Abstract
Objective
Previous studies suggested that magnesium (Mg) might protect against atherosclerosis, but data were scarce in an Asian population. We examined the association of Mg levels in serum and urine with carotid intima-media thickness (cIMT) and serum lipids in Chinese adults.
Methods
This community-based cross-sectional study recruited 2,837 participants aged 40–75 years in Guangzhou, China. General information, lifestyle factors, serum and urinary concentrations of Mg and cardiometabolic factors were determined. The cIMTs of the common carotid artery (CCA) and the carotid bifurcation (BIF) were measured ultrasonographically.
Results
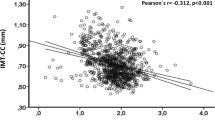
The mean (SD) concentration of serum Mg was 0.85 (0.07) mmol/L and median (IQR) for urinary Mg excretion was 2.29 (1.56–3.51) mmol/L. After adjustment for potential covariates, both serum and the urinary concentrations of Mg were inversely associated with CCA-IMT, but not with BIF-IMT. The regression coefficients (standard errors) were −100 (29) µm (total), −86 (34) µm (women) and −117 (52) µm (men) CCA-IMT per 1 mmol/L of serum Mg, and −41 (8) µm (total), −41 (10) µm (women) and −44 (15) µm (men) CCA-IMT per 1 unit of urinary Mg/creatinine (log mmol/mmol) (all p < 0.05), respectively. Higher serum Mg levels were associated with higher total cholesterol, HDLc, LDLc and triglyceride, but lower non-HDLc/HDLc in total population (all p < 0.05). Similar relationships of urinary Mg with lipoproteins were also found in total population (all p < 0.05).
Conclusion
Higher levels of serum and urinary Mg are associated with lower CCA-IMTs, and the role of Mg in lipid metabolism needs further investigation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Magnesium (Mg), abundant in whole grains, green leafy vegetables and nuts, is a critical cofactor for hundreds of enzymes and a natural antagonist of intracellular calcium, known to affect many aspects of the cardiovascular system [1]. Many studies showed that lower serum or urinary Mg was associated with increased risk of ischemic stroke [2], coronary heart disease (CHD) and cardiovascular mortality [3, 4]. Atherosclerosis is the fundamental pathologic change of cardiovascular diseases (CVDs). In vitro and animal studies showed that Mg prevents atherosclerosis by maintaining endothelial function and improving the lipid profile [5, 6]. However, limited studies examined the association of Mg with atherosclerosis in human.
The measurement of carotid intima-media thickness (cIMT) as an indicator of subclinical atherosclerosis allows the progress of CVD to be evaluated [7]. Previous epidemiologic studies investigating the relationship between Mg and vascular atherosclerosis were mostly aimed at hemodialysis patients, in whom the atherosclerotic process progresses more dramatically than in the general population [8]. These studies suggested that serum Mg concentration was inversely associated with general atherosclerosis, as indicated by cIMT [9, 10]. Compared with general population, more CVD risk factors were found [11] and serum Mg levels were largely influenced by the dialysate in dialysis patients [12]. Little is known about the Mg-cIMT association in healthy individuals and the findings in dialysis patients can not be generalized to general population. To the authors’ knowledge, only the ARIC (Atherosclerosis Risk in Communities) study and another cross-sectional study (n = 728) have examined this association and found higher serum Mg levels to be inversely associated with cIMT in the general population [13, 14]. In ARIC study, the associations of Mg levels with cIMT [14] and ischemic stroke [15] were more pronounced among Whites than Blacks, while data were scarce in the Chinese population. Moreover, it was found that dietary Mg was associated with increased CHD mortality in Chinese women with a lower dietary calcium/Mg ratio (≤1.7) but not in those with a higher calcium/Mg ratio (>1.7) in a population-based cohort study [16]. Thus, the results obtained from Western populations might not be well generalized to Chinese populations with a much lower calcium/Mg ratio (1.7) than that in US populations (around 3.0) [16]. Since ethnic and environmental factors may modify the Mg-atherosclerosis associations, more studies in a variety of populations are needed to get the final conclusion on this issue. Therefore, we tested the hypothesis that higher Mg levels were associated with lower levels of atherosclerosis and better lipid profiles, by examining Mg levels in the serum and urine in relation to cIMT and serum lipid profile in middle-aged and elderly Chinese people.
