Abstract
Purpose
Gestational diabetes mellitus (GDM) represents the most frequent metabolic disorder in pregnancy. Since dietary intake plays an important role in obesity and type 2 diabetes development, it is likely to be for the susceptibility to GDM too. Food preferences, driving partly the diet composition, are changing during pregnancy. Taste and genetic variability in taste receptors is an important factor in determining food preferences. Aims of our study were (1) to characterize dietary habits of pregnant women and to find possible differences in food preferences between healthy pregnant women and those with GDM and (2) to ascertain possible association of several single nucleotide polymorphisms (SNPs) in taste receptor (TR) genes with GDM.
Methods
A total of 363 pregnant women (293 with GDM and 70 with physiologic pregnancy) were included in the study. Dietary pattern spanning the period of approx. 6 months preceding the time of GDM screening was assessed using a semi-quantitative food frequency questionnaire. A total of five SNPs in TR genes were selected for genotyping based on their functionality or previous associations.
Results
Women with GDM exhibited significantly more frequent meat consumption (esp. poultry, pork and smoked meat), dairy products and sweet beverages consumption. The legumes consumption was found to be inversely correlated with fasting glycaemia (P = 0.007, Spearman). CC genotype in TAS2R9 gene (SNP rs3741845) was significantly associated with GDM (P = 0.0087, Chi-square test).
Conclusions
Our study showed differences in dietary intake of selected food items between healthy pregnant women and those with GDM and genetic association of bitter taste receptor allele with GDM.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Gestational diabetes mellitus (GDM) defined as any degree of glucose intolerance with the onset or first recognition during pregnancy [1] represents the most common medical complication of pregnancy nowadays [2] with prevalence reaching up to 18 % depending on the population and diagnostic criteria used [3]. Pregnancies affected by GDM pose an increased risk of perinatal complications for both mother (preeclampsia, need for Caesarean delivery and subsequent post-surgery complications) and child (macrosomia, shoulder dystocia, neonatal hypoglycaemia and hyperbilirubinemia) [4]. Although glucose abnormality usually resolves soon after birth, women with a history of GDM are at an increased risk of recurrent GDM in subsequent pregnancies, long-term risk of development of type 2 diabetes mellitus (T2DM) and cardiovascular diseases later in life and their children are more likely to be obese and to have impaired glucose tolerance (IGT) and T2DM in early adulthood [5, 6].
Healthy diet is an integral component of not only diabetes management, but also powerful tool for its prevention [7, 8]. Diet is also the cornerstone of the management of GDM [9] and most likely for its prevention too [10]. Recently, several studies attempted to clarify the role of consumption diet in not only quantitative, but also qualitative terms in the development of GDM. Consumption of legumes, fish, nuts [11–13], fruits and vegetables [14–16] appeared to be protective in relation to GDM, while higher intake of red meat [17], animal fats (saturated fatty acids and cholesterol) [17, 18] and sweet beverages [19] had increased the risk of GDM.
Taste is a powerful driver of eating that can play a non-negligible role in the long-term establishment of food preferences and therefore overall lifestyle. There are several taste modalities identified—bitter, sweet, umami, sour and salty—and inter-individual differences in intensity of taste perception in these categories may influence dietary habits and nutritional status and contribute to the development of complex lifestyle-related chronic disease risk. In addition to these traditional taste modalities, there is growing evidence that “fat taste” may represent a sixth modality [20]. Taste perception occurs when chemical molecules from food reach microvilli located at the apical tip of taste receptor cells (taste receptors, TRs). TRs congregate in groups on taste buds, which are located in epithelial surfaces of the tongue, palate, pharynx, larynx and upper oesophagus [21]. Several TRs have been identified within taste cell membranes on the surface of the tongue, and they include: T2R family of bitter TRs, the T1Rs associated with sweet and umami taste perception, the ion channels PKD1L3 and PKD2L1 linked to sour taste, and the integral membrane protein CD36, which is a putative “fat taste” receptor. Additionally, epithelial sodium channels and vanilloid receptor may account for salty taste perception [20].
