such as the skeletal muscle, and could cause insulin resistance. Surprisingly, a recent collaboration by our laboratory and that of Debbie Muoio and colleagues has shown that this is not the case [27]. Obesity and insulin resistance induced by a high fat diet are actually associated with elevated rates of incomplete fatty acid oxidation, as opposed to impaired fatty acid oxidation [27]. We also demonstrated that the MCD deficient mouse is protected from obesity induced insulin resistance, which is associated with a reduction in incomplete fatty acid oxidation rates. Furthermore, our most recent work has shown that hearts deficient for MCD following chronic high fat feeding have a marked preservation of insulin-stimulated glucose metabolism, and no signs of cardiac dysfunction ex vivo and in vivo (unpublished data).
As mentioned earlier, MCD is a target gene of PPARα [31, 63], and in previous studies we have shown that hearts from PPARα deficient mice have a reduced MCD expression and increased malonyl CoA levels [6]. These animals subsequently have lower rates of fatty acid oxidation, increased rates of glucose oxidation, and exhibit improved recovery of cardiac power during reperfusion following ischemia, whereas PPARα over-expressing mice hearts have an opposite metabolic profile and a decreased recovery of cardiac power under identical conditions [49].
Targeting AMPK to treat ischemia/reperfusion injury in the heart
Since its initial discovery in 1988 by Grahame Hardie [52], AMPK has become a protein with wide interest among many laboratories, due to its ability to regulate energy metabolism in times of stress. AMPK activation has a number of effects on cardiac energy metabolism, which includes an increase in glucose uptake and glycolysis, as well as an increase in fatty acid oxidation. The increase in fatty acid oxidation is due to AMPK phosphorylation and inhibition of ACC, which leads to a decrease in malonyl CoA levels in the heart [28, 29]. In 1995, we showed that AMPK is rapidly activated during ischemia, leading to a phosphorylation and inhibition of ACC [28]. This is accompanied by an increase in fatty acid oxidation and a decrease in cardiac function and efficiency during reperfusion of hearts following ischemia. However, whether this AMPK stimulation of fatty acid oxidation contributes to ischemic injury is controversial. A number of groups have proposed that ischemic-induced stimulation of AMPK is beneficial, due to an increase in glucose uptake and glycolysis, which can provide an anaerobic source of ATP for the energy starved heart [3, 4, 45]. However, an AMPK-induced decrease in cardiac malonyl CoA levels and increase in fatty acid oxidation can decrease glucose oxidation, which has the potential to increase proton production and decrease cardiac efficiency [28]. We therefore hypothesized that AMPK inhibition would benefit the heart during ischemia/reperfusion by lowering malonyl CoA levels and fatty acid oxidation rates, thereby increasing glucose oxidation and preventing myocardial acidosis. However, despite this it has been reported that hearts from mice expressing a dominant negative (DN) AMPKα2 with nearly a complete loss of myocardial AMPK activity recover worse during reperfusion following a low-flow ischemic insult [46]. Hearts from the transgenic DN-AMPKα2 mice are also unable to increase glucose uptake during low-flow ischemia/reperfusion, and have increased rates of apoptosis as determined via TUNEL staining. However, these DN-AMPKα2 transgenic mice have significant left ventricular dysfunction in the normal setting, which may explain why they did not recover as well during ischemia/reperfusion. Moreover, there were no changes in cardiac metabolism between wild type and DN-AMPKα2 transgenic mice, which suggests that there may not have been changes in malonyl CoA levels from the hearts of these animals. In a recent study, we found that if hearts from DN-AMPKα2 transgenic mice were perfused with high levels of fatty acids and subjected to ischemia, heart function recovered to a greater extent, which was associated with an increase in glucose oxidation [22]. As a result, there is still confusion as to whether AMPK inhibition or stimulation is beneficial in the setting of ischemia and reperfusion.
Adding to this controversy, a recent study in AMPKα2 subunit deficient mice showed that while AMPKα2 deficiency accelerates the appearance of contracture during ischemia, there is no effect of AMPKα2 deficiency on reperfusion recovery of these hearts, suggesting that AMPK inhibition is not detrimental in the heart [65]. This study utilized glucose-only perfusions, and thus high rates of fatty acid oxidation would not present a problem during ischemia/reperfusion. It is possible that the benefit of reducing the extremely high rates of fatty acid oxidation during reperfusion may have been masked in this study.