Methods
Study population
This cross-sectional study was based on the first follow-up of a community-based cohort study designed to assess the determinants of cardiometabolic outcomes and osteoporosis. A total of 3,216 participants aged 40–75 years were recruited in local communities in the city of Guangzhou, China, between July 2008 and June 2010 [17]. Participants with previously confirmed CHD, stroke, chronic renal failure or cirrhosis were excluded at baseline, giving 3,169 participants. Of these initial participants, 2,458 were followed up between April 2011 and January 2013. During this period, an additional 850 participants were recruited in the same way as before. A total of 2,912 participants, comprising the 2,458 who were followed up and 454 newly recruited participants, with completed cIMT measurements were included in this cross-sectional study. We excluded participants with missing data for cIMT (n = 11), laboratory parameters (n = 48), and the questionnaire (n = 16). We further excluded those who reported implausible total energy intakes (<500 or >3,500 kcal/day; n = 18) [18]. After this process, 2,837 participants remained. In analyses of relationship between Mg and serum lipids and blood pressure, we respectively excluded individuals under treatment for dyslipidemia (n = 244), or for hypertension (n = 521). The study protocol was approved by the Ethics Committee of the School of Public Health at Sun Yat-sen University. Written informed consent was obtained from all of the participants at initial enrollment and at follow-up. The study has therefore been performed in accordance with ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Data collection
Participant information was collected by experienced interviewers via a face-to-face interview based on a structured questionnaire covering sociodemographic data, lifestyle factors, physical activity, habitual dietary intake and female reproductive history.
Dietary intake was assessed using a quantitative food frequency questionnaire (FFQ) containing 81 items [19]. For each food item, the participants were asked how often they had consumed a commonly used portion size over the previous year. Cooking oil was not including in this FFQ due to poor validity. Dietary nutrients were calculated by multiplying the consumption of each food by its nutrient content based on the China Food Composition Table [20].
Weight and height were measured while the participants were wearing lightweight clothing and without shoes. Body mass index (BMI, in kg/m2) was calculated. Blood pressure was measured twice, with a 5 min interval, after the participants had rested for ≥10 min. Blood pressure was measured a third time if the systolic blood pressure (SBP) or diastolic blood pressure (DBP) differed by ≥4 or ≥3 mmHg, respectively, between the first two measurements. The average blood pressure was calculated and used in the final analysis.
Laboratory assay
Detailed instructions and tools for urine sample collection were given to each participant at the first visit of the survey in 2011–2013. Participants were asked to collect the middle part of the first void morning urine at home on the day of fasting blood collection, and then carry the urine specimen to our laboratory before 9:30 a.m. A venous blood sample and a urine sample were obtained from each participant on the morning after a 12 h fast. Serum was obtained by centrifuging at 3,000 rev/min for 10 min at 4 °C and was separated into several aliquots and stored at −80 °C within 2 h. The determination of the Mg levels in the serum and urine was based on the reaction of Mg with xylidyl blue in an alkaline solution using a commercial kits (Roche Diagnostics, Mannheim, Germany) with a coefficient variation of 1.2 % for human serum and 3.2 % for human urine. The Mg concentration was estimated photometrically via decrease in the xylidyl blue absorbance. The serum levels of total cholesterol (TC), high-density lipoprotein cholesterol (HDLc), low-density lipoprotein cholesterol (LDLc), and triglycerides (TG) were measured using commercial kits and a Hitachi 7600-010 automatic analyzer (Hitachi, Tokyo, Japan), as described elsewhere [21]. A pooling sample was analyzed with each batch of analyses for quality control. The coefficient variation for the serum measurements in our study was 6.58 % for serum Mg, 4.53 % for TC, 5.92 % for HDLc, 3.45 % for LDLc and 5.76 % for TG. The atherogenic index (log TG/HDLc) was calculated and used as a marker of atherogenicity [22]. The urinary Mg levels in the first morning urine were measured using a Hitachi 7180 automatic analyzer giving a coefficient variation of 3.4 %.
