Abstract
Introduction
The influence of sex and gender is particularly relevant in cardiovascular diseases (CVD) as well as in several aspects of drug pharmacodynamics and pharmacokinetics. Anatomical and physiological differences between the sexes may influence the activity of many drugs, including the possibility of their interaction with other drugs, bioactive compounds, foods and beverages. Phenolic compounds could interact with our organism at organ, cellular, and molecular levels triggering a preventive action against chronic diseases, including CVD.
Results
This article will review the role of sex on the activity of these bioactive molecules, considering the existence of sex differences in oxidative stress. It describes the pharmacokinetics of phenolic compounds, their effects on vessels, on cardiovascular system, and during development, including the role of nuclear receptors and microbiota.
Conclusions
Although there is a large gap between the knowledge of the sex differences in the phenolic compounds’ activity and safety, and the urgent need for more research, available data underlie the possibility that plant-derived phenolic compounds could differently influence the health of male and female subjects.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Until now, women have been underrepresented or excluded in medicine, except in the field of female reproduction, leading the male body to be considered the “norm”. Currently, there are relevant clinical examples of the impact of the variables sex (biological differences between males and females in terms of genetics, epigenetics, endocrinology, etc.) and gender (sociocultural aspects), including cardiovascular diseases (CVD), diabetes mellitus, autoimmune diseases, and alcoholism ([1, 2] and cited literature). As it is very difficult to separate sex and gender because of their multiple and varied interactions in the time and in different countries and societies, the simultaneous use of the two terms is more appropriate. The influence of sex and gender is particularly relevant in CVD [3, 4] as well as in several aspects of drug pharmacodynamics and pharmacokinetics [5]. There are, in fact, numerous anatomical and physiological differences between the sexes (body dimension and composition, gastric and intestinal differences, metabolism, renal functions) that may influence the activity of many drugs, including the possibility of their interaction with other drugs, bioactive compounds, foods and beverages [6].
Although investigations on sex-related differences have recently increased, and sex and gender are pivotal determinants of health [3], the awareness of their relevance and importance is still lacking. Little is known, for example, about the influence of sex and gender on the imbalance between oxidants and antioxidants, i.e., the so-called oxidative stress [7], although some sex differences have emerged [4, 8, 9].
Oxidative stress is a known hallmark of many diseases, including CVD (ischemic diseases, cardiac hypertrophy, stroke, etc.), and cardiovascular risk factors (CVR), such as hypercholesterolemia, hypertension, diabetes, and atherosclerosis [10, 11]. Moreover, it is important to recall that cardiovascular homeostasis depends on the balance between the generation of nitric oxide (NO) and reactive oxygen species (ROS): NO has beneficial effects against damage induced by ROS, and vice versa, ROS reduces the effect of NO [11].
Oxidizing agents include O2·, peroxynitrite, hydroxyl radicals, and non-radicals, such as hydrogen peroxide (H2O2). They are physiologically generated intracellularly, extracellularly, or in specific intracellular compartments [12] through the activity of numerous enzymes, such as xanthine oxidase, cyclo-oxygenases, lipo-oxygenases, myeloperoxidases, cytochrome P450 mono-oxygenases (CYP), uncoupled NO synthase, peroxidases, and NADPH oxidase [12]. However, the human body has evolved different physiological defences against ROS [7], including superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPx), glutathione transferases (GST), albumin, bilirubin and glutathione (GSH) [8], which act as ROS scavengers. In addition, ROS activate the intracellular molecular pathway of Nrf2/ARE-mediated antioxidant gene expression [7, 13]. However, under pathophysiological conditions, production of ROS might exceed the natural antioxidant defence of the cells. Although oxidative stress plays a crucial role in CVD, antioxidant therapy with supplements of micronutrients, such as vitamin E, has not resulted, to date, in a clear efficacy in the prevention of CVD [14, 15]. Notably, some authors report a slight elevation in the risk of angina pectoris [14].
Another class of food-derived bioactive molecules that acts as antioxidants and thus may have health benefits for CVD is phenolic compounds, which are a ubiquitous class of secondary plant metabolites. Some phenolic compounds are responsible for colour and others for the organoleptic properties of fruits and vegetables. The potential health benefits of phenolic compounds are attributed, in part, to their antioxidant activity [16, 17]. In vitro, they scavenge O2 as well as hydroxyl, peroxyl, alkoxyl radicals, and NO [18], and some of them may also chelate transition metal ions, inhibiting the iron- and copper-catalysed formation of initiating radical species [18]. However, phenolic compounds act through multiple pathways and may interact with different cellular functions at concentrations much lower than that required for effective antioxidant activity [19]. In brief, phenolic compounds bind to both oestrogen receptors (ER), ERα and ERβ [19, 20]. Regrettably, very few papers have addressed the effects of these substances on the androgen receptor (AR) [21].
There are evidences, although not unequivocal, that link high consumption of fruits and vegetables to a lower risk of CVD [22,23,24,25]. Multiple factors could be used to explain the heterogeneity of the results in the literature, and among them, are the gut microbiome, body mass index, age, genetics, the country where people live and the sex [26,27,28,29].
The goal of this review is to demonstrate the effect of sex on the activity of these bioactive molecules.
Phenolic compounds
Phenolic compounds classification
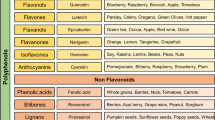
Phenolic compounds are among the most important and abundant phytochemicals in plants and are found in many foods and beverages, such as beverages made from fruits, vegetables and chocolate, as well as in coffee, tea, beer, and wine ([30] and cited literature). Chemically, phenolic compounds are characterized by one or more phenolic rings and are classified into different groups (mainly flavonoids, phenolic acids, stilbenes and lignans) as a function of the number of phenol rings that they contain as well as of the structural elements that bind these rings to others [31] (Fig. 1). In plants, phenolic compounds are mainly conjugated with sugars, forming glycosides. The most studied class, due to its potential and preventive role in degenerative diseases, is that of flavonoids, which are characterized by two aromatic rings connected through an oxygenated heterocycle (Fig. 1). They can be further subdivided into six main subclasses, isoflavones, flavones, flavanols, flavanones, anthocyanidins, and flavonols (catechins and proanthocyanidins) ([30] and cited literature), which differ in the characteristics of the heterocyclic ring.
Sex-dependent bioavailability of phenolic compounds
In foods and beverages, phenolic compounds are mainly stored as a glycone or, mostly, as glycosidic conjugates. In the organism, they are widely metabolized [32]. Briefly, their metabolism starts in the oral cavity, where they come into contact with saliva and bacteria, which may possess glycosidases that induce hydrolysation of glycosylated phenolic compounds [33]. In the intestine, they are further hydrolysed [34] by lactase phlorizin hydrolase and by β-glucosidases of intestinal bacteria (microbiome; the importance of sex on microbiome is described below). The resulting aglycones generally enter into the enterocyte by passive diffusion. However, some particular flavonoids might also be transported into intestinal epithelial cells by the active sodium-dependent glucose transporter SGLT1 and hydrolysed inside the cell by a cytosolic β-glucosidase [35]. Furthermore, some phenolic compounds, such as genistein and daidzein, can also be biotransformed by CYP, which are responsible for the oxidative metabolism of various and numerous endogenous and exogenous molecules. It is well-known that some CYPs are differently expressed in males and females: CYP2B6, CYP2A6, and CYP3A have higher activity in women than in men, while CYP2D6, CYP2E1, and CYP1A2 have slightly higher activity in men than in women ([5, 36, 37] and the references therein). The activity of CYP can be regulated by inhibitors and inducers, including nuclear receptors, such as constitutive androstane receptor (CAR), and pregnane X receptor (PXR), and peroxisome proliferator-activated receptor α (PPARα) [38]. CAR also regulates phase III transporters, such as multidrug resistance-associated proteins 2 and 3. Interestingly, androgens seem to inhibit CAR, and CAR mRNA expression is greater in females [39]. CAR-regulated CYP2B6 has higher activity in women than men and is especially higher in Hispanic females than Caucasian or African–American females [40]. In addition, genetic polymorphisms appear to be sex-specific. For example, the 1459C>T SNP and intron-3 15582C>T SNP are associated with the lowest level of CYP2B6 activity in the livers of females [40]. The above findings suggest the importance of sex and ethnicity in the regulation of CYP2B6 polymorphism. The effects of rutin, myricetin, quercetin, isorhamnetin, p-coumaric acid, gallic acid, and caffeic acid on CYP1A, CYP2A, CYP2E1, and CYP3A are sex-related. In female pigs, rutin, myricetin, quercetin, isorhamnetin, p-coumaric acid, gallic acid, and caffeic acid mainly inhibit the activity of CYP1A. Quercetin inhibition of CYP1A is not sex-dependent, but quercetin-induced inhibition of CYP2E1 only occurs in male pigs. Regarding CYP3A activity, myricetin only inhibits CYP3A in male pigs, whereas isorhamnetin is a competitive inhibitor of CYP3A activity in both sexes [41]. In humans, the sex effect is less clear [42]. Finally, it is relevant to recall that phenolic compounds may regulate the activity of transcription factors. In vitro, genistein, daidzein, and equol can activate human and mouse PXR, with equol being more active compared to human PXR [43].
Another important aspect is that phenolic compounds can interact with the pharmacokinetics of endogenous and exogenous compounds, including drugs [44, 45]. The above results indicate that at least isoflavones may modify the metabolizing activity of CYP in a sex-dependent manner. Thus, more detailed investigations are needed, because many individuals combine drugs and isoflavone-containing supplements.
The aglycones are biotransformed by conjugation (phase II of metabolism) in the small intestine and in the liver. With only a few exceptions, the metabolism of dietary phenolic compounds leads to plasma conjugates (glucuronates or sulphates, with or without O-methylation). Rats, mice and humans differ in plasma metabolites after ingestion of phenolic compounds [46]. Moreover, the levels of these metabolites are also influenced by sex. In particular, in male rats, the main molecule is represented by disulphates, followed by 7-sulpho-4′-glucuronides, while female rats have a higher concentration of 7-glucuronides [46]. The conjugation with glucuronic acid occurs via numerous tissue-specific families of UDP-glucuronosyltransferases (UGTs) ([30] and cited literature). There is some evidence that some UGTs, such as UGTA1, may be influenced by sex [47]. For example, UGT2B17 is more expressed in men than women ([30] and cited literature); therefore, potentially, phenolic compounds might be more conjugated in males than females. In humans and animals, UGTs are induced by foods, such as citrus fruits [48], cruciferous vegetables [49], and soy (e.g., genistein) [50]. Consequentially, increased ingestion of these foods may lead to variable UGT activity, altering substrate metabolism. UGTs are also activated by the aryl hydrocarbon receptor (AhR), hepatocyte nuclear factor 4 alpha, CAR, and PXR [47]. In rodents, their activity seems to depend on steroid hormones [51]. In particular, AhR modulates ERα and ERβ and AR [52]. For example, AhR, which regulates the UGT1A gene, may interact with ERα, suppressing CYP1A1 transcription [53, 54].
