Abstract
Aim
For the treatment of complex pelvic organ prolapse, many different surgical procedures are described without any comparative studies available. Laparoscopic ventral mesh rectopexy after D’Hoore is one of the methods, which is publicized to treat patients with symptomatic rectocele, enterocele and rectal prolapse.
Method
All patients who received ventral mesh rectopexy since 07/10 for symptomatic rectocele, enterocele and possible rectal prolapse I ° or II ° in terms of a complex pelvic floor disorder were included in this follow-up study. The Wexner score for incontinence was recorded (range 0–20), the constipation score of Herold (r6-30) was evaluated as well as supplementary questions compiled by D’Hoore concerning outlet symptoms (r0-20). In addition, the quality of life (SF-12) was requested.
Results
Thirty-one women were operated in the period, and 27 were eligible to be included in the present study. Median follow-up was 22 months (2–39). The preoperative Wexner score was in median 8 (0–20), going down to 6 (0–20) without significance (p = 0.735). The constipation score decreased significantly from median 14 (9–21) to 11 (6–25) (p = 0.007). The median score after D’Hoore was preoperatively 8 (4–16) and 4.5 (0–17) postoperatively (p = 0.004). The SF-12 values were preoperatively significantly reduced compared to the normal population; postoperatively, they equalized.
Conclusion
Two years after laparoscopic ventral mesh rectopexy, constipation and quality of life improve significantly in patients with complex pelvic organ prolapse. The grade of incontinence remains essentially the same, but was not the dominant clinical problem in the treated patients of our study.
Statement
The improvement in constipation and quality of life after laparoscopic ventral mesh rectopexy for obstructive defecation is encouraging. However, the impact on sexual life differs; some patients improve but a relevant number reports a change for the worse.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Obstructive defecation is a term for functional disorder resulting from different kinds of pelvic pathologies. The cause may be internal obstruction of the lumen, e.g. intussusception or rectal prolapse, external compression, e.g. by an enterocele or cul-de-sac, or a bulge with an obstructive angulation, e.g. rectocele [1]. Patients typically show incomplete evacuation and fragmented defecation; straining paradoxically obstructs the evacuation which gave the disorder its name. Evacuation of stool may require manual support. Dynamic MRI and conventional defecography can illustrate the changes in real time and, thus, confirm the diagnosis [2]. However, even when the morphological findings are impressive, they do not automatically warrant surgery as a high percentage of healthy people without symptoms show such morphologic changes [3]. The indication for surgery depends on the associated functional problems. There are dozens of therapeutic approaches, conservative and surgical, which reflects the low-evidence level in this field. The ideal surgical procedure for obstructive defecation remains unclear [4–6].
While rectal prolapse is a disease of predominantly elder women, obstructive defecation also affects younger patients [7]. This calls for special attention not only for restoration of bowel function but also for the preservation of sexual function or simultaneous restoration if impaired.
This study analyses functional outcome and quality of life in 27 patients having undergone ventral mesh rectopexy according to D’Hoore for complex pelvic floor disorders [8].
Patients and methods
The study protocol was reviewed and approved by the local institutional ethics committee of the University Hospital Mannheim, Germany. The study was performed according to the Declaration of Helsinki. All patients provided written informed consent for participation in the study.
Eligibility criteria
Patients with complex pelvic floor disorders are all discussed in an interdisciplinary setting in our institution at the monthly held pelvic floor conference, and patients are selected for surgery only after having undergone maximal conservative treatment. Obstipation was the main symptom in patients finally operated. Patients with exclusive rectal prolapse III° do not undergo ventral mesh rectopexy in our department as according to our treatment algorithm, such patients are treated with either resection rectopexy or Delorme’s procedure. Both of these surgical treatments of external prolapse have been proven effective in multiple studies, and no adequately powered comparative study has yet shown any distinct advantage for ventral mesh rectopexy for this indication. On the other hand, mesh implantation in the pelvis has been associated with rare but severe complications. As the above other techniques are good options for external prolapse, our concept here does not include ventral mesh rectopexy in the primary treatment. Patients with only morphologic changes without any significant clinical complaints of obstipation are generally not operated. All consecutive patients undergoing ventral mesh rectopexy for functional complex pelvic floor disorders over a 4-year period were included in this follow-up study.
Pretreatment evaluation
All patients underwent physical examination, digital rectal examination, colonoscopy, dynamic pelvic NMR (in selected cases conventional defecography), urological, and gynaecological examination.