Pregnant women commonly experience changes in taste at the onset and throughout the pregnancy. In physiological pregnancy, craving and a consumption of sweet foods culminate during the second trimester and normalize after delivery [22]. Furthermore, pregnant women are supposed to have a higher threshold for a salty taste in the third trimester with subsequent increased consumption of salty foods. Finally, sensitivity for bitter taste is reported to increase during human pregnancy [23]. Exaggerated preference for sweet foods could alter a dietary behaviour and contribute to the manifestation of underlying glucose abnormality. While several studies focusing on taste preferences were conducted in both type 1 diabetes mellitus [24, 25] and T2DM [26], only very few studies have investigated taste preferences (preferentially sweet) and dietary behaviour specifically in women with GDM [22, 27]. Nonetheless, results suggest a trend towards higher preference of sweet taste in GDM-affected women [27]. Park et al. [28] studied snack consumption, night eating and nutrient intake in GDM and non-GDM women and observed higher preference for salty and greasy taste in GDM patients.
Noticeably, genetic variability [i.e. common single nucleotide polymorphisms (SNPs)] in genes involved in taste perception may account for some of the observed inter-individual differences in food preferences and dietary habits in general population [20]. However, only few studies so far specifically investigated a putative association of genetic polymorphisms in genes for TR with diabetes, namely T2DM. Dotson et al. [29] studied SNPs in bitter TR genes using candidate gene study within the Amish Family Diabetes Study and found minor alleles of TAS2R7 (rs619381) and TAS2R9 (rs3741845) to be associated with T2DM. Study of Ma et al. [30] reported minor allele of variant rs1527479 in CD36 gene significantly overrepresented in T2DM patients. In another study, higher sugar intake was found to be associated with SNP rs35874116 in TAS1R2 gene for sweet TR in overweight and obese individuals [31]. Moreover, SNP rs5400 in SLC2A2 gene for glucose transporter type 2 (GLUT2) that facilitates the first step in glucose-induced insulin secretion (and is expressed in the pancreas, liver, small intestine, kidney and brain) was associated with higher sugar intake [32] and T2DM [33]. Nevertheless, no study so far investigated possible relationship between genetic variability in TR genes and GDM.
Therefore, given the well-documented changes of taste and food intake in pregnancy and the importance of taste receptors in the regulation of alimentary responses to chemical stimuli and thus food intake, we hypothesized that not only quantitative (influencing body weight and composition), but also qualitative differences in food intake may contribute to the development of GDM and thus constitute a modifiable environmental risk factor. Furthermore, we assumed that part of the inter-individual variability in food intake and preferences may be due to genetic variability in loci encoding taste receptors and, therefore, constitute an unmodifiable risk factor affecting susceptibility to GDM. Specific aims of our study were (1) to characterize dietary habits and to find possible differences in food preferences between healthy pregnant women and those with GDM and (2) to ascertain a possible association of selected SNPs in genes for taste receptors with GDM or/and anthropometric parameters previously associated with a higher risk of GDM development.
Materials and methods
Study design and subjects
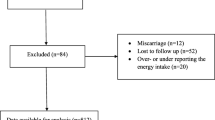
Case–control study included a total of 363 pregnant women of Caucasian origin, geographically derived from South Moravia region of Czech Republic. Of those, 293 women had GDM (consenting subjects positively diagnosed with GDM during routine mid-gestational screening and then followed from the time of GDM diagnosis till the birth at the Diabetes Centre of the University Hospital Brno) and 70 had physiological pregnancy (those were consenting women who passed mid-gestational GDM screening with negative result and were followed in several outpatient prenatal centres in the city of Brno until delivery). Exclusion criteria were (1) a prior history of type 1 or type 2 diabetes, (2) a multiple gestation (twins or higher-order multiple gestation) and (3) severe comorbidities. Study was approved by the Ethical Committee of Faculty of Medicine, Masaryk University, and was conducted in accordance with Helsinki Declaration. Each participant provided an informed consent.