Sulphate conjugation occurs through sulphatases (SULTs), which belong to two families: SULT1 and SULT 2. SULT1A enzymes preferentially metabolize phenolic substrates [55] and are regulated by PPARα, RXR, CAR, vitamin D receptor, farnesoid X receptor, retinoid-related orphan receptors, ERs, and oestrogen-related receptors (ERR) [55]. Sex plays a role in the activity of SULTs, and some isoforms are oestrogen-dependent, suggesting a variation in their activity during the menstrual cycle or oestrous phase. Indeed, in rodents, oral bioavailability of genistein appears to be dependent on the oestrous phase and ovariectomy: in ovariectomized female rats, oestrogen administration reduces the oral bioavailability of genistein, mostly via increased sulphation ([30] and cited literature). In the mouse liver, sulphation is more efficient in the female liver versus male liver, with the exception of SULT1c1, which is dominant in male mice [56]. The opposite is observed in rats [57].
Catechol-O-methyltransferase (COMT) plays a role in methylation, a metabolic pathway that is implicated in the metabolism of epigallocatechin and quercetin [58, 59]. Notably, COMT has lower activity (approximately 25%) in women than in men ([30] and cited literature). Considering the sexual dimorphism in COMT activity, sex differences in methylation could be plausible.
Some conjugation reactions might also involve GSH. These reactions may occur either spontaneously or be catalysed by GST [60]. Sex-specific (female mice more than male mice) induction of GST is observed with the flavone 2-phenyl benzopyrone, but not with morin, naringenin, catechin, and quercetin. Notably, the effect depends on the isozyme [61]. Sex differences are also present in the rat liver, which displays a higher expression of GST in males than in females [62]. Finally, phenolic compounds may be subjected to enterohepatic transport in the bile [63].
Some sex differences are observed in daidzein metabolism. Daidzein can be biotransformed in equol ([30] and cited literature). However, this does not occur in all humans (on average, 30–35% among the Caucasian population). Relevantly, after 15 days of soy ingestion, only women develop the capacity to metabolize daidzein into equol ([30] and cited literature), which is more active than daidzein. This is in line with the results of a 2-year study in postmenopausal women. In particular, a major hypocholesterolemic effect is found in women who produce equol versus non-producers. Beyond equol, O-desmethylangolensin can also be produced, and its plasmatic level is higher in men than women [64].
Recently, it has been shown that the activity of some transcription factors may be influenced by circadian rhythms, which in turn, are influenced by sex [65], suggesting the importance of timing on the influence of sex on the metabolism of phenolic compounds.
Excretion of conjugated phenols occurs via renal (7–30%) and biliary (10%) routes ([30, 66] and cited literature). Sex differences are described for some molecules that are eliminated via the renal route: adult female rats eliminate a higher amount of the mono-glucuronoconjugate than mono-sulphoconjugate of apigenin after oral intake [67]. The basal urinary and plasma total phenolic contents are lower in women than in men [68]. Indeed, during 1 month of daily soy ingestion, women initially excreted more isoflavone conjugates in urine than men, but they also experienced a progressive decrease in urinary excretion of genistein and daidzein, unlike men [69]. Urinary isoflavone excretion following ingestion of different soy foods has been examined by Faughnan et al. [70]: they observed that women eliminate more genistein after higher ingestion of soy milk versus textured vegetable protein, while urinary excretion in men is not influenced by the food matrix. Urinary daidzein excretion does not depend on sex or on the food matrix. Finally, equol excretion (in equol producers) requires the food matrix to be higher following textured vegetable protein intake. Because women have lower renal excretion versus men [5], it is plausible that renal excretion of phenolic compounds occurs in a sex-specific manner. Thus, isoflavone metabolism and disposition are affected by the duration of soy ingestion in women, but not in men.
In conclusion, the awareness of bioavailability, metabolism, distribution and excretion of these compounds in humans should be clearly established and linked with their biological effects. The interindividual variability in their pharmacokinetics suggest an interindividual variability in biological response, but interindividual variability in efficacy has been little explored [71, 72]. The understanding of inter-individuality is necessary to clarify their role in wellbeing and health.
Phenolic compounds effects from a sex perspective
Effect on vascular function
The effect of genistein and quercetin on blood pressure was recently reviewed [73, 74]; however, the effect of sex was not analysed. Nevertheless, some sex differences have been described in animals and humans. In short, genistein (600 mg genistein/kg food for 4 weeks) reduces systolic blood pressure, pulse pressure, and cardiac work only in female normotensive mice [75]. The inhibitory effect of the endocannabinoid: anandamide on the contractility induced by noradrenaline is only potentiated by oral genistein in female rats [76]. This effect is prevented by the concomitant administration of the oestrogen receptor antagonist fulvestrant. Phenolic compounds of red wine relax aortic rings, especially in female rats [77], this action is linked with redox-sensitive PI3-kinase/Akt-dependent NO-mediated relaxations, which is higher in the aorta of female than male aorta [77]. In rabbits, genistein and daidzein inhibit the formation of neointimal cells, and genistein is more active in male aortic rings [78].
A recent meta-analysis that included subjects supplemented with flavanols from 18 randomized studies (9 trials enrolled men and women, while 5 and 4 studies included only males and only female subjects, respectively) demonstrates that there are significant decreases in blood pressure, fasting plasma glucose, total cholesterol, low-density lipoproteins, and triglycerides, whereas high-density lipoproteins were significantly increased. A comparison of studies that enrolled only men or only women is inconclusive due to the low number of subjects [79].
The supplementation with flavonoids seems to improve vascular function and reduce reactive protein C, VCAM, and E-selectin, whereas the plasma NO concentration is higher, especially in men with an increased risk of CVD [80]. The SU.VI.MAX study demonstrates that there is an inverse relationship between flavonoids present in the diet and blood pressure in women [81]. These discrepancies could be attributed to the limited consumption of soy in Western populations, to short-term and small-size studies, to ethnicity, and to pharmacokinetics. For example, only 25% of non-Asians and 50% of Asians lack the intestinal bacteria that convert daidzein into equol [82], and it has been reported that individuals who do not have these bacteria may benefit from soy consumption [83]. However, other studies suggest that subjects that are able to produce more equol are more sensitive to the beneficial effects of isoflavone ingestion [84].
Uric acid is the major antioxidant of plasma and hydrophilic environments and is also able to propagate oxidative damage. Moreover, it is considered to be a CVD risk factor [85]. Blueberries are rich in phenolic compounds and are able to decrease uric acid both in men and women, even if the effect lasts for a shorter period in women than in men [86].
A recent paper shows that flavan-3-ol, which is present in dark chocolate, only reduces platelet aggregation and P-selectin expression induced by adenosine diphosphate in men. Moreover, it reduces platelet aggregation induced by thrombin receptor-activating peptide and increases thrombin receptor-activating peptide-induced fibrinogen binding in women [87]. In addition, the ex vivo bleeding time induced by collagen/epinephrine is increased both in women and men. White chocolate only reduces P-selectin expression induced by adenosine diphosphate and elevates collagen/epinephrine-induced ex vivo bleeding time in men [87].
Chronic supplementation with quercetin, one of the most potent antioxidants of plant origin, also significantly reduced systolic blood pressure [88, 89] in women with type 2 diabetes mellitus [90]. Moreover, Alonso and Martinez-Gonzalez [91] showed that the antihypertensive effect of extra-virgin olive oil is only present in men, and Nagata et al. [92] found an inverse correlation between soy intake and diastolic blood pressure in Japanese men as well as a marginally positive association in peri-and postmenopausal women. In humans of both sexes with mild to moderate hypertension, consumption of soy milk for 3 months induces a modest but significant hypotensive effect [93]. The hypotensive effect is also present in healthy normotensive men and postmenopausal women (50–76 years old), in which 3 months of soy dietary supplementation also significantly reduce the lipid profile [94, 95]. However, other authors have not confirmed hypotensive effects in either hypertensive or normotensive subjects [73]. Globally, the above data suggest that sex is important for the effects of phenolic compounds in vessels (Table 1).
In humans, the results of a placebo-controlled intervention study conducted in healthy men showed that quercetin can improve endothelial function by increasing endogenous NO and reducing endothelin-1 production through inhibition of NADPH oxidase and activation of endothelial NO synthase [96].
In addition, soy products improve the lipid profile, especially in Chinese men [97], and two meta-analyses [98, 99] demonstrated that soybean isoflavones ameliorate endothelial function in postmenopausal women. Subgroup analysis shows that this effect is more evident in women who have impaired endothelial function at baseline [98].
Effect on CVD prevention
Studies conducted in animals and humans indicate a potential beneficial effect of flavonoids on heart health. In isolated guinea pig myocytes, an electrophysiological study shows that the genistein effects on the intracellular Ca2+ cycle differs in male and female cells [100]. The results suggest that genistein may afford greater cardioprotection in females than males.
An in vivo study performed on transgenic mice with a mutation in the alpha-myosin heavy chain gene, usually fed with a soy enriched diet, shows that while female mice preserve cardiac contractile function and continue to increase their cardiac mass, male mice develop thin ventricular walls and have poorly contractile hearts[101, 102]. When the diet of these mice were substituted with a casein-based diet, males no longer deteriorate versus cardiomyopathy [103], suggesting that the soy-based diet worsens hypertrophic cardiomyopathy more in male mice than female mice [104]. The supplementation with genistein and daidzein into casein-based diet does not recapitulate the entire cardiac phenotype of the young animals consuming the soy diet, but it augments the hypertrophy in the transgenic males and attenuates the myocardial growth in the females [105]. In addition, the hearts of female mice experience hypertrophy in response to the exercise stimulus regardless of diet [106].
Notably, these results also show that dietary manipulation of the heart can lead to adaptation. These results strongly suggest that caution should be used before recommending oestrogenic compound supplementation, at least in patients with cardiac disease [104].
The beneficial health effects of phytoestrogens are not clearly demonstrated regarding CVD prevention. The American Heart Association reversed its endorsement of soy, because it did not find a significant beneficial effect of soy on lipids, blood pressure, or menopausal changes [107], and it has been observed that dietary isoflavones do not reduce the risk of CVD or stroke in Dutch women [108]. However, some clinical studies indicate a potential beneficial effect of soy intake on cardiovascular risk factors or CVD mortality [109,110,111,112,113,114,115]. The Shanghai Women’s Health Study shows that higher soy protein ingestion is linked with a lower risk of incident CVD in Chinese women [116], whereas there is a positive association in men [117]. In a Japanese study, total soy intake and soy isoflavones are only associated with a lower risk of CVD and stroke in women [118], but this association is not present in another Japanese cohort that included men and women aged > 35 years [119]. In a large cohort study in Chinese men and women, a soy-rich diet is not significantly associated with CVD mortality [120]. Notably, in men, the highest quartile of soy protein intake is linked with a slightly elevated risk of CVD mortality. However, a systematic meta-analysis shows little evidence of the beneficial effects of soy consumption on the risk of stroke and CVD [121].
