Abstract
Background
Amyloid light chain (AL) amyloidosis is a rare disease with poor prognosis and limited therapeutic alternatives. Recently, one clinical case with cardiac involvement, as well as a compelling evidence of green tea polyphenol, epigallocatechin-3-gallate (EGCG), inducing the formation of benign aggregation products that do not polymerize into fibrils were published. This is a report of the cardiac effects of green tea consumption in these patients.
Methods
Patients with known cardiac involvement in AL amyloidosis were examined by routine cardiovascular examinations that took place every 3–6 months. Of 59 patients with cardiac involvement, 11 revealed a decrease of at least 2 mm of interventricular wall thickness, after initiation of regular green tea consumption (GT). A matched historic control group (n = 22) was selected. Comprehensive echocardiography was conducted at every control examination and analyzed offline by two independent examiners.
Results
GT patients showed an improvement in New York Heart Association (NYHA) class from a median of 3 (25th, 75th percentiles: 2, 3) to 2 (2, 3), P = 0.038. Septal thickness decreased from 18 (18, 20) to 16 (16, 17) mm, P = 0.021. Left ventricular mass index decreased from 175 (154, 180) to 133 (128, 154) g/m2, P = 0.007. Comparing both groups, an increase in left ventricular ejection fraction could be found in the GT group, 65 (51, 73) versus 53 (47, 59)%, P = 0.012. These changes could not be observed in the control group.
Conclusion
Consumption of green tea polyphenol EGCG in patients with cardiac involvement with AL amyloidosis causes a significant decrease in left ventricular wall thickness and mass, as well as an improvement in NYHA functional classification and left ventricular ejection fraction.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Systemic immunoglobulin light-chain (AL) amyloidosis is a rare disease with very poor prognosis and limited therapeutic alternatives. Clinical outcome and even the likelihood of a response to therapy are in large part determined by the extent of cardiac involvement at diagnosis [1]. The most common cause of death is a progressive congestive cardiomyopathy or sudden death due to ventricular fibrillation [2]. Even after heart transplantation for severe, therapy refractory cardiac involvement, prognosis remains poor [3].
Green tea (Camellia sinensis) has long been ascribed as a health promoting substance with no known toxicities and ubiquitous properties. Recently, a retired member of our faculty, suffering from AL amyloidosis himself, alerted us on the positive effect on cardiac involvement of the main green tea polyphenol, epigallocatechin-3-gallate (EGCG) [4]. Experimental proof of the ability of EGCG as an agent with potent and specific antimyeloma activity [5], to induce apoptotic cell death in the leukemic B-cells [6, 7], or to reduce cerebral amyloidosis in Alzheimer transgenic mice through modulation of amyloid precursor protein cleavage were reported previously [8]. Experimental evidence showing EGCG acting as a positive intropic substance and preventing cardiac hypertrophy induced by pressure overload were published recently [9, 10]. Furthermore, in vitro experiments have shown that EGCG very efficiently prevents amyloidogenesis and modulates protein complex formation by promoting the formation of benign aggregation products that do not polymerize into fibrils [11, 12], whereas clinical observations support of these experimental findings [4, 13], more convincing evidence of clinical benefit, as well as possible side effects and optimal dosage is still lacking.
This is a report of a longitudinal observation of patients with cardiac amyloidosis who decided to consume daily large amounts of green tea, encouraged by a publication on a first successful self-treatment published recently [4].
Methods
Study population and design
This is a longitudinal observational study, with prospective offline analysis of digitally stored echocardiographic data. Consecutive patients with known cardiac involvement in AL amyloidosis were examined in the setting of routine cardiovascular assessments at our department. Follow-up examinations took place every 3–6 months, also in a usual manner. Patients that showed a decrease in at least 2 mm of interventricular wall thickness were asked on potential changes in lifestyle since the last examination, as well as their current functional capacity. Common to this cohort was a significant change in lifestyle, since they have started to drink large daily amounts of green tea (1.5–2 L/daily) after they got information based on the publication of a case report [4]. Two patients did not tolerate the taste of green tea, so they went on commercial available capsules with green tea extracts, taking a total daily doses of an equivalent of 600–800 mg EGCG. Retrospective anamnesis on green tea consumption was conducted in all other consecutive patients with cardiac involvement in AL amyloidosis examined during the same period of time. It was not possible to perform assessment of plasma concentrations of EGCG in these patients.