Measurement of cIMT
Carotid intima-media thickness was measured by well-trained technicians. The common carotid artery (CCA; 1 cm proximal to the dilation of the carotid bulb) and the carotid bifurcation (BIF; 1 cm proximal to the flow divider) were measured bilaterally at the far wall of the artery using a 7.5–12.5 MHz linear-array probe (Sonoscape SSI-5500). All of the participants were examined in the supine position with the head slightly extended and turned away from the carotid artery being measured. B-mode images at the CCA and BIF were recorded and the technician then used an electronic caliper to measure the wall thickness on a computer. The common carotid artery intima-media thickness (CCA-IMT) and carotid bifurcation intima-media thickness (BIF-IMT) were defined as the averages of the bilateral values. Site-specific reliability coefficients based on test–retest using a new image from 21 participants selected at random on the same day were estimated at 0.935 for CCA-IMT and 0.917 for BIF-IMT.
Statistical analysis
Continuous variables were presented as means and standard deviation (SD) or medians and interquartile range (IQR), as appropriate. A t test and non-parametric test were performed to compare the means and medians between the women and the men. Categorical variables were presented as number and percentage, and the difference in the frequency of the categorical variables was assessed using Pearson’s Chi square test. The urinary Mg levels were expressed relative to urinary creatinine excretion (UMg/Cr, mmol/mmol). The distributions of the serum TG, UMg/Cr, and dietary nutrients intake were skewed, and logarithmic transformation was thus used to improve the normality and model fit. All of the dietary nutrient intakes were adjusted for total energy intake using a residual method.
Multiple linear regression was used to estimate the regression coefficients and standard error (SE) between the serum and urine Mg levels and the cIMT after adjusting for potential covariates. Model 1 adjusted for age and total energy intake. Model 2 further adjusted for BMI, physical activity, smoking status, alcohol intake, tea intake, supplementary multivitamins, menopausal status (women only), dietary intake of fiber and cholesterol, and ratio of polyunsaturated to saturated fat in the diet, urinary sodium and potassium to creatinine ratio. To examine the independent effects of Mg levels on cIMT, we adjusted for traditional CVD risk factors, including the ratio of serum TC to HDLc, TG level, and SBP, in Model 3. Variance inflation factors were calculated across all of the models to evaluate potential multicollinearity. Model 2 was also used to evaluate the relationships between serum and urine Mg levels and serum lipids. Conclusions were made based on the results of Model 2.
Interactions between Mg and gender, smoking status (never or ever during the past year), alcohol intake (yes or no), tea intake (yes or no), and anti-hypertension or anti-diabetic medication (taking or not taking) were examined. In sensitivity analyses, we repeated the analyses after excluding participants with diagnosed gastric or duodenal ulcer or diabetes mellitus, which may alter Mg homeostasis due to potential medications [23]. All of the analyses were performed using SPSS Statistics (version 20.0, SPSS Inc, Chicago, IL). The reported p values were two-tailed and results with p < 0.05 were considered statistically significant.
Results
The participant characteristics are presented by gender in Table 1. Our study involved 2,837 participants, of whom 1,926 were female with a mean age of 59.8 years and 911 were male with a mean age of 62.3 years. The mean (±SD) concentration of serum Mg was 0.85 (±0.07) mmol/L for both genders. The median excretion of UMg/Cr (mmol/mmol) was 0.45 for women and 0.32 for men (p < 0.001). The means (±SE) of cIMT was lower in women than in men, with 909 (±2.62) µm versus 956 (±4.31) µm in common carotid artery and 950 (±2.87) µm versus 998 (±4.37) µm in carotid bifurcation, respectively (all p < 0.001).
As shown in Table 2, both the serum and the urinary concentrations of Mg were inversely associated with CCA-IMT in Models 1 and 2 for both women and men (p < 0.05 for all results). Similar associations were observed after further adjusting for cardiometabolic factors using Model 3. In Model 2, CCA-IMT decreased by 100 (±29) µm in total participants (p < 0.001), 86 (±34) µm in women (p = 0.012) and 117 (±52) µm in men (p = 0.024) per 1 mmol/L of serum Mg, and by 41 (±8) µm in total subjects (p < 0.001), 41 (±10) µm in women (p < 0.001) and 44 (±15) µm (p = 0.004) in men per 1 unit of urinary Mg/Cr (log mmol/mmol). No significant association was found between the serum or urinary Mg levels and BIF-IMT.
