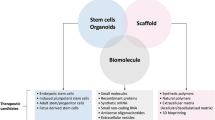
Abstract
Despite the advancements that have been made in treating infants with congenital malformations, these still represent a major cause of disease and death during the first years of life and childhood. Regeneration of natural tissue from living cells to restore damaged tissues and organs is the main purpose of regenerative medicine. This relatively new field has emerged by the combination of tissue engineering and stem cell transplantation as a possible strategy for the replacement of damaged organs or tissues. This review would like to offer an insight on the latest evolution of stem cells with a glance at their possible application for regenerative medicine, particularly in the Paediatric Surgery field.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Congenital malformations represent a major cause of disease and death during the first years of life and childhood and this is mostly due to complex conditions in which prosthetic materials are used because of the lack of biocompatible tissues able to replace or regenerate damaged organs. Besides the risk of infection, the major drawback of using a prosthetic patch closure is the risk of dislodgment and subsequent recurrence of the initial problem. Moreover, foreign body reactions and implant rejection occur when synthetic polymers are used. Regeneration of natural tissue from living cells to restore damaged tissues and organs is the main purpose of regenerative medicine. This relatively new field has emerged by the combination of tissue engineering and cell transplantation as a possible strategy for the replacement of damaged organs or tissues. So far, most of the attention has been focused on degenerative diseases such as Parkinson or Alzheimer, while very little has been done for the treatment of congenital conditions. However, the knowledge acquired in the last years from stem cell biology and regenerative medicine strategies could lead to new ways of repairing or replacing injured organs and systems, even during fetus development and therefore paediatric patients could largely benefit from the evolution of this new exciting field. In order to give rise to a new functional organ-like structure, several variables, such as local environment, nutrients, and metabolites are pivotal. These variables, in the contest of tissue engineering, are mainly dependent on the provision of a three-dimensional growth structure termed “scaffold” [1]. Scaffolds are usually made by natural materials, which are essentially bioactive but lack mechanical strength, or synthetic materials, which lack inherent bioactivity but are mechanically strong and can be engineered with the desirable macro-, microstructure, and might possess desired bioactive properties to make possible cellular growth and organogenesis [2]. Despite scaffolds could ultimately represent the exclusive tool for tissue engineering and several attempts to generate whole organs, such as liver, have been done by developing structures with vascular channels to ensure an adequate network of vascular supply [3], major developments in regenerative medicine have been achieved after the discovery of stem cells (SCs). These cells are unspecialized or undifferentiated cells with the capacity of self-renewal and the power to give rise to multiple different specialized cell types [4]. Three are the main sources SCs in human and animals: from embryonic, fetal, and adult tissues.
Adult SCs have a limited cellular regeneration or turnover that could represent a limitation for tissue engineering application where a large number of cells is necessary [5]. They can be identified in many adult mammalian tissues, such as bone marrow, skeletal muscle, skin, and adipose tissue, where they contribute to the replenishment of cells lost through normal cellular senescence or injury [6–10]. In contrast, SCs derived from embryonic sources have the ability to give rise to cells that not only proliferate and replace themselves indefinitely, but also have the potential to form any cell type [11, 12]. ES cells are derived from the inner cell mass of pre-implantation embryos, are pluripotent and demonstrate germ-line transmission in experimentally produced chimeras [13, 14]. More recently, cells with intermediate potential could be derived from the amniotic fluid (fetal SCs) [15] or reprogramed from adult SCs using various factors implicated with the maintenance of pluripotent potential of ES cells [16]. This review would like to offer an insight on the latest evolution of SCs with a glance at their possible application for regenerative medicine, particularly in the Paediatric Surgery field.
