Abstract
Background
We are presenting an extension of a previously published trial on the efficacy and safety of a paclitaxel-coated balloon in coronary ISR in a larger patient population and after a complete follow-up of 2 years.
Methods
Hundred eight patients were enrolled in two separately randomized, double-blind multicenter trials on efficacy and safety using an identical protocol. Patients were treated by the paclitaxel-coated (3 µg/mm2 balloon surface; Paccocath) or an uncoated balloon. The main inclusion criteria were a diameter stenosis of ≥70% and <30 mm length with a vessel diameter of 2.5–3.5 mm. The primary endpoint was angiographic late lumen loss in-segment. Secondary endpoints included binary restenosis rate and major adverse cardiovascular events (MACE).
Results
Quantitative coronary angiography revealed no differences in baseline parameters. After six months in-segment late lumen loss was 0.81 ± 0.79 mm in the uncoated balloon group vs. 0.11 ± 0.45 mm (P < 0.001) in the drug-coated balloon group resulting in a binary restenosis rate of 25/49 vs. 3/47 (P < 0.001). Until 12 months post procedure 20 patients in the uncoated balloon group compared to two patients in the coated balloon group required target lesion revascularization (P = 0.001). Between 12 and 24 only two MACE were recorded, a stroke in the uncoated and a target lesion revascularization in the coated balloon group.
Conclusion
Treatment of coronary ISR with paclitaxel-coated balloon catheters persistently reduces repeat restenosis up to 2 years. (ClinicalTrials.gov Identifier: NCT00106587, NCT00409981).
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Drug-eluting stents (DES) have become widely accepted and are used for a wide spectrum of clinical indications. Clinical trials of drug-eluting stents show excellent results in reducing the need for target lesion re-intervention [15, 20, 33]. Nevertheless, concerns have been raised that such drug-releasing stents while being effective may be associated with an increased incidence of late thrombotic complications [2, 5, 10, 15, 16, 18, 21, 22], especially in high-risk patient populations [5, 10, 31].
Although DES are used in the treatment of in-stent restenosis [12, 14] they further reduce the flexibility of the vessel and limit the repeatability of the procedure. Drug-coated balloon catheters represent an alternative option for the treatment of coronary and peripheral arteries. A drug coated balloon delivers an initially homogenous drug concentration to the arterial wall, which has been shown to be an effective substitute for sustained release [27]. Preclinical trials demonstrated the efficacy of drug-coated balloons in inhibiting neointimal proliferation [26, 32]. These results have been confirmed by first clinical evidence in patients with coronary in-stent restenosis [24] and peripheral artery disease [35]. However, these initial clinical findings need to be corroborated by further investigation and longer follow-up data.
In this article we present an extension of the already published trial by a separately randomized group of patients (ISR II trial) and the results of two-year follow-up of the patients enrolled in the ISR I and II trials after treatment of coronary in-stent restenosis with a paclitaxel-coated balloon compared to its uncoated counterpart. The Paccocath ISR II trial was conducted with an identical protocol to increase the probability of detecting coating-related adverse events and test the reproducibility of the result of ISR I.
Methods
Study design
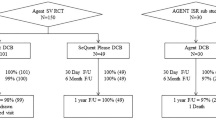
A total of 108 patients were enrolled in two separately randomized, double-blind multicenter trials (Paccocath ISR I [24] and ISR II) investigating the efficacy and safety of a paclitaxel-coated balloon (3 µg/mm2 balloon surface; Paccocath), using an identical protocol. The trials were conducted at five departments of cardiology at the medical schools of the universities of Berlin, Freiburg, Homburg/Saar, and Mannheim/Heidelberg in Germany. Financial support was provided by Bavaria Medizintechnik GmbH, Oberpfaffenhoven, Germany, the manufacturer of the balloon angioplasty catheters used in this study. The sponsor had no role in the design or conduct of the study, in the analysis of the results, in the decision to publish, or in the drafting of the manuscript. The authors vouch for the accuracy and completeness of the data presented.
The study was performed according to the Declaration of Helsinki and WHO guidelines. Furthermore, the requirements of sections 20–22 of the German Medical Device Law as well as the European standard EN 540 were followed. All patients gave written informed consent. The study was approved by the local ethics committees.