Methods
GDM screening was carried out using oGTT with 75 g of glucose performed between 24th and 30th week of pregnancy. GDM diagnosis was established according to the modified WHO criteria recommended by the Czech Diabetes Society at the time of the recruitment (2012): fasting plasma glucose (FPG) ≥5.6 mmol/l, 1 h post-load glucose ≥8.9 mmol/l and 2 h post-load glucose ≥7.7 mmol/l (any one of the three above cut-off values qualified for the GDM diagnosis). Dietary pattern spanning the period of approx. 6 months preceding the time of GDM screening (i.e. beginning as close as possible to the supposed conception date up to 24th–30th week of pregnancy) was assessed using a semi-quantitative food frequency questionnaire (SFFQ) in Czech language complying with previous recommendations [34–38] and moderately modified to local conditions. For each food item, a standard serving size was specified and food consumption frequency was self-reported on a daily, weekly or monthly basis. In particular, SFFQ used in the study included 46 items divided into eight categories (see Table 1) according to current local dietary guidelines—“food pyramid” [39]. Initially, eight frequencies of consumption were used that were subsequently converted to daily frequencies for the purpose of statistical analyses (see Table 2). SFFQ was completed by 266 (73.3 %) subjects (67 % GDM and 100 % controls) at the time of enrolment in the study (between 24th and 30th week of pregnancy). Any specific dietary counselling was provided by outpatient prenatal centres before routine GDM screening and event diagnosis. Following GDM diagnosis, all women in the GDM group followed in Diabetes Centre of the University Hospital Brno were counselled equally during their regular visits by diabetologist/diabetic nurse. Recommendations corresponded with those provided to any diabetic patients in qualitative terms and were adjusted to ensure optimal energy intake for a given period of gestation.
Blood samples
Sample of peripheral venous blood was taken from each subject at the time of enrolment in the study. Blood samples were centrifuged immediately after the collection at 3000 g for 10 min. DNA was extracted from peripheral blood leucocytes by the phenol–chloroform method and together with plasma frozen and stored at −20 or −80 °C, respectively, until further analysis. DNA was available from all studied subjects. Five SNPs in TR genes—TAS1R2 gene (rs35874116) for sweet TR, TAS2R7 (rs619381) and TAS2R9 (rs3741845) gene for bitter TR, CD36 (rs1527479) gene for “fat” TR and finally a gene SLC2A2 (rs5400) for glucose transporter GLUT2—were selected for genotyping based on previously reported association or functionality (see Table 3). Genotyping was performed by real-time PCR (ABI PRISM 7000) using TaqMan SNP Genotyping Assays (Applied Biosystems).
Statistical analyses
Data were expressed as medians and interquartile ranges [IQR] or proportions for between-group comparisons. Differences between groups were compared using nonparametric Mann–Whitney or Pearson Chi-square tests for continuous or categorical variables, respectively. Kolmogorov–Smirnov test was used to test for the normality of distribution of food consumption frequencies. Two methods of SFFQ evaluation were used: (1) for the whole categories of the “food pyramid”, we evaluated a mean frequency of food intake, and then (since “food pyramid” is typically constructed as daily recommendation), we evaluated also daily frequencies of consumption (i.e. binary variable: (1) less than once a day vs. (2) once a day or more). (2) For the particular items in categories, a mean frequency was used only. Fisher’s exact test and odds ratio test were used subsequently for more detailed analyses comparing daily frequencies of food consumption between two groups and to estimate the GDM risk. Spearman test was used to assess correlations between selected variables. Statistica for Windows (Statsoft Inc., Tulsa, OK, USA) served as statistic software. P ≤ 0.05 was considered statistically significant.
Results
Characteristics of study subjects in the two groups are shown in Table 4. Although women with GDM were not significantly older, they were significantly heavier, but had smaller weight gain at the end of pregnancy (while a weight gain until the second trimester, i.e. period between the conception and the time of the enrolment to the study/event. GDM diagnosis was similar). This probably reflects the effect of diet instituted in GDM subjects following diagnosis. Surprisingly, both groups did not statistically differ in an incidence of obesity at baseline (BMI ≥ 30 kg/m2); therefore, the higher prevalence of overweight in GDM group accounted for the observed difference in baseline weight/BMI. Offspring’s birth weight was also similar in both groups. The two groups significantly differed in the prevalence of positive family history of DM and GDM history in multiparous women (both categories being significantly higher in GDM group). Focusing specifically on the GDM group women with recurrent GDM had significantly smaller weight gain until the second trimester compared to those without GDM history and primiparous. So we can assume a positive effect of a diet counselling during previous pregnancy.
