Abstract
Background
Stricturing Crohn’s disease is accompanied by a high-risk for bowel resection and subsequent short bowel syndrome. Strictureplasty (SP) and endoscopic balloon dilatation (EBD) have been developed to prevent, or at least delay, the requirement for resection. The goal of this study was to compare the outcome of these two procedures with regard to complications and disease recurrence.
Methods
We conducted a MEDLINE literature search to give a current overview about the safety and efficacy of EBD and SP.
Results
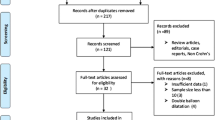
The initial search yielded 744 articles. Case reports, reviews and meta-analyses were excluded. Finally, 63 articles (SP, 40 articles; EBD, 23 articles) were used for the review. None of the studies compared the two methods directly. A total of 2,532 patients (SP, n = 1,958; EBD, n = 574) were included. The incidence of perioperative complications after SP was 11% and the incidence of major complications was 5%. The median surgical recurrence rate was 24% after a median follow-up of 46 months. The median technical success for EBD was 90%. Major complications occurred in 3% of the cases. According to an intention-to-treat protocol, the median surgical recurrence rate was 27.6%. Per-protocol analysis revealed a median surgical recurrence rate of 21.4% after a median follow-up of 21 months.
Conclusion
Due to the lack of comparable data, there is currently no reliable information on whether one treatment option is superior to the other. Regarding the limited applicability of EBD in strictures of the small bowel, only a controlled trial would provide evidence as a basis for clinical decision making in CD strictures that are potentially treatable by EBD and SP.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Stricturing Crohn’s disease (CD) is accompanied by a high rate of surgical bowel resection with a risk of short bowel syndrome. Therefore, strictureplasty (SP) and endoscopic balloon dilatation (EBD) have been applied to avoid, or at least delay, the need for bowel resection. This MEDLINE review gives an overview of the epidemiology as well as the natural and therapeutical course of stricturing CD and about the safety and efficacy of SP and EBD.
Epidemiology of Crohn’s disease
Crohn’s disease is a relapsing, transmural inflammatory disease of the gastrointestinal tract with an incidence of up to 20.2 cases per 100,000 persons per year in Western societies [1, 2]. The European Collaborative Study on Inflammatory Bowel Disease (EC-IBD) reported an incidence of nine cases per 100,000 persons per year in Europe [3]. Similar numbers were observed in the US (7/100,000/year) [4]. Ott et al. [5] recently reported 6.6 newly diagnosed cases per 100,000 persons per year (95% confidence interval: 5.6-7.7) in the region of Oberpfalz, Germany (1,089,000 inhabitants). The prevalence of CD in Germany is estimated to be between 1/500 and 1/800 or approximately 100,000–150,000 patients [6, 7]. Similar numbers have been reported from North America where 400,000-600,000 patients are affected by CD [8].
According to data from the EC-IBD, the highest age-specific incidence is observed between 15 and 34 years of age [3]. This means that CD starts during the educational phase and lasts through the entire work life of most of the patients. Thus, total costs of CD are composed of direct as well as indirect costs (non-productive time, sick payments, etc.). The German Evidence-based Consensus Conference on CD has estimated an annual cost of 3 billion Euros for treating CD patients in Germany [9].
Natural course of Crohn’s disease
CD can affect the entire gastrointestinal tract from the mouth to the anus and the localization as well as the clinical behavior of CD (stricturing, penetrating as well as non-stricturing, non-penetrating type) can change over time. The anatomical localization of CD and its behavior over time were analyzed in 297 patients regularly followed-up at the University Hospital of Liège, Belgium [10]. The authors did a 10-year evaluation (n = 125) of CD localization and CD behavior according to the Vienna Classification System [11]. Affection of the colon and upper gastrointestinal tract were stable over the time while affection of the ileal region decreased and affection of the ileocolonic segment increased during the observation period. After 10 years of CD history, 43.3% suffered from CD in the terminal ileum, 30% from ileocolonic, 23.3% from colonic disease, and 3.3% from upper gastrointestinal affection. CD behavior also changed over time: at the time of diagnosis the majority of patients (73.7%) suffered from non-stricturing, non-penetrating disease. After 10 years, 37.2% suffered from penetrating and 27.1% from stricturing CD. The vast majority of Crohn strictures (98.8%) were found in the colon, ileocolonic region, and ileum after 10 years.