Flavanols are strong antioxidants in plant foods, and tea is a major dietary source. There is evidence from prospective cohort studies that tea and flavanols are inversely related to stroke incidence [122]. Finally, a systematic review and meta-analysis performed with apple, tea and cocoa containing flavanols in cohorts coming from different countries shows that low-density lipoprotein and body mass index are reduced in women and men, respectively [29], whereas total white cell reduction and triglycerides are not influenced by sex. These results again show a great interindividual response, indicating that beneficial effect may partially depends on sex of individual in combination with other individual characteristic such as genetical, hormonal, and metabolic status.
Phenolic compound mechanisms from a sex perspective
Oxidative stress among males and females
Sex differences in oxidative stress and inflammation have already been reviewed, and they seem to depend on species and tissues and cells ([4, 8, 9, 123,124,125] and cited literature). Oxidative stress and inflammation play a crucial role in the pathogenesis of atherosclerosis vascular inflammation leading to infarction and ischemia. Oxidized low-density lipoproteins (oxLDL) are internalized by macrophage, vascular endothelial and smooth muscle cells, and these cells are the source of foam cells [9, 125]. Internalization of oxLDL activates the inflammasome, which in part is regulated by sexual hormones [125] through ERα, ERβ, and G-protein-coupled receptor-30 (GPR30) (see below) as well as AR. We currently do not understand how the immune response and oxidative stress in women differ from men and in atherosclerosis. However, some differences have been found in animal models and humans.
Oxidative stress seems to be higher in male than female rats, being that the levels of SOD, GPx and lipid peroxidation are lower and higher in males versus females, respectively [126]. In rats, H2O2 and O2· production, at baseline, is lower in females than in males in aortic vascular smooth muscle cells (VSMCs) and isolated aortas [127,128,129] and lipid peroxidation is higher in male cells versus female cells [129]. In addition, male VSMCs have less SOD and CAT activity [127, 129], and the in vivo SOD levels are also lower in male rat carotid arteries than in female ones [127].
Furthermore, sex differences also emerge or persist after different treatments: exposure to NO, for example, only reduces O2· and elevates SOD activity as well as gene expression in male rat VSMCs [127]. In aortic rat VSMCs, UVB radiation significantly increases production of the 4-hydroxynonenal, H2O2 and \({{\text{O}}^ - }_{{\text{2}}}\), the effect of which is larger in male VSMCs than in female VSMCs. Further UVB exposure induces a higher reduction of GPx in female VSMCs [130].
SOD activity is higher in female rat hearts than in male [126], whereas male rats and mice have a higher level of GPx [126, 131]. Moreover, at the cardiac level, in young and aged male rats, cardiac protein oxidation is higher in males than in females, whereas cardiac lipid peroxidation is not different between sexes [132, 133]. In spontaneously hypertensive rats, the SOD, CAT and trolox equivalent antioxidant capacities are higher in males than in females, whereas GPx is not different, while sex-dependent differences are not observed in normotensive and borderline hypertensive rats of both sexes, with lipid peroxidation being similar in both sexes [134]. Finally, sex differences have been described in response to antioxidant therapy. Fortepiani and Reckelhoff [135] demonstrate that the antioxidant tempol decreases blood pressure only in male spontaneously hypertensive rats. In rats, sex differences are also present when enzymes that produce ROS are considered. As a subunit-specific example, Nox1 and Nox4 of NAPH oxidase are higher in male than in female rats, while Nox2 does not differ between males and females [136, 137].
In mice, SOD activity is higher in females than in males in the brain and lung, but not at the renal and cardiac levels [131]. Moreover, CAT activity is similar in the two sexes in the brain, heart, and lung of mice, but is higher in female kidneys [131]. The activities of GPx and glutamate-cysteine ligase (GCL), a key enzyme in the synthesis of GSH, are more elevated in the female mouse kidney and brain versus the male equivalents. By contrast, the activities of GCL and GPx are more elevated in the male heart than in the female heart [131]. Notably, at least in the mouse brain, age increases oxidation, which is greater in females than in males [138].
Nrf2 activates cytoprotective genes during periods of oxidative stress, and interestingly, many of cytoprotective genes are more highly expressed in the livers of female than male mice [139, 140]. Furthermore, male and female Nrf2 knockout mice have different phenotypes, because Nrf2-dependent antioxidant enzymes are only expressed in female bones [141]. Oestrogens can up-regulate Nrf2 and increase the expression of antioxidant enzymes during ischemic and reperfusion injury [142].
In humans, at baseline, H2O2 production is higher in male human umbilical vein endothelial cells (HUVECs) than in female ones [143]. Clinically, the biomarkers of oxidative stress are higher in young men than in women of the same age [144], although this is not a univocal result, because another study reports that women present a higher oxidative status than males [145]. Tóthová et al. [146] demonstrated that MDA and advanced glycation end-products are significantly higher in healthy adult women, whereas advanced oxidation protein and the ferric-reducing ability of plasma are greater in men. Young fertile women present higher levels of MDA than men, whereas no differences are detected between postmenopausal women and men of the same age [147]. Interestingly, when data are corrected for body weight, the difference in the MDA levels between fertile women and young men persists and extends to postmenopausal women and men > 45 years old [147]. The same study shows that protein oxidation does not diverge between men and women, but when the body weight correction is applied to carbonyl groups, fertile and postmenopausal women have higher levels of carbonyls than men of both ages [147].
As previously noted, plant-derived phenolic compounds might protect against heart disease through inhibition of oxidative damage [19]. Flavonoids can be used as an illustrative example of such activity. Their chemical structure is compatible with a one-electron donor activity. They function as antioxidants in vitro in both cell cultures and cell-free systems by scavenging superoxide anion, singlet oxygen, and lipid peroxy-radicals and/or by stabilizing free radicals that are involved in oxidative processes through hydrogenation or complexing with oxidizing species ([32], and cited literature). As a product of this reaction, flavonoids themselves become free radicals, but their conjugated structure allows the remaining orbital electron to be relatively inactive. Consequently, the beneficial effect of bioactive phenolic compounds has been widely attributed to the putative antioxidant capacity of these molecules. Although in vitro experiments clearly sustain the positive influence of phenolic compounds on oxidative stress, the effects of these substances in living systems are inconclusive or could not confirm the in vitro data at all [148]. More recently, the real impact of phenolic compounds as antioxidants has been reconsidered and questioned based on evidence that shows that the molecular basis of their activity is much larger than originally considered [149]. For example, it has been reported that the phenolic compounds of extra-virgin olive oil, in a sex-specific manner, increase glutathione reductase activity and GPx and GSH levels by activating the Nrf2 pathway in rat VSMCs [150]. Moreover, the effect of red wine phenolic compounds on increasing aorta ring relaxation is greater in females than males, probably due to the high expression of endothelial NOS in females [77].
These data may, in part, provide a partial explanation for the variability in the literature that indicates the importance of sex on phenolic compounds effects. Moreover, sex differences may vary according to a specific species, organ or tissue, as well as the studied parameters. Globally, the above findings urge for future detailed studies that focus on the evaluation of the influence of sex on redox homeostasis, also considering that the deterioration of the redox status seems to be a link between psychological stress and cardiovascular risk [151].
Nuclear receptors
Although the affinity of phenolic compounds for both ERα and ERβ is lower than oestrogen, competition-binding studies have confirmed that nutritional molecules (e.g., genistein, coumestrol, daidzein, and equol) showed a distinct preference for ERβ [152]. By contrast, in comparison with genistein, 8-prenylnaringenin, which only occurs in hops, was found to be 100 times more potent as an ERα agonist [153]. The ability to modulate ER activities led to the inclusion of phenolic compounds in the class of selective oestrogen receptor modulators (SERM) [19]. However, accumulating data on phenolic compounds’ action mechanisms indicate that these compounds have a very complex spectrum of activities. Thus, they should not be considered to be “natural” SERMs, which function as ER agonists in some tissues and antagonists in other organs. Rather, phenolic compounds could function as ER agonists on certain pathways (i.e., membrane-initiating pathways) in all organs, eliciting effects downstream of these pathways. We recently defined these substances as mechanism-specific ligands that differentially modify ER signalling and physiological outcomes. Although the available data are currently limited, they may be important in providing a conceptual background for the putative protective effect of phenolic compounds against CVD. Apart from the direct effect on sex-hormone receptors, phenolic compounds could affect the levels of these receptors. As example, quercetin inhibits AR protein levels in a dose-dependent manner [21], while naringenin maintains a high level of ER in cells [154].
To evaluate whether a different susceptibility of male and female sex-hormone receptors to phytochemicals exists, we evaluated the impact of naringenin on rat VSMC motility. Naringenin inhibits VSMC migration, a protective effect against atherosclerosis insurgence in both female- and male-derived cells, via ERβ [155]. Naringenin requires ERβ and p38 activation to affect female VSMC motility. Intriguingly, this naringenin effect is also present in male VSMC in which ERβ is expressed. Thus, these data indicate that ERβ signals are prone to flavonoid modulation in both male and female VSMC [155]. In humans, ERα, ERβ, and GPR30 are differently expressed in males and females, whereas AR presents the same level of expression.
Microbiota
Numerous studies link the gut microbiota (the microorganisms living in the digestive tracts of humans and other animals) to the so-called metabolic syndrome, obesity, type 2 diabetes, and CVD [156,157,158,159]. Metabolic syndrome is also associated with autoimmune disorders, such as diabetes type I [160], depression and anxiety [161].
The dominant phyla in the human gastrointestinal tract are Firmicutes (including Clostridium, Enterococcus, Lactobacillus, Ruminococcus, and Faecalibacterium genera) and Bacteroidetes (including Bacteroides and Prevotella genera) [162, 163], with phyla such as Actinobacteria, Proteobacteria, Synergistetes, Fusobacteria, and Verrucomicrobia is also present [162]. The human adult microbiota is relatively stable from adulthood to old age [163]. Notably, sex differences are described in the gut microbiota [164, 165] and could be implicated in sexual dimorphic patterns in energy and nutritional requirements [166,167,168,169,170]. In particular, an American study demonstrates that Bacteroidetes are lower in the gut of men than women [168]. This is line with a European study that shows that Bacteroides–Prevotella is more prominent in males than females [171], whereas Bacteroides thetaiotaomicron is higher in Chinese men in Chinese women [170]. Indeed, the abundance of the Bacteroides is lower in obese men than obese women [172]. The maternal gut microbiome varies during pregnancy, with bacterial diversity more pronounced during the third trimester, when oestrogens are at maximum levels [173].