After adjusting for potential confounding factors in Model 2, higher levels of serum Mg were associated with higher TC, HDLc, LDLc and TG, but lower non-HDLc/HDLc in total population (all p < 0.05). Positive associations of urinary Mg with TC, HDLc, and LDLc were also found in total population (all p < 0.05). Similar trends in these associations were observed between women and men (p interaction range: 0.186–0.848). No significant association of Mg with blood pressure was found (Table 3).
No significant interaction between the Mg levels in serum or urine and gender, smoking status, alcohol intake, tea intake, or anti-hypertension, or anti-diabetic medication in relation to cIMT or lipids was observed (p interaction range: 0.223–0.929). After excluding participants with diagnosed gastric or duodenal ulcer or diabetes mellitus, the results on the associations of serum or urinary Mg levels with cIMT or serum lipids were not substantially different from those in the total study population (data not shown).
Discussion
In this large, community-based cross-sectional study in middle-aged and elderly Chinese people, Mg levels in serum and urine were found to be inversely associated with CCA-IMT, an indicator of general atherosclerosis.
To our knowledge, only two cross-sectional studies have investigated the relationship between serum Mg level and cIMT in the general population. The ARIC study, which involved 9,572 participants aged 45–64 years, reported that the mean level of serum Mg (range 0.79–0.83 mmol/L) was inversely associated with age-adjusted average cIMT (range 0.68–0.82 mm) in women and White men but not in Black men [14]. In another cross-sectional study including 728 Japanese people, the mean CCA-IMT was significantly lower (0.71 vs. 0.75 mm) in participants in the top (vs. bottom) quartile of serum Mg (>0.96 vs. <0.88 mmol/L) [13]. Consistent with previous studies, we found that Mg levels in serum and urine were inversely associated with cIMT in a middle-aged and elderly Chinese population. A magnesium supplementation trail has not yet been conducted in general population with respect to atherosclerosis as indicated by cIMT. However, a small pilot study in patients with chronic hemodialysis (at particularly high risk for atherosclerosis progression) reported cIMT decreased by 9.3 % compared with baseline in the group treated orally with 440 mg of Mg oxide 3 times per week for 6 months (p = 0.001) [10]. In addition to Mg acting as an antagonist of calcium in inhibiting hydroxyapatite and crystal precipitation [24], low Mg levels are reported to be associated with a proinflammatory state, oxidative stress, endothelial dysfunction and platelet aggregation, which may potentially accelerate the progression of atherosclerosis [6]. The inverse relationships of the Mg levels in serum and urine with cIMT suggest a protective role of Mg in the process of atherosclerosis.
It has long been argued whether the protective role of Mg is its improvement of lipid metabolism. An animal study has postulated a favorable effect of Mg in lipid metabolism in the conversion of TG to HDLc [25]. This result was supported by the cross-sectional ARIC study, in which serum Mg level was inversely associated with TG and positively associated with HDLc and LDLc in 4,308 White men [14]. Another cross-sectional study linked low levels of serum Mg to high levels of TG and TC and low levels of HDLc in 290 participants with type 2 diabetes [26]. However, we found that the Mg levels in both serum and urine were positively associated with TC, HDLc and LDLc in the middle-aged and elderly Chinese population. Similarly, a cross-sectional study involving 1,318 middle-aged participants suggested that serum Mg concentration was positively associated with TG, TC, HDLc, and LDLc [27]. A theory suggesting a simple binding interaction between serum Mg and lipoprotein particles may partially explain our findings. According to this theory, Mg was given for the affinity of certain phospholipids head groups as divalent cation, resulting in a positive correlation between Mg and possible all lipoprotein species [28]. Nevertheless, considering the significantly inverse relationship of the serum Mg level with the non-HDLc/HDLc ratio observed in our study, we cannot rule out a possible mechanism in which Mg has a favorable effect in terms of improving lipid composition. Future studies are needed to elucidate the role of Mg in lipid metabolism.