All patients signed a statement previously approved by the internal institutional review board, in which patients agreed to the use of their medical information in an anonymized form for research purposes.
Historic control group
This cohort was built based on a databank of comprehensive echocardiography examinations and reports that took place before September 2007, of patients with confirmed AL amyloidosis under conventional therapy at the Department of Hematology from our institution. This time period was well ahead of the first publication on a possible beneficial effect of green tea on cardiac amyloidosis [4]. There was no evidence of consumption of significant amounts of green tea in this cohort. Patients were matched to age, gender, body mass index (BMI), New York Heart Association (NYHA) functional classification, interventricular wall thickness and indexed left ventricular mass.
Echocardiography
Comprehensive echocardiography examination at rest was conducted according to current recommendations of the American Society of Echocardiography [14] with conventional equipment (Vivid 7 Dimension, GE Medical systems, Horten, Norway) by experienced cardiac sonographers, which were blinded to patient’s symptoms. Diastolic left ventricular function (E/E′ and LVEFP) was assessed according to current guidelines [15]. Left ventricular long axis function was assessed by mitral annular systolic plane excursion (MAPSE) and the mitral annular systolic velocity (MASV). Pulmonary artery systolic pressure (PASP) was estimated from peak tricuspid regurgitation jet velocities according to the modified Bernoulli equation: PASP = 4 × V 2 + right atrial pressure, where V is the peak velocity (m/s) of tricuspid valve regurgitant jet. In the absence of obstruction to right ventricular outflow, right ventricular systolic pressure equals PASP [16]. Right atrial pressure was estimated from characteristics of the inferior vena cava [17]. Offline analysis of echocardiography examinations of both cohorts were conducted on a commercial available workstation (Centricity Cardiology CA1000 2.0, GE Medical Systems, Milwaukee, WI, USA). Three cardiac cycles were stored in cine loop format for offline analysis. All data were analyzed by two independent expert examiners who were unaware of the patients’ clinical status.
Cardiac biomarkers
Serum levels of N-terminal prohormone brain natriuretic peptide (NT-proBNP) and cardiac troponin T (cTnT) were measured routinely in all patients at first assessment and then at every control examination.
Statistical methods
Continuous variables are expressed as median (25th, 75th percentiles). Normality was assessed with the Kolmogorov–Smirnov test. Comparisons between groups were made using Student’s t test or Mann–Whitney U test as appropriate. Categorical variables were compared using the χ2 test. P values <0.05 were considered statistically significant.
Results
Study population
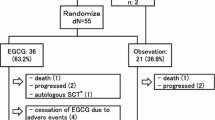
Of 59 consecutive patients with new diagnosed cardiac involvement in AL amyloidosis, 11 selected patients, median age 52 (51, 65) years, 3 females, with known cardiac involvement in AL amyloidosis showed a decrease in at least 2 mm of interventricular septum thickness in a period of 6 (3, 10) months between digitally stored comprehensive echocardiographic examinations (Tables 1, 2, GT group). Two patients received high doses intravenous melphalan followed by autologous stem cell transplantation, eight patients were treated with oral melphalan and dexamethasone and one patient was in an evaluation period, receiving fortuitously two echocardiographic examinations within a 1 month interval and had no specific therapy (Table 1). Retrospective anamnesis on green tea consumption in the remaining 48 patients was negative. Patients in the GT group showed an improvement in the functional classification at the follow-up examination compared to the first assessment, with a median decrease of 3 (2, 3) to 2 (2, 3), P = 0.038 (Table 3).
In the historic control (HC, n = 22) group with cardiac involvement in AL amyloidosis, eight patients showed no change in septal wall thickness, in nine patients an increase of 1 mm and in one patient even of 2 mm in septal wall thickness was observed. A decrease in 1 mm thickness was also observed in four patients. All these changes were not significant to initial measurements or compared to similar changes in the GT group (Table 3). Nine patients received high doses intravenous melphalan followed by autologous stem cell transplantation, eight patients were treated with oral melphalan and dexamethasone and five patients with high doses melphalan alone. No difference in functional classification could be observed within the HC group at start compared to follow-up examination (Table 3).