Surgery
Two surgeons (P.K. and S.P.) performed all surgical procedures. The surgical procedure was performed in a standardized fashion in accordance with the originally published technique by D’Hoore [8]. The mesh was always sutured and not tacked to the proximal sacrum/promontory with non-resorbable sutures (0 silk or 0 Ethibond, Ethicon Inc., Somerville, NJ).
Wexner score
The Wexner score consists of five items describing the dimension of incontinence in the presence of “formed stool”, “liquid stool”, “gas”, the “need for pads” and “limitations of social life”. The time-dependent severity of the items can be indicated on a scale ranging from 0 to 4: “never” (0), “rare” (1; less than once per month), “sometimes” (2; less than once per week but more than once per month), “most of the time” (3; less than once a day but more than once per week), “always” (4; more than once a day). Thus, the range of the Wexner score is from 0 (full continence) to 20 (complete incontinence).
Modified constipation score by Herold
The validated outlet obstipation score by Herold [9] is a modification of the Cleveland Clinic Obstipation Score. The score consists of six items. Four questions clarify the existence and severity of constipation; two questions centre the differentiation of outlet versus slow transit constipation. Each question ranges from 1 to 5, the maximum score being 30 (Table 1). This score is able to distinguish between slow transit and outlet symptoms [9].
SF-12 questionnaire
The SF-12 questionnaire comprises 12 questions that target frequency-independent symptoms being incorporated into eight items describing different aspects of quality of life. The questionnaire is a generic quality of life instrument with good reliability and validity. It generates one continuous summary score each for subjective physical and mental health. Scores range from 0 to 100, with higher values indicating better quality of life. SF-12 contains values of a German norm population for comparison.
Symptoms of rectocele score
A specific scoring system for symptoms of a rectocele, designed by D’Hoore, was also used [10] (Table 2).
Additional questions
All patients were asked if they were sexually active before surgery and if they were, if their sexual life improved, worsened or did not change since the operation. At last, they were asked if their discomfort all over improved, worsened or did not change since the operation.
Statistical analysis
Data were analysed with SPSS version 19.0 (SPSS Inc., Chicago, IL). Comparison of symptoms was performed with a chi-square test for categorical data and a Mann–Whitney U-test for nonparametric variables. All tests were two-sided, and a p value of <0.05 was considered as statistically significant.
SF-12® questionnaire was purchased from Hogrefe Verlag (Testzentrale GmbH, Göttingen, Germany) that includes the software for statistical analysis.
Results
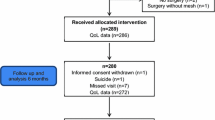
Thirty-one consecutive female patients were operated from 07/2010 to 08/2013, and there were no male patients. Two patients refused to take part in an interview; one patient was lost to follow-up. One patient was excluded because of dementia. Twenty-seven patients could clearly remember their preoperative state and were included in the study.
Preoperative characteristics are listed in Table 3. In nine cases, an enterocele in combination with a rectocele was the indication for the operation; six patients in addition had rectal prolapse (I and II°). There was a wide range of preoperative symptoms. Surgery in median took 208 min (range 127–339). Follow-up was 22 months (median, range 2–39).
Patients were asked if they generally felt better, worse or unchanged. Twenty (74 %) felt better, one patient worse (3.5 %) and six unchanged (22.2 %).
The preoperative Wexner score was 8 (median, range 0–20), going down to 6 (range 0–20), postoperatively. The difference was not statistically significant (p = 0.735).
The preoperative Herold obstipation score was 14 (median, range 9–21), and the postoperative value fell to 11 (range 6–25); this difference was statistically significant (p = 0.007).
The rectocele score by D’Hoore before surgery was 8 (median, range 4–16), decreasing to 4.5 postoperatively (range 0–17); this difference was statistically significant (p = 0.004). The results of the functional scores are displayed in Fig. 1.
Results of the SF-12 are displayed in Table 4. The patients preoperatively had a significantly lower estimate of physical and mental summary scales compared to the norm population. Postoperatively, the values were not anymore different from the norm population. The comparison from preoperative to postoperative summary scales of the cohort showed a significant difference in regard to the physical but not to the mental summary scale.
Fifteen women reported sexual activity before surgery; six women felt their sexual life improved in the postoperative course. Eight women indicated a change for the worse; one patient refused to answer this question.
Until now, one of the patients had a clinical relapse. She reported a sudden snapping sensation (“as if something was torn apart”) while squatting to do garden works 2 months after ventral mesh rectopexy. After that, she had identical complaints as before the surgery. During surgical revision one month later, the mesh was not attached to the sacrum anymore; a refixation of the mesh to the sacrum in combination with a Wells mesh rectopexy was performed, and she has not had a recurrence since then.