Another study investigating the beneficial effects of adiponectin during ischemia/reperfusion injury observed a reduced phosphorylation of AMPK at its activating threonine 172 residue in an adiponectin deficient mouse model 48 h after a 30 min ligation of the left anterior descending coronary artery (LAD) [50]. This study showed that inhibition of AMPK prevented the anti-apoptotic effects of adiponectin on cardiac myocytes subjected to hypoxia/reoxygenation. Nonetheless, the anti-apoptotic effects of AMPK were studied only in culture, and the beneficial effects of adiponectin during reperfusion following LAD ligation were also proposed to be explained via cyclooxygenase II (COX-II) dependent mechanisms.
Interestingly, a recent study showing the beneficial effects of short-term caloric restriction (CR) on cardiac ischemia/reperfusion observed that short-term CR increased plasma adiponectin levels and reduced infarct size [51]. However, inhibition of AMPK or use of an adiponectin antisense transgenic mouse strain prevented the observed cardioprotection. Unfortunately, these studies were limited due to the absence of fatty acids from the perfusate in their ischemia/reperfusion protocol, and the AMPK inhibitor used (i.e. adenosine [9-D arabinofuranoside] as an AMPK agonist is very nonspecific) [11]. In addition, other pathways, such as the aforementioned COX-II pathway, may also be contributing to the cardioprotective actions of adiponectin observed during short-term CR.
AMPK can also be inhibited via insulin administration, and a number of studies have examined the role of glucose–insulin–potassium (GIK) for the treatment of acute myocardial infarction [18, 58, 64]. Our initial hypothesis was that insulin would benefit the aerobically reperfused ischemic heart via inhibition of AMPK, thereby reducing fatty acid oxidation rates. This would increase glucose oxidation rates, preventing myocardial acidosis and improving functional recovery. To our surprise, we discovered that fatty acids in the perfusate interfere with insulin’s ability to inhibit AMPK, and although insulin still was able to reduce fatty acid oxidation, a greater stimulation of glycolysis than glucose oxidation actually increased proton production and worsened functional recovery during reperfusion [20]. Therefore, it is unlikely that the beneficial effects of insulin during reperfusion involve an inhibition of AMPK, and it is possible that our results can explain the lack of mortality improvement with GIK for patients experiencing an acute myocardial infarction (AMI) during the recent multi-centre CREATE-ECLA trial [39]. In fact, results from a recent study suggest that GIK may actually increase mortality in the early post AMI period [10].
Due to the mixed results of the discussed studies, we believe that there is insufficient evidence to state that AMPK activation is beneficial or harmful during cardiac ischemia/reperfusion. What is needed to reconcile these differences is more in-depth studies using in vivo ischemia/reperfusion models to investigate the effects of AMPK on myocardial function, as well as the need for actual measurements of cardiac malonyl CoA levels in these systems.
Summary
Optimization of energy metabolism represents a potential exciting novel approach for the treatment of cardiac ischemia/reperfusion. An example of this approach includes increasing cardiac levels of malonyl CoA, a potent endogenous inhibitor of fatty acid oxidation, secondary to its inhibition of CPT 1, the gatekeeper of mitochondrial fatty acid uptake. Inhibition of MCD is one effective approach to increase cardiac malonyl CoA levels. MCD inhibition can improve functional recovery of the heart during ischemia/reperfusion injury, which can be attributed to an increased glucose oxidation and a decreased proton production. Thus, targeting the malonyl CoA axis represents a potential exciting new avenue to explore for the treatment of patients suffering from ischemic heart disease.