These observations were confirmed by a prospective study from Oslo, Norway, with 197 CD patients completing a 10-year follow-up. A total of 53% of patients had developed stricturing or penetrating CD after 10 years [12]. This means that stricturing CD is relatively uncommon at the time of diagnosis but will become more likely over the years. This phenomenon is probably due to the chronic inflammation in CD, which is accompanied by cell death, consecutive scarring [13], and contraction of the scar.
The need for intervention in stricturing Crohn’s disease
An analysis of 2,002 CD patients from Paris, France showed that 13% of them suffered from stricturing CD behavior at the time of diagnosis [14]. Of these patients, 646 were enrolled in a prospective follow-up study in 1995 with an observation period until 2000. Seventy-nine percent of the patients with stricturing behavior of CD had already undergone surgery prior to their inclusion in the follow-up study. Twenty-two percent underwent (further) surgical intervention. This observation was verified by a population-based study from Oslo, Norway [12]: The authors observed a 64% cumulative rate of surgery in patients with stricturing CD after 10 years, and stricturing CD behavior was an independent risk factor for the need of surgery. These data indicate that stricturing CD leads to the need for intervention in the majority of patients.
The impact of immunosuppressants on the need for intervention
The impact of modern immunosuppressants on the need for surgery in CD was evaluated by a French group in 2005 [15]. This work shows that there was a significant increase in the use of immunosuppressive drugs after 1982 and 1998. The absolute number of intestinal resections and the percentage of CD patients with a need for resection did not change during the same observation period. Although immunosuppressants had been used more frequently over the last 25 years, there was no significant decrease in the need for surgery. Recent evidence seems to support the hypothesis that medical treatment [16] and cessation of smoking [17] after surgery help to avoid a surgical recurrence of CD.
Material and methods
We performed a MEDLINE review of the German and English literature on SP and EBD in CD from 1980 to 2009. The search term was ‘crohn AND strictur*’ in order to increase sensitivity of the search. The initial search yielded 744 articles. Case reports, reviews, and meta-analyses were excluded. Finally, 40 papers dealing with SP and 23 papers dealing with EBD were used for the review.
Treatment options for stricturing Crohn’s disease
Surgical resection
The traditional surgical strategy for structuring disease is resection of the affected segment. In the early 1980s, several groups found that the presence or absence of microscopic disease at the resection margins of CD specimens did not influence the probability of recurrence [18, 19]. This finding was affirmed by a randomized, controlled trial in 1996 [20], where patients with CD were randomized to either ‘limited resection’ (2 cm resection margins) or ‘extended resection’ (12 cm resection margins). The probability of recurrence-free survival did not differ between the two groups. Extensive resection of the terminal ileum, which is the most frequently affected region in CD, may lead to short bowel syndrome, as the remaining intestine is not able to compensate the absorption of vitamins B12, A, D, E, or Kor bile salts. Thus, the surgical strategy in CD changed towards ‘bowel-sparing’ resections. Nevertheless, recurrence rates 15 years after first surgery are reported to be between 28% and 45% [21]. The type of anastomosis (end-to-end vs. side-to-side) also does not seem to influence the probability of recurrence, as recently shown in a randomized, controlled trial (n = 139): After a mean follow-up of 11.9 months, the symptomatic recurrence rate was 21.9% in the end-to-end anastomosis group compared to 22.7% in the side-to-side anastomosis group (p = 0.92) [22].
It was thus reasonable to search for alternatives to bowel resection in stricturing CD. The development of SP, which will be described in detail in the next chapter, provided an opportunity to widen CD strictures without bowel resection.
According to the German Clinical Practice Guideline on Diagnosis and Treatment of Crohn’s Disease [9], bowel resection in stricturing CD is only indicated in case of non-feasibility of SP or EBD. The European evidence-based consensus on the diagnosis and management of Crohn’s disease recommends conventional SP when the length of the stricture is <10 cm [23].
Strictureplasty—technical considerations
The Heineke-Mikulicz (H-M) pyloroplasty, named for Walter Hermann Heineke (1834-1901) and Johann von Mikulicz-Radecki (1850-1905), is the basic principle of traditional SP. It consists of a longitudinal enterotomy at the side of stricture followed by transverse closure with avoidance of bowel resection and anastomoses (Fig. 1). This is important as recurrent resection will potentially lead to short bowel syndrome, and anastomoses bear a higher risk of anastomotic leakage [24].