Discussion
Obstructive defecation results from various reasons. The vast majority of the patients are female with pelvic descent; our collective also reflects this with exclusively female patients. Patients with complex pelvic disorder often suffer from symptoms that affect quality of life in a relevant extent. The patients in our study showed a significant reduction of their physical and mental status before surgery compared to the norm population. In the follow-up interview after surgical repair (median 22 months postoperatively), the quality of life of the patients does not differ anymore from the norm population.
The modified obstipation score as well as the “cele score” by D’Hoore showed impressive improvements. The Wexner incontinence score did not show a significant improvement in our cohort. This is well explicable as incontinence was not the major complaint of the women operated on as reflected in a relative low preoperative Wexner score value. Predominant symptom of our patients with complex pelvic floor dysfunction was obstructed defecation. The impressive change of the scores shows that ventral mesh rectopexy can improve quality of life in most of these patients.
The etiology as well as the ideal treatment of obstructed defecation remains controversial. While some consider intussusception as a precursor of external rectal prolapse, others regard the pathologies as separate ones. If intussusception is indeed the precursor of external prolapse, treatment obviously should be identical for both conditions [11]. However, this may not address the other potential morphological causes (e.g. rectocele, enterocele) of obstructed defecation adequately.
In the literature, the indication for ventral mesh rectopexy is inconsistent. While in some studies intussusception served as indication [12–14], others made the indication by the complaints of obstructive defecation [15]. In this latter study, a rectocele was present in all patients, in most of them an enterocele too, and only one had had an internal prolapse. The distribution of obstruction and incontinence was similar to our study while the former studies report about a higher percentage of significant complaints about incontinence [12–14]. Other studies performed a ventral mesh rectopexy because of complex rectocele or obstructive defecation [16, 17], and did not even mention the rate of incontinence before surgery but focused exclusively on the symptoms of obstipation.
Obstipation after resection rectopexy—as an alternative procedure for outlet obstruction—improves in 53–84 % of the patients [18, 19]. However, the comparison of functional results is difficult, as the published results are often not stratified by indication and the used scores differ between the publications [19, 20]. The functional results after transanal repair by the STARR procedure seem to be comparable to those after ventral mesh rectopexy [6, 21]. However, samples sizes are small, the quality of the studies suboptimal, and long-term results rare. Moreover, both alternative procedures—resection rectopexy as well as transanal repair by STARR—are undertaken in obstructive defecation but they are both not able to reinforce the rectovaginal septum and lift the pelvic floor like it is both done by ventral mesh fixation [22]. Besides this, most surgeons would agree that enterocele is a contraindication for STARR procedure. But even this was disagreed by some authors [23].
Special attention has to be paid to complications after D’Hoore’s procedures. Badrek-Al Amoudi published severe complications that were treated in a tertiary referral centre after ventral mesh rectopexy [24]. However, in this report, the most frequent major complication was mesh erosion in postmenopausal women, a complication that we did not had to deal with. Pain and dyspareunia after mesh rectopexy are there reported only half as often [24]. In our collective, more than half of the women showed an impairment of their sexual life after the operation, whereas fewer than 50 % stated an improvement. Comparison with the literature is difficult, as there are only few reports on sexual function of the affected patients. There are reports on improvement of sexual function after laparoscopic ventral mesh rectopexy [7]. However, in this study, 18 of 22 preoperatively sexually active women remained so after surgery but only 12 of them could be evaluated after surgery by a questionnaire [7, 16]. This inadequate follow-up results in a high risk of selection bias and reflects the problem of analysing sexual function, as especially female patients are often reluctant to answer specific questions in this field due to shame. Either way, the number of female patients unpleasant with their sexual lifer after mesh implantation is worrying.
Hypothetically, the problem could be induced by the dissection of the spatium rectovaginale and consecutive fibrosis. This is in accordance with data from D’Hoore who found worsening of dyspareunia after combined laparoscopic and perineal approach for complex rectocele [10]. However, also in exclusively laparoscopic repair, the dissection to the lowest part of the pelvis is necessary to anchor the mesh there. Probably, the consequences should be pointed out more in detail to the patients. At least, we have to consider that the patients may be very young and sexual life plays a significant role. Three patients in our series were less than 30 years old, and another two were under 50. In summary, it is difficult to foresee how the sexual life of patients with functional pelvic disorders develops after mesh implantation in ventral rectopexy. Nonetheless, the relevant number of female patients in our studies stating a change for the worse in their sexual life is worrying and should result in patients being counselled on this issue.