None of the patients had a history of systemic arterial hypertension. Body mass index (BMI), a predictor for increased left ventricular mass [18], with a median of 23.9 (21.5, 24.4) kg/m2 in the GT group and 23.4 (22.5, 26.5, P = ns) kg/m2 in the HC group showed no significant obesity any patient of both groups.
The time to echocardiographic end-point in the GT group, 6 (3, 10) months and time between two examinations under therapy in the HC group, 6 (4, 8) months were similar (Table 3, TEP, P = ns).
Echocardiography
All patients met echocardiography criteria for cardiac involvement in histologically confirmed and immunohistochemically typified AL amyloidosis, with left ventricular wall thickness >12 mm with otherwise no other cardiac cause [1]. Other common characteristics for cardiac amyloidosis were also present: diffuse hyperrefractile “granular sparkling” [19], impaired left ventricular diastolic [20–22] and long axis and function [23].
None of the patients had a significant valvular stenosis, and none showed more than mild valvular regurgitation.
Wall thickness and left ventricular mass
Interventricular septum (IVS) thickness decreased significantly in the GT group compared to first examination, 18 (18, 20) to 16 (16, 17) mm, P = 0.021 (Table 3, Fig. 1). Posterior wall thickness decreased also in the same manner, from 17 (16, 18) to 15 (14, 16) mm, P = 0.047. Left ventricular mass, indexed to the body surface area (LVMI) decreased correspondingly from 175 (154, 180) to 133 (128, 154) g/m2, P = 0.006 (Table 3; Fig. 2).
Dimensions of the interventricular septum measured at examination before start of green tea consumption (a) and at the echocardiographic end-point (b) in the green tea patient group (GT, n = 11, left). Same measurements in the historic control group (HC, n = 22, right) conducted at first diagnosis (a) and at the next echocardiographic control after 3–6 months under conventional therapy. GT versus HC, *P = 0.013
Left ventricular mass indexed to body surface area at examination before start of green tea consumption (a) and at the echocardiographic end-point (b) in the green tea patient group (GT, n = 11, left). Same measurements in the historic control group (HC, n = 22, right) conducted at first diagnosis (a) and at the next echocardiographic control after 3–6 months under conventional therapy. GT versus HC, *P = 0.006
Changes in the HC group were not statistically significant. Median values of IVS thickness were even increased compared to the first examination, from 17 (17, 18) to 18 (17, 19) mm, P = ns (Table 3). Similar findings could be observed at the calculation of the LVMI.
Left ventricular wall thickness and mass decreased also significantly at follow-up examination in the GT group compared to the HC group (Table 3; Figs. 1, 2).
Left atrium
Left atrial (LA) volume indexed to body surface area showed a decreasing trend at the follow-up examination in the GT group, 35 (20, 24) to 34 (19, 37) mL/m2. However, when comparing second examinations of both groups, a significant decrease in LA volume could be documented in the GT group, 34 (19, 37) versus HC group, 43 (29, 48) mL/m2, P = 0.042 (Table 3).
Left ventricular function and PASP
When comparing second examinations of both groups, a significant increase in left ventricular ejection fraction (LVEF) could be found in the GT group, 65 (51, 73) versus HC group, 53 (47, 59)%, P = 0.012 (Table 3). This change went correspondingly to the decrease in left ventricular end-systolic diameter in the GT cohort. Furthermore, no changes in LVEF within the HC group could be assessed, and an increase in LVEF observed within the GT group, 53 (49, 64) versus 65 (51, 73)% was not statistically significant.
Mitral annular systolic plane excursion and systolic velocities were similar in both groups at the first examination, as well as at the follow-up examination (Table 3). Values were compatible with a severe impairment of left ventricular long axis function. No significant changes could be determined in each group, nor at any examination time. Similar findings could be observed concerning diastolic function, as assessed with the parameters E/E′ and left ventricular end-diastolic pressure [15]. A high prevalence of severe diastolic dysfunction, as suggested in the former guideline (E/E′ > 15), could be documented in both groups (GT 45.5 vs. HC 45.3%, P = ns), but no significant change could be observed in the follow-up examination.