References
Abu-Elheiga L, Matzuk MM, Abo-Hashema KA, Wakil SJ (2001) Continuous fatty acid oxidation and reduced fat storage in mice lacking acetyl-CoA carboxylase 2. Science 291:2613–2616
Antman EM, Hand M, Armstrong PW, Bates ER, Green LA, Halasyamani LK, Hochman JS, Krumholz HM, Lamas GA, Mullany CJ, Pearle DL, Sloan MA, Smith SC Jr, Anbe DT, Kushner FG, Ornato JP, Pearle DL, Sloan MA, Jacobs AK, Adams CD, Anderson JL, Buller CE, Creager MA, Ettinger SM, Halperin JL, Hunt SA, Lytle BW, Nishimura R, Page RL, Riegel B, Tarkington LG, Yancy CW (2008) 2007 focused update of the ACC/AHA 2004 guidelines for the management of patients with ST-elevation myocardial infarction: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol 51:210–247
Baron SJ, Li J, Russell RR 3rd, Neumann D, Miller EJ, Tuerk R, Wallimann T, Hurley RL, Witters LA, Young LH (2005) Dual mechanisms regulating AMPK kinase action in the ischemic heart. Circ Res 96:337–345
Beauloye C, Marsin AS, Bertrand L, Krause U, Hardie DG, Vanoverschelde JL, Hue L (2001) Insulin antagonizes AMP-activated protein kinase activation by ischemia or anoxia in rat hearts, without affecting total adenine nucleotides. FEBS Lett 505:348–352
Bianchi A, Evans JL, Iverson AJ, Nordlund AC, Watts TD, Witters LA (1990) Identification of an isozymic form of acetyl-CoA carboxylase. J Biol Chem 265:1502–1509
Campbell FM, Kozak R, Wagner A, Altarejos JY, Dyck JR, Belke DD, Severson DL, Kelly DP, Lopaschuk GD (2002) A role for peroxisome proliferator-activated receptor alpha (PPARalpha) in the control of cardiac malonyl-CoA levels: reduced fatty acid oxidation rates and increased glucose oxidation rates in the hearts of mice lacking PPARalpha are associated with higher concentrations of malonyl-CoA and reduced expression of malonyl-CoA decarboxylase. J Biol Chem 277:4098–4103
Choi CS, Savage DB, Abu-Elheiga L, Liu ZX, Kim S, Kulkarni A, Distefano A, Hwang YJ, Reznick RM, Codella R, Zhang D, Cline GW, Wakil SJ, Shulman GI (2007) Continuous fat oxidation in acetyl-CoA carboxylase 2 knockout mice increases total energy expenditure, reduces fat mass, and improves insulin sensitivity. Proc Natl Acad Sci USA 104:16480–16485
Cuthbert KD, Dyck JR (2005) Malonyl-CoA decarboxylase is a major regulator of myocardial fatty acid oxidation. Curr Hypertens Rep 7:407–411
Dennis SC, Gevers W, Opie LH (1991) Protons in ischemia: where do they come from; where do they go to? J Mol Cell Cardiol 23:1077–1086
Diaz R, Goyal A, Mehta SR, Afzal R, Xavier D, Pais P, Chrolavicius S, Zhu J, Kazmi K, Liu L, Budaj A, Zubaid M, Avezum A, Ruda M, Yusuf S (2007) Glucose-insulin-potassium therapy in patients with ST-segment elevation myocardial infarction. JAMA 298:2399–2405
Dyck JR (2007) The ischemic heart: starving to stimulate the adiponectin-AMPK signaling axis. Circulation 116:2779–2781
Dyck JR, Barr AJ, Barr RL, Kolattukudy PE, Lopaschuk GD (1998) Characterization of cardiac malonyl-CoA decarboxylase and its putative role in regulating fatty acid oxidation. Am J Physiol 275:H2122–H2129
Dyck JR, Cheng JF, Stanley WC, Barr R, Chandler MP, Brown S, Wallace D, Arrhenius T, Harmon C, Yang G, Nadzan AM, Lopaschuk GD (2004) Malonyl coenzyme a decarboxylase inhibition protects the ischemic heart by inhibiting fatty acid oxidation and stimulating glucose oxidation. Circ Res 94:e78–e84
Dyck JR, Hopkins TA, Bonnet S, Michelakis ED, Young ME, Watanabe M, Kawase Y, Jishage K, Lopaschuk GD (2006) Absence of malonyl coenzyme A decarboxylase in mice increases cardiac glucose oxidation and protects the heart from ischemic injury. Circulation 114:1721–1728
Dyck JR, Lopaschuk GD (2006) AMPK alterations in cardiac physiology and pathology: enemy or ally? J Physiol 574:95–112
Dyck JR, Lopaschuk GD (2002) Malonyl CoA control of fatty acid oxidation in the ischemic heart. J Mol Cell Cardiol 34:1099–1109