In this context, it is important to recall that a two-way interaction exists between phenolic compounds and the gut microbiota, being that the gut microbiome is involved in the metabolism of phenolic compounds (i.e., bioavailability), and the phenols and their microbial metabolites can promote factors for proliferation of beneficial gut inhabitants and can inhibit the pathogenic species [174, 175]. Microbiota can metabolize oestrogen-like compounds, which in turn, may induce the proliferation and growth of certain types of bacteria. For instance, daidzein is metabolized by gut bacteria to O-desmethylangolensin and S-equol [176], which can activate ERα or ERβ[177, 178]. Additionally, genistein and glycitin can vary the composition of the faecal bacterial community in postmenopausal women by increasing and decreasing the concentration of the Bifidobacterium and Clostridiaceae, respectively [176, 177]. Notably, Clostridium scindens may convert glucocorticoids into androgens by side-chain cleavage [179]. Intriguingly, some bacteria that have β-glucuronidase activity might increase intestinal reabsorption of oestrogens (see above). As a whole, the modulation exerted by phenolic compounds on the composition of the gut microbiota should be considered to be possible novel action mechanisms that are activated by these molecules to exert effects on cardiovascular functions.
Phenolic compounds’ effect during development
Early life experiences have profound influences on health in adult life; therefore, we report the sex differences with phenolic compounds observed in early life, even if only a few studies are available. Daidzein and genistein are detected in amniotic fluid, and genistein can cross human (in vitro) and rat (in vivo) placentae [180,181,182]. Notably, amniotic fluid containing female foetuses can store more daidzein and genistein than amniotic fluid containing male foetuses [183]. Moreover, a mother of a female baby presents a higher total antioxidant status and lower plasma hydroperoxides than a mother of a male baby: umbilical cords obtained from female babies have, indeed, a higher total antioxidant capacity, lower plasma membrane hydroperoxides and higher CAT, GPx, and SOD activities than male samples [184]. It has been observed that HUVECs obtained from umbilical cord of males produce more H2O2 than female ones [143]. The importance of the perinatal environment on the development of modifying the risk profile for disease in adulthood is now well-recognized [185]. Epidemiological studies underline the association between perinatal factors, such as breastfeeding versus formula milk. It is relevant to recall that soy-based milk composes 13% of the infant formula marketed in the United States [186]. Neonates fed with soy milk have higher soy isoflavones than Japanese men consuming a soy-based diet and much higher soy isoflavones than in US adults [186, 187].
Animal studies demonstrate that ingestion of genistein in the first 22 days after birth increases the fat/lean mass ratio, fat mass, adipocyte size and number as well as decreases the muscle fibre perimeter in females [188]. These findings suggest that postnatal exposure induces sex-specific effects. Indeed, environmentally relevant ingestion of genistein may disrupt female reproductive development and function [189], as genistein is a reproductive toxicant and endocrine disruptor in rodents [190, 191]. Some consequences (irregular oestrous cycles, early reproductive senescence, infertility) appear late in life [189]. Male rats also appear to be sensitive to phytoestrogens during development, because the central nervous system-gonadal axis and male sexual behaviour are altered [192]. All of these data, as also demonstrated by others [193], suggest that differences in male and female susceptibility to phytoestrogen could be present.
Conclusions
CVD is the first cause of death in developed countries; thus, primary prevention of CVD is a major public-health priority. Regarding phenolic compounds, a recent meta-analysis shows that some phenolic compounds, such as flavanols, may be beneficial in the prevention of CVD. This is not an univocal result, because although CVD is associated with increased oxidative stress, data on the efficacy of antioxidant therapy is still lacking [14]. In addition, many, if not all, trials are of low quality, and this does not permit any firm conclusions.
Considering that dietary intake is not sufficient to reach pharmacological concentrations of phenolic compounds, the problem of dosage must be resolved. Data on the influence of sex on the activity of phenolic compounds are even scarcer; nevertheless, sex differences in the biotransformation of phenolic compounds have been described. The difficulty of reaching a firm conclusion is further aggravated by the fact that sex differences in oxidative stress and the response to antioxidants are species (mouse, rat, humans)-specific, making data not transferable from animals to men and women. It is evident that there are still large gaps in our knowledge regarding the sex differences of phenolic compounds’ activity and safety, and significantly more research is urgently required.
References
Legato MJ, Johnson PA, Manson JE (2016) Consideration of sex differences in medicine to improve health care and patient outcomes. JAMA 316(18):1865–1866
Agabio R, Pisanu C, Gessa GL, Franconi F (2016) Sex differences in alcohol use disorder. Curr Med Chem 24:2661–2670
Regitz-Zagrosek V, Oertelt-Prigione S, Prescott E, Franconi F, Gerdts E, Foryst-Ludwig A, Maas AH, Kautzky-Willer A, Knappe-Wegner D, Kintscher U, Ladwig KH, Schenck-Gustafsson K, Stangl V (2016) Gender in cardiovascular diseases: impact on clinical manifestations, management, and outcomes. Eur Heart J 37(1):24–34
Kander MC, Cui Y, Liu Z (2016) Gender difference in oxidative stress: a new look at the mechanisms for cardiovascular diseases. J Cell Mol Med:1024–1032
Franconi F, Campesi I (2014) Sex and gender influences on pharmacological response: an overview. Expert Rev Clin Pharmacol 7(4):469–485
Marino M, Masella R, Bulzomi P, Campesi I, Malorni W, Franconi F (2011) Nutrition and human health from a sex-gender perspective. Mol Asp Med 32(1):1–70
Lemire J, Alhasawi A, Appanna VP, Tharmalingam S, Appanna VD (2017) Metabolic defense against oxidative stress: the road less travelled-so far. J Appl Microbiol 173:798–809
Malorni W, Campesi I, Straface E, Vella S, Franconi F (2007) Redox features of the cell: a gender perspective. Antioxid Redox Signal 9(11):1779–1801
Franconi F, Rosano G, Basili S, Montella A, Campesi I (2017) Human cells involved in atherosclerosis have a sex. Int J Cardiol 228:983–1001
Egea J, Fabregat I, Frapart YM, Ghezzi P, Gorlach A, Kietzmann T, Kubaichuk K, Knaus UG, Lopez MG, Olaso-Gonzalez G, Petry A, Schulz R, Vina J, Winyard P, Abbas K, Ademowo OS, Afonso CB, Andreadou I, Antelmann H, Antunes F, Aslan M, Bachschmid MM, Barbosa RM, Belousov V, Berndt C, Bernlohr D, Bertran E, Bindoli A, Bottari SP, Brito PM, Carrara G, Casas AI, Chatzi A, Chondrogianni N, Conrad M, Cooke MS, Costa JG, Cuadrado A, My-Chan Dang P, De Smet B, Debelec-Butuner B, Dias IHK, Dunn JD, Edson AJ, El Assar M, El-Benna J, Ferdinandy P, Fernandes AS, Fladmark KE, Forstermann U, Giniatullin R, Giricz Z, Gorbe A, Griffiths H, Hampl V, Hanf A, Herget J, Hernansanz-Agustin P, Hillion M, Huang J, Ilikay S, Jansen-Durr P, Jaquet V, Joles JA, Kalyanaraman B, Kaminskyy D, Karbaschi M, Kleanthous M, Klotz LO, Korac B, Korkmaz KS, Koziel R, Kracun D, Krause KH, Kren V, Krieg T, Laranjinha J, Lazou A, Li H, Martinez-Ruiz A, Matsui R, McBean GJ, Meredith SP, Messens J, Miguel V, Mikhed Y, Milisav I, Milkovic L, Miranda-Vizuete A, Mojovic M, Monsalve M, Mouthuy PA, Mulvey J, Munzel T, Muzykantov V, Nguyen ITN, Oelze M, Oliveira NG, Palmeira CM, Papaevgeniou N, Pavicevic A, Pedre B, Peyrot F, Phylactides M, Pircalabioru GG, Pitt AR, Poulsen HE, Prieto I, Rigobello MP, Robledinos-Anton N, Rodriguez-Manas L, Rolo AP, Rousset F, Ruskovska T, Saraiva N, Sasson S, Schroder K, Semen K, Seredenina T, Shakirzyanova A, Smith GL, Soldati T, Sousa BC, Spickett CM, Stancic A, Stasia MJ, Steinbrenner H, Stepanic V, Steven S, Tokatlidis K, Tuncay E, Turan B, Ursini F, Vacek J, Vajnerova O, Valentova K, Van Breusegem F, Varisli L, Veal EA, Yalcin AS, Yelisyeyeva O, Zarkovic N, Zatloukalova M, Zielonka J, Touyz RM, Papapetropoulos A, Grune T, Lamas S, Schmidt H, Di Lisa F, Daiber A (2017) European contribution to the study of ROS: a summary of the findings and prospects for the future from the COST action BM1203 (EU-ROS). Redox Biol 13:94–162
Ritchie RH, Drummond GR, Sobey CG, De Silva TM, Kemp-Harper BK (2017) The opposing roles of NO and oxidative stress in cardiovascular disease. Pharmacol Res 116:57–69
Holmstrom KM, Finkel T (2014) Cellular mechanisms and physiological consequences of redox-dependent signalling. Nat Rev Mol Cell Biol 15(6):411–421
Jaiswal AK (2004) Nrf2 signaling in coordinated activation of antioxidant gene expression. Free Radic Biol Med 36(10):1199–1207
Myung SK, Ju W, Cho B, Oh SW, Park SM, Koo BK, Park BJ (2013) Efficacy of vitamin and antioxidant supplements in prevention of cardiovascular disease: systematic review and meta-analysis of randomised controlled trials. BMJ 346:f10
Vivekananthan DP, Penn MS, Sapp SK, Hsu A, Topol EJ (2003) Use of antioxidant vitamins for the prevention of cardiovascular disease: meta-analysis of randomised trials. Lancet 361(9374):2017–2023