Details of the methods have been previously published [24]. Patients at least 18 years of age with clinical evidence of stable or unstable angina or a positive functional study and a single restenotic lesion in a stented coronary artery with bare metal stents oder drug eluting stents, were considered for enrollment. Major clinical exclusion criteria were acute myocardial infarction within the past 72 h, chronic renal insufficiency with serum creatinine levels >2.0 mg per deciliter, known hypersensitivity or contraindications to aspirin, heparin, clopidogrel, abciximab, or paclitaxel, and sensitivity to contrast media not amenable to premedication. Cardiac catheterization premedication and medication during the intervention was carried out according to hospital practice. Glycoprotein IIb/IIIa antagonists were administered at operator’s discretion.
Baseline angiography of the target vessel was performed in at least two near-orthogonal views showing the target lesion free of foreshortening or vessel overlap. After assessment for angiographic exclusion criteria, each suitable patient was randomly assigned to undergo balloon angioplasty of the target lesion with either a paclitaxel-coated or an uncoated balloon catheter. Standard angioplasty catheters (Orbus X, Bavaria Medizin Technologie GmbH, Oberpfaffenhofen, Germany) were supplied either uncoated or coated with a paclitaxel dose of 3 µg/mm2 balloon surface. Balloon catheters were supplied as usual as sterile medical devices, six pieces of different size per patient. At that stage they were perfectly blinded. Only after the patient has been included in the study, the envelop of the selected balloon catheter was opened. Small visual differences between coated and non coated balloons became visible and could have been recognized after the same investigator has seen one or more balloons of each type. Investigators were not informed about visual differences. QCA was done by an independent core lab with no information on the balloons used in the individual patients. Thus, patient selection and core-lab data were done while investigators were perfectly blinded whereas differences in the appearance of the devices could at least theoretically have resulted in unblinding in some of the patients in the clinical course of the study.
Pre-dilatation of the target lesion was usually performed prior to the study intervention, using a non-study balloon catheter with a diameter 0.5 mm smaller than the study balloon. Study balloon inflation was performed in the same fashion as the inflation of a conventional balloon catheter. Recommended balloon inflation time was 60 s. Immediately following the procedure, heparin was discontinued. Vascular sheaths were removed according to usual hospital practice. After the performance of the procedure, the study balloon was saved for determination of residual paclitaxel content as previously reported [26].
Quantitative coronary angiography
Angiography was performed before and after all interventions and at angiographic follow-up using identical projections and analyses. Quantitative analysis of the coronary angiographic images was performed by an independent, blinded core laboratory. The CAAS II Research System (Pie Medical Imaging, Maastricht, The Netherlands) was used for automated contour detection and quantification. Measurements were obtained in the inner stenotic area, in the stented area with measurement shoulder to shoulder (in-stent), and in the total stented area plus 5 mm proximally and distally (in-segment). Restenosis was defined as ≥50% diameter stenosis at angiographic follow-up. Patterns of in-stent restenosis were defined according to the Mehran classification [19].
Follow-up and end points
100 mg aspirin and 75 mg clopidogrel were continued orally for one month, followed by treatment with aspirin alone. Patients underwent follow-up angiography after 6 months (up to 9 months) and were followed up for 24 months by clinical observation. All endpoints and adverse events were evaluated in consensus by the investigators. The investigators and the core lab remained blinded until the database was closed.
Angiographic late lumen loss (difference between the post-procedural and 6-month follow-up in-segment minimal lumen diameter; evaluated by quantitative coronary angiography) was the primary end point. Secondary end points included binary angiographic restenosis rate (diameter stenosis of at least 50% assessed by quantitative coronary angiography at 6-month follow-up) and combined clinical end points with a follow-up of 24 months including acute and subacute stent thrombosis, target lesion revascularization, myocardial infarction, cerebral stroke, and death.
Acute stent thrombosis was defined as the occurrence of new severely reduced flow (TIMI grade 0 or 1) within the target vessel during the intervention that persisted and required rescue by a non-assigned treatment strategy or resulted in myocardial infarction or death. Subacute stent thrombosis was defined as vessel closure occurring during follow-up. Target lesion revascularization was defined as percutaneous reintervention or coronary artery bypass graft surgery involving the target lesion. The decision to perform a reintervention procedure was based upon symptoms, anatomic findings at follow-up angiography, or both.