Originally strictureplasty was applied as pyloroplasty of the stomach, done to enlarge the outlet of the stomach and make it non-functional. In 1977, Katariya et al. [25] published their results of strictureplasties performed in nine patients suffering from multiple tubercular strictures in the lower gastrointestinal tract. In 1982, Lee and Papaioannou [26] published nine cases of H-M strictureplasties for CD of the small intestine. Since then, H-M strictureplasty has evolved as the most frequently performed SP for short-segment stenosis in CD. In a meta-analysis from 2007, 81% (2,499/3,259) of all strictureplasties were H-M procedures [27]. As an alternative surgical strategy, the Judd procedure is a modification of the H-M strictureplasty for fistulas arising from the strictured segment with elliptical excision of the fistula and transverse closure as in H-M strictureplasty. The Moskel-Walske-Neumayer strictureplasty is applied in cases of severe prestenotic small bowel dilatation. It consists of a Y-shaped enterotomy, advancement of the proximal intestinal wall over the stricture and final transverse closure. The Finney procedure, Jaboulay procedure, and the Michelassi side-to-side isoperistaltic strictureplasty’ involve a side-to-side enterostomy and are favored for longer stenoses of up to 25-90 cm.
Strictureplasty—evaluation of safety and efficacy
To evaluate the safety and efficacy of SP in CD, we performed a MEDLINE review on the existing German and English literature. Table 1 gives an overview of the retrieved studies. There were reports of 5,896 strictureplasties in 1,958 patients. We limited the statistical analysis to the calculation of medians and means and their comparison without hypothesis tests, as the heterogeneity of the retrieved studies does not allow any meta-analysis. To assess the safety of SP, we noted the frequency of perioperative complications. The median incidence of perioperative complications was 11% (mean 11.2%). The median incidence of major complications, i.e., anastomotic leakage, intraabdominal abscess, fistulas, sepsis, hemorrhage, ileus, or other events requiring relaparotomy was 5%; the mean major complication rate was 5.7%. To evaluate the efficacy of SP, we noted the frequency of the need for recurrent surgery after SP. We preferentially used cumulative recurrence estimates from survival analyses rather than simple frequencies for our analysis. The median surgical recurrence rate was 24% (mean: 24.0%) after a median follow-up of 46 months (mean, 57.7 months).
The major drawback of these studies is that very few authors provided information about site-specific recurrence and that many studies evaluated different techniques of SP.
Endoscopic balloon dilatation
After EBD had become an established treatment option in esophageal, gastric, colonic, and biliary tree strictures [67], R. A. Brower in 1986 was one of the first authors to report about the successful hydrostatic balloon dilatation of a terminal ileal stricture secondary to CD in a 24-year-old woman with refractory symptoms of bowel obstruction that refused surgery [68]. In the following two decades, several institutions reported their results of the endoscopic dilatation of CD strictures. EBD is normally restricted to CD strictures in the colon and terminal ileum. Newer techniques like double-balloon endoscopy are able to reach more orally localized regions of the small bowel but their application is still experimental.
To assess the safety and efficacy of EBD in CD, we performed a MEDLINE literature review and noted the frequency of complications as well as the rate of the need for re-dilatation and surgical recurrence, i.e., the need for surgery after initial EBD. We found 23 papers reporting about 1,003 treatment sessions in 574 patients on an intention-to-treat basis. Results are shown in Table 2.
Median technical success was 90% (mean, 90.3%). The median rate of major complications like bowel perforation or severe bleeding was 3% (mean, 4%). According to an intention-to-treat protocol, the median surgical recurrence rate was 27.6% (mean, 28.5%). After excluding patients in whom EBD was not possible due to technical reasons (e.g., analysis per protocol), the median surgical recurrence rate was 21.4% (mean, 21.5%) after a median follow-up of 21.0 months (mean, 27.3 months).
The drawback of these studies is that different authors applied different treatment protocols. They used different balloons with different dilatation pressures and different times of insufflation. Some used intralesional steroid injection, while others did not. As in SP, very few authors reported site-specific recurrences. Some authors reported in detail the number of dilatations per patient while others only reported if they were successful or not.
Comparison of results
Table 3 lists reported results of strictureplasty and EBD. These data can only give a rough overview due to the epistemological reasons mentioned above. Meta-analysis and direct comparison of the two methods was not possible due to differing treatment protocols. One can state that there is more literature about SP and that the median/mean length of follow-up is longer for those patients. There is no difference between the incidence of major complications after SP and EBD.
