Abstract
Objectives
T1 mapping by cardiac magnetic resonance imaging (CMR) is able to determine the extracellular volume fraction. Wild-type transthyretin amyloidosis (WT-ATTR) is characterized by extracellular amyloid deposition in the heart. Recent reports indicated a reduction of left ventricular (LV) myocardial mass in WT-ATTR after consumption of epigallocatechin-3-gallate, the main catechin in green tea. It remained unclear, whether reduction of LV myocardial mass reflects decrease of amyloid load or progressive atrophy of cardiomyocytes.
Methods
This study included 7 male patients with CMR repetitively performed before and 12 months after daily consumption of green tea extract (600 mg epigallocatechin-3-gallate). Short axis slices as well as 2-, 3-, and 4-chamber views were acquired using SSFP sequences. T1 mapping was created out of 11 mid-ventricular short axis views with increasing inversion times using a single breath-hold modified look-locker inversion recovery sequence before and 15 min after Gadolinium contrast administration.
Results
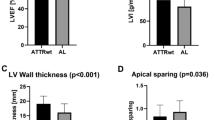
After 12 months, a significant decrease of LV myocardial mass [198 (160; 212) vs. 180 (142; 204) g; p < 0.05] was observed. Moreover, a significant decrease of native [T1 1110 (1072; 1150) ms vs. 1080 (970; 1101), p < 0.05 or p = 0.03] was noticed. The calculated extracellular volume decreased in 5 patients (62.5 %) by 7 % and increased in 2 patients (37.5 %) by 9.5 %, in trend resulting in a (not significant) decrease of median ECV by 2.4 %. Left ventricular ejection fraction (LVEF) [57 (48; 65) vs. 55 (47; 64) %; p = 0.3] remained unchanged.
Conclusions
This study provided further evidence of LV myocardial mass reduction in patients with WT-ATTR daily consuming green tea extract. Additionally, this study gave first insights into the histomorphological correlate of LV mass reduction using T1 mapping. LV mass reduction appeared to be rather due to a decrease of amyloid load than atrophy of cardiomyocytes.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Wild-type transthyretin amyloidosis (WT-ATTR) is an age-related type of amyloid disease characterized by extracellular deposition of wild-type transthyretin amyloid almost exclusively in the heart. Thus, patients primarily present with symptoms of heart failure [1, 2]. Up to now causative treatment options are lacking. Standard heart failure medication is not able to halt progression of the disease [3]. Tafamidis (Vyndaqel®), a stabilizer of the TTR tetramer [4], has been approved for treatment of stage 1 familial amyloid polyneuropathy in Germany in 2011 [5, 6]. However, the effect of tafamidis on TTR amyloid cardiomyopathy has not been elucidated yet.
In the past, in vitro experiments gave evidence that epigallocatechin-3-gallate (EGCG), the most abundant catechin in green tea, appeared to inhibit fibril formation of diverse amyloidogenic proteins [7–9]. This therapeutic effect was further confirmed in an animal study [10]. Moreover, a recent observational study from our group in patients with TTR-related cardiac amyloidosis demonstrated a reduction of left ventricular (LV) myocardial mass by approximately 10 % after daily consumption of 450 mg EGCG over a 1-year period [11]. However, the morphological correlate of LV myocardial mass reduction remained unknown since mass reduction could either be related to atrophy of cardiomyocytes or for reduction of amyloid load.
With respect to this, CMR tissue characterization offers a unique perspective for further clarification of the histological correlate resulting in decrease of LV myocardial mass: the emerging technique of T1 mapping allows a precise analysis of the myocardial interstitial space [12], including quantification of the extracellular volume (ECV) [13, 14] that expands due to edema, fibrosis or even amyloid deposition.
In this study, we sought to investigate the morphological correlate of LV myocardial mass reduction observed in a small group of WT-ATTR patients after consumption of EGCG by the use of the CMR T1 mapping technique. None of the study patients has been included in our previous study (11).