There are some limitations in our study. First, this is a single-centre observational study. Therefore, it is difficult to generalize the results or draw conclusions in regard to other surgical methods. Second, patients filled out all questionnaires after the operation, which is a major limitation. Although we had the impression that patients remembered well their condition before the operation, also because we principally only operate patients after a long period of conservative treatment, a relevant bias cannot be ruled out.
The low rate of recurrence observed in our study also needs to be seen critically as the follow-up was short. In order to evaluate the “real” recurrence rate after such a procedure, a longer follow-up time of at least 5 years is necessary, especially when wanting to compare it to other competing procedures such as resection rectopexy. In this context also, the rare but potentially severe complications of mesh implantation in the pelvis such as mesh erosion into the rectum or vagina have to be discussed [24, 25]. These complications may occur after a long period of time, which is why the FDA has already published on the risks of mesh implantation in the pelvis. Biological meshes may be an alternative here, but comparative data is still lacking.
In a recently published critical appraisal of the increasing practice of laparoscopic ventral mesh rectopexy based on low-level evidence, the authors argue that high-level evidence needs to be generated and attention in further studies should focus not only on restoration of bowel function but also on sexual problems [11]. This is a justifiable request; however, although randomized controlled trials are of great importance for defining the role of new procedures, they are difficult to perform. Surgeons are often reluctant to randomize patients, as they seem confident to know which treatment is optimal for their patient, resulting in many important studies failing to reach their recruitment goal [26]. Therefore, single-centre observational studies as the presented one often remain the only available basis for evaluation of a new method.
In conclusion, the improvement in obstipation and quality of life after ventral mesh rectopexy for obstructive defecation is encouraging. However, the impact on sexual life differs; some patients improve but a relevant number has a change for the worse. The recurrence rate after 2 years is low, but longer follow-up is mandatory.
References
DeLancey JO (1999) Structural anatomy of the posterior pelvic compartment as it relates to rectocele. Am J Obstet Gynecol 180(4):815–823
Schafer AO, Burk J, Baumann T, Langer M (2012) MR defaecography for the diagnosis of obstructive defaecation disorders. Zentralbl Chir 137(4):352–356. doi:10.1055/s-0032-1315103
Shorvon PJ, McHugh S, Diamant NE, Somers S, Stevenson GW (1989) Defecography in normal volunteers: results and implications. Gut 30(12):1737–1749
Zbar AP, Lienemann A, Fritsch H, Beer-Gabel M, Pescatori M (2003) Rectocele: pathogenesis and surgical management. Int J Colorectal Dis 18(5):369–384. doi:10.1007/s00384-003-0478-z
Gagliardi G, Pescatori M, Altomare DF, Binda GA, Bottini C, Dodi G, Filingeri V, Milito G, Rinaldi M, Romano G, Spazzafumo L, Trompetto M, Italian Society of Colo-Rectal S (2008) Results, outcome predictors, and complications after stapled transanal rectal resection for obstructed defecation. Dis Colon Rectum 51(2):186–195. doi:10.1007/s10350-007-9096-0, discussion 195
Thornton MJ, Lam A, King DW (2005) Laparoscopic or transanal repair of rectocele? A retrospective matched cohort study. Dis Colon Rectum 48(4):792–798. doi:10.1007/s10350-004-0843-1
Abet E, Lehur PA, Wong M, Rigaud J, Darnis E, Meurette G (2012) Sexual function and laparoscopic ventral rectopexy for complex rectocoele. Colorectal Dis 14(10):e721–e726. doi:10.1111/j.1463-1318.2012.03113.x
D’Hoore A, Cadoni R, Penninckx F (2004) Long-term outcome of laparoscopic ventral rectopexy for total rectal prolapse. Br J Surg 91(11):1500–1505. doi:10.1002/bjs.4779
Braun M (2005) Evaluierung eines Scores zur Unterscheidung verschiedener Obstipationsformen (hier: Outlet-Obstruction und Slow-Transit-Obstipation). Karl-Ruprechts-Universität Heidelberg, Heidelberg