First, we evaluated differences in food intake between women with and without GDM in the whole categories of “food pyramid” (i.e. cereals, vegetables, fruit, dairy products, protein food, goodies, sweet beverages, alcoholic beverages). When comparing mean frequency of consumption, women with GDM reported higher consumption of protein food and sweet beverages (P = 0.005 and P = 0.025, respectively, Kolmogorov–Smirnov test). In terms of daily consumption (i.e. frequency of intake less than once a day vs. once a day or more), women with GDM exhibited significantly more frequent daily consumption of dairy products, goodies and sweet beverages (all P < 0.05, Fisher’s exact test). Furthermore, we calculated odds ratios and respective 95 % CI using odds ratio test showing “odds” of developing GDM between the two categories of food consumption, i.e. “once a day or more” and “less than once a day”. For summary of results and odds ratios, see Table 5. Finally, comparison of particular food items revealed statistically significant differences in consumption of fresh vegetables (P = 0.034), smoked meat (P = 0.033), pork (P = 0.015), poultry (P = 0.02), fresh fruit (P = 0.006) and syrup (P = 0.031, all Mann–Whitney test). Women with GDM consumed significantly less fresh vegetables compared to controls, while for the remaining items, the consumption was significantly higher.
Furthermore, we assessed correlations between selected parameters (BMI, weight gain during pregnancy, offspring birth weight and glycaemia in oGTT test) and frequency of food consumption and found inverse correlation between frequency of legumes consumption and FPG in oGTT test (r = −0.17, P = 0.007) and positive correlation between frequency of smoked meat consumption and pre-pregnancy BMI (r = 0.17, P = 0.006), BMI in mid-trimester of gravidity (r = 0.14, P = 0.02), FPG in oGTT test (r = 0.15, P = 0.02) and glycaemia in 2 h post-load glucose in oGTT (r = 0.18, P = 0.004, all Spearman). Finally, we found no differences in food consumption between women with or without positive family history of DM or GDM in previous pregnancies.
Genotype frequencies of all selected SNPs were in Hardy–Weinberg equilibrium (P > 0.05, χ 2 test). Allele frequencies of SNPs studied are shown in Table 3. Statistically significant difference in allele frequencies of rs3741845 in TAS2R9 gene was found between the groups with T allele significantly less frequent in GDM subjects. Moreover, comparison of rs3741845 genotype frequencies revealed significant difference between the two groups with CC genotype more frequent in GDM group (P = 0.0087, χ 2 test). Comparison of selected parameters (BMI, weight gain during pregnancy, offspring birth weight and glycaemia in oGTT test) between carriers of particular alleles or genotypes of SNPs studied did not reveal statistically significant differences (all P > 0.05, Kruskal–Wallis and Mann–Whitney test). Finally, carriers of particular alleles or genotypes did not differ in the frequencies of particular food consumption categories (all P > 0.05, Kruskal–Wallis and Mann–Whitney test).
Discussion
Given the high prevalence and steadily rising incidence of GDM, the lifelong increased diabetes risk it poses on affected woman and, above all, potentially negative metabolic imprint of GDM on the offspring, there is a strong need of its early diagnosis, effective management and, ideally, a prevention. Given the available data on the role of nutritional factors in T2DM prevention and results of the RADIEL study showing efficacy of lifestyle (incl. dietary) interventions in GDM prevention [10], we were interested in replicating/confirming event, differences in food preferences in another population and, at the same time, in the degree of their association with genetic variability in taste receptor genes. In the current case–control study comprising 363 metabolically well-characterized pregnant subjects, we therefore compared dietary patterns of women with and without GDM using SFFQ reflecting immediate post-conception plus first- and second-trimester pregnancy period (i.e. time period directly preceding event. GDM diagnosis) and analysed its association with metabolic and anthropometric traits and genetic polymorphism in selected TR genes.
This study was a part of larger study aimed at differences between normal pregnancies and GDM-affected pregnancies in several metabolic pathways, food intake, micronutrients and pregnancy outcomes.
Major findings of the current study could be summarized as follows: (1) higher BMI and certain dietary patterns (namely more frequent consumption of dairy products, sweet beverages and goodies and lower consumption of fresh vegetables) constitute the major pre-gestational nutritional feature associated with GDM. Furthermore, (2) glycaemic indices (oGTT values) ascertained during the second trimester correlated with certain dietary patterns (smoked meat consumption positively with FPG and 2 h post-load glucose and legumes inversely with FPG). Finally, (3) a CC genotype of rs3741845 in TAS2R9 gene encoding bitter taste receptor was associated with GDM independent of its association with any particular food preference.
Body weight is an extremely relevant trait in the context of GDM. Pre-conception overweight and obesity are established risk factors for GDM.