Materials and methods
Study subjects
The initial study cohort consists of 9 male WT-ATTR patients [72(70; 73) years, Table 1] with histologically proven TTR amyloid by endomyocardial biopsy (Congo-red staining, immunohistochemistry) and exclusion of any TTR gene variant by moleculargenetical testing [11]. Patients were recruited between 2011 and 2012 at the Heidelberg Amyloidosis center. All patients had compensated heart failure for at least 4 months prior to study inclusion. Patients were observed over a period of 12 months while daily consuming four capsules of green tea extract (praevent® Loges, Dr. Loges & Co, Winsen/Luhe, Germany; 600 mg EGCG, extraction solvent ethyl acetate/ethanol/water). Detailed methodological aspects have been described previously [11]. The study was approved by the local ethics committee and has been performed in accordance with the ethical standards according to the declaration of Helsinki.
Laboratory testing
Blood samples were taken on the day of CMR examination for the determination of haematocrit (HCT), serum creatinine, serum albumin, troponin T and NT-proBNP. For NT-proBNP and troponin T determination electrochemiluminescence immunoassays (Elecsys-proBNP/troponin T, Roche Diagnostics, Mannheim, Germany) were used.
Echocardiography
Transthoracic echocardiograms were performed using commercially available ultrasound diagnostic systems (Vivid 7, GE Healthcare, Milwaukee, WI, USA) and examinations were analyzed offline on a commercially available workstation (Centricity Cardiology CA1000 2.0, GE Medical Systems, Milwaukee, WI, USA).
The LV cavity diameter was scaled in end diastole and end systole, thickness of interventricular septum (IVS) and posterior wall were measured in late diastole. M-mode imaging from the apical 4-chamber view was used to obtain mitral (MAPSE) and tricuspid annular plane systolic excursion (TAPSE). For measurements of the systolic velocity, pulsed-wave Doppler tissue imaging of the mitral annulus was performed.
CMR protocol
CMR assessment was performed before consumption of green tea extract and at the end of a 1-year period with daily intake of green tea extract. All CMR exams were performed on a 1.5-Tesla scanner (Philips Achieva, Best, The Netherlands) with a 32-element cardiac receiver coil as reported previously [11]. Vector ECG-gated standard steady state free precession (SSFP) cine sequences were acquired (a) in short axis planes covering the whole LV and (b) in three long axis views (2-, 3-, 4-chamber views).
For calculation of T1 mapping, 11 raw images with increasing inversion times (TI 100–4400 ms) were acquired before and 15 min after injection of 0.2 mmol per kg body weight gadolinium-diethylene triamine pentaacetic acid (DTPA) contrast agent (Magnevist®, Bayer Healthcare, Leverkusen, Germany) using a modified look-locker inversion recovery sequence (MOLLI) during breath-hold in expiration (TR/TE: 3.5/1.8 ms, flip angle 35°).
CMR data analysis
All CMR data analyses were performed independently by two experienced investigators (HS, FS) blinded to all clinical data. A commercially available workstation (Philips Viewforum, Version 3.4, Best, The Netherlands) was used. Mean values were reported in the manuscript. Analyses included assessment of end-systolic and end-diastolic volumes and LV ejection fraction (LVEF). LV myocardial mass was measured in short axis image stacks of late diastole by manual delineation of endocardial and epicardial borders excluding papillary muscles. Mitral annular plane systolic excursion (MAPSE) and tricuspid annular plane systolic excursion (TAPSE) were obtained in 4-chamber views by measuring the distance traveled by the lateral annulus from end diastole to end systole.
Pre- and post-contrast T1 mappings with motion correction in x- and y-axis were generated using the open-source software tool “MRmap”. All maps were analyzed with OsiriX (v5.5.2 32 bit, PixmeoSarl). Myocardial T1 values were determined by tracing endocardial and epicardial borders manually and T1 values for blood were gathered by manually drawing a region of interest (ROI) in the LV cavity.
