Abstract
Background
The electrical activity of atria can be demonstrated by P waves on surface electrocardiogram (ECG). Atrial electromechanical delay (AEMD) measured with tissue Doppler imaging (TDI) echocardiography can be a useful non-invasive method for evaluating atrial conduction features. We investigated whether AEMD is prolonged in patients with chronic obstructive pulmonary disease (COPD).
Patients and methods
Study consisted of 41 (15 female, 26 male, mean age 62 + 12 years) patients with COPD and 41 healthy subjects. Pulmonary function tests,12 lead surface ECG and echocardiographic examination were performed and recorded. P wave changes on surface ECG, minimum (P min) and maximum (P max) duration of P wave and its difference as P wave dispersion (P wd) were measured and recorded. Atrial electromechanic delay (AEMD) was calculated from colored-TDI recordings.
Results
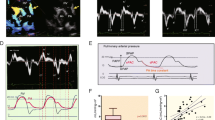
Pulmonary functions were significantly lower in COPD group than the control group as expected. Right atrial areas and pulmonary arterial systolic pressures (PAP) were significantly higher in COPD group than the controls (right atrial area: 11.9 ± 3.4 cm2 and 8.2 ± 2.2 cm2, p < 0.0001 and PAP: 38.4 ± 12.2 and 19.0 ± 3.2 mmHg p < 0.0001, respectively). P wave intervals on surface ECG were significantly increased in COPD patients than the control group (P max: 105 ± 11 and 90 ± 12 ms, p < 0.0001; P min: 60 ± 12 and 51 ± 10 ms, p = 0.003 and P wd: 39 ± 10 and 31 ± 7 ms, p < 0.0001). According to the AEMD measurements from different sites by TDI, there was a significant delay between the onset of the P wave on surface ECG and the onset of the late diastolic wave in patients with COPD when compared with controls measured from tricuspid lateral septal annulus (TAEMD) (COPD: 41.3 ± 9.8 ms, control: 36 ± 4.5 ms; p = 0.005). There was a positive correlation between TAEMD and right atrial area (r = 0.63, p < 0.0001) and also between TAEMD and PASP (r = 0.43, p < 0.0005) and a negative correlation between TAEMD and forced expiratory volume (FEV1) (r = −0.44, p = 0.04).
Conclusions
Right atrial electromechanical delay is significantly prolonged in patients with COPD. The right atrial area, PAP and FEV1 levels are important factors of this prolonged delay. Also the duration of atrial depolarization is significantly prolonged and propagation of depolarization is inhomogeneous in patients with COPD. These may be the underlying mechanisms to explain the atrial premature beats, multifocal atrial tachycardia, atrial flutter and fibrillation often seen in patients with COPD secondary to these changes.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Chronic obstructive pulmonary disease (COPD) is characterized by bronchial wall fibrosis and obstruction. It is a progressive disease that affects anatomy and physiology of the heart, eventually [1–3]. The most common cardiac pathologies secondary to pulmonary disorders may be recalled as dilatation and hypertrophy. Atrial premature beats, multifocal atrial tachycardia, atrial flutter and fibrillation are often seen in patients with COPD secondary to the morphological changes [4–9]. The pathophysiology of those arrhythmias is not well defined; however, may be summarized as ‘increased atrial automaticity’. Slowing down of electrical conduction due to fibrosis is another important factor that plays role in arrhythmia formation in such circumstances [7–10]. To precisely name the conduction abnormality, electrophysiological studies are required. On the other hand, their widespread applicability is limited due to their invasive nature and high costs [4–6]. A non-invasive, safe, cheap and clinically relevant method that shows parallel data would be very helpful.
P wave on surface electrocardiogram (ECG) demonstrates the depolarization of atria and influenced by electrical conduction changes of atrial tissue. The previous studies have shown prolongation of P wave duration and P wave dispersion was assosiated with paroxysmal atrial fibrillation (PAF) in patients undergoing coronary artery bypass surgery, patients with hypertension, hypertrophic cardiomyopathy, right atrial dilatation with atrial septal defect, diastolic dysfunction and COPD [9, 11–15].