Del Rio D, Rodriguez-Mateos A, Spencer JP, Tognolini M, Borges G, Crozier A (2013) Dietary (poly)phenolics in human health: structures, bioavailability, and evidence of protective effects against chronic diseases. Antioxid Redox Signal 18(14):1818–1892
Rodriguez-Mateos A, Vauzour D, Krueger CG, Shanmuganayagam D, Reed J, Calani L, Mena P, Del Rio D, Crozier A (2014) Bioavailability, bioactivity and impact on health of dietary flavonoids and related compounds: an update. Arch Toxicol 88(10):1803–1853
Cueva C, Gil-Sanchez I, Ayuda-Duran B, Gonzalez-Manzano S, Gonzalez-Paramas AM, Santos-Buelga C, Bartolome B, Moreno-Arribas MV (2017) An integrated view of the effects of wine polyphenols and their relevant metabolites on gut and host health. Molecules 22(1):99
Virgili F, Marino M (2008) Regulation of cellular signals from nutritional molecules: a specific role for phytochemicals, beyond antioxidant activity. Free Radic Biol Med 45(9):1205–1216
Pandjaitan N, Hettiarachchy N, Ju ZY, Crandall P, Sneller C, Dombek D (2000) Evaluation of genistin and genistein contents in soybean varieties and soy protein concentrate prepared with three basic methods. J Food Sci 65:399–402
Marcoccia D, Pellegrini M, Fiocchetti M, Lorenzetti S, Marino M (2017) Food components and contaminants as (anti)androgenic molecules. Genes Nutr 12:6
Woodside JV, Young IS, McKinley MC (2013) Fruit and vegetable intake and risk of cardiovascular disease. Proc Nutr Soc 72(4):399–406
Wang X, Ouyang Y, Liu J, Zhu M, Zhao G, Bao W, Hu FB (2014) Fruit and vegetable consumption and mortality from all causes, cardiovascular disease, and cancer: systematic review and dose-response meta-analysis of prospective cohort studies. BMJ 349:g4490
Rangel-Huerta OD, Pastor-Villaescusa B, Aguilera CM, Gil A (2015) A systematic review of the efficacy of bioactive compounds in cardiovascular disease: phenolic compounds. Nutrients 7(7):5177–5216
Anthony MS, Clarkson TB, Bullock BC, Wagner JD (1997) Soy protein versus soy phytoestrogens in the prevention of diet-induced coronary artery atherosclerosis of male cynomolgus monkeys. Arterioscler Thromb Vasc Biol 17(11):2524–2531
Chong MF, Macdonald R, Lovegrove JA (2010) Fruit polyphenols and CVD risk: a review of human intervention studies. Br J Nutr 104(Suppl 3):S28–S39
Alissa EM, Ferns GA (2017) Dietary fruits and vegetables and cardiovascular diseases risk. Crit Rev Food Sci Nutr 57(9):1950–1962
Guo Y, Bruno RS (2015) Endogenous and exogenous mediators of quercetin bioavailability. J Nutr Biochem 26(3):201–210
González-Sarrías A, Combet E, Pinto P, Mena P, Dall’Asta M, Garcia-Aloy M, Rodríguez-Mateos A, Gibney ER, Dumont J, Massaro M, Sánchez-Meca J, Morand C, García-Conesa MT (2017) A systematic review and meta-analysis of the effects of flavanol-containing tea, cocoa and apple products on body composition and blood lipids: exploring the factors responsible for variability in their efficacy. Nutrients 9:746
Campesi I, Romani A, Marino M, Franconi F (2014) Phenolic compounds from a sex-gender perspective. In: Romani A, Lattanzio S, Quideau S (eds) Recent advances in polyphenol research. Wiley, Chichester, pp 327–339
Quideau S, Deffieux D, Douat-Casassus C, Pouysegu L (2011) Plant polyphenols: chemical properties, biological activities, and synthesis. Angew Chem Int Ed Engl 50(3):586–621
Galluzzo P, Marino M (2006) Nutritional flavonoids impact on nuclear and extranuclear estrogen receptor activities. Genes Nutr 1(3–4):161–176
Walle T, Browning AM, Steed LL, Reed SG, Walle UK (2005) Flavonoid glucosides are hydrolyzed and thus activated in the oral cavity in humans. J Nutr 135(1):48–52
Marin L, Miguelez EM, Villar CJ, Lombo F (2015) Bioavailability of dietary polyphenols and gut microbiota metabolism: antimicrobial properties. Biomed Res Int 2015:905215
Gee JM, DuPont MS, Day AJ, Plumb GW, Williamson G, Johnson IT (2000) Intestinal transport of quercetin glycosides in rats involves both deglycosylation and interaction with the hexose transport pathway. J Nutr 130(11):2765–2771
Franconi F, Brunelleschi S, Steardo L, Cuomo V (2007) Gender differences in drug responses. Pharmacol Res 55(2):81–95
Franconi F, Chessa MS,ES, Rosano R G (2011) Pharmacokinetics and pharmacodynamics: the role of sex and gender. In: Oertelt-Prigione S, Regitz-Zagrosek V (eds) Sex and gender aspects in clinical medicine. Springer, London, pp 183–194
Gerbal-Chaloin S, Iankova I, Maurel P, Daujat-Chavanieu M (2013) Nuclear receptors in the cross-talk of drug metabolism and inflammation. Drug Metab Rev 45(1):122–144
Petrick JS, Klaassen CD (2007) Importance of hepatic induction of constitutive androstane receptor and other transcription factors that regulate xenobiotic metabolism and transport. Drug Metab Dispos 35(10):1806–1815
Lamba V, Lamba J, Yasuda K, Strom S, Davila J, Hancock ML, Fackenthal JD, Rogan PK, Ring B, Wrighton SA, Schuetz EG (2003) Hepatic CYP2B6 expression: gender and ethnic differences and relationship to CYP2B6 genotype and CAR (constitutive androstane receptor) expression. J Pharmacol Exp Ther 307(3):906–922
Ekstrand B, Rasmussen MK, Woll F, Zlabek V, Zamaratskaia G (2015) In vitro gender-dependent inhibition of porcine cytochrome p450 activity by selected flavonoids and phenolic acids. Biomed Res Int 2015:387918
Ostlund J, Zlabek V, Zamaratskaia G (2017) In vitro inhibition of human CYP2E1 and CYP3A by quercetin and myricetin in hepatic microsomes is not gender dependent. Toxicology 381:10–18
Li Y, Ross-Viola JS, Shay NF, Moore DD, Ricketts ML (2009) Human CYP3A4 and murine Cyp3A11 are regulated by equol and genistein via the pregnane X receptor in a species-specific manner. J Nutr 139(5):898–904
Wahajuddin SN, Taneja I, Arora S, Raju KS, Siddiqui N (2013) Disposition of pharmacologically active dietary isoflavones in biological systems. Curr Drug Metab 14(4):369–380
Zhou S, Gao Y, Jiang W, Huang M, Xu A, Paxton JW (2003) Interactions of herbs with cytochrome P450. Drug Metab Rev 35(1):35–98
Soukup ST, Helppi J, Muller DR, Zierau O, Watzl B, Vollmer G, Diel P, Bub A, Kulling SE (2016) Phase II metabolism of the soy isoflavones genistein and daidzein in humans, rats and mice: a cross-species and sex comparison. Arch Toxicol 90(6):1335–1347
Strassburg CP, Kalthoff S, Ehmer U (2008) Variability and function of family 1 uridine-5′-diphosphate glucuronosyltransferases (UGT1A). Crit Rev Clin Lab Sci 45(6):485–530
Saracino MR, Bigler J, Schwarz Y, Chang JL, Li S, Li L, White E, Potter JD, Lampe JW (2009) Citrus fruit intake is associated with lower serum bilirubin concentration among women with the UGT1A1*28 polymorphism. J Nutr 139(3):555–560
Navarro SL, Peterson S, Chen C, Makar KW, Schwarz Y, King IB, Li SS, Li L, Kestin M, Lampe JW (2009) Cruciferous vegetable feeding alters UGT1A1 activity: diet- and genotype-dependent changes in serum bilirubin in a controlled feeding trial. Cancer Prev Res (Phila) 2(4):345–352
Saracino MR, Lampe JW (2007) Phytochemical regulation of UDP-glucuronosyltransferases: implications for cancer prevention. Nutr Cancer 59(2):121–141
Zhang Y, Klein K, Sugathan A, Nassery N, Dombkowski A, Zanger UM, Waxman DJ (2011) Transcriptional profiling of human liver identifies sex-biased genes associated with polygenic dyslipidemia and coronary artery disease. PLoS One 6(8):e23506
Matthews J, Wihlen B, Thomsen J, Gustafsson JA (2005) Aryl hydrocarbon receptor-mediated transcription: ligand-dependent recruitment of estrogen receptor alpha to 2,3,7,8-tetrachlorodibenzo-p-dioxin-responsive promoters. Mol Cell Biol 25(13):5317–5328
Monostory K, Pascussi JM, Kobori L, Dvorak Z (2009) Hormonal regulation of CYP1A expression. Drug Metab Rev 41(4):547–572
Ohtake F, Takeyama K, Matsumoto T, Kitagawa H, Yamamoto Y, Nohara K, Tohyama C, Krust A, Mimura J, Chambon P, Yanagisawa J, Fujii-Kuriyama Y, Kato S (2003) Modulation of oestrogen receptor signalling by association with the activated dioxin receptor. Nature 423(6939):545–550
Runge-Morris M, Kocarek TA, Falany CN (2013) Regulation of the cytosolic sulfotransferases by nuclear receptors. Drug Metab Rev 45(1):15–33
Alnouti Y, Klaassen CD (2011) Mechanisms of gender-specific regulation of mouse sulfotransferases (Sults). Xenobiotica 41(3):187–197
Yang CH, Tang L, Lv C, Ye L, Xia BJ, Hu M, Liu ZQ (2011) Sulfation of selected mono-hydroxyflavones by sulfotransferases in vitro: a species and gender comparison. J Pharm Pharmacol 63(7):967–970
Wang P, Heber D, Henning SM (2012) Quercetin increased bioavailability and decreased methylation of green tea polyphenols in vitro and in vivo. Food Funct 3(6):635–642
Manach C, Scalbert A, Morand C, Remesy C, Jimenez L (2004) Polyphenols: food sources and bioavailability. Am J Clin Nutr 79(5):727–747
Hong YJ, Mitchell AE (2004) Metabolic profiling of flavonol metabolites in human urine by liquid chromatography and tandem mass spectrometry. J Agric Food Chem 52(22):6794–6801
Mitchell AE, Burns SA, Rudolf JL (2007) Isozyme- and gender-specific induction of glutathione S-transferases by flavonoids. Arch Toxicol 81(11):777–784
Igarashi T, Satoh T, Ueno K, Kitagawa H (1983) Sex-related difference in the hepatic glutathione level and related enzyme activities in rat. J Biochem 93(1):33–36
Donovan JL, Manach C, Faulks RM, Kroon PA (2006) Absorption and metabolism of dietary plant secondary metabolites. In: Crozier A, Clifford MN, Ashihara H (eds) Plant secondary metabolites: occurrence, structure and role in the human diet. Wiley Online Library, Hoboken, pp 303–351