ECV values were created according to the following formula:
Statistical analysis
Analyses were performed using SPSS (Version 20.0, IBM Corp.). Statistical significance was defined for p values <0.05. Differences between the non-parametric, continuous parameters assessed at study inclusion and at 12-month follow-up were compared by the Wilcoxon matched-pairs signed rank test. Continuous data were expressed as median (interquartile range). Categorical variables were expressed as absolute numbers (percent).
Results
Complete CMR datasets were obtained from seven study patients; one patient did not receive Gadolinium contrast agent due to markedly reduced renal function (MDRD < 30 ml/min × 1.73 m2); another dataset was excluded due to poor image quality. Three patients had a history of mild arterial hypertension, but normotensive blood pressure due to medical treatment. One patient had a moderate aortic stenosis. None of the patients received pacemaker or ICD prior to study inclusion or during follow-up. Clinical characteristics are given in detail in Table 1. Mean time from first diagnosis of amyloid disease to assessment by CMR was 16 ± 27 months.
No changes in NYHA class and renal function calculated by modified diet in renal disease [MDRD; 69 (64; 75) vs. 73 (61; 77) ml/min × 1.73 m2; p = 0.9] were observed after 12 months. Median NT-proBNP plasma levels [2313 (763; 5864) vs. 3920 (2079; 6436) ng/ml; p = 0.11] and NYHA functional class remained unchanged and no hospitalization for heart failure was observed during the study period.
Detailed data for all the seven study patients at the beginning and the end of the observation period are given in Table 2 (Supplemental material).
Morphological and functional CMR analysis
After 12 months, a significant decrease of median LV myocardial mass by 6 % was observed in these patients [198 (160; 212) vs. 180 (142; 204) g; p = 0.03; Fig. 1]. Overall, in 5 (71 %) patients LV myocardial mass decreased during 1 year of green tea consumption, in 1 (14 %) patient LV myocardial mass remained unchanged. Only 1 (14 %) patient presented as a non-responder with increase of LV myocardial mass. LV ejection fraction [57 (48; 65) vs. 55 (47; 64) %; p = 0.3], right ventricular longitudinal function [TAPSE: 15.0 (8.8; 16) vs. 12.5 (10.3; 15.8) mm; p = 0.7] and LV longitudinal function [MAPSE: 6.5 (5.0; 8.8) vs. 5.5 (3.3; 6.0) mm; p = 0.06] remained unchanged during the observation period.
Tissue characterization by T1 mapping
Native T1 time decreased in 6 (86 %) patients after 12 months of green tea consumption resulting in a median decrease of 4 % [1110 (1072; 1150) vs. 1080 (971; 1101), p < 0.05; Fig. 2]. Moreover, post-contrast T1 time remained unchanged [347 (324; 396) vs. 356 (294; 394) ms; p = 0.8]. The calculated ECV decreased in 5 patients (62.5 %) by 7 % and increased in 2 patients (37.5 %) by 9.5 %. One of those patients with increase of ECV (+17 %) presented with a decrease of LV myocardial mass (−16 %), while in the other one an increase of ECV by 2 % was accompanied by an increase of LV myocardial mass by 6 %. In total, a trend for lower median ECV was observed (42 vs. 41 %) after consumption of green tea for 12 months, but statistical significance was not reached in this small patient population (p = 0.47).
The inter-observer mean differences in T1 determination were 9.3 ms in native, 5.6 ms in post-contrast scans and 0.5 % in ECV calculation. Mean intra-observer differences were 6.5 ms for native, 4.7 ms for post-contrast T1 values and 0.3 % for ECV values (Figs. 3, 4).