Essop MF, Camp HS, Choi CS, Sharma S, Fryer RM, Reinhart GA, Guthrie PH, Bentebibel A, Gu Z, Shulman GI, Taegtmeyer H, Wakil SJ, Abu-Elheiga L (2008) Reduced heart size and increased myocardial fuel substrate oxidation in ACC2 mutant mice. Am J Physiol Heart Circ Physiol 295:H256–H265
Fath-Ordoubadi F, Beatt KJ (1997) Glucose-insulin-potassium therapy for treatment of acute myocardial infarction: an overview of randomized placebo-controlled trials. Circulation 96:1152–1156
Finck BN, Lehman JJ, Leone TC, Welch MJ, Bennett MJ, Kovacs A, Han X, Gross RW, Kozak R, Lopaschuk GD, Kelly DP (2002) The cardiac phenotype induced by PPARalpha overexpression mimics that caused by diabetes mellitus. J Clin Invest 109:121–130
Folmes CD, Clanachan AS, Lopaschuk GD (2006) Fatty acids attenuate insulin regulation of 5′-AMP-activated protein kinase and insulin cardioprotection after ischemia. Circ Res 99:61–68
Folmes CD, Lopaschuk GD (2007) Role of malonyl-CoA in heart disease and the hypothalamic control of obesity. Cardiovasc Res 73:278–287
Folmes CDL, Wagg CS, Shen M, Clanachan AS, Tian R, Lopaschuk GD (2008) Suppression of AMP-Activated Protein Kinase Activity Does Not Impair Recovery of Contractile Function During Reperfusion of Ischemic Hearts. Submitted
Fontaine KR, Redden DT, Wang C, Westfall AO, Allison DB (2003) Years of life lost due to obesity. Jama 289:187–193
Hubert HB, Feinleib M, McNamara PM, Castelli WP (1983) Obesity as an independent risk factor for cardiovascular disease: a 26-year follow-up of participants in the Framingham Heart Study. Circulation 67:968–977
Kenchaiah S, Evans JC, Levy D, Wilson PWF, Benjamin EJ, Larson MG, Kannel WB, Vasan RS (2002) Obesity and the risk of heart failure. N Engl J Med 347:305–313
Kewalramani G, An D, Kim MS, Ghosh S, Qi D, Abrahani A, Pulinilkunnil T, Sharma V, Wambolt RB, Allard MF, Innis SM, Rodrigues B (2007) AMPK control of myocardial fatty acid metabolism fluctuates with the intensity of insulin-deficient diabetes. J Mol Cell Cardiol 42:333–342
Koves TR, Ussher JR, Noland RC, Slentz D, Mosedale M, Ilkayeva O, Bain J, Stevens R, Dyck JR, Newgard CB, Lopaschuk GD, Muoio DM (2008) Mitochondrial overload and incomplete Fatty Acid oxidation contribute to skeletal muscle insulin resistance. Cell metabolism 7:45–56
Kudo N, Barr AJ, Barr RL, Desai S, Lopaschuk GD (1995) High rates of fatty acid oxidation during reperfusion of ischemic hearts are associated with a decrease in malonyl-CoA levels due to an increase in 5′-AMP-activated protein kinase inhibition of acetyl-CoA carboxylase. J Biol Chem 270:17513–17520
Kudo N, Gillespie JG, Kung L, Witters LA, Schulz R, Clanachan AS, Lopaschuk GD (1996) Characterization of 5′AMP-activated protein kinase activity in the heart and its role in inhibiting acetyl-CoA carboxylase during reperfusion following ischemia. Biochim Biophys Acta 1301:67–75
Kurien VA, Oliver MF (1971) Free fatty acids during acute myocardial infarction. Prog Cardiovasc Dis 13:361–373
Lee GY, Kim NH, Zhao ZS, Cha BS, Kim YS (2004) Peroxisomal-proliferator-activated receptor alpha activates transcription of the rat hepatic malonyl-CoA decarboxylase gene: a key regulation of malonyl-CoA level. Biochem J 378:983–990
Liu B, Clanachan AS, Schulz R, Lopaschuk GD (1996) Cardiac efficiency is improved after ischemia by altering both the source and fate of protons. Circ Res 79:940–948
Liu B, el Alaoui-Talibi Z, Clanachan AS, Schulz R, Lopaschuk GD (1996) Uncoupling of contractile function from mitochondrial TCA cycle activity and MVO2 during reperfusion of ischemic hearts. Am J Physiol 270:H72–H80
Liu Q, Docherty JC, Rendell JC, Clanachan AS, Lopaschuk GD (2002) High levels of fatty acids delay the recovery of intracellular pH and cardiac efficiency in post-ischemic hearts by inhibiting glucose oxidation. J Am Coll Cardiol 39:718–725