Wiseman H, Casey K, Bowey EA, Duffy R, Davies M, Rowland IR, Lloyd AS, Murray A, Thompson R, Clarke DB (2004) Influence of 10 wk of soy consumption on plasma concentrations and excretion of isoflavonoids and on gut microflora metabolism in healthy adults. Am J Clin Nutr 80(3):692–699
Lu YF, Jin T, Xu Y, Zhang D, Wu Q, Zhang YK, Liu J (2013) Sex differences in the circadian variation of cytochrome p450 genes and corresponding nuclear receptors in mouse liver. Chronobiol Int 30(9):1135–1143
Kelly GE, Nelson C, Waring MA, Joannou GE, Reeder AY (1993) Metabolites of dietary (soya) isoflavones in human urine. Clin Chim Acta 223(1–2):9–22
Gradolatto A, Basly JP, Berges R, Teyssier C, Chagnon MC, Siess MH, Canivenc-Lavier MC (2005) Pharmacokinetics and metabolism of apigenin in female and male rats after a single oral administration. Drug Metab Dispos 33(1):49–54
Wruss J, Lanzerstorfer P, Huemer S, Himmelsbach M, Mangge H, Hoglinger O, Weghuber D, Weghuber J (2015) Differences in pharmacokinetics of apple polyphenols after standardized oral consumption of unprocessed apple juice. Nutr J 14:32–42
Lu LJ, Anderson KE (1998) Sex and long-term soy diets affect the metabolism and excretion of soy isoflavones in humans. Am J Clin Nutr 68(6 Suppl):1500S–1504S
Faughnan MS, Hawdon A, Ah-Singh E, Brown J, Millward DJ, Cassidy A (2004) Urinary isoflavone kinetics: the effect of age, gender, food matrix and chemical composition. Br J Nutr 91(4):567–574
Milenkovic D, Morand C, Cassidy A, Konic-Ristic A, Tomas-Barberan F, Ordovas JM, Kroon P, De Caterina R, Rodriguez-Mateos A (2017) Interindividual variability in biomarkers of cardiometabolic health after consumption of major plant-food bioactive compounds and the determinants involved. Adv Nutr 8(4):558–570
Manach C, Milenkovic D, Van de Wiele T, Rodriguez-Mateos A, de Roos B, Garcia-Conesa MT, Landberg R, Gibney ER, Heinonen M, Tomas-Barberan F, Morand C (2017) Addressing the inter-individual variation in response to consumption of plant food bioactives: towards a better understanding of their role in healthy aging and cardiometabolic risk reduction. Mol Nutr Food Res 61:(6)
Sureda A, Sanches Silva A, Sanchez-Machado DI, Lopez-Cervantes J, Daglia M, Nabavi SF, Nabavi SM (2017) Hypotensive effects of genistein: from chemistry to medicine. Chem Biol Interact 268:37–46
Serban MC, Sahebkar A, Zanchetti A, Mikhailidis DP, Howard G, Antal D, Andrica F, Ahmed A, Aronow WS, Muntner P, Lip GY, Graham I, Wong N, Rysz J, Banach M (2016) Effects of quercetin on blood pressure: a systematic review and meta-analysis of randomized controlled trials. J Am Heart Assoc 5(7):e00273
Al-Nakkash L, Martin JB, Petty D, Lynch SM, Hamrick C, Lucy D, Robinson J, Peterson A, Rubin LJ, Broderick TL (2012) Dietary genistein induces sex-dependent effects on murine body weight, serum profiles, and vascular function of thoracic aortae. Gend Med 9(5):295–308
Peroni RN, Abramoff T, Neuman I, Podesta EJ, Adler-Graschinsky E (2012) Phytoestrogens enhance the vascular actions of the endocannabinoid anandamide in mesenteric beds of female rats. Int J Hypertens 2012:647856
Kane MO, Anselm E, Rattmann YD, Auger C, Schini-Kerth VB (2009) Role of gender and estrogen receptors in the rat aorta endothelium-dependent relaxation to red wine polyphenols. Vascul Pharmacol 51(2–3):140–146
Finking G, Wohlfrom M, Lenz C, Wolkenhauer M, Eberle C, Hanke H (1999) The phytoestrogens Genistein and Daidzein, and 17 beta-estradiol inhibit development of neointima in aortas from male and female rabbits in vitro after injury. Coron Artery Dis 10(8):607–615
Menezes R, Rodriguez-Mateos A, Kaltsatou A, Gonzalez-Sarrias A, Greyling A, Giannaki C, Andres-Lacueva C, Milenkovic D, Dumont ER, Schar M, Garcia-Aloy M, Palma-Duran SA, Ruskovska T, Maksimova V, Combet E, Pinto P (2017) Impact of flavonols on cardiometabolic biomarkers: a meta-analysis of randomized controlled human trials to explore the role of inter-individual variability. Nutrients 9(2):117
Macready AL, George TW, Chong MF, Alimbetov DS, Jin Y, Vidal A, Spencer JP, Kennedy OB, Tuohy KM, Minihane AM, Gordon MH, Lovegrove JA (2014) Flavonoid-rich fruit and vegetables improve microvascular reactivity and inflammatory status in men at risk of cardiovascular disease—FLAVURS: a randomized controlled trial. Am J Clin Nutr 99(3):479–489
Mennen LI, Sapinho D, de Bree A, Arnault N, Bertrais S, Galan P, Hercberg S (2004) Consumption of foods rich in flavonoids is related to a decreased cardiovascular risk in apparently healthy French women. J Nutr 134(4):923–926
Setchell KD, Clerici C (2010) Equol: pharmacokinetics and biological actions. J Nutr 140(7):1363S–1368S
Setchell KD, Brown NM, Lydeking-Olsen E (2002) The clinical importance of the metabolite equol-a clue to the effectiveness of soy and its isoflavones. J Nutr 132(12):3577–3584
Hazim S, Curtis PJ, Schar MY, Ostertag LM, Kay CD, Minihane AM, Cassidy A (2016) Acute benefits of the microbial-derived isoflavone metabolite equol on arterial stiffness in men prospectively recruited according to equol producer phenotype: a double-blind randomized controlled trial. Am J Clin Nutr 103(3):694–702
Lippi G, Montagnana M, Franchini M, Favaloro EJ, Targher G (2008) The paradoxical relationship between serum uric acid and cardiovascular disease. Clin Chim Acta 392(1–2):1–7
Cheatham CL, Vazquez-Vidal I, Medlin A, Voruganti VS (2016) Blueberry consumption affects serum uric acid concentrations in older adults in a sex-specific manner. Antioxidants (Basel) 5 (4)
Ostertag LM, Kroon PA, Wood S, Horgan GW, Cienfuegos-Jovellanos E, Saha S, Duthie GG, de Roos B (2013) Flavan-3-ol-enriched dark chocolate and white chocolate improve acute measures of platelet function in a gender-specific way—a randomized-controlled human intervention trial. Mol Nutr Food Res 57(2):191–202
Egert S, Bosy-Westphal A, Seiberl J, Kurbitz C, Settler U, Plachta-Danielzik S, Wagner AE, Frank J, Schrezenmeir J, Rimbach G, Wolffram S, Muller MJ (2009) Quercetin reduces systolic blood pressure and plasma oxidised low-density lipoprotein concentrations in overweight subjects with a high-cardiovascular disease risk phenotype: a double-blinded, placebo-controlled cross-over study. Br J Nutr 102(7):1065–1074
Edwards RL, Lyon T, Litwin SE, Rabovsky A, Symons JD, Jalili T (2007) Quercetin reduces blood pressure in hypertensive subjects. J Nutr 137(11):2405–2411
Zahedi M, Ghiasvand R, Feizi A, Asgari G, Darvish L (2013) Does quercetin improve cardiovascular risk factors and inflammatory biomarkers in women with type 2 diabetes: a double-blind randomized controlled clinical trial. Int J Prev Med 4(7):777–785
Alonso A, Martinez-Gonzalez MA (2004) Olive oil consumption and reduced incidence of hypertension: the SUN study. Lipids 39(12):1233–1238
Nagata C, Shimizu H, Takami R, Hayashi M, Takeda N, Yasuda K (2003) Association of blood pressure with intake of soy products and other food groups in Japanese men and women. Prev Med 36(6):692–697
Rivas M, Garay RP, Escanero JF, Cia P Jr, Cia P, Alda JO (2002) Soy milk lowers blood pressure in men and women with mild to moderate essential hypertension. J Nutr 132(7):1900–1902
Teede HJ, Dalais FS, Kotsopoulos D, Liang YL, Davis S, McGrath BP (2001) Dietary soy has both beneficial and potentially adverse cardiovascular effects: a placebo-controlled study in men and postmenopausal women. J Clin Endocrinol Metab 86(7):3053–3060
Liang YL, Teede H, Dalais F, McGrath BP (2006) The effects of phytoestrogen on blood pressure and lipids in healthy volunteers. Zhonghua Xin Xue Guan Bing Za Zhi 34(8):726–729
Loke WM, Hodgson JM, Proudfoot JM, McKinley AJ, Puddey IB, Croft KD (2008) Pure dietary flavonoids quercetin and (−)-epicatechin augment nitric oxide products and reduce endothelin-1 acutely in healthy men. Am J Clin Nutr 88(4):1018–1025
Ho SC, Woo JL, Leung SS, Sham AL, Lam TH, Janus ED (2000) Intake of soy products is associated with better plasma lipid profiles in the Hong Kong Chinese population. J Nutr 130(10):2590–2593
Li SH, Liu XX, Bai YY, Wang XJ, Sun K, Chen JZ, Hui RT (2010) Effect of oral isoflavone supplementation on vascular endothelial function in postmenopausal women: a meta-analysis of randomized placebo-controlled trials. Am J Clin Nutr 91(2):480–486
Beavers DP, Beavers KM, Miller M, Stamey J, Messina MJ (2012) Exposure to isoflavone-containing soy products and endothelial function: a Bayesian meta-analysis of randomized controlled trials. Nutr Metab Cardiovasc Dis 22(3):182–191
Liew R, Stagg MA, Chan J, Collins P, MacLeod KT (2004) Gender determines the acute actions of genistein on intracellular calcium regulation in the guinea-pig heart. Cardiovasc Res 61(1):66–76
Olsson MC, Palmer BM, Leinwand LA, Moore RL (2001) Gender and aging in a transgenic mouse model of hypertrophic cardiomyopathy. Am J Physiol Heart Circ Physiol 280(3):H1136-1144
Vikstrom KL, Factor SM, Leinwand LA (1996) Mice expressing mutant myosin heavy chains are a model for familial hypertrophic cardiomyopathy. Mol Med 2(5):556–567
Haines CD, Harvey PA, Leinwand LA (2012) Estrogens mediate cardiac hypertrophy in a stimulus-dependent manner. Endocrinology 153(9):4480–4490
Haines CD, Harvey PA, Luczak ED, Barthel KK, Konhilas JP, Watson PA, Stauffer BL, Leinwand LA (2012) Estrogenic compounds are not always cardioprotective and can be lethal in males with genetic heart disease. Endocrinology 153(9):4470–4479