Myocardial infarction was assumed if two of the following five criteria applied: (1) chest pain lasting longer than 30 min; (2) significant ECG changes typical of acute MI (0.1 mV ST elevation in at least two adjacent ECG leads or new occurrence of a complete left bundle branch block); (3) significant increase (three times above normal) of creatinine kinase or its MB-isoform; (4) new significant Q-waves; or (5) chest pain leading to angiography up to 6 h after the onset of symptoms and showing a totally occluded vessel compared with the previous angiogram. Deaths were documented and confirmed from hospital records or by contacting the patient’s relatives or the treating physician.
The definition of a significant adverse event followed international (ICH) guidelines [11]. Target lesion revascularization was considered a significant adverse event because it involved patient hospitalization in each case.
Statistical analysis
The Paccocath ISR I trial was designed to investigate if balloon coating influences late lumen loss [24]. The Paccocath ISR II trial was conducted with an identical protocol to increase the probability of detecting coating-related adverse events and test the reproducibility of the result of ISR I.
Analysis of the data for all end points was performed according to intention-to-treat. Continuous data are expressed as mean ± standard deviation. Categorical variables were compared using the two-sided χ 2 test, and continuous variables were compared using two-sided Student’s t test. Confidence intervals for the difference of proportions were calculated using normal approximation of the binominal distribution without correction for continuity. Event-free survival was compared by Kaplan-Meier analysis using a log rank test (Mantel-Cox) (SPSS 15.0). P-values were adjusted according to Fisher’s method of combining independent tests. A two-sided P-value of 0.05 was considered significant.
Results
A total of 108 patients were enrolled, 52 patients in ISR I and 56 patients in ISR II. Fifty-four were randomly assigned to the uncoated balloon group, and 54 to the coated balloon group. Baseline parameters were similar in both groups (Tables 1 and 2). The mean age of the study population was 66 years. Most patients had multi-vessel coronary artery disease. Patients enrolled in the ISR II trial were older, were more often female, had a higher incidence of diabetes mellitus, and had longer lesions than the patients included in the ISR I study.
The pattern of in-stent restenosis was predominantly diffuse. Procedural data including the size of the study balloon, the use of additional stents, and administration of glycoprotein IIb/IIIa antagonists were also similar in the two groups (Table 1). One patient assigned to the uncoated balloon group was erroneously treated with a drug-coated balloon catheter taken from a non-assigned set but was analyzed by intention-to-treat with the uncoated balloon group.
Complete clinical follow-up is available in all 108 patients. One patient from the uncoated and the coated balloon group each suffered cardiac death; two further non-cardiac deaths in the uncoated and one in the coated group occurred. Myocardial infarction occurred somewhat more frequently (P > 0.05) in the uncoated balloon group. The incidence of major adverse cardiac events was reduced from 46% in the control group to 11% in patients treated with the drug-coated balloon. This difference was mainly driven by the reduction of target lesion revascularization from 37% to 6% (Table 3). The Kaplan-Meier curves of major adverse cardiovascular events for the two groups over the 24 months of the trial are shown in Fig. 1.
Angiographic follow-up was available in 49 of 54 patients (91%) in the uncoated balloon group and in 47 of 54 patients (87%) in the drug-coated balloon group. After 6 months, in-segment late lumen loss was reduced from 0.80 ± 0.79 mm with the uncoated balloon to 0.11 ± 0.44 mm in the paclitaxel-coated balloon group (P = 0.001). This reduction was reflected in a reduction of binary restenosis rate from 51% to 6% (P = 0.001) (Table 2).
Originally, ISR I and ISR II population were separately evaluated as the trials were separately randomized. Late lumen loss as the primary end point proved to be statistically significantly reduced in the coated balloon groups in both trials (P < 0.01 in each case) as were binary restenosis rate (P < 0.01) and target lesion revascularization (P < 0.01) (for details see Table 4).