A decent weight gain is also important for prevention of GDM. Several studies confirmed adequate weight gain during pregnancy can facilitate fast weight reduction after delivery, normalize offspring’s birth weight and decrease the risk of development of children obesity. Pregnant women with BMI above 25 kg/m2 are recommended to have as low as possible weight gain during pregnancy, but they should not reduce the weight [40]. Results of our study showed lower weight gain during the whole pregnancy in GDM-affected women, but the weight gain in the pregnancy period before enrolment to the study was similar in both groups. This could be due to lifestyle intervention in subjects following GDM diagnosis as part of the regular diabetic education. We should emphasize the counselling was performed after GDM diagnosis, so did not influence the answers in the SFFQ. Our results are in line with previous findings of lower weight gain in GDM-affected women from the time of GDM diagnosis in comparison with women with physiological pregnancy, e.g. by Horosz et al. [41] or Stewart et al. [42]. Furthermore, we found no difference in the occurrence of macrosomia in the offspring between women with and without GDM in the current study.
Several studies in the past focused on food consumption either before or during pregnancy with the aim to find relationships between various dietary patterns and the risk of GDM. No such study has been done in the Czech Republic so far though. Using SFFQ, we ascertained statistically significant differences in consumption of protein food (esp. smoked meat, pork and poultry meat) with a higher preference in GDM group. We also found an association between consumption of smoked meat and both BMI and glucose levels during oGTT test. Our results basically correspond with previous findings of other studies in GDM observing higher consumption of red meat [11, 12, 17].
We also found an inverse correlation of legumes frequency consumption and fasting glycaemia measured in oGTT test in mid-trimester of pregnancy. Consumption of poultry, fish, nuts and legumes instead of red meat is perceived as a protective factor in relation to GDM [11, 12]. The distinct effects could be due to variations of amino acid composition in these foods. Fish, nuts and legumes are rich in monounsaturated fatty acids and polyunsaturated fatty acids and have beneficial effects in the setting of glucose intolerance compared to saturated fatty acids. Moreover, nuts have a relatively low glycaemic index. These factors, either individually or in combination, have been associated with improved insulin sensitivity and lower diabetes risk [11].
Several studies showed a protective effect of fruit and vegetable consumption in relation to GDM risk [12, 14, 15]. Both have a high antioxidant and fibre content as well as relatively low energy density and low glycaemic load. In addition, fruit contains numerous bioactive components, which have been suggested to be beneficial in insulin sensitivity and/or pancreatic β-cell function by relieving oxidative stress. It has also been previously hypothesized that individuals with increased bitter taste sensitivity might avoid antioxidant-rich vegetables because of their perceived bitterness. Increased sensitivity to bitterness has also been associated with heightened taste acuity, which may prevent food overconsumption in general [20]. Results of our study suggest a similar trend since we observed healthy pregnant women consuming more vegetables and less fruit compared to GDM.
Taste sensation has been reported reduced not only in type 1 diabetes [24, 25], but decreased taste acuity for glucose has been observed also in patients with T2DM [26]. Chen et al. in 10-year follow-up study focusing on pre-pregnancy sugar-sweetened beverage consumption and the subsequent risk of GDM observed Coca-Cola consumption was significantly and positively associated with GDM risk. High-sugar foods or diets were observed to induce a greater postprandial plasma glucose response. Higher sugar intake itself may lead to impaired pancreatic β-cell function by an accumulation of reactive oxygen species in β-cells that will subsequently cause β-cell damage [19]. Our study confirmed a higher preference of sweet beverages in GDM group. Moreover, focusing on daily food intake, women with GDM consumed significantly more goodies.
At the genetic part of the study, we focused on the possible relationship of selected SNPs in TR genes to food preferences of pregnant women and GDM. That is, we believe a first study aimed at this topic. Several previous studies aimed at SNPs in selected TR genes in connection to T2DM or glucose metabolism. Dotson et al. found T (minor) allele in TAS2R9 gene for bitter TR (rs3741845) significantly associated with T2DM [29]. This SNP has been shown to alter an amino acid sequence within a region of TAS2R9 that is predicted to influence ligand binding and postulated that T allele is responsible for a loss of function in taste response to an array of bitter stimuli. The likely mechanism explaining the association of this variant with glucose metabolism and insulin secretion related is by modifying GLP-1 secretion from entero-endocrine L cells. Association of the CC genotype with GDM in the current study could seem plausible—aversion to bitterness reflected in the eating pattern in carriers of functional C allele—however, such hypothesis might be challenged by findings of more recent study by Allen et al. [43], postulating minor T allele as a gain of function variant. Given the plethora of bitter taste receptors and their likely functionality and the wide spectrum of ligands, more detailed and comprehensive analysis is warranted to explain the possible pathogenic role of bitter taste aversion in GDM.