In our study, the favorable association between Mg and cIMT was more pronounced in the CCA than the BIF. Such a segment-specific association of Mg with cIMT has not previously been reported. The reasons for these different magnitudes of association are not clear, although pathophysiologic differences may provide some clues. Both of the sites differ in their geometry, shear stress, and cell composition. The shear stress rates are cyclically constant in the CCA but have more complex oscillations in the BIF, resulting in a preponderance of foam cells in the CCA and a primary deposition of LDLc observed in the wall at the BIF [29]. Previous work has indicated that this pathophysiologic difference is the key to segmental differences in risk factors for vascular alteration [30]. Therefore, the positive relationship between the Mg level and the LDLc level observed in our study and in others [13, 27] may weaken the protective effect of Mg in the BIF. In addition, the predominance of foam cells in the CCA makes this the preferred site for measurements of cIMT, and the use of cIMT as a marker of atherosclerosis makes more sense if foam cells are responsible for the dynamics of cIMT [31].
In this study, we evaluated the associations of Mg levels in serum and urine with cIMT, and observed consistent results for serum and urinary Mg. Although Mg is mostly present in the intracellular space [32], serum Mg concentrations show a moderate correlation (r = 0.54) with intracellular free Mg [33], and thus serum levels may represent the nutritional status of Mg in the body. The urinary excretion of Mg has also been suggested as an objective estimate of the amount of Mg that is systemically absorbed because of its ability to capture the amount of Mg in food and supplement that has been available for biological activity in tissues and cells [34]. The estimation of dietary Mg was suffer from limitations of poor precision and uncertainty of intestinal absorption. Although our FFQ was validated before [19], the reliability and validity of the assessment of dietary Mg intakes were unknown in this study. Serum and urinary markers would be superior to measurements of dietary intake in assessing the nutritional status of Mg. Thus, the consistent results for Mg in serum and in urine in relation to cIMT in our study make the favorable association of Mg with atherosclerosis more credible.
Some limitations of our study merit consideration. First, the cross-sectional study design made it difficult to determine the direction of causality. Replication of these findings in randomized controlled trials and additional research into the casual mechanism are warranted. Second, the participants in our study were recruited from the community through volunteer recruitment rather than random selection, which may have affected the representativeness of the results. However, the gender- and age-specific distributions of cardiometabolic risk factors in our study are consistent with those in the China National Diabetes and Metabolic Disorders Study [35]; and gender, smoking status, alcohol intake, tea intake, and medication for hypertension or diabetes did not significantly modify the Mg-cIMT association found in this study. Third, we only collected the first void morning urine instead of 24-h urine. Although 24-h urine is a more accurate surrogate for habitual dietary Mg intake [36], previous study showed the creatinine-adjusted Mg in first void morning urine was highly correlated with the total Mg excretion in 24-h urine (r = 0.80) [37]. Fourth, we did not have data on serum or urinary concentration of calcium, and the possible modifying effect of calcium/Mg ratio could not be tested in this study. Finally, Mg is mainly obtained by the body through food, and a higher Mg level in the serum or urine may be representative of a healthy diet in general. Although we adjusted carefully for some other dietary factors, residual confounding cannot be completely controlled in observational studies.
In conclusion, high levels of Mg in serum and urine were inversely associated with CCA-IMT and non-HDL/HDL (serum Mg only), but not with BIF-IMT in middle-aged and elderly Chinese adults. Despite positive associations of Mg with serum TC and LDL were also found in our study, the inverse relationships between serum Mg and cIMT and non-HDL/HDL suggested Mg might be benefit to atherosclerosis prevention in this population. Special attention should be given to Mg and randomized controlled trials are needed to test the casual relationship of Mg with the cIMT and lipid profile in the general population.