Pulmonary artery systolic pressure (PASP) at rest at first examination was elevated, i.e., >30 mmHg, up to almost half of the patients (GT 45.5 vs. HC 22.7%, P = ns). A decreasing trend in PASP at rest could be seen at the follow-up examination in the GT group. Moreover, when comparing second examinations of both groups, a significant decrease in PASP could be found in the GT group, 30 (23, 35) versus HC group, 35 (30, 41)%, P = 0.041 (Table 3). At follow-up examination, prevalence of patients with pulmonary hypertension at rest in the GP group was unchanged. However, a significant increase could be observed in the HC group (22.7–54.5%, P = 0.030).
Cardiac biomarkers
NT-proBNP, a sensitive marker for myocardial dysfunction was massively elevated in both groups at first assessment, with no significant change at the follow-up examination. Serum cardiac troponin T (cTnT), a marker for myocardial damage was mildly elevated and only slightly higher in the GT group, showing a trend in lower values at the follow-up examination (Table 3).
Discussion
This is the first longitudinal observation in a group of patients with AL amyloidosis receiving current therapy options, who in addition started a daily EGCG consumption from green tea after available information of a first case in the literature [4]. A significant decrease in left ventricular wall thickness and mass, as well as an improvement in NYHA functional classification and left ventricular ejection fraction could be assessed in the green tea drinking group, confirming the former original observation.
Concentric wall thickening due to infiltration of the heart by extracellular deposition from insoluble protein deposits, i.e., β-amyloid fibrils, with subsequent myocardial disarray and necrosis, posterior local and/or diffuse fibrosis, constitute the complex leading to architectural changes and functional impairment. Thus, treatment targets are both, reduce the production, as well as disaggregate “seeds” of β-pleaded amyloid fibrils.
Reduction in left ventricular wall thickness under chemotherapy is reported in only one former study, where in 3 of 20 of patients with cardiac response a reduction in septal or mean left ventricular wall thickness could be found [24]. In our observation, all patients showed a wall thickness reduction of at least 2 mm, despite a more severe phenotype than the historical group. In this group, a higher proportion of patients were eligible for autologous stem cell transplantation, since a better clinical stratification for reduction treatment-related mortality was aimed [25]. Furthermore, 4 patients in the GT group came to the next routine follow-up examination and showed further reduction in LV wall thickness with a median of 5 (5, 7) mm. The patient of the first case published [4, 13], not included in this report, has a constant interventricular septal thickness of 12 mm since the last 22 months, showing a total reduction of almost 5 mm, under daily therapy with EGCG. Last administered chemotherapy was 36 months ago and no further cardiac/hematological medication was administered since that time. These clinical findings are supported by structural and molecular biology results on the effects of EGCG on amyloid formation [11, 12].
The improvement in wall thickness in patients of the GT group was accompanied with functional changes, such as an increase in left ventricular ejection fraction. This may be due to one or more of the following factors: a decrease in β-amyloid infiltration of the heart caused by EGCG [11], an EGCG calcium-dependent positive inotropic effect [9], and/or a modulation of contraction through a binding of EGCG to a cardiac myofilament, cTnC [26].
Severe impairment of diastolic function (restrictive pattern) did not change significantly during follow-up in the GT group, similar findings in patients with advanced cardiac involvement and severe left ventricular diastolic dysfunction at the first examination were reported before [22]. Restrictive filling pattern in cardiac amyloidosis is possibly due to amyloid infiltration itself [27], but also alterations in cardiomyocyte metabolism, cellular edema, intracellular calcium handling, myocardial necrosis due to excessive extracellular mechanically induced architectural disarray and remodeling of the cardiac matrix and fibrosis. Although it has been shown that EGCG inhibits proliferation of cardiac fibroblasts in vivo and in vitro, thus preventing myocardial fibrosis in cardiac hypertrophy [28], this action may not play a significant role in well established cardiac amyloidosis.
Pulmonary artery systolic pressure (PASP) estimated through tricuspid regurgitation velocity was not severely elevated in all patients, despite severe impairment of diastolic left ventricular function. PASP can show normal values at rest, but an excessive hypertensive response to exercise even in individuals without dyspnea on exertion [29]. However, an assessment of PASP under exercise was not part of the routine examinations in these patients. The trend in decrease of PASP at rest in the GT group as assessed in follow-up examinations, as well as a significant decrease compared to the HC group, are in agreement with other signs of improvement in left ventricular hemodynamics.