A systematic review and meta-analysis of studies describing the safety and efficacy of SP from 2000 (n = 506 patients) onward reported a pooled surgical recurrence rate of 25.5% [92]. In a meta-analysis published in 2007 (n = 1,112 patients), the pooled recurrence rate was 23.4% (95% confidence interval: 16.9-30%) [27]. These numbers are very close to the mean/median recurrence rate in the present review. For EBD in stricturing CD, there is one systematic review published in 2007 [93]. In this review, the long-term success rate of EBD is 58% with a recurrence rate of 42% after a mean follow-up of 33 months.
The need for a trial
Both methods, SP and EBD, have now been applied for more than two decades [26, 68] in parallel and have become well-established treatment options in short-segment stricturing CD [9]. Unfortunately, there is no clinical study or even a multicenter, randomized, and controlled study that directly compares their efficacy and safety. Clinical reports about EBD exclusively derive from internal departments while results of SP are only investigated by surgeons. Internists have proven EBD to be safe and effective while surgeons did so for SP, and both seem to have good reasons for applying their method.
There is no valid information at all as to whether one treatment option is superior to the other with regard to the applicability of both. Assignment to one of the treatment options strongly depends on whether the patient is primarily seen at an internal/endoscopic or surgical department. A multicenter, randomized controlled trial would provide evidence as a basis for clinical decision making.
References
Bernstein CN, Blanchard JF, Rawsthorne P et al (2006) The epidemiology of inflammatory bowel disease in Canada: a population-based study. Am J Gastroenterol 101:1559–1568
Pavlovic-Calic N, Salkic NN, Gegic A, Smajic M, Alibegovic E (2008) Crohn’s disease in Tuzla region of Bosnia and Herzegovina: a 12-year study (1995-2006). Int J Colorectal Dis 23:957–964
Shivananda S, Lennard-Jones J, Logan R, Fear N, Price A, Carpenter L, van Blankenstein M (1996) Incidence of inflammatory bowel disease across Europe: is there a difference between north and south? Results of the European Collaborative Study on Inflammatory Bowel Disease. Gut 39:690–697
Jess T, Loftus EV Jr, Harmsen WS, Zinsmeister AR, Tremaine WJ, Melton LJ 3rd, Munkholm P, Sandborn WJ (2006) Survival and cause specific mortality in patients with inflammatory bowel disease: a long-term outcome study in Olmsted County, Minnesota, 1940-2004. Gut 55:1248–1254
Ott C, Obermeier F, Thieler S, Kemptner D, Bauer A, Schölmerich J, Rogler G, Timmer A (2008) The incidence of inflammatory bowel disease in a rural region of Southern Germany: a prospective population-based study. Eur J Gastroenterol Hepatol 20:917–923
Stange EF, Schreiber S, Fölsch UR, von Herbay A, Schölmerich J, Hoffmann J, Zeitz M, Fleig WE, Buhr HJ, Kroesen AJ, Moser G, Matthes H, Adler G, Reinshagen M, Stein J, German Society for Digestive and Metabolic Diseases (2003) Diagnostics and treatment of Crohn’s disease—results of an evidence-based consensus conference of the German Society for Digestive and Metabolic Diseases. Z Gastroenterol 41:19–20
Timmer A, Breuer-Katschinski B, Goebell H (1999) Time trends in the incidence and disease location of Crohn’s disease 1980-1995: a prospective analysis in an urban population in Germany. Inflamm Bowel Dis 5:79–84
Loftus EV, Schoenfeld P, Sandborn WJ (2002) The epidemiology and natural history of Crohn’s disease in population-based patient cohorts from North America: a systematic review. Aliment Pharmacol Therap 16:51–60
Hoffmann JC, Preiss JC, Autschbach F, Buhr HJ, Häuser W, Herrlinger K, Höhne W, Koletzko S, Krieglstein CF, Kruis W, Matthes H, Moser G, Reinshagen M, Rogler G, Schreiber S, Schreyer AG, Sido B, Siegmund B, Stallmach A, Bokemeyer B, Stange EF, Zeitz M (2008) Clinical practice guideline on diagnosis and treatment of Crohn’s disease. Z Gastroenterol 46:1094–1146