D’Hoore A, Vanbeckevoort D, Penninckx F (2008) Clinical, physiological and radiological assessment of rectovaginal septum reinforcement with mesh for complex rectocele. Br J Surg 95(10):1264–1272. doi:10.1002/bjs.6322
Lundby L, Laurberg S (2014) Laparoscopic ventral mesh rectopexy for ODS—time for a critical appraisal. Colorectal Dis. doi:10.1111/codi.12830
Collinson R, Wijffels N, Cunningham C, Lindsey I (2010) Laparoscopic ventral rectopexy for internal rectal prolapse: short-term functional results. Colorectal Dis 12(2):97–104. doi:10.1111/j.1463-1318.2009.02049.x, CDI2049 [pii]
Portier G, Kirzin S, Cabarrot P, Queralto M, Lazorthes F (2011) The effect of abdominal ventral rectopexy on faecal incontinence and constipation in patients with internal intra-anal rectal intussusception. Colorectal Dis 13(8):914–917. doi:10.1111/j.1463-1318.2010.02327.x, CDI2327 [pii]
Slawik S, Soulsby R, Carter H, Payne H, Dixon AR (2008) Laparoscopic ventral rectopexy, posterior colporrhaphy and vaginal sacrocolpopexy for the treatment of recto-genital prolapse and mechanical outlet obstruction. Colorectal Dis 10(2):138–143. doi:10.1111/j.1463-1318.2007.01259.x, CDI1259 [pii]
van den Esschert JW, Van Geloven AA, Vermulst N, Groenedijk AG, De Wit LT, Gerhards MF (2008) Laparoscopic ventral rectopexy for obstructed defecation syndrome. Surg Endosc 22(12):2728–2732. doi:10.1007/s00464-008-9771-9
Wong MT, Abet E, Rigaud J, Frampas E, Lehur PA, Meurette G (2011) Minimally invasive ventral mesh rectopexy for complex rectocoele: impact on anorectal and sexual function. Colorectal Dis 13(10):e320–e326. doi:10.1111/j.1463-1318.2011.02688.x
Oom DM, Gosselink MP, Van Wijk JJ, Van Dijl VR, Schouten WR (2008) Rectocele repair by anterolateral rectopexy: long-term functional outcome. Colorectal Dis 10(9):925–930. doi:10.1111/j.1463-1318.2008.01538.x, CDI1538 [pii]
Von Papen M, Ashari LH, Lumley JW, Stevenson AR, Stitz RW (2007) Functional results of laparoscopic resection rectopexy for symptomatic rectal intussusception. Dis Colon Rectum 50(1):50–55. doi:10.1007/s10350-006-0781-1
Laubert T, Kleemann M, Roblick UJ, Burk C, Schorcht A, Hildebrand P, Bruch HP (2012) Laparoscopic resection rectopexy as treatment for obstructive defecation syndrome. Zentralbl Chir 137(4):357–363. doi:10.1055/s-0032-1315125
Kellokumpu IH, Vironen J, Scheinin T (2000) Laparoscopic repair of rectal prolapse: a prospective study evaluating surgical outcome and changes in symptoms and bowel function. Surg Endosc 14(7):634–640. doi:10.1007/s004640000017 [pii]
Borie F, Bigourdan JM, Pissas MH, Guillon F (2014) Laparoscopic ventral rectopexy for the treatment of outlet obstruction associated with recto-anal intussusception and rectocele: a valid alternative to STARR procedure in patients with anal sphincter weakness. Clinics and research in hepatology and gastroenterology 38(4):528–534. doi:10.1016/j.clinre.2013.12.010
Festen S, Van Geloven AA, D’Hoore A, Lindsey I, Gerhards MF (2011) Controversy in the treatment of symptomatic internal rectal prolapse: suspension or resection? Surg Endosc 25(6):2000–2003. doi:10.1007/s00464-010-1501-4
Reibetanz J, Boenicke L, Kim M, Germer CT, Isbert C (2011) Enterocele is not a contraindication to stapled transanal surgery for outlet obstruction: an analysis of 170 patients. Colorectal Dis 13(6):e131–e136. doi:10.1111/j.1463-1318.2011.02554.x
Badrek-Al Amoudi AH, Greenslade GL, Dixon AR (2013) How to deal with complications after laparoscopic ventral mesh rectopexy: lessons learnt from a tertiary referral centre. Colorectal Dis 15(6):707–712. doi:10.1111/codi.12164
Van Geluwe B, Wolthuis A, Penninckx F, D’Hoore A (2013) Lessons learned after more than 400 laparoscopic ventral rectopexies. Acta Chir Belg 113(2):103–106
Senapati A, Gray RG, Middleton LJ, Harding J, Hills RK, Armitage NC, Buckley L, Northover JM, Group PC (2013) PROSPER: a randomised comparison of surgical treatments for rectal prolapse. Colorectal Dis 15(7):858–868. doi:10.1111/codi.12177
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Horisberger, K., Rickert, A., Templin, S. et al. Laparoscopic ventral mesh rectopexy in complex pelvic floor disorder. Int J Colorectal Dis 31, 991–996 (2016). https://doi.org/10.1007/s00384-016-2545-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-016-2545-2