Lopaschuk GD, Belke DD, Gamble J, Itoi T, Schonekess BO (1994) Regulation of fatty acid oxidation in the mammalian heart in health and disease. Biochim Biophys Acta 1213:263–276
Lopaschuk GD, Collins-Nakai R, Olley PM, Montague TJ, McNeil G, Gayle M, Penkoske P, Finegan BA (1994) Plasma fatty acid levels in infants and adults after myocardial ischemia. Am Heart J 128:61–67
Lydell CP, Chan A, Wambolt RB, Sambandam N, Parsons H, Bondy GP, Rodrigues B, Popov KM, Harris RA, Brownsey RW, Allard MF (2002) Pyruvate dehydrogenase and the regulation of glucose oxidation in hypertrophied rat hearts. Cardiovasc Res 53:841–851
Mao J, DeMayo FJ, Li H, Abu-Elheiga L, Gu Z, Shaikenov TE, Kordari P, Chirala SS, Heird WC, Wakil SJ (2006) Liver-specific deletion of acetyl-CoA carboxylase 1 reduces hepatic triglyceride accumulation without affecting glucose homeostasis. Proc Natl Acad Sci USA 103:8552–8557
Mehta SR, Yusuf S, Diaz R, Zhu J, Pais P, Xavier D, Paolasso E, Ahmed R, Xie C, Kazmi K, Tai J, Orlandini A, Pogue J, Liu L (2005) Effect of glucose-insulin-potassium infusion on mortality in patients with acute ST-segment elevation myocardial infarction: the CREATE-ECLA randomized controlled trial. JAMA 293:437–446
Mueller HS, Ayres SM (1978) Metabolic response of the heart in acute myocardial infarction in man. Am J Cardiol 42:363–371
Oh W, Abu-Elheiga L, Kordari P, Gu Z, Shaikenov T, Chirala SS, Wakil SJ (2005) Glucose and fat metabolism in adipose tissue of acetyl-CoA carboxylase 2 knockout mice. Proc Natl Acad Sci USA 102:1384–1389
Petersen KF, Shulman GI (2006) Etiology of insulin resistance. Am J Med 119:S10–S16
Randle PJ, Garland PB, Hales CN, Newsholme EA (1963) The glucose fatty-acid cycle. Its role in insulin sensitivity and the metabolic disturbances of diabetes mellitus. Lancet 1:785–789
Reszko AE, Kasumov T, Comte B, Pierce BA, David F, Bederman IR, Deutsch J, Des Rosiers C, Brunengraber H (2001) Assay of the concentration and 13C-isotopic enrichment of malonyl-coenzyme A by gas chromatography-mass spectrometry. Anal Biochem 298:69–75
Russell R 3rd (2006) Stress signaling in the heart by AMP-activated protein kinase. Curr Hypertens Rep 8:446–450
Russell RR 3rd, Li J, Coven DL, Pypaert M, Zechner C, Palmeri M, Giordano FJ, Mu J, Birnbaum MJ, Young LH (2004) AMP-activated protein kinase mediates ischemic glucose uptake and prevents postischemic cardiac dysfunction, apoptosis, and injury. J Clin Invest 114:495–503
Saddik M, Gamble J, Witters LA, Lopaschuk GD (1993) Acetyl-CoA carboxylase regulation of fatty acid oxidation in the heart. J Biol Chem 268:25836–25845
Saha AK, Schwarsin AJ, Roduit R, Masse F, Kaushik V, Tornheim K, Prentki M, Ruderman NB (2000) Activation of malonyl-CoA decarboxylase in rat skeletal muscle by contraction and the AMP-activated protein kinase activator 5-aminoimidazole-4-carboxamide-1-beta -D-ribofuranoside. J Biol Chem 275:24279–24283
Sambandam N, Morabito D, Wagg C, Finck BN, Kelly DP, Lopaschuk GD (2006) Chronic activation of PPARalpha is detrimental to cardiac recovery after ischemia. Am J Physiol Heart Circ Physiol 290:H87–H95
Shibata R, Sato K, Pimentel DR, Takemura Y, Kihara S, Ohashi K, Funahashi T, Ouchi N, Walsh K (2005) Adiponectin protects against myocardial ischemia-reperfusion injury through AMPK- and COX-2-dependent mechanisms. Nat Med 11:1096–1103
Shinmura K, Tamaki K, Saito K, Nakano Y, Tobe T, Bolli R (2007) Cardioprotective effects of short-term caloric restriction are mediated by adiponectin via activation of AMP-activated protein kinase. Circulation 116:2809–2817
Sim AT, Hardie DG (1988) The low activity of acetyl-CoA carboxylase in basal and glucagon-stimulated hepatocytes is due to phosphorylation by the AMP-activated protein kinase and not cyclic AMP-dependent protein kinase. FEBS Lett 233:294–298