Discussion
This is the first study using repetitive CMR scans with T1 mapping in a small cohort of patients with WT-ATTR. This study provided first insights into the potential role of serial CMR assessments and the histomorphological correlate of LV myocardial mass reduction that has recently been reported after daily consumption of green tea for 1 year [11]. According to the present data, it appears that a decrease of LV myocardial mass might rather be caused by reduction of amyloid load than hypotrophy or atrophy of cardiomyocytes as a decrease of ECV was observed in the vast majority of patients. A trend for lower median ECV after green tea consumption was observed, however, no statistical significance was reached in this small study cohort of seven patients.
In general, hereditary ATTR amyloidosis is claimed to be a rare disease with an incidence of 0.1–1 patients per 1,000,000 inhabitants. Pre-mortem diagnosis of WT-ATTR amyloidosis is infrequent, but appears to be an underestimated cause of left ventricular hypertrophy. It is assumed that approximately in 0.5–1 % of patients above 60 years of age ATTR amyloid deposition will be present [15]. Although survival of patients with WT-ATTR is much better than survival of patients with light-chain amyloidosis [16, 17], the clinical course of the disease is (slowly) progressive. Recently, an 8 % increase of LV myocardial mass has been described within 12 months [18]. Additionally, TRACS data indicated a decrease of LV ejection fraction by 3.2 % for every 6 months increment [19]. Moreover, causative treatment approaches are lacking in WT-ATTR as common heart failure treatment is neither able to stop amyloid deposition nor to disaggregate amyloid fibrils.
Promising in vitro results [8, 9] and the letter by Hunstein [20] prompted us to evaluate the effect of EGCG on TTR amyloid. Later on a reduction of LV wall thickness was observed retrospectively in a larger cohort of patients with light-chain amyloidosis [21]. Recently, we reported on a decrease of LV myocardial mass in ATTR patients after consumption of green tea for 12 months [11].
Our present study, although obtained in a very small cohort, is well in line with previous reports. It supports the potential effects of green tea on the clinical course of WT-ATTR as again a decrease of LV myocardial mass was observed in this cohort with WT-ATTR. Only one patient presented as a non-responder regarding reduction of LV myocardial mass. A potential explanation might be an irregular or incorrect intake of GT capsules or a very advanced amyloid disease. Additionally, excessively high levels of NT-proBNP and ECV were observed in this patient.
Focusing on the potential histological correlates of LV myocardial mass reduction, T1 mapping by CMR offers a unique diagnostic tool for a more specific analysis of myocardial tissue by analyzing signals from intra- and extracellular space separately. Thus, the technique might help to differentiate, whether the changes in LV myocardial mass are caused by progressive atrophy of cardiomyocytes or by decrease of extracellular amyloid load. While the native T1 time represents a combination of cellular and interstitial information [22], the relative changes of T1 time of the myocardium and of the blood pool after administration of Gadolinium indicate the extracellular volume fraction as a percentage of the whole myocardium [23–25]. The usefulness of CMR extracellular volume quantification has recently been demonstrated for early diagnosis [22, 26, 27] and prognosis [28] in different forms of cardiac amyloidosis. However, its potential role for monitoring of treatment effects in cardiac amyloidosis is lacking. In contrast to the well-established late gadolinium enhancement that can solely be analyzed semi-quantitatively, T1 mapping allows quantitative analysis of the myocardial tissue composition.
According to the preliminary results of the present study, reduction of LV myocardial mass observed in WT-ATTR patients after 12 months of green tea consumption appears to be associated with a decrease of cardiac amyloid burden. Tissue characterization by T1 mapping might be a promising tool and even more sensitive biomarker for monitoring of pharmaceutical treatment effects as compared with functional parameters of MAPSE, TAPSE or even LV ejection fraction, respectively. Almost all of the patients with LV myocardial mass reduction after 12 months of green tea intake presented with decrease of ECV. As the changes in general are small in present patients the results are prone to errors. This might potentially explain the mismatch between reduction of LV myocardial mass and increase of ECV as observed in a single patient of the present study. The potential reasons for this mismatch are manifold. First, image quality was suboptimal. Moreover, the patient’s comorbidities including moderate aortic stenosis and arterial hypertension are associated with myocardial fibrosis that impacts native and post-contrast T1 times and finally results in increase of ECV [29, 30]. Additionally, body composition and renal function have been reported to impact post-contrast T1 time [12]. However, this appears to be highly unlikely as only a minimal change of body weight (1 %) and glomerular filtration rate (7 %) was observed in this particular patient. Post-contrast T1 time was notably low (240 ms) in comparison to the remaining patients. Thus, T1 mapping and ECV calculation might be of limited value in this case.