In normal hearts, depolarization is followed by contraction of the myofibers. The relationship between the spatiotemporal pattern of ventricular electrical activation and the pattern and degree of mechanical response is important in understanding the function of the diseased heart. Electromechanical delay (EMD) is defined simply in heart physiology as the time interval between electrical depolarization and ventricular contraction. Tissue Doppler imaging (TDI) is a noninvasive and simple echocardiographic techique that can be used to evaluate mechanical properties and also EMD of both atrial and ventricular myocardium [16–21]. Published studies have shown increment of EMD by TDI in different patient groups like mitral stenosis, paroxysmal atrial fibrillation, coronary slow-flow pattern, ankylosing spondylitis and diabetes mellitus [22–28]. In this study, we aimed to evaluate atrial conduction features obtained by TDI technique in patients with COPD.
Patients and methods
Patient popuation
The study was performed at our instituion between 2009–2010. Ethical approval was obtained from the local ethics committee. Prior to inclusion to the research, the study protocol was explained in details to all individuals and patients were included in the study following their consent. Study group consisted of 41 (15 females, 26 males) patients with COPD who were followed up at chest disease out-patient clinics. Mean age was 62 ± 12 years. Control group was chosen as to be compatible in terms of age and sex with the study group. In the study group, the diagnosis of COPD in patients was first made 4–12 years before. Patients with known right or left ventricular failure, atrial fibrillation, right and left bundle branch block, moderate–severe valve abnormalities, known coronary artery disease, diabetes mellitus, chronic renal failure, anemia, thyroid dysfunction and rheumatic diseases were excluded from the study. None were receiving pharmacological cardiovascular drugs. Pulmonary function tests were performed before echocardiographic examination to all patients. Patients with serious obstructive disease who according to the prediction of echocardiography were difficult to assess were excluded
Pulmonary function tests (spirometric evaluation)
Pulmonary function test was performed with Zan 300 spirometer (ZAN Messgerate GmbH, Oberthulba, Germany). Patients were made to sit 90° upright and the nose off. At least three challenging expiratory maneuvers were made and the maneuver that has the best values was recorded. Global Initiative for Chronic Obstructive Lung Disease (GOLD) 2006 criteria were used for the diagnosis of the COPD.
Electrocardiographic examination
Twelve-lead ECG in supine position with speed of 50 mm/s and gain with 0.5 cm/mV was recorded. P wave durations were measured manually by two of the investigators who were blind about clinical status of the patients and patient groups. Measurements were performed with calipers and under magnifying lenses for greater accuracy. Maximum (P max) and minimum (P min) P wave durations were measured and recorded. The difference between P max and P min was calculated as P wave dispersion (P wd).
Echocardiographic examination
Transthoracic echocardiography (TTE) recordings were performed while the patients were at left lateral decubitus position. Apical four chambers, two chambers and parasternal long axis images were recorded at expiratory apnea for three consecutive cardiac cycles with Vivid 7 (GE Vingmed Ultrasound, Horten, Norway) echocardiography device. Records were analyzed by two physicians, who were blind to the study. Electrocardiography was continuously recorded during echocardiographic studies. Two-dimensional and M-mode TTE imaging were performed according to American Society of Echocardiography guidelines. M-mode measurements of left ventricular end-diastolic diameter (LVEDD), end-systolic diameter (LVESD), interventricular septum (IVS), posterior wall (PW) and left atrial diameter (LA) measurements were obtained from parasternal long axis view. Mitral early diastolic (E) and late diastolic maximal (A m) flow rates were measured from apical four-chamber view with pulse-wave Doppler. Left atrial maximal volume (LA-vol) was calculated at the end of ventricular systole by ventricular biplane area length method from apical 4- and 2-chamber views. Teicholz method was used for left ventricular ejection fraction (EF) calculation. Pulmonary artery systolic pressure was measured by Bernoulli equation (p = 4v 2) from tricuspid regurgitation jet flow. Estimated pulmonary artery systolic pressure then calculated as adding 5–20 mmHg; according to inferior vena cava width; to pulmonary artery systolic pressure (PAP). Colored-TDI images were recorded from the apical 4-chamber window with possible highest frame number, correction angle kept below 30°, using a 2.5-mm sample volume.