Serious adverse events
A total of 62 patients suffered from one or more serious adverse events according to the ICH definition which includes any hospitalization or prolongation of hospitalization (Table 5) [11]. Of these, 37 occurred in patients treated with uncoated balloons, in 20 of these patients due to repeated intervention of the target lesion. Serious adverse events occurred in 25 patients treated with the drug-coated balloon (P = 0.032 versus uncoated balloon group), in 21 of them classified as unrelated to treatment. One event classified as possibly related to treatment in the drug-coated balloon arm was death following myocardial infarction 11 months after the intervention. The three other events possibly related to the treatment in this group were target lesion reinterventions 6–18 months after the index procedure.
Discussion
Restenosis inhibition in coronary arteries by local drug delivery mediated by the angioplasty balloon is a new concept which differs from the well known and clinically established drug-eluting stents in a variety of features. A critical difference is the very short exposure time of the vessel wall to the drug during the time a balloon is inflated. Preclinical studies have demonstrated that even brief contact between antiproliferative agents and vascular smooth muscle cells results in prolonged inhibition of neointimal proliferation [7, 28, 29]. First-in-man data in the treatment of coronary in-stent restenosis with paclitaxel-coated balloon catheters showed clinical efficacy and safety over 12 months [24]. However, reproducibility of the results observed in the small number of patients enrolled in the first clinical study as well as persistence of restenosis inhibition beyond 12 months was questioned.
The data reported in the present paper extends the initial findings in the Paccocath ISR I trial. They indicate perfect reproducibility of the results in additional 56 patients with similar baseline clinical and angiographic data. In each of the two separately randomized patient populations the primary end-point late lumen loss as well as the secondary clinical end point ‘target lesion revascularization’ reached statistically significant differences to the control group. Overall, the primary endpoint late lumen loss was reduced from 0.8 ± 0.8 mm in the uncoated balloon group to 0.1 ± 0.4 mm in the drug-coated balloon group (P = 0.001). Furthermore, the follow-up of the patients enrolled in the initial trial and the 56 patients of ISR II support the assumption that preventing the balloon-dilatation related acute response to injury is sufficient to achieve a persistent benefit if no polymer coated stent is implanted. The clinical benefit (target lesion revascularization, MACE) was found to be maintained over a two-year follow-up period. The reduction in angiographic parameters of restenosis and clinical events was more pronounced than previously reported with drug eluting stents and brachytherapy. Reintervention rates after treatment of coronary in-stent restenosis with drug eluting stents ranges from 10 to 19%, with brachytherapy from 17 to 28% [8, 14, 30, 34], and 6% after two years in this trial with the PACCOCATH balloon. In contrast to drug eluting stents, combined antiplatelet therapy was continued only for one month, followed by treatment with aspirin alone. Although late thrombosis of the target lesion in the patients treated with the coated balloons can not be ruled out with certainty the larger number of myocardial infarctions and the equal number of cardiac deaths (Table 5) do not indicate that it was a problem in this trial.
Restenosis caused by neointimal proliferation is a slow process, suggesting that prolonged local drug administration is necessary for effective inhibition. This can be achieved by stent-based sustained drug delivery due to special features for slow release, mostly polymer matrixes [25, 36]. About 85% of the stented vessel wall area is not covered by the stent struts. Sustained drug release is essential probably because drug distribution from a drug-eluting stent to the arterial wall is inhomogeneous and consequently tissue concentrations are very low more distant to the struts [9, 27]. Long lasting drug release from the stent struts is associated with delayed and incomplete endothelialization and an increased risk for stent thrombosis [3, 13, 17, 23]. Furthermore, polymers embedding the antiproliferative agent can directly induce thrombosis and have been reported to cause chronic inflammatory reactions [4, 6, 13, 23, 36]. The concept of implanting drug-eluting stents in a restenotic in-stent lesion involves insertion of a second layer of metal in a native coronary artery. The repeatability of this approach is limited. Sustained drug release and permanent implantation of polymer coated stents are avoided if the drug is administered by the balloon surface.
Limitations
There was a non-significant difference of gender and diabetes mellitus in baseline data. However this had no impact on angiographic and clinical outcomes (Table 6). The Paccocath ISR I and II trial was a preliminary study limited in scope and observation period. It is unclear, if these positive findings in the treatment of coronary in-stent restenosis can be transferred to restenosis prevention in atherosclerotic coronary lesions. So far, efficacy in restenosis prevention has been demonstrated for peripheral vessels [35]. Further clinical trials are warranted to study the drug coated balloon in different indications and in direct comparison with drug eluting stents or brachytherapy.