Native T1 time is independent of these factors and thus might be more reliable in some conditions. In contrast, native T1 reflects composition of the extra- and intracellular space and thus might not be a sufficient and specific biomarker indicating amyloid burden exclusively localized extracellularly. Future (in an optimal way placebo controlled) studies need to focus on long-term observation and the correlation of histopathological findings (e.g. amount of amyloid) and T1 mapping parameters (e.g. native T1, ECV).
Limitations
We are fully aware of the limitations of the present study. However, this is the first report on the potential role of repetitive T1 mapping by cardiac MRI for assessment of response to treatment. First, this study is limited by the small number of patients and by its lack of a control group. WT-ATTR is an orphan disease with a limited number of diagnosed patients available for clinical trials. Additionally, there is a widespread knowledge of the results of Prof. Hunstein’s experiment among patients with different types of amyloidosis. Green tea extract is available as over the counter supplements and reveals an excellent safety profile. This results in a very low acceptance to renounce on the use of a potential beneficial effect of green tea in the absence of any specific treatment approach for WT-ATTR up to now. Due to novelty of T1 mapping, we also were not able to create a control group from retrospective data. Currently, T1 mapping is an experimental tool and no general consensus concerning sequence and post-processing methods was made. Consistent normal values for T1 relaxation times or ECV values are lacking, most likely due to variability among different MRI scanner types.
Second, the observation period of 1 year is relatively short. It needs to be demonstrated that changes of ECV might be more pronounced with longer follow-up. Nevertheless, lack of increase of LV myocardial mass during follow-up of 1 year potentially indicates any positive effect when compared to non-interventional reports in cardiac ATTR [18, 19].
Thirdly, we are familiar with the complexity and the potential sources of error of T1 mapping measurements and their related calculations. Nevertheless, a low inter-study variability and an excellent reproducibility were recently reported, when using the same MRI scanner and the same protocol [31, 32]. Regrettably, up to now, no data exist on the consistency of T1 and ECV values in normal healthy volunteers reassessed after 1 year. Unfortunately, we were not able to obtain these data due to a recent exchange of the MRI scanner at our department. Moreover, technical limitations included that T1 mapping was only created in a mid-ventricular short axis, representing only 6 segments of the AHA 16-segment model. Though amyloid deposition is a global process, regional differences in T1 values or ECV cannot be fully excluded. The heterogeneity of amyloid deposition might even impact ECV measurements, even if the scans were performed in the same region and multiple measurements of ECV were performed.
Fourth, the clinical benefit of green tea consumption would be strengthened by demonstration of an increase of the individual exercise capacity. Unfortunately, cardiopulmonary exercise testing or 6 min walk test has not been performed in this study that was primarily focused on imaging aspects. However, none of the study subjects worsened regarding left ventricular function or NYHA functional class during the study period. No hospitalization for heart failure became necessary.
Conclusions
In summary, the present study confirms LV myocardial mass reduction in patients with cardiac TTR amyloidosis in a small, but more uniform cohort of patients with WT-ATTR daily consuming green tea. Moreover, this preliminary proof of principle data—although not statistically significant and limited by the low number of patients—gave first evidence that the decrease of LV myocardial mass could potentially rather be caused by reduction of the amyloid load than atrophy of cardiomyocytes. Thus, this study stimulates future research focused on the potential role of T1 mapping for monitoring of progression of amyloid deposition or even treatment effects. Further multi-center (in an optimal way placebo controlled) studies with longer follow-up in a larger patient cohorts are urgently needed to draw final conclusions from the present hypothesis generating study.