Atrial electromechanical delay measurement
Atrial electromechanical delay (AEMD) was calculated from colored-TDI recordings. Atrial electromechanical delay was defined as the time between the beginning of electrocardiographic P wave to the initial of Am wave in TDI recordings. Atrial electromechanic delay was measured from mitral lateral, mitral septal and tricuspid lateral annulus from apical-4-chamber views and named, respectively, as LAEMD, SAEMD and TAEMD.
Statistical analyis
The statistical analyses were performed using Statistical package for Social Sciences (SPSS Inc, Chicago, IL, USA) for Windows 15.0 computer program. Besides descriptive statistical methods, for quantitative comparisons paired samples t test was used for parameters that are normally distributed within groups and Wilcoxon test was used for parameters that are not distributed normally within groups. Non-normally distributed parameters between groups are compared by Mann–Whitney U test. For relations between parameters, Pearson correlation analysis was used for normally distributed parameters and Spearman’s correlation analysis was used for non-normal parameters. Results were evaluated at 95% confidence interval, and a p value less than 0.05 was accepted significant.
Results
Patient demographics
There were 41 patients in both the COPD and the control groups. Mean ages were 62 ± 12 (46–76) years and 63 ± 15 (44–79) years, respectively. There were 26 males and 15 females in the study group and 27 males and 14 females in the control group. Body mass indices were 28.7 ± 4.4 kg/m2 (24.2–33.8) in COPD group and 28.2 ± 4.5 kg/m2 (23.5–33.2) in the control group. Body surface areas were similar between both groups as 1.8. ± 0.17 m2 (1.7–1.9) and 1.8 ± 0.25 m2 (1.7–1.9). There were 10 smokers in each group. Eighteen patients were hypertensive and received various anti-hypertensives in the COPD group and there were 19 hypertensive patients in the control group. Anti-hypertensive medications were similar. Patients received bronchodilator agents according to the prescription of the pulmonology specialists. The demographic characteristics of COPD and control groups are shown on Table 1. Age, sex, history of hypertension, smoking status, medications, body mass index and body surface area comparisons were not significantly different between two groups.
Conventional echocardiographic examination and spirometric evaluation
Standard TTE measurements and spirometric assessments are presented in Table 2. Pulmonary function values of FVC, FEV1, PEF, FEV1% measured with the spirometer were significantly lower in the COPD group than the control group. LVEDD, LVESD, IVS, PW, mitral A and E wave flow rate measurements did not differ between groups. Right atrial area dimension and PAP were significantly higher in COPD group than control group [(RAd: 11.9 ± 3.4, 8.2 ± 2.2 mm, respectively; p < 0.0001) and (PAP: 38.4 ± 12.2, 19 ± 3.2 mmHg, respectively; p < 0.0001)]. LA volumes were significantly lower in COPD group than control group (COPD group LA vol: 39.5 ± 8.6 mL, Control group LA vol: 47.3 ± 11.6 mL; p = 0.001).
Atrial electromechanical delay
P wave measurements are shown in Table 1. There was a significant prolongation of P wave durations of COPD group than controls, (P max: 105 ± 11 and 90 ± 12 ms, p < 0.0001 and P min: 60 ± 12 and 51 ± 10 ms, p = 0.003) and also heterogenity of P wave duration; P wd; is significantly higher in COPD group (P wd: 39 ± 10 and 31 ± 7 ms, p < 0.0001).
Atrial electromechanical delay measurements from different sites by TDI are noted in Table 2. There was significant delay between the onset of the P wave on surface ECG and the onset of the late diastolic wave obtained by tissue Doppler echocardiography (Fig. 1) in patients with COPD compared with controls measured from tricuspid lateral septal annulus (COPD: 41.3 ± 9.8 ms, control: 36 ± 4.5 ms; p = 0.005), (Fig. 2). The two groups were similar with respect to SAEMD and LAEMD. There was a positive correlation between TAEMD and right atrial area (r = 0.63, p < 0.0001; Fig. 3) and PAP (r = 0.43, p = 0.005; Fig. 4) in COPD group, and a significant negative correlation between TAEMD and FEV1 (r = −0.44, p = 0.04; Fig. 5); however, the level of significance was weak. TAEMD was not correlated with age, blood pressure, heart rate, left ventricular EF, LVEDD, LVESD, and the LA volume.