Although cardiac biomarkers were markedly elevated (NT-proBNP), multiple mechanisms may lead to their elevation in blood, e.g., chronic renal failure. A trend in decrease of values of cardiac troponin T in the GT group suggests a reduction in myocardial damage in these patients.
Limitations
Since serum EGCG levels were not assessed in these patients, it was not possible to determine the percentage of “non responders” in this cohort, a very important fact, since the effect of EGCG from green tea occurs a dose-dependent way [11] and the bioavailability of EGCG is very limited [30, 31].
Conclusions
In this longitudinal observation, new-onset daily green tea polyphenol EGCG consumption in patients with AL amyloidosis and cardiac involvement, under current standard therapy options, causes a significant decrease in left ventricular wall thickness and mass, as well as an improvement in NYHA functional classification and left ventricular ejection fraction. Prospective clinical studies are needed to assess effect, define doses and possible side effects of EGCG in the therapy of AL amyloidosis.
References
Gertz MA, Comenzo R, Falk RH et al (2005) Definition of organ involvement and treatment response in immunoglobulin light chain amyloidosis (AL): a consensus opinion from the 10th International Symposium on Amyloid and Amyloidosis, Tours, France, 18–22 April 2004. Am J Hematol 79:319–328
Gertz MA, Lacy MG, Dispenzieri A (2004) Therapy for immunoglobulin light chain amyloidosis: the new and the old. Blood Rev 18:17–37
Roig E, Almenar L, González-Vílchez F et al (2009) Outcomes of heart transplantation for cardiac amyloidosis: subanalysis of the Spanish registry for heart transplantation. Am J Transplant 9:1414–1419
Hunstein W (2007) Epigallocatechin-3-gallate in AL amyloidosis: a new therapeutic option? Blood 110:2216
Shammas MA, Neri P, Koley H et al (2006) Specific killing of multiple myeloma cells by (−)-epigallocatechin-3-gallate extracted from green tea: biologic activity and therapeutic implications. Blood 108:2804–2810
Shanafelt TD, Lee YK, Call TG et al (2006) Clinical effects of oral green tea extracts in four patients with low grade B-cell malignancies. Leuk Res 30:707–712
Shanafelt TD, Call TG, Zent CS et al (2009) Phase I trial of daily oral polyphenon E in patients with asymptomatic Rai Stage 0 to II chronic lymphocytic leukemia. J Clin Oncol 27:3808–3814
Rezai-Zadeh K, Shytle D, Sun N et al (2005) Green tea epigallocatechin-3-gallate (EGCG) modulates amyloid precursor protein cleavage and reduces cerebral amyloidosis in Alzheimer transgenic mice. J Neurosci 25:8807–8814
Lorenz M, Hellige N, Rieder P et al (2008) Positive inotropic effects of epigallocatechin-3-gallate (EGCG) involve activation of Na+/H+ and Na+/Ca2 + exchangers. Eur J Heart Fail 10:439–445
Hao J, Kim CH, Ha TS, Ahn HY (2007) Epigallocatechin-3 gallate prevents cardiac hypertrophy induced by pressure overload in rats. J Vet Sci 8:121–129
Ehrnhoefer DE, Bieschke J, Boeddrich A et al (2008) EGCG redirects amyloidogenic polypeptides into unstructured, off-pathway oligomers. Nat Struct Mol Biol 15:558–566
Hauber I, Hohenberg H, Holstermann B et al (2009) The main green tea polyphenol epigallocatechin-3-gallate counteracts semen-mediated enhancement of HIV infection. Proc Natl Acad Sci USA 106:9033–9038
Mereles D, Wanker EE, Katus HA (2008) Therapy effects of green tea in a patient with systemic light-chain amyloidosis. Clin Res Cardiol 97:341–344
Lang RM, Bierig M, Devereux RB et al (2005) Recommendations for chamber quantification: a report from the American Society of Echocardiography’s Guidelines and Standards Committee and the Chamber Quantification Writing Group, developed in conjunction with the European Association of Echocardiography, a branch of the European Society of Cardiology. J Am Soc Echocardiogr 18:1440–1463