References
Cornwell GG 3rd, Murdoch WL, Kyle RA, Westermark P, Pitkanen P (1983) Frequency and distribution of senile cardiovascular amyloid. A clinicopathologic correlation. Am J Med 75(4):618–623
Pomerance A (1965) Senile cardiac amyloidosis. Br Heart J 27(5):711–718
Mohty D, Damy T, Cosnay P, Echahidi N, Casset-Senon D, Virot P, Jaccard A (2013) Cardiac amyloidosis: updates in diagnosis and management. Arch Cardiovasc Dis 106(10):528–540. doi:10.1016/j.acvd.2013.06.051
Bulawa CE, Connelly S, Devit M, Wang L, Weigel C, Fleming JA, Packman J, Powers ET, Wiseman RL, Foss TR, Wilson IA, Kelly JW, Labaudiniere R (2012) Tafamidis, a potent and selective transthyretin kinetic stabilizer that inhibits the amyloid cascade. Proc Natl Acad Sci USA 109(24):9629–9634. doi:10.1073/pnas.1121005109
Coelho T, Maia LF, Martins da Silva A, Waddington Cruz M, Plante-Bordeneuve V, Lozeron P, Suhr OB, Campistol JM, Conceicao IM, Schmidt HH, Trigo P, Kelly JW, Labaudiniere R, Chan J, Packman J, Wilson A, Grogan DR (2012) Tafamidis for transthyretin familial amyloid polyneuropathy: a randomized, controlled trial. Neurology 79(8):785–792. doi:10.1212/WNL.0b013e3182661eb1
Nencetti S, Rossello A, Orlandini E (2013) Tafamidis (Vyndaqel): a light for FAP patients. Chem Med Chem. doi:10.1002/cmdc.201300245
Bieschke J, Russ J, Friedrich RP, Ehrnhoefer DE, Wobst H, Neugebauer K, Wanker EE (2010) EGCG remodels mature alpha-synuclein and amyloid-beta fibrils and reduces cellular toxicity. Proc Natl Acad Sci USA 107(17):7710–7715. doi:10.1073/pnas.0910723107
Ehrnhoefer DE, Bieschke J, Boeddrich A, Herbst M, Masino L, Lurz R, Engemann S, Pastore A, Wanker EE (2008) EGCG redirects amyloidogenic polypeptides into unstructured, off-pathway oligomers. Nat Struct Mol Biol 15(6):558–566. doi:10.1038/nsmb.1437
Ferreira N, Cardoso I, Domingues MR, Vitorino R, Bastos M, Bai G, Saraiva MJ, Almeida MR (2009) Binding of epigallocatechin-3-gallate to transthyretin modulates its amyloidogenicity. FEBS Lett 583(22):3569–3576. doi:10.1016/j.febslet.2009.10.062
Ferreira N, Saraiva MJ, Almeida MR (2012) Epigallocatechin-3-gallate as a potential therapeutic drug for TTR-related amyloidosis: “in vivo” evidence from FAP mice models. PLoS One 7(1):e29933. doi:10.1371/journal.pone.0029933
Kristen AV, Lehrke S, Buss S, Mereles D, Steen H, Ehlermann P, Hardt S, Giannitsis E, Schreiner R, Haberkorn U, Schnabel PA, Linke RP, Rocken C, Wanker EE, Dengler TJ, Altland K, Katus HA (2012) Green tea halts progression of cardiac transthyretin amyloidosis: an observational report. Clin Res Cardiol 101(10):805–813. doi:10.1007/s00392-012-0463-z
White SK, Sado DM, Flett AS, Moon JC (2012) Characterising the myocardial interstitial space: the clinical relevance of non-invasive imaging. Heart 98(10):773–779. doi:10.1136/heartjnl-2011-301515
Jerosch-Herold M, Kwong RY (2014) Cardiac T(1) imaging. Top Magn Reson Imaging 23(1):3–11. doi:10.1097/RMR.0000000000000013