Stanley WC, Morgan EE, Huang H, McElfresh TA, Sterk JP, Okere IC, Chandler MP, Cheng J, Dyck JR, Lopaschuk GD (2005) Malonyl-CoA decarboxylase inhibition suppresses fatty acid oxidation and reduces lactate production during demand-induced ischemia. Am J Physiol Heart Circ Physiol 289:H2304–H2309
Stanley WC, Recchia FA, Lopaschuk GD (2005) Myocardial substrate metabolism in the normal and failing heart. Physiol Rev 85:1093–1129
Tani M, Neely JR (1990) Na+ accumulation increases Ca2+ overload and impairs function in anoxic rat heart. J Mol Cell Cardiol 22:57–72
Tani M, Neely JR (1989) Role of intracellular Na+ in Ca2+ overload and depressed recovery of ventricular function of reperfused ischemic rat hearts. Possible involvement of H+-Na+ and Na+-Ca2+ exchange. Circ Res 65:1045–1056
Thampy KG (1989) Formation of malonyl coenzyme A in rat heart. Identification and purification of an isozyme of A carboxylase from rat heart. J Biol Chem 264:17631–17634
Timmer JR, van der Horst IC, Ottervanger JP, De Luca G, van ‘t Hof AW, Bilo HJ, Zijlstra F (2004) Glucose-insulin-potassium infusion as adjunctive therapy in myocardial infarction: current evidence and potential mechanisms. Ital Heart J 5:727–731
Ussher JR, Lopaschuk GD (2006) Clinical implications of energetic problems in cardiovascular disease. Heart Metab 32:9–17
Ussher JR, Lopaschuk GD (2008) The malonyl CoA axis as a potential target for treating ischaemic heart disease. Cardiovasc Res 79:259–268
Wang W, Ussher JER, Wang S, Dyck JRB, Lopaschuk GD (2007) Malonyl CoA decarboxylase deficient mice display minimal infarct during in vivo ischemia/reperfusion. J Mol Cell Cardiol 42:S194–S195
Whitmer JT, Idell-Wenger JA, Rovetto MJ, Neely JR (1978) Control of fatty acid metabolism in ischemic and hypoxic hearts. J Biol Chem 253:4305–4309
Young ME, Goodwin GW, Ying J, Guthrie P, Wilson CR, Laws FA, Taegtmeyer H (2001) Regulation of cardiac and skeletal muscle malonyl-CoA decarboxylase by fatty acids. Am J Physiol Endocrinol Metab 280:E471–E479
Yusuf S, Mehta SR, Diaz R, Paolasso E, Pais P, Xavier D, Xie C, Ahmed RJ, Khazmi K, Zhu J, Liu L (2004) Challenges in the conduct of large simple trials of important generic questions in resource-poor settings: the CREATE and ECLA trial program evaluating GIK (glucose, insulin and potassium) and low-molecular-weight heparin in acute myocardial infarction. Am Heart J 148:1068–1078
Zarrinpashneh E, Carjaval K, Beauloye C, Ginion A, Mateo P, Pouleur AC, Horman S, Vaulont S, Hoerter J, Viollet B, Hue L, Vanoverschelde JL, Bertrand L (2006) Role of the alpha2-isoform of AMP-activated protein kinase in the metabolic response of the heart to no-flow ischemia. Am J Physiol Heart Circ Physiol 291:H2875–H2883
Zhang D, Liu ZX, Choi CS, Tian L, Kibbey R, Dong J, Cline GW, Wood PA, Shulman GI (2007) Mitochondrial dysfunction due to long-chain Acyl-CoA dehydrogenase deficiency causes hepatic steatosis and hepatic insulin resistance. Proc Natl Acad Sci USA 104:17075–17080
NHLBI morbidity and mortality chart-book (2004) National Heart, Lung, and Blood Institute, Bethesda, May 2004 (Accessed October 23, 2008, at http://www.nhlbi.nih.gov/resources/docs/cht-book.htm)
Acknowledgments
Supported by a grant from the Canadian Institutes for Health Research Grant to GDL. JRU is a trainee of the Alberta Heritage Foundation for Medical Research and Tomorrow’s Research Cardiovascular Health Professionals (TORCH). GDL is a Medical Scientist of the Alberta Heritage Foundation for Medical Research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ussher, J.R., Lopaschuk, G.D. Targeting malonyl CoA inhibition of mitochondrial fatty acid uptake as an approach to treat cardiac ischemia/reperfusion. Basic Res Cardiol 104, 203–210 (2009). https://doi.org/10.1007/s00395-009-0003-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00395-009-0003-9