Louis E, Collard A, Oger AF, Degroote E, Aboul Nasr El Yafi FA, Belaiche J (2001) Behaviour of Crohn’s disease according to the Vienna classification: changing pattern over the course of the disease. Gut 49:777–782
Gasche C, Schölmerich J, Brynskov J, D’Haens G, Hanauer SB, Irvine EJ (2002) A simple classification of Crohn’s disease: report of Working Party for the World Congress of Gastroenterology, Vienna 1998. Inflamm Bowel Dis 6:8–15
Solberg IC, Vatn MH, Høie O, Stray N, Sauar J, Jahnsen J, Moum B, Lygren I, IBSEN Study Group (2007) Clinical course in Crohn’s disease: results of a Norwegian population-based ten-year follow-up study. Clin Gastroenterol Hepatol 5:1430–1438
Brenmoehl J, Falk W, Göke M, Schölmerich J, Rogler G (2008) Inflammation modulates fibronectin isoform expression in colonic lamina propria fibroblasts (CLPF). Int J Colorectal Dis 23:947–955
Cosnes J, Cattan S, Blain A, Beaugerie L, Carbonnel F, Parc R, Gendre JP (2002) Long-term evolution of disease behavior of Crohn’s disease. Inflamm Bowel Dis 8:244–250
Cosnes J, Nion-Larmurier I, Beaugerie L, Afchain P, Tiret E, Gendre JP (2005) Impact of the increasing use of immunosuppressants in Crohn’s disease on the need for intestinal surgery. Gut 54:237–241
Cao Y, Gao F, Liao C, Tan A, Mo Z (2009) Meta-analysis of medical treatment and placebo treatment for preventing postoperative recurrence in Crohn’s disease (CD). Int J Colorectal Dis 24:509–520
Reese GE, Nanidis T, Borysiewicz C, Yamamoto T, Orchard T, Tekkis PP (2008) The effect of smoking after surgery for Crohn’s disease: a meta-analysis of observational studies. Int J Colorectal Dis 23:1213–1221
Heuman R, Boeryd B, Bolin T, Sjödahl R (1983) The influence of disease at the margin of resection on the outcome of Crohn’s disease. Br J Surg 70:519–521
Pennington L, Hamilton SR, Bayless TM, Cameron JL (1980) Surgical management of Crohn’s disease. Influence of disease at margin of resection. Ann Surg 192:311–318
Fazio VW, Marchetti F, Church M, Goldblum JR, Lavery C, Hull TL, Milsom JW, Strong SA, Oakley JR, Secic M (1996) Effect of resection margins on the recurrence of Crohn’s disease in the small bowel. A randomized controlled trial. Ann Surg 224:563–571
Wolters FL, Russel MG, Stockbrügger RW (2004) Systematic review: has disease outcome in Crohn’s disease changed during the last four decades? Aliment Pharmacol Ther 20:483–496
McLeod RS, Wolff BG, Ross S, Parkes R, McKenzie M, Investigators of the CAST Trial (2009) Recurrence of Crohn’s disease after ileocolic resection is not affected by anastomotic type: results of a multicenter, randomized, controlled trial. Dis Colon Rectum 52:919–927
Travis SP, Stange EF, Lémann M, Oresland T, Chowers Y, Forbes A, D’Haens G, Kitis G, Cortot A, Prantera C, Marteau P, Colombel JF, Gionchetti P, Bouhnik Y, Tiret E, Kroesen J, Starlinger M, Mortensen NJ, European Crohn’s and Colitis Organisation (2006) European evidence based consensus on the diagnosis and management of Crohn’s disease: current management. Gut 55(Suppl 1):i16–i35
Iesalnieks I, Kilger A, Glass H, Müller-Wille R, Klebl F, Ott C, Strauch U, Piso P, Schlitt HJ, Agha A (2008) Intraabdominal septic complications following bowel resection for Crohn’s disease: detrimental influence on long-term outcome. Int J Colorectal Dis 23:1167–1174
Katariya RN, Sood S, Rao PG, Rao PL (1977) Stricture-plasty for tubercular strictures of the gastro-intestinal tract. Br J Surg 64:496–498
Lee EC, Papaioannou N (1982) Minimal surgery for chronic obstruction in patients with extensive or universal Crohn’s disease. Ann R Coll Surg Engl 64:229–233
Yamamoto T, Fazio VW, Tekkis PP (2007) Safety and efficacy of strictureplasty for Crohn’s disease: a systematic review and meta-analysis. Dis Colon Rectum 50:1968–1986