Conclusions
In conclusion, we demonstrated significant differences in dietary intake of selected food items between healthy pregnant women and those with GDM in the period before event diagnosis of GDM and their correlation with selected glycaemic indices during oGTT at the time of GDM screening. Furthermore, in accordance with several previous reports, we ascertained different weight change pattern in GDM subjects compared to controls, likely as a consequence of successful diabetic diet institution. These results—in line with results of interventional lifestyle studies in GDM—support the concept of targeting diet and weight management very early in overweight/obese women when pregnancy is confirmed. While complex lifestyle interventions in pre-conception period are most likely unfeasible (given their limited effectiveness and long-term adherence in general population), targeted, personalized and concise short-term intervention at the beginning of pregnancy seems more realistic scenario with potential far-reaching impacts considering high lifetime diabetes risk in women with a history of GDM and consequences of altered metabolic programming in their offspring.
Abbreviations
- BMI:
-
Body mass index
- DBP:
-
Diastolic blood pressure
- DM:
-
Diabetes mellitus
- DNA:
-
Deoxyribonucleic acid
- FPG:
-
Fasting plasma glucose
- GDM:
-
Gestational diabetes mellitus
- IGT:
-
Impaired glucose tolerance
- IQR:
-
Interquartile ranges
- oGTT:
-
Oral glucose tolerance test
- PCR:
-
Polymerase chain reaction
- SBP:
-
Systolic blood pressure
- SFFQ:
-
Semi-quantitative food frequency questionnaire
- SNP:
-
Single nucleotide polymorphism
- TR:
-
Taste receptor
- T2DM:
-
Type 2 diabetes mellitus
- WHO:
-
World Health Organization
References
Harlev A, Wiznitzer A (2010) New insights on glucose pathophysiology in gestational diabetes and insulin resistance. Curr Diabetes Rep 10(3):242–247
Alfadhli EM (2015) Gestational diabetes mellitus. Saudi Med J 36(4):399–406
Weinert LS (2010) International Association of Diabetes and Pregnancy Study Groups recommendations on the diagnosis and classification of hyperglycemia in pregnancy: comment to the International Association of Diabetes and Pregnancy Study Groups Consensus Panel. Diabetes Care 33(7):e97 (author reply e98)
Catalano PM, Ehrenberg HM (2006) The short- and long-term implications of maternal obesity on the mother and her offspring. BJOG 113(10):1126–1133
Group HSCR (2002) The hyperglycemia and adverse pregnancy outcome (HAPO) study. Int J Gynaecol Obstet 78(1):69–77
Retnakaran R, Qi Y, Connelly PW, Sermer M, Zinman B, Hanley AJ (2010) Glucose intolerance in pregnancy and postpartum risk of metabolic syndrome in young women. J Clin Endocrinol Metab 95(2):670–677
Bantle JP, Wylie-Rosett J, Albright AL, Apovian CM, Clark NG, Franz MJ, Hoogwerf BJ, Lichtenstein AH, Mayer-Davis E, Mooradian AD et al (2008) Nutrition recommendations and interventions for diabetes: a position statement of the American Diabetes Association. Diabetes Care 31(Suppl 1):S61–S78
Reader DM (2007) Medical nutrition therapy and lifestyle interventions. Diabetes Care 30(Suppl 2):S188–S193
Reader D, Splett P, Gunderson EP (2006) Group DCaEDP: impact of gestational diabetes mellitus nutrition practice guidelines implemented by registered dietitians on pregnancy outcomes. J Am Diet Assoc 106(9):1426–1433
Koivusalo SB, Rönö K, Klemetti MM, Roine RP, Lindström J, Erkkola M, Kaaja RJ, Pöyhönen-Alho M, Tiitinen A, Huvinen E et al (2016) Gestational diabetes mellitus can be prevented by lifestyle intervention: the finnish gestational diabetes prevention study (RADIEL): a randomized controlled trial. Diabetes Care 39(1):24–30
Bao W, Bowers K, Tobias DK, Hu FB, Zhang C (2013) Prepregnancy dietary protein intake, major dietary protein sources, and the risk of gestational diabetes mellitus: a prospective cohort study. Diabetes Care 36(7):2001–2008