References
Alfonso F, Perez-Vizcayno MJ, Hernandez R, Bethencourt A, Marti V, Lopez-Minguez JR, Angel J, Mantilla R, Moris C, Cequier A, Sabate M, Escaned J, Moreno R, Banuelos C, Suarez A, Macaya C, RIBS-II Investigators (2006) A randomized comparison of sirolimus-eluting stent with balloon angioplasty in patients with in-stent restenosis: results of the restenosis intrastent: balloon angioplasty Vs. elective sirolimus-eluting stenting (RIBS-II) trial. J Am Coll Cardiol 47:2152–2160
Camenzind E, Steg PG, Wijns W (2007) Stent thrombosis late after implantation of first-generation drug-eluting stents: a cause for concern. Circulation 115:1440–1455; discussion 1455, Epub 2007 Mar 7
Carlsson J, von Wagenheim B, Linder R, Anwari TM, Qvist J, Petersson I, Magounakis T, Lagerqvist B (2007) Is late stent thrombosis in drug-eluting stents a real clinical issue?: a single-center experience and review of the literature. Clin Res Cardiol 96:86–93
Carter AJ, Aggarwal M, Kopia GA, Tio F, Tsao PS, Kolata R, Yeung AC, Llanos G, Dooley J, Falotico R (2004) Long-term effects of polymer-based, slow-release, sirolimus-eluting stents in a porcine coronary model. Cardiovasc Res 63:617–624
Daemen J, Wenaweser P, Tsuchida K, Abrecht L, Vaina S, Morger C, Kukreja N, Juni P, Sianos G, Hellige G, van Domburg RT, Hess OM, Boersma E, Meier B, Windecker S, Serruys PW (2007) Early and late coronary stent thrombosis of sirolimus-eluting and paclitaxel-eluting stents in routine clinical practice: data from a large two-institutional cohort study. Lancet 369:667–678
Farb A, Heller PF, Shroff S, Cheng L, Kolodgie FD, Carter AJ, Scott DS, Froehlich J, Virmani R (2001) Pathological analysis of local delivery of paclitaxel via a polymer-coated stent. Circulation 104:473–479
Herdeg C, Oberhoff M, Baumbach A, Blattner A, Axel DI, Schroder S, Heinle H, Karsch KR (2000) Local paclitaxel delivery for the prevention of restenosis: biological effects and efficacy in vivo. J Am Coll Cardiol 35:1969–1976
Holmes DR Jr, Teirstein P, Satler L, Sketch M, O’Malley J, Popma JJ, Kuntz RE, Fitzgerald PJ, Wang H, Caramanica E, Cohen SA, SISR Investigators (2006) Sirolimus-eluting stents vs vascular brachytherapy for in-stent restenosis within bare-metal stents: the SISR randomized trial. JAMA 295:1264–1273; Epub 2006 Mar 12
Hwang CW, Wu D, Edelman ER (2001) Physiological transport forces govern drug distribution for stent-based delivery. Circulation 104:600–605
Iakovou I, Schmidt T, Bonizzoni E, Ge L, Sangiorgi GM, Stankovic G, Airoldi F, Chieffo A, Montorfano M, Carlino M, Michev I, Corvaja N, Briguori C, Gerckens U, Grube E, Colombo A (2005) Incidence, predictors, and outcome of thrombosis after successful implantation of drug-eluting stents. JAMA 293:2126–2130
ICH Harmonized Tripartite Guideline. Clinical safety data management: definitions and standards for expedited reporting E2A. International conference on harmonisation of technical requirements for registration of pharmaceuticals for human use. October 1994. Available on-line at http://www.ich.org /LOB/media/MEDIA436.pdf. Accessed 23 Jun 2006
Iofina E, Haager PK, Radke PW, Langenberg R, Blindt R, Ortlepp J, Kuhl H, Hanrath P, Hoffmann R (2005) Sirolimus- and paclitaxel-eluting stents in comparison with balloon angioplasty for treatment of in-stent restenosis. Catheter Cardiovasc Interv 64:28–34
Joner M, Finn AV, Farb A, Mont EK, Kolodgie FD, Ladich E, Kutys R, Skorija K, Gold HK, Virmani R (2006) Pathology of drug-eluting stents in humans: delayed healing and late thrombotic risk. J Am Coll Cardiol 48:193–202