Discussion
Supraventricular tachyarrhythmias are common in patients with COPD. Several factors such as hypoxemia, hypercapnia, acid–base disturbances and autonomic dysfunction may contribute to development of arrhythmias in these patients [5, 29, 30]. In addition, structural changes in myocardium may be implicated for development of atrial fibrillation in COPD groups [31–33].
In our study, patients with COPD had similar left atrial electromechanical delay, but prolonged right atrial electromechanical delay measurements than patients in control group. This prolongation of SAEMD was significantly correlated with right atrial area and PAP and negatively correlated with FEV1 measurements. Also, we found out that patients with COPD had bigger right atrium and smaller left atrium dimensions probably secondary to the increase in the PAP. Moreover, we found that P wave durations and P wave dispersion are significantly higher in COPD patients than controls.
Previous studies have shown that prolongation of P wave duration and P wave dispersion were assosiated with PAF in patients undergoing coronary artery bypass surgery, patients with hypertension, hypertrophic cardiomyopathy, right atrial dilatation with atrial septal defect and COPD [9, 11–15]. Moreover, P wave dispersion has been used for prediction of idiopathic PAF [34]. Dilaveris et al. [35] also found prolonged maximum P wave durations, as a predictor of frequently relapsing atrial fibrillation.
Atrial electromechanical delay (AEMD) can be measured with TDI by measuring temporal relation between atrial myocardial regional motions simultaneous with electrocardiographic P waves [16–19]. AEMD has been evaluated by this method in some cardiac disorders such as mitral stenosis, paroxysmal atrial fibrillation (PAF) and congestive heart failure (CHF) [22, 23, 26]. Omi et al. [24] showed that patients with PAF had longer TAEMD and bigger right atrial dimensions than control group. However, in another study by Pala et al. [36] which was planned to show prolongation of AEMD in patients with non-ischemic dilated cardiomyopathy, researchers pointed out that while SAEMD and LAEMD were significantly prolonged in cardiomyopathy group than the control group, there was no significant prolongation of TAEMD between groups. Multivariate analysis in that study showed left atrial maximal volume was an independent predictor for LAEMD while right atrial dimensions were similar between groups [36]. Ozer et al. [22] assessed AEMD in patients with mitral stenosis and also revealed a significant positive correlation between prolonged LAEMD and increase in left atrial size. This indicates atrial size as an important factor for determining AEMD. Similarly, in our study there was moderate positive correlation between right atrial area and TAEMD. Even left atrial dimensions were similar between groups, left atrial electromechanical delay was observed in the COPD group.
In order to create an effective ventricular contraction relevant atrial or ventricular myocard should be totally depolarized. The size of the tissue that will depolarize is the most important factor that determines speed of depolarization. Atrial size is an important factor to determine the degree of AEMD and plays an important role in depolarization; hence, effective cardiac output.
Pulmonary arterial intimal and medial cellular hypertrophy, and hyperplasia are the reasons of increaed PAP levels in COPD patients [37–41]. Moreover, decrease in prostacyclin synthetase and nitric oxide secretion in lungs, high levels of endothelin-1, impaired serotonin metabolism also play role in PAP increment. Pulmonary hypertension, as a result of high PAP levels, is the beginning of right ventricular hypertrophy [37–40]. Right ventricular hypertrophy together with right ventricular enlargement leads to tricuspid annulus enlargement, functional tricuspid valve failure and right atrial dilatation. Right atrial remodeling, as a result of all these factors, impairs the electrical conduction pathways and leads to slow down and that may explain why SAEMD prolongs under such circumstances. Besides these; increased atrial tension secondary to high PAP levels, delays atrial depolarization and so may prolong AEMD [37–40].