Stauffer BL, Konhilas JP, Luczak ED, Leinwand LA (2006) Soy diet worsens heart disease in mice. J Clin Investig 116(1):209–216
Konhilas JP, Chen H, Luczak E, McKee LA, Regan J, Watson PA, Stauffer BL, Khalpey ZI, McKinsey TA, Horn T, LaFleur B, Leinwand LA (2015) Diet and sex modify exercise and cardiac adaptation in the mouse. Am J Physiol Heart Circ Physiol 308(2):H135-145
Sacks FM, Lichtenstein A, Van Horn L, Harris W, Kris-Etherton P, Winston M (2006) Soy protein, isoflavones, and cardiovascular health: a summary of a statement for professionals from the american heart association nutrition committee. Arterioscler Thromb Vasc Biol 26(8):1689–1692
van der Schouw YT, Kreijkamp-Kaspers S, Peeters PH, Keinan-Boker L, Rimm EB, Grobbee DE (2005) Prospective study on usual dietary phytoestrogen intake and cardiovascular disease risk in Western women. Circulation 111(4):465–471
Maskarinec G, Aylward AG, Erber E, Takata Y, Kolonel LN (2008) Soy intake is related to a lower body mass index in adult women. Eur J Nutr 47(3):138–144
Anderson JW, Johnstone BM, Cook-Newell ME (1995) Meta-analysis of the effects of soy protein intake on serum lipids. N Engl J Med 333(5):276–282
Welty FK, Lee KS, Lew NS, Zhou JR (2007) Effect of soy nuts on blood pressure and lipid levels in hypertensive, prehypertensive, and normotensive postmenopausal women. Arch Intern Med 167(10):1060–1067
Nagata C, Takatsuka N, Kurisu Y, Shimizu H (1998) Decreased serum total cholesterol concentration is associated with high intake of soy products in Japanese men and women. J Nutr 128(2):209–213
Cheng SY, Shaw NS, Tsai KS, Chen CY (2004) The hypoglycemic effects of soy isoflavones on postmenopausal women. J Women’s Health (Larchmt) 13(10):1080–1086
Azadbakht L, Kimiagar M, Mehrabi Y, Esmaillzadeh A, Padyab M, Hu FB, Willett WC (2007) Soy inclusion in the diet improves features of the metabolic syndrome: a randomized crossover study in postmenopausal women. Am J Clin Nutr 85(3):735–741
Zamora-Ros R, Jimenez C, Cleries R, Agudo A, Sanchez MJ, Sanchez-Cantalejo E, Molina-Montes E, Navarro C, Chirlaque MD, Maria Huerta J, Amiano P, Redondo ML, Barricarte A, Gonzalez CA (2013) Dietary flavonoid and lignan intake and mortality in a Spanish cohort. Epidemiology 24(5):726–733
Zhang X, Shu XO, Gao YT, Yang G, Li Q, Li H, Jin F, Zheng W (2003) Soy food consumption is associated with lower risk of coronary heart disease in Chinese women. J Nutr 133(9):2874–2878
Yu D, Zhang X, Xiang YB, Yang G, Li H, Fazio S, Linton M, Cai Q, Zheng W, Gao YT, Shu XO (2014) Association of soy food intake with risk and biomarkers of coronary heart disease in Chinese men. Int J Cardiol 172(2):e285–e287
Kokubo Y, Iso H, Ishihara J, Okada K, Inoue M, Tsugane S (2007) Association of dietary intake of soy, beans, and isoflavones with risk of cerebral and myocardial infarctions in Japanese populations: the Japan Public Health Center-based (JPHC) study cohort I. Circulation 116(22):2553–2562
Nagata C, Takatsuka N, Shimizu H (2002) Soy and fish oil intake and mortality in a Japanese community. Am J Epidemiol 156(9):824–831
Talaei M, Koh WP, van Dam RM, Yuan JM, Pan A (2014) Dietary soy intake is not associated with risk of cardiovascular disease mortality in Singapore Chinese adults. J Nutr 144(6):921–928
Lou D, Li Y, Yan G, Bu J, Wang H (2016) Soy consumption with risk of coronary heart disease and stroke: a meta-analysis of observational studies. Neuroepidemiology 46(4):242–252
Hollman PC, Geelen A, Kromhout D (2010) Dietary flavonol intake may lower stroke risk in men and women. J Nutr 140(3):600–604
Giergiel M, Lopucki M, Stachowicz N, Kankofer M (2012) The influence of age and gender on antioxidant enzyme activities in humans and laboratory animals. Aging Clin Exp Res 24(6):561–569
Klein SL, Flanagan KL (2016) Sex differences in immune responses. Nat Rev Immunol 16(10):626–638
Fairweather D (2014) Sex differences in inflammation during atherosclerosis. Clin Med Insights Cardiol 8(Suppl 3):49–59
Barp J, Araujo AS, Fernandes TR, Rigatto KV, Llesuy S, Bello-Klein A, Singal P (2002) Myocardial antioxidant and oxidative stress changes due to sex hormones. Braz J Med Biol Res 35(9):1075–1081
Morales RC, Bahnson ES, Havelka GE, Cantu-Medellin N, Kelley EE, Kibbe MR (2015) Sex-based differential regulation of oxidative stress in the vasculature by nitric oxide. Redox Biol 4:226–233
Matarrese P, Colasanti T, Ascione B, Margutti P, Franconi F, Alessandri C, Conti F, Riccieri V, Rosano G, Ortona E, Malorni W (2011) Gender disparity in susceptibility to oxidative stress and apoptosis induced by autoantibodies specific to RLIP76 in vascular cells. Antioxid Redox Signal 15(11):2825–2836
Straface E, Vona R, Gambardella L, Ascione B, Marino M, Bulzomi P, Canu S, Coinu R, Rosano G, Malorni W, Franconi F (2009) Cell sex determines anoikis resistance in vascular smooth muscle cells. FEBS Lett 583(21):3448–3454
Malorni W, Straface E, Matarrese P, Ascione B, Coinu R, Canu S, Galluzzo P, Marino M, Franconi F (2008) Redox state and gender differences in vascular smooth muscle cells. FEBS Lett 582(5):635–642
Chen Y, Ji LL, Liu TY, Wang ZT (2011) Evaluation of gender-related differences in various oxidative stress enzymes in mice. Chin J Physiol 54(6):385–390
Kayali R, Cakatay U, Uzun H, Genc H (2007) Gender difference as regards myocardial protein oxidation in aged rats: male rats have increased oxidative protein damage. Biogerontology 8(6):653–661
Campesi I, Straface E, Occhioni S, Montella A, Franconi F (2013) Protein oxidation seems to be linked to constitutive autophagy: a sex study. Life Sci 93(4):145–152
Horvathova M, Zitnanova I, Kralovicova Z, Balis P, Puzserova A, Muchova J, Kluknavsky M, Durackova Z, Bernatova I (2016) Sex differences in the blood antioxidant defense system in juvenile rats with various genetic predispositions to hypertension. Hypertens Res 39(2):64–69
Fortepiani LA, Reckelhoff JF (2005) Role of oxidative stress in the sex differences in blood pressure in spontaneously hypertensive rats. J Hypertens 23(4):801–805
Miller AA, Drummond GR, Mast AE, Schmidt HH, Sobey CG (2007) Effect of gender on NADPH-oxidase activity, expression, and function in the cerebral circulation: role of estrogen. Stroke 38(7):2142–2149
Zhang R, Thor D, Han X, Anderson L, Rahimian R (2012) Sex differences in mesenteric endothelial function of streptozotocin-induced diabetic rats: a shift in the relative importance of EDRFs. Am J Physiol Heart Circ Physiol 303(10):H1183-1198
Ali SS, Xiong C, Lucero J, Behrens MM, Dugan LL, Quick KL (2006) Gender differences in free radical homeostasis during aging: shorter-lived female C57BL6 mice have increased oxidative stress. Aging Cell 5(6):565–574
Dai G, He L, Chou N, Wan YJ (2006) Acetaminophen metabolism does not contribute to gender difference in its hepatotoxicity in mouse. Toxicol Sci 92(1):33–41
Masubuchi Y, Nakayama J, Watanabe Y (2011) Sex difference in susceptibility to acetaminophen hepatotoxicity is reversed by buthionine sulfoximine. Toxicology 287(1–3):54–60
Pellegrini GG, Cregor M, McAndrews K, Morales CC, McCabe LD, McCabe GP, Peacock M, Burr D, Weaver C, Bellido T (2017) Nrf2 regulates mass accrual and the antioxidant endogenous response in bone differently depending on the sex and age. PLoS One 12(2):e0171161
Yu J, Zhao Y, Li B, Sun L, Huo H (2012) 17beta-estradiol regulates the expression of antioxidant enzymes in myocardial cells by increasing Nrf2 translocation. J Biochem Mol Toxicol 26(7):264–269
Addis R, Campesi I, Fois M, Capobianco G, Dessole S, Fenu G, Montella A, Cattaneo MG, Vicentini LM, Franconi F (2014) Human umbilical endothelial cells (HUVECs) have a sex: characterisation of the phenotype of male and female cells. Biol Sex Differ 5(1):18
Ide T, Tsutsui H, Ohashi N, Hayashidani S, Suematsu N, Tsuchihashi M, Tamai H, Takeshita A (2002) Greater oxidative stress in healthy young men compared with premenopausal women. Arterioscler Thromb Vasc Biol 22(3):438–442
Brunelli E, Domanico F, La Russa D, Pellegrino D (2014) Sex differences in oxidative stress biomarkers. Curr Drug Targets 15(8):811–815
Tothova L, Ostatnikova D, Sebekova K, Celec P, Hodosy J (2013) Sex differences of oxidative stress markers in young healthy subjects are marker-specific in plasma but not in saliva. Ann Hum Biol 40(2):175–180
Campesi I, Occhioni S, Tonolo G, Cherchi S, Basili S, Carru C, Zinellu A, Franconi F (2016) Ageing/menopausal status in healthy women and ageing in healthy men differently affect cardiometabolic parameters. Int J Med Sci 13(2):124–132
Hollman PC, Cassidy A, Comte B, Heinonen M, Richelle M, Richling E, Serafini M, Scalbert A, Sies H, Vidry S (2011) The biological relevance of direct antioxidant effects of polyphenols for cardiovascular health in humans is not established. J Nutr 141(5):989S–1009S
Halliwell B (2007) Dietary polyphenols: good, bad, or indifferent for your health? Cardiovasc Res 73(2):341–347
Ambrosini G, Romani A, Scazzocchio B, Varì R, Vona R, Campesi I, Banelli L, Straface E, Masella R, Malorni W, Franconi F (2012) Sex-gender influences the expression of antioxidant/detoxifying enzyme induced by extra rirgin olive oil extracts in rat vascular smooth muscle cells. In: XXVIth international conference on polyphenols 22–26 July 2012, Florence Italy 2012, pp 437–438
Kamper EF, Chatzigeorgiou A, Tsimpoukidi O, Kamper M, Dalla C, Pitychoutis PM, Papadopoulou-Daifoti Z (2009) Sex differences in oxidant/antioxidant balance under a chronic mild stress regime. Physiol Behav 98(1–2):215–222