Tobias DK, Zhang C, Chavarro J, Bowers K, Rich-Edwards J, Rosner B, Mozaffarian D, Hu FB (2012) Prepregnancy adherence to dietary patterns and lower risk of gestational diabetes mellitus. Am J Clin Nutr 96(2):289–295
Bao W, Bowers K, Tobias DK, Olsen SF, Chavarro J, Vaag A, Kiely M, Zhang C (2014) Prepregnancy low-carbohydrate dietary pattern and risk of gestational diabetes mellitus: a prospective cohort study. Am J Clin Nutr 99(6):1378–1384
Chen L, Hu FB, Yeung E, Tobias DK, Willett WC, Zhang C (2012) Prepregnancy consumption of fruits and fruit juices and the risk of gestational diabetes mellitus: a prospective cohort study. Diabetes Care 35(5):1079–1082
Ali HI, Jarrar AH, El Sadig M, Yeatts KB (2013) Diet and carbohydrate food knowledge of multi-ethnic women: a comparative analysis of pregnant women with and without gestational diabetes mellitus. PLoS ONE 8(9):e73486
Zhang C, Schulze MB, Solomon CG, Hu FB (2006) A prospective study of dietary patterns, meat intake and the risk of gestational diabetes mellitus. Diabetologia 49(11):2604–2613
Bowers K, Yeung E, Williams MA, Qi L, Tobias DK, Hu FB, Zhang C (2011) A prospective study of prepregnancy dietary iron intake and risk for gestational diabetes mellitus. Diabetes Care 34(7):1557–1563
Park S, Kim MY, Baik SH, Woo JT, Kwon YJ, Daily JW, Park YM, Yang JH, Kim SH (2013) Gestational diabetes is associated with high energy and saturated fat intakes and with low plasma visfatin and adiponectin levels independent of prepregnancy BMI. Eur J Clin Nutr 67(2):196–201
Chen L, Hu FB, Yeung E, Willett W, Zhang C (2009) Prospective study of pre-gravid sugar-sweetened beverage consumption and the risk of gestational diabetes mellitus. Diabetes Care 32(12):2236–2241
Garcia-Bailo B, Toguri C, Eny KM, El-Sohemy A (2009) Genetic variation in taste and its influence on food selection. OMICS 13(1):69–80
Chandrashekar J, Hoon MA, Ryba NJ, Zuker CS (2006) The receptors and cells for mammalian taste. Nature 444(7117):288–294
Belzer LM, Smulian JC, Lu SE, Tepper BJ (2009) Changes in sweet taste across pregnancy in mild gestational diabetes mellitus: relationship to endocrine factors. Chem Senses 34(7):595–605
Faas MM, Melgert BN, de Vos P (2010) A brief review on how pregnancy and sex hormones interfere with taste and food intake. Chemosens Percept 3(1):51–56
Hardy SL, Brennand CP, Wyse BW (1981) Taste thresholds of individuals with diabetes mellitus and of control subjects. J Am Diet Assoc 79(3):286–289
Khobragade RS, Wakode SL, Kale AH (2012) Physiological taste threshold in type 1 diabetes mellitus. Indian J Physiol Pharmacol 56(1):42–47
Perros P, MacFarlane TW, Counsell C, Frier BM (1996) Altered taste sensation in newly-diagnosed NIDDM. Diabetes Care 19(7):768–770
Tepper BJ, Seldner AC (1999) Sweet taste and intake of sweet foods in normal pregnancy and pregnancy complicated by gestational diabetes mellitus. Am J Clin Nutr 70(2):277–284
Park HJ, Lee J, Kim JM, Lee HA, Kim SH, Kim Y (2013) A study of snack consumption, night-eating habits, and nutrient intake in gestational diabetes mellitus. Clin Nutr Res 2(1):42–51
Dotson CD, Zhang L, Xu H, Shin YK, Vigues S, Ott SH, Elson AE, Choi HJ, Shaw H, Egan JM et al (2008) Bitter taste receptors influence glucose homeostasis. PLoS ONE 3(12):e3974
Ma X, Bacci S, Mlynarski W, Gottardo L, Soccio T, Menzaghi C, Iori E, Lager RA, Shroff AR, Gervino EV et al (2004) A common haplotype at the CD36 locus is associated with high free fatty acid levels and increased cardiovascular risk in Caucasians. Hum Mol Genet 13(19):2197–2205
Eny KM, Wolever TM, Corey PN, El-Sohemy A (2010) Genetic variation in TAS1R2 (Ile191Val) is associated with consumption of sugars in overweight and obese individuals in 2 distinct populations. Am J Clin Nutr 92(6):1501–1510