Kastrati A, Mehilli J, von Beckerath N, Dibra A, Hausleiter J, Pache J, Schuhlen H, Schmitt C, Dirschinger J, Schomig A, ISAR-DESIRE Study Investigators (2005) Sirolimus-eluting stent or paclitaxel-eluting stent vs balloon angioplasty for prevention of recurrences in patients with coronary in-stent restenosis: a randomized controlled trial. JAMA 293:165–171
Kastrati A, Mehilli J, Pache J, Kaiser C, Valgimigli M, Kelbaek H, Menichelli M, Sabate M, Suttorp MJ, Baumgart D, Seyfarth M, Pfisterer ME, Schomig A (2007) Analysis of 14 trials comparing sirolimus-eluting stents with bare-metal stents. N Engl J Med 356:1030–1039; Epub 2007 Feb 12
Lagerqvist B, James SK, Stenestrand U, Lindback J, Nilsson T, Wallentin L, SCAAR Study Group (2007) Long-term outcomes with drug-eluting stents versus bare-metal stents in Sweden. N Engl J Med 356:1009–1019; Epub 2007 Feb 12
Maisel WH (2007) Unanswered questions–drug-eluting stents and the risk of late thrombosis. N Engl J Med 356:981–984
Mauri L, Hsieh WH, Massaro JM, Ho KK, D’Agostino R, Cutlip DE (2007) Stent thrombosis in randomized clinical trials of drug-eluting stents. N Engl J Med 356:1020–1029; Epub 2007 Feb 12
Mehran R, Dangas G, Abizaid AS, Mintz GS, Lansky AJ, Satler LF, Pichard AD, Kent KM, Stone GW, Leon MB (1999) Angiographic patterns of in-stent restenosis: classification and implications for long term outcome. Circulation 100:1872–1878
Morice MC, Serruys PW, Sousa JE, Fajadet J, Ban Hayashi E, Perin M, Colombo A, Schuler G, Barragan P, Guagliumi G, Molnar F, Falotico R, RAVEL Study Group (2002) Randomized study with the sirolimus-coated Bx velocity balloon-expandable stent in the treatment of patients with de Novo Native coronary artery lesions. A randomized comparison of a sirolimus-eluting stent with a standard stent for coronary revascularization. N Engl J Med 346:1773–1780
Nordmann AJ, Briel M, Bucher HC (2006) Mortality in randomized controlled trials comparing drug-eluting Vs. bare metal stents in coronary artery disease: a meta-analysis. Eur Heart J 27:2784–814; Epub 2006 Oct 4
Pfisterer M, Brunner-La Rocca HP, Buser PT, Rickenbacher P, Hunziker P, Mueller C, Jeger R, Bader F, Osswald S, Kaiser C, BASKET-LATE Investigators (2006) Late clinical events after clopidogrel discontinuation may limit the benefit of drug-eluting stents: an observational study of drug-eluting versus bare-metal stents. J Am Coll Cardiol 48:2584–2591; Epub 2006 Nov 2
Scheller B, Grandt A, Wnendt S, Kuttler B, Bolz U, Lorenz G, Böhm M, Nickenig G (2005) Comparative study of tacrolimus and paclitaxel stent coating to prevent restenosis after coronary intervention. Z Kardiol 94:445–452
Scheller B, Hehrlein C, Bocksch W, Rutsch W, Haghi D, Dietz U, Böhm M, Speck U (2006) Treatment of in-stent restenosis with a paclitaxel-coated balloon catheter. N Engl J Med 355:2113–2124
Scheller B, Kühler M, Cremers B, Mahnkopf D, Böhm M, Boxberger M (2008) Short- and long-term effects of a novel paclitaxel coated stent in the porcine coronary model. Clin Res Cardiol 97:118–123
Scheller B, Speck U, Abramjuk C, Bernhardt U, Böhm M, Nickenig G (2004) Paclitaxel balloon coating—a novel method for prevention and therapy of restenosis. Circulation 110:810–814
Scheller B, Speck U, Böhm M (2007) Prevention of restenosis—is angioplasty the answer? Heart 93:539–541