Nagueh SF, Appleton CP, Gillebert TC et al (2009) Recommendations for the evaluation of left ventricular diastolic function by echocardiography. J Am Soc Echocardiogr 22:107–133
Yock PG, Popp RL (1984) Noninvasive estimation of right ventricular systolic pressure by Doppler ultrasound in patients with tricuspid regurgitation. Circulation 70:657–662
Ommen SR, Nishimura RA, Hurrel DG et al (2000) Assessment of right atrial pressure with 2-dimensional and Doppler-echocardiography: a simultaneous catheterization and echocardiography study. Mayo Clin Proc 75:24–29
Levy D, Anderson KM, Savage DD et al (1988) Echocardiographically detected left ventricular hypertrophy: prevalence and risk factors. The Framingham Heart Study. Ann Intern Med 108:7–13
Siqueira-Filho AG, Cunha CL, Tajik AJ et al (1981) M-mode and two-dimensional echocardiographic features in cardiac amyloidosis. Circulation 63:188–196
Hongo M, Ikeda S (1986) Echocardiographic assessment of the evolution of amyloid heart disease: a study with familial amyloid polyneuropathy. Circulation 73:249–256
Klein AL, Hatle LK, Burstow DJ et al (1990) Comprehensive Doppler assessment of right ventricular diastolic function in cardiac amyloidosis. J Am Coll Cardiol 15:99–108
Klein AL, Hatle LK, Taliercio CP et al (1990) Serial Doppler echocardiographic follow-up of left ventricular diastolic function in cardiac amyloidosis. J Am Coll Cardiol 16:1135–1141
Bellavia D, Pellikka PA, Abraham TP et al (2008) Evidence of impaired left ventricular systolic function by Doppler myocardial imaging in patients with systemic amyloidosis and no evidence of cardiac involvement by standard two-dimensional and Doppler echocardiography. Am J Cardiol 101:1039–1045
Palladini G, Lavatelli F, Russo P et al (2006) Circulating amyloidogenic free light chains and serum N-terminal natriuretic peptide type B decrease simultaneously in association with improvement of survival in AL. Blood 107:3854–3858
Jantunen E, Itälä M, Lehtinen T et al (2006) Early treatment-related mortality in adult autologous stem cell transplant recipients: a nation-wide survey of 1,482 transplanted patients. Eur J Haematol 76:245–250
Robertson IM, Li MX, Sykes BD (2009) The solution structure of human cardiac troponin C in complex with the green tea polyphenol; (−)-epigallocatechin-3-gallate. J Biol Chem 284:23012–23023
Liao R, Jain M, Teller P et al (2001) Infusion of light chains from patients with cardiac amyloidosis causes diastolic dysfunction in isolated mouse hearts. Circulation 104:1594–1597
Sheng R, Gu ZL, Xie ML et al (2009) EGCG inhibits proliferation of cardiac fibroblasts in rats with cardiac hypertrophy. Planta Med 75:113–120
Grünig E, Janssen B, Mereles D et al (2000) Abnormal pulmonary artery pressure response in asymptomatic carriers of primary pulmonary hypertension gene. Circulation 102:1145–1150
Henning SM, Niu Y, Liu Y et al (2005) Bioavailability and antioxidant effect of epigallocatechin gallate administered in purified form versus as green tea extract in healthy individuals. J Nutr Biochem 16:610–616
Chow HH, Hakim IA, Vining DR et al (2005) Effects of dosing condition on the oral bioavailability of green tea catechins after single-dose administration of Polyphenon E in healthy individuals. Clin Cancer Res 11:4627–4633
Author information
Authors and Affiliations
Corresponding author
Additional information
D. Mereles and S. J. Buss contributed equally to this work.
Rights and permissions
About this article
Cite this article
Mereles, D., Buss, S.J., Hardt, S.E. et al. Effects of the main green tea polyphenol epigallocatechin-3-gallate on cardiac involvement in patients with AL amyloidosis. Clin Res Cardiol 99, 483–490 (2010). https://doi.org/10.1007/s00392-010-0142-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00392-010-0142-x