Escande A, Pillon A, Servant N, Cravedi JP, Larrea F, Muhn P, Nicolas JC, Cavailles V, Balaguer P (2006) Evaluation of ligand selectivity using reporter cell lines stably expressing estrogen receptor alpha or beta. Biochem Pharmacol 71(10):1459–1469
Mueller SO, Simon S, Chae K, Metzler M, Korach KS (2004) Phytoestrogens and their human metabolites show distinct agonistic and antagonistic properties on estrogen receptor alpha (ERalpha) and ERbeta in human cells. Toxicol Sci 80(1):14–25
Acconcia F, Fiocchetti M, Marino M (2016) Xenoestrogen regulation of ERalpha/ERbeta balance in hormone-associated cancers. Mol Cell Endocrinol 457:3–12
Pellegrini M, Bulzomi P, Lecis M, Leone S, Campesi I, Franconi F, Marino M (2014) Endocrine disruptors differently influence estrogen receptor beta and androgen receptor in male and female rat VSMC. J Cell Physiol 229(8):1061–1068
Forslund K, Hildebrand F, Nielsen T, Falony G, Le Chatelier E, Sunagawa S, Prifti E, Vieira-Silva S, Gudmundsdottir V, Krogh Pedersen H, Arumugam M, Kristiansen K, Voigt AY, Vestergaard H, Hercog R, Igor Costea P, Kultima JR, Li J, Jorgensen T, Levenez F, Dore J, Nielsen HB, Brunak S, Raes J, Hansen T, Wang J, Ehrlich SD, Bork P, Pedersen O (2015) Disentangling type 2 diabetes and metformin treatment signatures in the human gut microbiota. Nature 528(7581):262–266
Tang WH, Hazen SL (2014) The contributory role of gut microbiota in cardiovascular disease. J Clin Investig 124(10):4204–4211
Gregory JC, Buffa JA, Org E, Wang Z, Levison BS, Zhu W, Wagner MA, Bennett BJ, Li L, DiDonato JA, Lusis AJ, Hazen SL (2015) Transmission of atherosclerosis susceptibility with gut microbial transplantation. J Biol Chem 290(9):5647–5660
Aron-Wisnewsky J, Clement K (2016) The gut microbiome, diet, and links to cardiometabolic and chronic disorders. Nat Rev Nephrol 12(3):169–181
Burrows MP, Volchkov P, Kobayashi KS, Chervonsky AV (2015) Microbiota regulates type 1 diabetes through Toll-like receptors. Proc Natl Acad Sci USA 112(32):9973–9977
Rieder R, Wisniewski PJ, Alderman BL, Campbell SC (2017) Microbes and mental health: a review. Brain Behav Immun 66:9–17
Donaldson GP, Lee SM, Mazmanian SK (2016) Gut biogeography of the bacterial microbiota. Nat Rev Microbiol 14(1):20–32
Salazar N, Arboleya S, Valdes L, Stanton C, Ross P, Ruiz L, Gueimonde M, de CG Los Reyes-Gavilan (2014) The human intestinal microbiome at extreme ages of life. Dietary intervention as a way to counteract alterations. Front Genet 5:406
Kaplan H, Hill K, Lancaster J, Hurtado AM (2000) A theory of human life history evolution: diet, intelligence, and longevity. Evol Anthropol 9:156–185
Bolnick DI, Snowberg LK, Hirsch PE, Lauber CL, Org E, Parks B, Lusis AJ, Knight R, Caporaso JG, Svanback R (2014) Individual diet has sex-dependent effects on vertebrate gut microbiota. Nat Commun 5:4500
Marino M, Masella R, Bulzomi P, Campesi I, Malorni W, Franconi F (2010) Nutrition and human health from a sex-gender perspective. Mol Asp Med 32(1):1–70
Markle JG, Frank DN, Mortin-Toth S, Robertson CE, Feazel LM, Rolle-Kampczyk U, von Bergen M, McCoy KD, Macpherson AJ, Danska JS (2013) Sex differences in the gut microbiome drive hormone-dependent regulation of autoimmunity. Science 339(6123):1084–1088
Dominianni C, Sinha R, Goedert JJ, Pei Z, Yang L, Hayes RB, Ahn J (2015) Sex, body mass index, and dietary fiber intake influence the human gut microbiome. PLoS One 10(4):e0124599
Yatsunenko T, Rey FE, Manary MJ, Trehan I, Dominguez-Bello MG, Contreras M, Magris M, Hidalgo G, Baldassano RN, Anokhin AP, Heath AC, Warner B, Reeder J, Kuczynski J, Caporaso JG, Lozupone CA, Lauber C, Clemente JC, Knights D, Knight R, Gordon JI (2012) Human gut microbiome viewed across age and geography. Nature 486(7402):222–227
Li M, Wang B, Zhang M, Rantalainen M, Wang S, Zhou H, Zhang Y, Shen J, Pang X, Zhang M, Wei H, Chen Y, Lu H, Zuo J, Su M, Qiu Y, Jia W, Xiao C, Smith LM, Yang S, Holmes E, Tang H, Zhao G, Nicholson JK, Li L, Zhao L (2008) Symbiotic gut microbes modulate human metabolic phenotypes. Proc Natl Acad Sci USA 105(6):2117–2122
Mueller S, Saunier K, Hanisch C, Norin E, Alm L, Midtvedt T, Cresci A, Silvi S, Orpianesi C, Verdenelli MC, Clavel T, Koebnick C, Zunft HJ, Dore J, Blaut M (2006) Differences in fecal microbiota in different European study populations in relation to age, gender, and country: a cross-sectional study. Appl Environ Microbiol 72(2):1027–1033
Haro C, Rangel-Zuniga OA, Alcala-Diaz JF, Gomez-Delgado F, Perez-Martinez P, Delgado-Lista J, Quintana-Navarro GM, Landa BB, Navas-Cortes JA, Tena-Sempere M, Clemente JC, Lopez-Miranda J, Perez-Jimenez F, Camargo A (2016) Intestinal microbiota is influenced by gender and body mass index. PLoS One 11(5):e0154090
Koren O, Goodrich JK, Cullender TC, Spor A, Laitinen K, Backhed HK, Gonzalez A, Werner JJ, Angenent LT, Knight R, Backhed F, Isolauri E, Salminen S, Ley RE (2012) Host remodeling of the gut microbiome and metabolic changes during pregnancy. Cell 150(3):470–480
Espin JC, Gonzalez-Sarrias A, Tomas-Barberan FA (2017) The gut microbiota: a key factor in the therapeutic effects of (poly)phenols. Biochem Pharmacol 139:82–93
Ozdal T, Sela DA, Xiao J, Boyacioglu D, Chen F, Capanoglu E (2016) The reciprocal interactions between polyphenols and gut microbiota and effects on bioaccessibility. Nutrients 8(2):78–113
Frankenfeld CL, Atkinson C, Wahala K, Lampe JW (2014) Obesity prevalence in relation to gut microbial environments capable of producing equol or O-desmethylangolensin from the isoflavone daidzein. Eur J Clin Nutr 68(4):526–530
Nakatsu CH, Armstrong A, Clavijo AP, Martin BR, Barnes S, Weaver CM (2014) Fecal bacterial community changes associated with isoflavone metabolites in postmenopausal women after soy bar consumption. PLoS One 9(10):e108924
Usui T, Tochiya M, Sasaki Y, Muranaka K, Yamakage H, Himeno A, Shimatsu A, Inaguma A, Ueno T, Uchiyama S, Satoh-Asahara N (2013) Effects of natural S-equol supplements on overweight or obesity and metabolic syndrome in the Japanese, based on sex and equol status. Clin Endocrinol (Oxf) 78(3):365–372
Ridlon JM, Ikegawa S, Alves JM, Zhou B, Kobayashi A, Iida T, Mitamura K, Tanabe G, Serrano M, De Guzman A, Cooper P, Buck GA, Hylemon PB (2013) Clostridium scindens: a human gut microbe with a high potential to convert glucocorticoids into androgens. J Lipid Res 54(9):2437–2449
Balakrishnan B, Thorstensen EB, Ponnampalam AP, Mitchell MD (2010) Transplacental transfer and biotransformation of genistein in human placenta. Placenta 31(6):506–511
Foster WG, Chan S, Platt L, Hughes CL Jr (2002) Detection of phytoestrogens in samples of second trimester human amniotic fluid. Toxicol Lett 129(3):199–205
Soucy NV, Parkinson HD, Sochaski MA, Borghoff SJ (2006) Kinetics of genistein and its conjugated metabolites in pregnant Sprague-Dawley rats following single and repeated genistein administration. Toxicol Sci 90(1):230–240
Jarrell J, Foster WG, Kinniburgh DW (2012) Phytoestrogens in human pregnancy. Obstet Gynecol Int 2012:850313
Diaz-Castro J, Pulido-Moran M, Moreno-Fernandez J, Kajarabille N, de Paco C, Garrido-Sanchez M, Prados S, Ochoa JJ (2016) Gender specific differences in oxidative stress and inflammatory signaling in healthy term neonates and their mothers. Pediatr Res 80(4):595–601
Hanson MA, Gluckman PD (2008) Developmental origins of health and disease: new insights. Basic Clin Pharmacol Toxicol 102(2):90–93
McCarver G, Bhatia J, Chambers C, Clarke R, Etzel R, Foster W, Hoyer P, Leeder JS, Peters JM, Rissman E, Rybak M, Sherman C, Toppari J, Turner K (2011) NTP-CERHR expert panel report on the developmental toxicity of soy infant formula. Birth Defects Res B Dev Reprod Toxicol 92(5):421–468
Setchell KD, Zimmer-Nechemias L, Cai J, Heubi JE (1997) Exposure of infants to phyto-oestrogens from soy-based infant formula. Lancet 350(9070):23–27
Strakovsky RS, Lezmi S, Flaws JA, Schantz SL, Pan YX, Helferich WG (2014) Genistein exposure during the early postnatal period favors the development of obesity in female, but not male rats. Toxicol Sci 138(1):161–174
Jefferson WN, Williams CJ (2011) Circulating levels of genistein in the neonate, apart from dose and route, predict future adverse female reproductive outcomes. Reprod Toxicol 31(3):272–279
Jefferson WN, Padilla-Banks E, Goulding EH, Lao SP, Newbold RR, Williams CJ (2009) Neonatal exposure to genistein disrupts ability of female mouse reproductive tract to support preimplantation embryo development and implantation. Biol Reprod 80(3):425–431
Newbold RR, Banks EP, Bullock B, Jefferson WN (2001) Uterine adenocarcinoma in mice treated neonatally with genistein. Cancer Res 61(11):4325–4328
Santti R, Makela S, Strauss L, Korkman J, Kostian ML (1998) Phytoestrogens: potential endocrine disruptors in males. Toxicol Ind Health 14(1–2):223–237
Bulzomi P, Marino M (2011) Environmental endocrine disruptors: does a sex-related susceptibility exist? Front Biosci 16:2478–2498
Acknowledgements
This research was partially funded by a Grant of INAIL “Bando BRIC 2016”.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Campesi, I., Marino, M., Cipolletti, M. et al. Put “gender glasses” on the effects of phenolic compounds on cardiovascular function and diseases. Eur J Nutr 57, 2677–2691 (2018). https://doi.org/10.1007/s00394-018-1695-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-018-1695-0