Atrial extrasystole, multifocal atrial tachycardia, atrial fibrillation and flutter are seen more frequently in COPD patients than normal population. Among these arrhythmias, especially atrial fibrillation is a major cause of morbidity and mortality [5–8, 33]. Prediction of atrial fibrillation earlier and elimination of triggering factors are important for the prognosis [5, 6, 41]. In a recently published study, basal AEMD has been recorded in 249 people with sinus rhythm. After nearly 2 years of follow-up, 15 patients (6%) developed AF. Of these patients those who developed AF, had prolonged AEMD than non-AF developers. Moreover, in sub-group analysis, the prevalence of COPD appeared to be higher in patients who developed AF. Consequently investigators reported prolonged AEMD by TDI as a predictor for development of AF [27]. A more recent study, Weijs et al. [20] measured total atrial conduction time with a newly developed transthoracic echocardiographic tool; PA-TDI (the time from the initiation of the P wave on ECG to the A’ wave on the lateral left atrial tissue Doppler tracing). As a conclusion, the researchers reported prolonged PA-TDI interval might predict the development of new-onset AF [20].
Study limitations
The major limitation of our study is its cross-sectional design and lack of follow-up of the patients. The sample size was also relatively small. Because of cross-sectional design, we could not evaluate much whether tricuspid annular electromechanical delay in COPD group that we showed predicts arrhythmia or not. Another limitation of the study is the relatively short duration of the patient follow-up. A documented arrythmia in any of the patients with COPD after the research period would definitely increase the significance of the study; however, during the controls, none of the patients were admitted with persistant arrythmias including AF.
Conclusions
Right atrial electromechanical delay is significantly prolonged in patients with COPD. The right atrial area, PAP, and FEV1 levels are important factors of this prolonged delay. The P wave durations and P wave dispersions in patients with COPD are higher which may indicate an impaired atrial depolarization. The study may explain the pathophysiology of the atrial rhythm disturbances seen in COPD patients. However, larger prospective long-term follow-up studies are warranted to reach a precise definition.
References
Rabe KF, Hurd S, Anzueto A, Barnes PJ, Buist SA, Calverley P, Fukuchi Y, Jenkins C, Rodriguez-Roisin R, van Weel C, Zielinski J (2007) Global initiative for chronic obstructive lung disease. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: GOLD executive summary. Am J Respir Crit Care Med 176(6):532–555 (Epub 2007 May 16)
Umut S, Erdinc E (2001) Torax Kitapları. Sayı:2; Kronik Obstruktif Akciğer Hastalığı. Turgut Yayıncılık, İstanbul, pp 11–18
Rodriguez Roisin R, MacNee W (1998) Pathophysiology of chronic obstructive pulmonary disease. In: Postma DS, Siafakas NM (eds) Management of chronic obsructive pulmonary disease. Eur Respir Monograph, vol 3, pp 107–126
Buch P, Friberg J, Scharling H, Lange P, Prescott E (2003) Reduced lung function and risk of atrial fibrillation in The Copenhagen City heart study. Eur Respir J 21:1012
Shih HT, Webb CR, Conway WA, Peterson E, Tilley B, Goldstein S (1988) Frequency and significance of cardiac arrhythmias in chronic obstructive lung disease. Chest 94(1):44–48
Hudson LD, Kurt TL, Petty TL, Genton E (1973) Arrhythmias associated with acute respiratory failure in patients with chronic airway obstruction. Chest 63(5):661–665
Tükek T, Yildiz P, Atilgan D, Tuzcu V, Eren M, Erk O, Demirel S, Akkaya V, Dilmener M, Korkut F (2003) Effect of diurnal variability of heart rate on development of arrhythmia in patients with chronic obstructive pulmonary disease. Int J Cardiol 88:199–206