References
Chakraborti S, Chakraborti T, Mandal M, Mandal A, Das S, Ghosh S (2002) Protective role of magnesium in cardiovascular diseases: a review. Mol Cell Biochem 238:163–179
Joosten MM, Gansevoort RT, Mukamal KJ et al (2013) Urinary and plasma magnesium and risk of ischemic heart disease. Am J Clin Nutr 97:1299–1306. doi:10.3945/ajcn.112.054114
Del Gobbo LC, Imamura F, Wu JH, de Oliveira Otto MC, Chiuve SE, Mozaffarian D (2013) Circulating and dietary magnesium and risk of cardiovascular disease: a systematic review and meta-analysis of prospective studies. Am J Clin Nutr 98:160–173. doi:10.3945/ajcn.112.053132
Qu X, Jin F, Hao Y et al (2013) Magnesium and the risk of cardiovascular events: a meta-analysis of prospective cohort studies. PLoS One 8:e57720. doi:10.1371/journal.pone.0057720
Altura BT, Brust M, Bloom S, Barbour RL, Stempak JG, Altura BM (1990) Magnesium dietary intake modulates blood lipid levels and atherogenesis. Proc Natl Acad Sci USA 87:1840–1844
Maier JA, Malpuech-Brugere C, Zimowska W, Rayssiguier Y, Mazur A (2004) Low magnesium promotes endothelial cell dysfunction: implications for atherosclerosis, inflammation and thrombosis. Biochim Biophys Acta 1689:13–21. doi:10.1016/j.bbadis.2004.01.002
Toth PP (2008) Subclinical atherosclerosis: what it is, what it means and what we can do about it. Int J Clin Pract 62:1246–1254. doi:10.1111/j.1742-1241.2008.01804.x
Amann K, Tyralla K, Gross ML, Eifert T, Adamczak M, Ritz E (2003) Special characteristics of atherosclerosis in chronic renal failure. Clin Nephrol 60(Suppl 1):S13–S21
Liu F, Zhang X, Qi H et al (2013) Correlation of serum magnesium with cardiovascular risk factors in maintenance hemodialysis patients—a cross-sectional study. Magnes Res off organ Int Soc Dev Res Magnes 26:100–108. doi:10.1684/mrh.2013.0344
Mortazavi M, Moeinzadeh F, Saadatnia M, Shahidi S, McGee JC, Minagar A (2013) Effect of magnesium supplementation on carotid intima-media thickness and flow-mediated dilatation among hemodialysis patients: a double-blind, randomized, placebo-controlled trial. Eur Neurol 69:309–316. doi:10.1159/000346427
Yerram P, Karuparthi PR, Hesemann L, Horst J, Whaley-Connell A (2007) Chronic kidney disease and cardiovascular risk. J Am Soc Hypertens 1:178–184. doi:10.1016/j.jash.2007.01.010
Spiegel DM (2011) Magnesium in chronic kidney disease: unanswered questions. Blood Purif 31:172–176. doi:10.1159/000321837
Hashimoto T, Hara A, Ohkubo T et al (2010) Serum magnesium, ambulatory blood pressure, and carotid artery alteration: the Ohasama study. Am J Hypertens 23:1292–1298. doi:10.1038/ajh.2010.168
Ma J, Folsom AR, Melnick SL et al (1995) Associations of serum and dietary magnesium with cardiovascular disease, hypertension, diabetes, insulin, and carotid arterial wall thickness: the ARIC study. Atherosclerosis Risk in Communities Study. J Clin Epidemiol 48:927–940
Ohira T, Peacock JM, Iso H, Chambless LE, Rosamond WD, Folsom AR (2009) Serum and dietary magnesium and risk of ischemic stroke: the atherosclerosis risk in communities study. Am J Epidemiol 169:1437–1444. doi:10.1093/aje/kwp071
Dai Q, Shu XO, Deng X et al (2013) Modifying effect of calcium/magnesium intake ratio and mortality: a population-based cohort study. BMJ Open. doi:10.1136/bmjopen-2012-002111
Liu YH, Xu Y, Wen YB et al (2013) Association of weight-adjusted body fat and fat distribution with bone mineral density in middle-aged chinese adults: a cross-sectional study. PLoS One 8:e63339. doi:10.1371/journal.pone.0063339
Joshipura KJ, Hu FB, Manson JE et al (2001) The effect of fruit and vegetable intake on risk for coronary heart disease. Ann Intern Med 134:1106–1114
Zhang CX, Ho SC (2009) Validity and reproducibility of a food frequency questionnaire among Chinese women in Guangdong province. Asia Pac J Clin Nutr 18:240–250
Yang YX, Wang GY, Pan XC (eds) (2002) China food composition tables. Peking University Medical Press, Beijing
Zhang ZQ, Deng J, He LP, Ling WH, Su YX, Chen YM (2013) Comparison of various anthropometric and body fat indices in identifying cardiometabolic disturbances in Chinese men and women. PLoS One 8:e70893. doi:10.1371/journal.pone.0070893