Scheller B, Speck U, Romeike B, Schmitt A, Sovak M, Böhm M, Stoll HP (2003) Contrast media as a carrier for local drug delivery: successful inhibition of neointimal proliferation in the porcine coronary stent model. Eur Heart J 24(15):1462–1467
Scheller B, Speck U, Schmitt A, Böhm M, Nickenig G (2003) Addition of paclitaxel to contrast media prevents restenosis after coronary stent implantation. J Am Coll Cardiol 42(8):1415–1420
Silber S, Popma JJ, Suntharalingam M, Lansky AJ, Heuser RR, Speiser B, Teirstein PS, Bass T, O’Neill W, Lasala J, Reisman M, Sharma SK, Kuntz RE, Bonan R (2005) Two-year clinical follow-up of 90Sr/90Y beta-radiation versus placebo control for the treatment of instent restenosis (START). Am Heart J 149:689–694
Spaulding C, Daemen J, Boersma E, Cutlip DE, Serruys PW (2007) A pooled analysis of data comparing sirolimus-eluting stents with bare-metal stents. N Engl J Med 356:989–997; Epub 2007 Feb 12
Speck U, Scheller B, Abramjuk C, Breitwieser C, Dobberstein J, Böhm M, Hamm B (2006) Neointima inhibition: comparison of effectiveness of non-stent-based local drug delivery and a drug-eluting stent in porcine coronary arteries. Radiology 240:411–418
Stone GW, Moses JW, Ellis SG, Schofer J, Dawkins KD, Morice MC, Colombo A, Schampaert E, Grube E, Kirtane AJ, Cutlip DE, Fahy M, Pocock SJ, Mehran R, Leon MB (2007) Safety and efficacy of sirolimus- and paclitaxel-eluting coronary stents. N Engl J Med 356:998–1008; Epub 2007 Feb 12
Stone GW, Ellis SG, O’Shaughnessy CD, Martin SL, Satler L, McGarry T, Turco MA, Kereiakes DJ, Kelley L, Popma JJ, Russell ME, TAXUS V ISR Investigators (2006) Paclitaxel-eluting stents vs vascular brachytherapy for in-stent restenosis within bare-metal stents: the TAXUS V ISR randomized trial. JAMA 295:1253–1263
Tepe G, Zeller T, Albrecht T, Heller S, Schwarzwälder U, Beregi JP, Claussen CD, Oldenburg A, Scheller B, Speck U (2008) Local taxane with short exposure for reduction of restenosis in distal arteries: thunder trial. N Engl J Med 358:689–699
van der Giessen WJ, Lincoff AM, Schwartz RS, van Beusekom HM, Serruys PW, Holmes DR Jr, Ellis SG, Topol EJ (1996) Marked inflammatory sequelae to implantation of biodegradable and nonbiodegradable polymers in porcine coronary arteries. Circulation 94:1690–1697
Acknowledgments
The authors thank Matthias Braeutigam, MD, Schering AG Berlin, for supporting basic experiments leading to the clinical studies. The authors further thank Wolfgang Gimbel, MD for clinical monitoring, Bettina Kelsch for critical data review and report writing, Bianca Werner and Nicole Hollinger for handling the database, and Kevin Wilhelmi for assistance in quantitative analysis of coronary angiographies. Statistical analysis was supported by BIAS GmbH, Berlin, Germany.
Disclosures: The study was supported by Bavaria Medizintechnik GmbH, Oberpfaffenhoven and Bayer-Schering Pharma AG, Berlin, both Germany. Dr. Scheller and Dr. Speck report being co-inventors of a patent application for various methods of restenosis inhibition, including the technique employed in this trial, by Charité University Hospital, Berlin. Dr. Speck reports serving as a consultant to Bayer-Schering AG, Berlin.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Scheller, B., Hehrlein, C., Bocksch, W. et al. Two year follow-up after treatment of coronary in-stent restenosis with a paclitaxel-coated balloon catheter. Clin Res Cardiol 97, 773–781 (2008). https://doi.org/10.1007/s00392-008-0682-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00392-008-0682-5