Yuan JG, Sachar DB, Koganei K, Greenstein AJ (1994) Enterolithiasis, refractory anemia, and strictures of Crohn’s disease. J Clin Gastroenterol 18:105–108
Michelassi F (1996) Side-to-side isoperistaltic strictureplasty for multiple Crohn’s strictures. Dis Colon Rectum 39:345–349
Poggioli G, Stocchi L, Laureti S et al (1997) Conservative surgical management of terminal ileitis: side-to-side enterocolic anastomosis. Dis Colon Rectum 40:234–239
Di Abriola GF, De Angelis P, Dall’oglio L, Di Lorenzo M (2003) Strictureplasty: an alternative approach in long segment bowel stenosis Crohn’s disease. J Pediatr Surg 38:814–818
Gaetini A, DeSimone M, Resegotti A (1989) Our experience with strictureplasty in the surgical treatment of Crohn’s disease. Hepatogastroenterology 36:511–515
Sasaki I, Shibata C, Funayama Y et al (2004) New reconstructive procedure after intestinal resection for Crohn’s disease: modified side-to-side isoperistaltic anastomosis with double Heineke-Mikulicz procedure. Dis Colon Rectum 47:940–943
Oliva L, Wyllie R, Alexander F et al (1994) The results of strictureplasty in pediatric patients with multifocal Crohn’s disease. J Pediatr Gastroenterol Nutr 18:306–310
Kendall GP, Hawley PR, Nicholls RJ, Lennard-Jones JE (1986) Strictureplasty. A good operation for small bowel Crohn’s disease? Dis Colon Rectum 29:312–316
Pritchard TJ, Schoetz DJ Jr, Caushaj FP et al (1990) Strictureplasty of the small bowel in patients with Crohn’s disease. An effective surgical option. Arch Surg 125:715–717
Yamamoto T, Bain IM, Connolly AB, Allan RN, Keighley MR (1999) Outcome of strictureplasty for duodenal Crohn’s disease. Br J Surg 86:259–262
Worsey MJ, Hull T, Ryland L, Fazio V (1999) Strictureplasty is an effective option in the operative management of duodenal Crohn’s disease. Dis Colon Rectum 42:596–600
Roy P, Kumar D (2006) Strictureplasty for active Crohn’s disease. Int J Colorectal Dis 21:427–432
Silverman RE, McLeod RS, Cohen Z (1989) Strictureplasty in Crohn’s disease. Can Assoc Clin Surg 32:19–22
Rentsch M, Beham A, Schlitt HJ, Jauch KW (2006) Crohn’s disease activity index and Vienna classification—is it worthwhile to calculate before surgery? Dig Surg 23:241–249
Laurent S, Detry O, Detroz B et al (2002) Strictureplasty in Crohn’s disease: short- and long-term follow-up. Acta Chir Belg 102:253–255
Keighley MR (1991) Stapled strictureplasty for Crohn’s disease. Dis Colon Rectum 34:945–947
Fichera A, Lovadina S, Rubin M, Cimino F, Hurst RD, Michelassi F (2006) Patterns and operative treatment of recurrent Crohn’s disease: a prospective longitudinal study. Surgery 140:649–654
Quandalle P, Gambiez L, Colombel JF, Paris JC, Cortot A (1994) Long-term follow-up of strictureplasty in Crohn’s disease. Acta Gastroenterol Belg 57:314–319
Roy P, Kumar D (2006) Intervention-free interval following strictureplasty for Crohn’s Disease. World J Surg 30:1020–1026
Broering DC, Eisenberger CF, Koch A et al (2001) Strictureplasty for large bowel stenosis in Crohn’s disease: quality of life after surgical therapy. Int J Colorectal Dis 16:81–87
Tonelli F, Fedi M, Paroli GM, Fazi M (2004) Indications and results of side-to-side isoperistaltic strictureplasty in Crohn’s disease. Dis Colon Rectum 47:494–501
Dasari BV, Maxwell R, Gardiner KR (2010) Assessment of complications following strictureplasty for small bowel Crohn’s Disease. Ir J Med Sci 179:201–205
Spencer MP, Nelson H, Wolff BG, Dozois RR (1994) Strictureplasty for obstructive Crohn’s disease: the Mayo experience. Mayo Clin Proc 69:33–36
Yamamoto T, Keighley MRB (1999) Follow up of more than 10 years after strictureplasty for jejunileal Crohn’s disease: long-term results and predictive factors for outcome. Colorectal Dis 1:101–106
Serra J, Cohen Z, McLeod RS (1995) Natural history of strictureplasty in Crohn’s disease: 9-year experience. Can J Surg 38:481–485
Tonelli F, Ficari F (2000) Strictureplasty in Crohn’s disease: surgical option. Dis Colon Rectum 43:920–926