Eny KM, Wolever TM, Fontaine-Bisson B, El-Sohemy A (2008) Genetic variant in the glucose transporter type 2 is associated with higher intakes of sugars in two distinct populations. Physiol Genom 33(3):355–360
Barroso I, Luan J, Middelberg RP, Harding AH, Franks PW, Jakes RW, Clayton D, Schafer AJ, O’Rahilly S, Wareham NJ (2003) Candidate gene association study in type 2 diabetes indicates a role for genes involved in beta-cell function as well as insulin action. PLoS Biol 1(1):E20
Cade JE, Burley VJ, Warm DL, Thompson RL, Margetts BM (2004) Food-frequency questionnaires: a review of their design, validation and utilisation. Nutr Res Rev 17(1):5–22
Welch AA, Luben R, Khaw KT, Bingham SA (2005) The CAFE computer program for nutritional analysis of the EPIC-Norfolk food frequency questionnaire and identification of extreme nutrient values. J Hum Nutr Diet 18(2):99–116
Weidl NM (2010) Development of a semi-quantitative food frequency questionnaire to assess dietary energy intake. University of Vienna, Diplomarbeit, Ernährungswissenschaften
Wakai K (2009) A review of food frequency questionnaires developed and validated in Japan. J Epidemiol 19(1):1–11
Willett WC, Reynolds RD, Cottrell-Hoehner S, Sampson L, Browne ML (1987) Validation of a semi-quantitative food frequency questionnaire: comparison with a 1-year diet record. J Am Diet Assoc 87(1):43–47
Brázdová Z, Fiala J, Bauerová J, Hrubá D (2000) Dietary guidelines in the Czech Republic. I.: theoretical background and development. Cent Eur J Public Health 8(3):186–190
Baci Y, Üstüner I, Keskin HL, Ersoy R, Avşar AF (2013) Effect of maternal obesity and weight gain on gestational diabetes mellitus. Gynecol Endocrinol 29(2):133–136
Horosz E, Bomba-Opon DA, Szymanska M, Wielgos M (2013) Maternal weight gain in women with gestational diabetes mellitus. J Perinat Med 41(5):523–528
Stewart ZA, Wallace EM, Allan CA (2012) Patterns of weight gain in pregnant women with and without gestational diabetes mellitus: an observational study. Aust NZ J Obstet Gynaecol 52(5):433–439
Allen AL, McGeary JE, Knopik VS, Hayes JE (2013) Bitterness of the non-nutritive sweetener acesulfame potassium varies with polymorphisms in TAS2R9 and TAS2R31. Chem Senses 38(5):379–389
Acknowledgments
The study was supported by Ministry of Health of the Czech Republic, Grant Nr. 16-28040A. The authors would like to thank M. Šindelářová, L. Kováčová and K. Kováčová (undergraduate students) for technical help with collecting SFFQ data and for genotyping our DNA samples, Dr. V. Dvořák for the help with recruitment of healthy pregnant controls and a diabetic nurse J. Michurová for the help with filling out the participants form. We also thank our study participants and for their time and participation.
Authors’ contributions
VB designed the study, analysed and interpreted the patient data and was a major contributor in writing the manuscript, KaKu did the genotyping and contributed to writing respective part of the manuscript, FZ participated at statistical analyses, JB collated and organized patient data and contributed to subjects characterization in Methods section of the manuscript, and KaKa was a major supervisor and coordinator of the study, repeatedly revised and finally completed the manuscript. All authors read and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Authors declare a compliance with ethics guidelines.
Informed consent
Study was approved by the Ethical Committee of Faculty of Medicine, Masaryk University, and was conducted in accordance with Helsinki Declaration. Each participant provided an informed consent.
Rights and permissions
About this article
Cite this article
Bartáková, V., Kuricová, K., Zlámal, F. et al. Differences in food intake and genetic variability in taste receptors between Czech pregnant women with and without gestational diabetes mellitus. Eur J Nutr 57, 513–521 (2018). https://doi.org/10.1007/s00394-016-1334-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-016-1334-6