Yildiz P, Tükek T, Akkaya V, Sözen AB, Yildiz A, Korkut F, Yilmaz V (2002) Ventricular arrhythmias in patients with COPD are associated with QT dispersion. Chest 122:2055–2061
Tükek T, Yildiz P, Akkaya V, Karan MA, Atilgan D, Yilmaz V, Korkut F (2002) Factors associated with the development of atrial fibrillation in COPD patients: the role of P-wave dispersion. Ann Noninvasive Electrocardiol 7(3):222–227
Hanrahan JP, Grogan DR, Baumgartner RA, Wilson A, Cheng H, Zimetbaum PJ, Morganroth J (2008) Arrhythmias in patients with chronic obstructive pulmonary disease (COPD): occurrence frequency and the effect of treatment with the inhaled long-acting beta2-agonists arformoterol and salmeterol. Medicine (Baltimore) 87:319–328
Weber UK, Osswald S, Huber M, Buser P, Skarvan K, Stulz P, Schmidhauser C, Pfisterer M (1998) Selective versus non-selective antiarrhythmic approach for prevention of atrial fibrillation after coronary surgery: is there a need for pre-operative risk stratification? A prospective placebo-controlled study using low-dose sotalol. Eur Heart J 19(5):794–800
Ozer N, Aytemir K, Atalar E, Sade E, Aksöyek S, Ovünç K, Açýl T, Nazlý N, Ozmen F, Oto A, Kes S (2000) P wave dispersion in hypertensive patients with paroxysmal atrial fibrillation. Pacing Clin Electrophysiol 23(11 Pt 2):1859–1862
Köse S, Aytemir K, Sade E, Can I, Ozer N, Amasyali B, Aksöyek S, Ovünç K, Ozmen F, Atalar E, Işik E, Kes S, Demirtaş E, Oto A (2003) Detection of patients with hypertrophic cardiomyopathy at risk for paroxysmal atrial fibrillation during sinus rhythm by P-wave dispersion. Clin Cardiol 26(9):431–434
Ho TF, Chia EL, Yip WC, Chan KY (2001) Analysis of P wave and P dispersion in children with secundum atrial septal defect. Ann Noninvasive Electrocardiol 6(4):305–309
Dagli N, Karaca I, Yavuzkir M, Balin M, Arslan N (2008) Are maximum P wave duration and P wave dispersion a marker of target organ damage in the hypertensive population? Clin Res Cardiol 97(2):98–104 Epub 2007 Oct 19
Sutherland GR, Stewart MJ, Groundstroem KW, Moran CM, Fleming A, Guell-Peris FJ, Riemersma RA, Fenn LN, Fox KA, McDicken WN (1994) Color Doppler myocardial imaging: a new technique for the assessment of myocardial function. J Am Soc Echocardiogr 7:441–458
McDicken WN, Sutherland GR, Moran CM, Gordon LN (1992) Color Doppler velocity imaging of the myocardium. Ultrasound Med Biol 18:651–654
Waggoner AD, Bierig SM (2001) Tissue Doppler imaging: a useful echocardiographic metod for the cardiac sonographer to assess systolic and diastolic ventricular function. J Am Soc Echocardiogr 14(12):1143–1152
Galiuto L, Ignone G, DeMaria AN (1998) Contraction and relaxation velocities of the normal left ventricle using pulsed-wave tissue Doppler echocardiography. Am J Cardiol 81(5):609–614
Weijs B, de Vos CB, Tieleman RG, Pisters R, Cheriex EC, Prins MH, Crijns HJ (2011) Clinical and echocardiographic correlates of intra-atrial conduction delay. Europace 13(12):1681–1687
Hummel YM, Klip IJ, de Jong RM, Pieper PG, van Veldhuisen DJ, Voors AA (2010) Diastolic function measurements and diagnostic consequences: a comparison of pulsed wave- and color-coded tissue Doppler imaging. Clin Res Cardiol 99(7):453–458
Ozer N, Yavuz B, Can I, Atalar E, Aksöyek S, Ovünç K, Ozmen F, Kes S (2005) Doppler tissue evaluation of intra-atrial and interatrial electromechanical delay and comparison with P-wave dispersion in patients with mitral stenosis. J Am Soc Echocardiogr 18:945–948
Omi W, Nagai H, Takamura M, Okura S, Okajima M, Furusho H, Maruyama M, Sakagami S, Takata S, Kaneko S (2005) Doppler tissue analysis of atrial electromechanical coupling in paroxysmal atrial fibrillation. J Am Soc Echocardiogr 18(1):39–44