Cristaldi M, Sampietro GM, Danelli PG, Bollani S, Bianchi Porro G, Taschieri AM (2000) Long-term results and multivariate analysis of prognostic factors in 138 consecutive patients operated on for Crohn’s disease using “bowel-sparing” techniques. Am J Surg 179:266–270
Stebbing JF, Jewell DP, Kettlewell MG, Mortensen NJ (1995) Recurrence and reoperation after strictureplasty for obstructive Crohn’s disease: long-term results [corrected]. Br J Surg 82:1471–1474, Erratum in. Br J Surg 1996;83:131
Hurst RD, Michelassi F (1998) Strictureplasty for Crohn’s disease: techniques and long-term results. World J Surg 22:359–363
Shatari T, Clark MA, Yamamoto T, Menon A, Keh C, Alexander-Williams J, Keighley M (2004) Long strictureplasty is as safe and effective as short strictureplasty in small-bowel Crohn’s disease. Colorectal Dis 6:438–441
Baba S, Nakai K (1995) Strictureplasty for Crohn’s disease in Japan. J Gastroenterol 30(Suppl 8):135–138
Greenstein AJ, Zhang LP, Miller AT, Yung E, Branco BC, Sachar DB, Greenstein AJ (2009) Relationship of the number of Crohn’s strictures and strictureplasties to postoperative recurrence. J Am Coll Surg 208:1065–1070
Fearnhead NS, Chowdhury R, Box B, George BD, Jewell DP, Mortensen NJ (2006) Long-term follow-up of strictureplasty for Crohn’s disease. Br J Surg 93:475–482
Sampietro GM, Cristaldi M, Maconi G et al (2004) A prospective, longitudinal study of nonconventional strictureplasty in Crohn’s disease. J Am Coll Surg 199:82
Futami K, Arima S (2005) Role of strictureplasty in surgical treatment of Crohn’s disease. J Gastroenterol 40(Suppl 16):35–39
Yamamoto T, Bain IM, Allan RN, Keighley MR (1999) An audit of strictureplasty for small-bowel Crohn’s disease. Dis Colon Rectum 42:797–803
Ozuner G, Fazio VW, Lavery IC, Church JM, Hull TL (1996) How safe is strictureplasty in the management of Crohn’s disease? Am J Surg 171:57–61
Michelassi F, Taschieri A, Tonelli F, Sasaki I, Poggioli G, Fazio V, Upadhyay G, Hurst R, Sampietro GM, Fazi M, Funayama Y, Pierangeli F (2007) An international, multicenter, prospective, observational study of the side-to-side isoperistaltic strictureplasty in Crohn’s disease. Dis Colon Rectum 50:277–284
Dietz DW, Laureti S, Strong SA et al (2001) Safety and longterm efficacy of strictureplasty in 314 patients with obstructing small bowel Crohn’s disease. J Am Coll Surg 192:330–338
Kozarek RA (1986) Hydrostatic balloon dilation of gastrointestinal stenoses: a national survey. Gastrointest Endosc 32:15–19
Brower RA (1986) Hydrostatic balloon dilation of a terminal ileal stricture secondary to Crohn’s disease. Gastrointest Endosc 32:38–40
Matsui T, Hatakeyama S, Ikeda K, Yao T, Takenaka K, Sakurai T (1997) Long-term outcome of endoscopic balloon dilation in obstructive gastroduodenal Crohn’s disease. Endoscopy 29:640–645
Williams AJ, Palmer KR (1991) Endoscopic balloon dilatation as a therapeutic option in the management of intestinal strictures resulting from Crohn’s disease. Br J Surg 78:453–454
Tsujikawa T, Saitoh Y, Andoh A, Imaeda H, Hata K, Minematsu H, Senoh K, Hayafuji K, Ogawa A, Nakahara T, Sasaki M, Fujiyama Y (2008) Novel single-balloon enteroscopy for diagnosis and treatment of the small intestine: preliminary experiences. Endoscopy 40:11–15
Junge U, Züchner H (1994) Endoscopic balloon dilatation of symptomatic strictures in Crohn’s disease. Dtsch Med Wochenschr 119:1377–1382
Pohl J, May A, Nachbar L, Ell C (2007) Diagnostic and therapeutic yield of push-and-pull enteroscopy for symptomatic small bowel Crohn’s disease strictures. Eur J Gastroenterol Hepatol 19:529–534
Ramboer C, Verhamme M, Dhondt E, Huys S, Van Eygen K, Vermeire L (1995) Endoscopic treatment of stenosis in recurrent Crohn’s disease with balloon dilation combined with local corticosteroid injection. Gastrointest Endosc 42:252–255