Moon JC, Messroghli DR, Kellman P, Piechnik SK, Robson MD, Ugander M, Gatehouse PD, Arai AE, Friedrich MG, Neubauer S, Schulz-Menger J, Schelbert EB (2013) Myocardial T1 mapping and extracellular volume quantification: a Society for Cardiovascular Magnetic Resonance (SCMR) and CMR Working Group of the European Society of Cardiology consensus statement. J Cardiovasc Magn Reson 15:92. doi:10.1186/1532-429X-15-92
Longhi S, Guidalotti PL, Quarta CC, Gagliardi C, Milandri A, Lorenzini M, Potena L, Leone O, Bartolomei I, Pastorelli F, Salvi F, Rapezzi C (2014) Identification of TTR-related subclinical amyloidosis with (99 m)Tc-DPD scintigraphy. JACC Cardiovasc Imaging 7(5):531–532. doi:10.1016/j.jcmg.2014.03.004
Rapezzi C, Quarta CC, Obici L, Perfetto F, Longhi S, Salvi F, Biagini E, Lorenzini M, Grigioni F, Leone O, Cappelli F, Palladini G, Rimessi P, Ferlini A, Arpesella G, Pinna AD, Merlini G, Perlini S (2013) Disease profile and differential diagnosis of hereditary transthyretin-related amyloidosis with exclusively cardiac phenotype: an Italian perspective. Eur Heart J 34(7):520–528. doi:10.1093/eurheartj/ehs123
Ng B, Connors LH, Davidoff R, Skinner M, Falk RH (2005) Senile systemic amyloidosis presenting with heart failure: a comparison with light chain-associated amyloidosis. Arch Intern Med 165(12):1425–1429. doi:10.1001/archinte.165.12.1425
Benson MD, Teague SD, Kovacs R, Feigenbaum H, Jung J, Kincaid JC (2011) Rate of progression of transthyretin amyloidosis. Am J Cardiol 108(2):285–289. doi:10.1016/j.amjcard.2011.03.040
Ruberg FL, Maurer, Judge DP, Zeldenrust S, Skinner M, Kim AY, Falk RH, Cheung KN, Patel AR, Pano A, Packman J, Grogan (2012) Prospective evaluation of the morbidity and mortality of wild-type and V122I mutant transthyretin amyloid cardiomyopathy: the transthyretin amyloidosis cardiac study (TRACS). Am Heart J 164(2):222–228e221. doi:10.1016/j.ahj.2012.04.015
Hunstein W (2007) Epigallocathechin-3-gallate in AL amyloidosis: a new therapeutic option? Blood 110(6):2216. doi:10.1182/blood-2007-05-089243
Mereles D, Buss SJ, Hardt SE, Hunstein W, Katus HA (2010) Effects of the main green tea polyphenol epigallocatechin-3-gallate on cardiac involvement in patients with AL amyloidosis. Clin Res Cardiol 99(8):483–490. doi:10.1007/s00392-010-0142-x
Fontana M, Banypersad SM, Treibel TA, Maestrini V, Sado DM, White SK, Pica S, Castelletti S, Piechnik SK, Robson MD, Gilbertson JA, Rowczenio D, Hutt DF, Lachmann HJ, Wechalekar AD, Whelan CJ, Gillmore JD, Hawkins PN, Moon JC (2014) Native T1 mapping in transthyretin amyloidosis. JACC Cardiovas Imaging 7(2):157–165. doi:10.1016/j.jcmg.2013.10.008
Jerosch-Herold M, Sheridan DC, Kushner JD, Nauman D, Burgess D, Dutton D, Alharethi R, Li D, Hershberger RE (2008) Cardiac magnetic resonance imaging of myocardial contrast uptake and blood flow in patients affected with idiopathic or familial dilated cardiomyopathy. Am J Physiol Heart Circ Physiol 295(3):H1234–H1242. doi:10.1152/ajpheart.00429.2008