Acar G, Sayarlioglu M, Akcay A, Sokmen A, Sokmen G, Altun B, Nacar AB, Gunduz M, Tuncer C (2009) Assessment of atrial electromechanical coupling characteristics in patients with ankylosing spondylitis. Echocardiography 26(5):549–557
Acar G, Akcay A, Sokmen A, Ozkaya M, Guler E, Sokmen G, Kaya H, Nacar AB, Tuncer C (2009) Assessment of atrial electromechanical delay, diastolic functions, and left atrial mechanical functions in patients with type 1 diabetes mellitus. J Am Soc Echocardiogr 22(6):732–738
Van Beeumen K, Duytschaever M, Tavernier R, Van de Veire N, De Sutter J (2007) Intra- and interatrial asynchrony in patients with heart failure. Am J Cardiol 99(1):79–83
De Vos CB, Weijs B, Crijns HJ, Cheriex EC, Palmans A, Habets J, Prins MH, Pisters R, Nieuwlaat R, Tieleman RG (2009) Atrial tissue Doppler imaging for prediction of new-onset atrial fibrillation. Heart 95(10):835–840
Akcay A, Acar G, Suner A, Sokmen A, Sokmen G, Nacar AB, Tuncer C (2009) Effects of slow coronary artery flow on P-wave dispersion and atrial electromechanical coupling. J Electrocardiol 42(4):328–333
Brashear RE (1984) Arrhythmias in patients with chronic obstructive pulmonary disease. Med Clin North Am 68(4):969–981 (Review)
Sarubbi B, Esposito V, Ducceschi V, Meoli I, Grella E, Santangelo L, Iacano A, Caputi M (1997) Effect of blood gas derangement on QTc dispersion in severe chronic obstructive pulmonary disease: evidence of an electropathy? Int J Cardiol 58(3):287–292
Incalzi RA, Pistelli R, Fuso L, Cocchi A, Bonetti MG, Giordano A (1990) Cardiac arrhythmias and left ventricular function in respiratory failure from chronic obstructive pulmonary disease. Chest 97(5):1092–1097
Vizza CD, Lynch JP, Ochoa LL, Richardson G, Trulock EP (1998) Right and left ventricular dysfunction in patients with severe pulmonary disease. Chest 113(3):576–583
Neuberger HR, Reil JC, Adam O, Laufs U, Mewis C, Böhm M (2008) Atrial fibrillation in heart failure: current treatment of patients with remodeled atria. Curr Heart Fail Rep 5(4):219–225 Review
Dilaveris PE, Gialafos EJ, Sideris SK, Theopistou AM, Andrikopoulos GK, Kyriakidis M, Gialafos JE, Toutouzas PK (1998) Simple electrocardiographic markers for the prediction of paroxysmal idiopathic atrial fibrillation. Am Heart J 135(5 Pt 1):733–738
Dilaveris PE, Gialafos EJ, Andrikopoulos GK, Richter DJ, Papanikolaou V, Poralis K, Gialafos JE (2000) Clinical and electrocardiographic predictors of recurrent atrial fibrillation. Pacing Clin Electrophysiol 23(3):352–358
Pala S, Tigen K, Karaahmet T, Dundar C, Kilicgedik A, Güler A, Cevik C, Kirma C, Basaran Y (2010) Assessment of atrial electromechanical delay by tissue Doppler echocardiography in patients with nonischemic dilated cardiomyopathy. J Electrocardiol 43(4):344–350
Biernacki W, Flenley DC, Muir AL, MacNee W (1988) Pulmonary hypertension and right ventricular function in patients with COPD. Chest 94(6):1169–1174
Salvaterra CG, Rubin LJ (1993) Investigation and management of pulmonary hypertension in COPD. Am Rev Respir Dis 148(5):1414–1417
Karabıyıkoğlu G (1993) KOAH’da pulmoner hemodinami. Tüberküloz Toraks 41:17–32
MacNee W (1994) State of the art: pathophysilogy of cor pulmonale in chronic obstructive pulmonary disease. Am J Respir Crit Care Med 150(3):part I 883–52, part II 1158-1168
Fuso L, Incalzi RA, Pistelli R, Muzzolon R, Valente S, Pagliari G, Gliozzi F, Ciappi G (1995) Predicting mortality of patients hospitalized for acutely exacerbated chronic obstructive pulmonary disease. Am J Med 98(3):272–277
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Caglar, I.M., Dasli, T., Turhan Caglar, F.N. et al. Evaluation of atrial conduction features with tissue doppler imaging in patients with chronic obstructive pulmonary disease. Clin Res Cardiol 101, 599–606 (2012). https://doi.org/10.1007/s00392-012-0431-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00392-012-0431-7