East JE, Brooker JC, Rutter MD, Saunders BP (2007) A pilot study of intrastricture steroid versus placebo injection after balloon dilatation of Crohn’s strictures. Clin Gastroenterol Hepatol 5:1065–1069
Brooker JC, Beckett CG, Saunders BP, Benson MJ (2003) Long-acting steroid injection after endoscopic dilation of anastomotic Crohn’s strictures may improve the outcome: a retrospective case series. Endoscopy 35:333–337
Singh VV, Draganov P, Valentine J (2005) Efficacy and safety of endoscopic balloon dilation of symptomatic upper and lower gastrointestinal Crohn’s disease strictures. J Clin Gastroenterol 39:284–290
Breysem Y, Janssens JF, Coremans G, Vantrappen G, Hendrickx G, Rutgeerts P (1992) Endoscopic balloon dilation of colonic and ileo-colonic Crohn’s strictures: long-term results. Gastrointest Endosc 38:142–147
Foster EN, Quiros JA, Prindiville TP (2008) Long-term follow-up of the endoscopic treatment of strictures in pediatric and adult patients with inflammatory bowel disease. J Clin Gastroenterol 42:880–885
Dear KL, Hunter JO (2001) Colonoscopic hydrostatic balloon dilatation of Crohn’s strictures. J Clin Gastroenterol 33:315–318
Fukumoto A, Tanaka S, Yamamoto H, Yao T, Matsui T, Iida M, Goto H, Sakamoto C, Chiba T, Sugano K (2007) Diagnosis and treatment of small-bowel stricture by double balloon endoscopy. Gastrointest Endosc 66:S108–S112
Stienecker K, Gleichmann D, Neumayer U, Glaser HJ, Tonus C (2009) Long-term results of endoscopic balloon dilatation of lower gastrointestinal tract strictures in Crohn’s disease: a prospective study. World J Gastroenterol 15:2623–2627
Hoffmann JC, Heller F, Faiss S, von Lampe B, Kroesen AJ, Wahnschaffe U, Schulzke JD, Zeitz M, Bojarski C (2008) Through the endoscope balloon dilation of ileocolonic strictures: prognostic factors, complications, and effectiveness. Int J Colorectal Dis 23:689–696
Blomberg B, Rolny P, Jarnerot G (1991) Endoscopic treatment of anastomotic strictures in Crohn’s disease. Endoscopy 23:195–198
Raedler A, Peters I, Schreiber S (1997) Treatment with azathioprine and budesonide prevents reoccurrence of ileocolonic stenoses after endoscopic dilatation in Crohn’s disease (Abstract). Gastroenterology 112:A1067
Ajlouni Y, Iser JH, Gibson PR (2007) Endoscopic balloon dilatation of intestinal strictures in Crohn’s disease: safe alternative to surgery. J Gastroenterol Hepatol 22:486–490
Sabate JM, Villarejo J, Bouhnik Y et al (2003) Hydrostatic balloon dilatation of Crohn’s strictures. Aliment Pharmacol Ther 18:409–413
Morini S, Hassan C, Lorenzetti R et al (2003) Long-term outcome of endoscopic pneumatic dilatation in Crohn’s disease. Dig Liver Dis 35:893–897
Ferlitsch A, Reinisch W, Puspok A et al (2006) Safety and efficacy of endoscopic balloon dilation for treatment of Crohn’s disease strictures. Endoscopy 38:483–487
Couckuyt H, Gevers AM, Coremans G, Hiele M, Rutgeerts P (1995) Efficacy and safety of hydrostatic balloon dilatation of ileocolonic Crohn’s strictures: a prospective longterm analysis. Gut 36:577–580
Thomas-Gibson S, Brooker JC, Hayward CM, Shah SG, Williams CB, Saunders BP (2003) Colonoscopic balloon dilation of Crohn’s strictures: a review of long-term outcomes. Eur J Gastroenterol Hepatol 15:485–488
Tichansky D, Cagir B, Yoo E, Marcus SM, Fry RD (2000) Strictureplasty for Crohn’s disease: meta-analysis. Dis Colon Rectum 43:911–919
Hassan C, Zullo A, De Francesco V, Ierardi E, Giustini M, Pitidis A, Taggi F, Winn S, Morini S (2007) Systematic review: endoscopic dilatation in Crohn’s disease. Aliment Pharmacol Ther 26:1457–1464
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wibmer, A.G., Kroesen, A.J., Gröne, J. et al. Comparison of strictureplasty and endoscopic balloon dilatation for stricturing Crohn’s disease—review of the literature. Int J Colorectal Dis 25, 1149–1157 (2010). https://doi.org/10.1007/s00384-010-1010-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-010-1010-x