Kehr E, Sono M, Chugh SS, Jerosch-Herold M (2008) Gadolinium-enhanced magnetic resonance imaging for detection and quantification of fibrosis in human myocardium in vitro. Int J Cardiovasc Imaging 24(1):61–68. doi:10.1007/s10554-007-9223-y
Pack NA, Dibella EV, Wilson BD, McGann CJ (2008) Quantitative myocardial distribution volume from dynamic contrast-enhanced MRI. Magn Reson Imaging 26(4):532–542. doi:10.1016/j.mri.2007.10.003
Karamitsos TD, Piechnik SK, Banypersad SM, Fontana M, Ntusi NB, Ferreira VM, Whelan CJ, Myerson SG, Robson MD, Hawkins PN, Neubauer S, Moon JC (2013) Noncontrast T1 mapping for the diagnosis of cardiac amyloidosis. JACC Cardiovascular Imaging 6(4):488–497. doi:10.1016/j.jcmg.2012.11.013
Brooks J, Kramer CM, Salerno M (2013) Markedly increased volume of distribution of gadolinium in cardiac amyloidosis demonstrated by T1 mapping. J Magn Reson Imaging 38(6):1591–1595. doi:10.1002/jmri.24078
Maceira AM, Prasad SK, Hawkins PN, Roughton M, Pennell DJ (2008) Cardiovascular magnetic resonance and prognosis in cardiac amyloidosis. J Cardiovasc Magn Reson 10:54. doi:10.1186/1532-429X-10-54
Flett AS, Hayward MP, Ashworth MT, Hansen MS, Taylor AM, Elliott PM, McGregor C, Moon JC (2010) Equilibrium contrast cardiovascular magnetic resonance for the measurement of diffuse myocardial fibrosis: preliminary validation in humans. Circulation 122(2):138–144. doi:10.1161/CIRCULATIONAHA.109.930636
Kuruvilla S, Janardhanan R, Antkowiak P, Keeley EC, Adenaw N, Brooks J, Epstein FH, Kramer CM, Salerno M (2014) Increased extracellular volume and altered mechanics are associated with LVH in hypertensive heart disease, not hypertension alone. JACC Cardiovasc Imaging. doi:10.1016/j.jcmg.2014.09.020
Sado DM, Maestrini V, Piechnik SK, Banypersad SM, White SK, Flett AS, Robson MD, Neubauer S, Ariti C, Arai A, Kellman P, Yamamura J, Schoennagel BP, Shah F, Davis B, Trompeter S, Walker M, Porter J, Moon JC (2014) Noncontrast myocardial T mapping using cardiovascular magnetic resonance for iron overload. J Magn Reson Imaging. doi:10.1002/jmri.24727
Puntmann VO, Voigt T, Chen Z, Mayr M, Karim R, Rhode K, Pastor A, Carr-White G, Razavi R, Schaeffter T, Nagel E (2013) Native T1 mapping in differentiation of normal myocardium from diffuse disease in hypertrophic and dilated cardiomyopathy. JACC Cardiovasc Imaging 6(4):475–484. doi:10.1016/j.jcmg.2012.08.019
Conflict of interest
AVK received financial support for research from Dr. Loges & Co GmbH, Winsen/Luhe Germany. All other authors state that they have no conflict of interest regarding this manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Siepen, F.a.d., Buss, S.J., Andre, F. et al. Extracellular remodeling in patients with wild-type amyloidosis consuming epigallocatechin-3-gallate: preliminary results of T1 mapping by cardiac magnetic resonance imaging in a small single center study. Clin Res Cardiol 104, 640–647 (2015). https://doi.org/10.1007/s00392-015-0826-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00392-015-0826-3