Dobiasova M, Frohlich J (2001) The plasma parameter log (TG/HDL-C) as an atherogenic index: correlation with lipoprotein particle size and esterification rate in apoB-lipoprotein-depleted plasma (FER(HDL)). Clin Biochem 34:583–588
Djurhuus MS, Skott P, Hother-Nielson O, Klitgaard NA, Beck-Nielsen H (1995) Insulin increases renal magnesium excretion: a possible cause of magnesium depletion in hyperinsulinaemic states. Diabet Med 12:664–669
Laurencin D, Almora-Barrios N, de Leeuw NH et al (2011) Magnesium incorporation into hydroxyapatite. Biomaterials 32:1826–1837. doi:10.1016/j.biomaterials.2010.11.017
Rayssiguier Y, Noe L, Etienne J, Gueux E, Cardot P, Mazur A (1991) Effect of magnesium deficiency on post-heparin lipase activity and tissue lipoprotein lipase in the rat. Lipids 26:182–186
Corica F, Corsonello A, Ientile R et al (2006) Serum ionized magnesium levels in relation to metabolic syndrome in type 2 diabetic patients. J Am Coll Nutr 25:210–215
Randell EW, Mathews M, Gadag V, Zhang H, Sun G (2008) Relationship between serum magnesium values, lipids and anthropometric risk factors. Atherosclerosis 196:413–419. doi:10.1016/j.atherosclerosis.2006.11.024
Merchant TE, Glonek T (1992) 31P NMR of tissue phospholipids: competition for Mg2+ , Ca2+ , Na+ and K+ cations. Lipids 27:551–559
Malek AM, Alper SL, Izumo S (1999) Hemodynamic shear stress and its role in atherosclerosis. JAMA 282:2035–2042
Polak JF, Person SD, Wei GS et al (2010) Segment-specific associations of carotid intima-media thickness with cardiovascular risk factors: the coronary artery risk development in young adults (CARDIA) study. Stroke 41:9–15. doi:10.1161/STROKEAHA.109.566596
Dalager S, Paaske WP, Kristensen IB, Laurberg JM, Falk E (2007) Artery-related differences in atherosclerosis expression: implications for atherogenesis and dynamics in intima-media thickness. Stroke 38:2698–2705. doi:10.1161/STROKEAHA.107.486480
Vormann J (2003) Magnesium: nutrition and metabolism. Mol Aspects Med 24:27–37
Ryzen E, Servis KL, DeRusso P, Kershaw A, Stephen T, Rude RK (1989) Determination of intracellular free magnesium by nuclear magnetic resonance in human magnesium deficiency. J Am Coll Nutr 8:580–587
Joosten MM, Gansevoort RT, Mukamal KJ et al (2013) Urinary magnesium excretion and risk of hypertension: the prevention of renal and vascular end-stage disease study. Hypertension 61:1161–1167. doi:10.1161/HYPERTENSIONAHA.113.01333
Yang ZJ, Liu J, Ge JP et al (2012) Prevalence of cardiovascular disease risk factor in the Chinese population: the 2007–2008 China National Diabetes and Metabolic Disorders Study. Eur Heart J 33:213–220. doi:10.1093/eurheartj/ehr205
Bohn T, Walczyk T, Davidsson L et al (2004) Comparison of urinary monitoring, faecal monitoring and erythrocyte analysis of stable isotope labels to determine magnesium absorption in human subjects. Br J Nutr 91:113–120
Tang NL, Cran YK, Hui E, Woo J (2000) Application of urine magnesium/creatinine ratio as an indicator for insufficient magnesium intake. Clin Biochem 33:675–678
Acknowledgments
The study was jointly supported by the 5,010 Program for Clinical Researches (No. 2007032) by the Sun Yat-sen University, Guangzhou, P. R. China and the Program of National Natural Science Foundation of China (Numbers 81472965 and 81130052). The funders played no role in study design; in the collection, analysis and interpretation of data; in the writing of the report; and in the decision to submit the article for publication.
Conflicts of interest
None of the authors had any conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Yi Cao and Cheng Wang have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Cao, Y., Wang, C., Guan, K. et al. Association of magnesium in serum and urine with carotid intima-media thickness and serum lipids in middle-aged and elderly Chinese: a community-based cross-sectional study. Eur J Nutr 55, 219–226 (2016). https://doi.org/10.1007/s00394-015-0839-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-015-0839-8