Embryonic Stem cells
Embryonic stem (ES) cells derive from the inner cell mass of a blastocyst stage embryo [17]. They are pluripotent and give rise during development to all derivatives of the three primary germ layers: ectoderm, endoderm, and mesoderm; hence, they possess the potential to develop into most of the cell types within the body [13, 18, 19]. The field of ES cell research began with the study of teratocarcinoma cells in 1950s, continued with first mouse ES cell lines derived from the inner cell mass of blastocysts using culture conditions (fibroblast feeder layers and serum) in 1981 and expanded in 1998 when Thomson et al. [13, 20, 21] first derived human ES (hES) cells. Optimal culture conditions have been developed employing both mouse embryonic stem (mES) or hES cells to evaluate and maintain both their proliferative and differentiative capacities. mES cells are grown on a layer of gelatin and require the presence of Leukemia Inhibitory Factor (LIF) while hES cells are grown on a feeder layer of mouse embryonic fibroblasts (MEFs) and require the presence of basic Fibroblast Growth Factor (bFGF) [18]. The maintenance of pluripotency in the hES is assured by the presence of different transcription factors like Oct-4, Nanog, and SOX2 that are essential to ensure the suppression of genes that lead to differentiation [22]. The cell surface antigens most commonly used to identify hES cells are the glycolipids Stem Cells Embryonic Antigen-3 and -4 (SSEA3 and SSEA4) and the keratan sulfate antigens Tra-1-60 and Tra-1-81 [9]. ES cells could be used not only to generate tissues, but also could be employed as “cellular models” to study a range of human diseases, and to test new drug candidates for efficacy and toxicity [23]. ES cells, being pluripotent, require specific signals for correct differentiation and if injected in vivo prior commitment, they will give rise to many different types of cells, causing teratomas. So far their potentials, together with the difficulties related to their allogenic origin, have limited their possible clinical applications [24]. In particular, the political debate surrounding SCs began suddenly after hES creation because of the destruction of the derivative embryo. Recently, researchers opened the possibility of generating ES cell lines without destroying embryos by deriving cells from the early development of the embryo without impairing their further development [25–27].
Somatic Cell Nuclear Transfer (SCNT) has also been adopted to create patient-specific SCs and avoid problems related to the creation of allogenic tissue. This procedure entails specifically the removal of an oocyte nucleus in culture, followed by its replacement with a nucleus derived from a somatic cell obtained from a patient. SCNT technique was first reported by Briggs and King [28] and some years after was used to obtain the first vertebrate (a frog) [29]. Cells yielded by this induction would be genetically identical to the donor and would not be rejected by the patient. SCNT can potentially be used for three purposes: (a) reproduction, leading to generation of an embryo for continuation of life (a notable example in 1996 was the generation of the first mammal, a sheep named Dolly, derived from an adult somatic cell by the use of this technique [30]); (b) therapy, generating blastocysts for SC derivation; and (c) research and regenerative medicine. The first is scientifically and ethical condemned. The second has important implications for the future of ES therapies, allowing the production of non-immunogenic ES lines. Besides, these cells could be stored and used subsequently for the treatment of future medical conditions. As a consequence this could be relevant for the creation of autologous tissues also in children who are born with complex malformations in which tissue viability represent a problem. Patient-specific cells could be created in vitro. ES cells derived using SCNT would have the same genetic background of the patient who has donated the initial genetic material and the tissue created would not be rejected after transplantation. ES cells have in fact the advantage of being extremely plastic facilitating the in vitro engineering of complex organ such as heart, liver, and kidney [31–33]. Nevertheless, in spite of the ethical considerations, the limitation of this technique is related both to the low efficiency, leading to a high loss in cell yield and the inadequate supply of human oocytes [34].
Induced Pluripotent Stem cells
Since the major objection to hES research is the destruction of embryos, it would be advantageous to develop a method of creating SCs that overcome this hurdle. A considerable step ahead was represented by the generation of the induced pluripotent stem (iPS) cells. The production of iPS cells with quasi-identical genetic and functional properties offers the possibility to bypass both moral conflicts and different genetic background inherent to the technologies mentioned above. iPS are pluripotent stem cell developed from a non-pluripotent cell, usually an adult somatic cell, by causing a forced expression of several genetic sequences and were first produced in 2006 by Takahashi and Yamanaka from mouse somatic cells. The key genes Oct3-4 (POU5F1), the transcription factor Sox2, c-Myc proto-oncogene protein and Klf4 (Krueppel-like factor 4) were sufficient to reprogram mouse fibroblasts to cells closely resembling mouse ES cells [16]. The insertion of these sequences is usually achieved through transfecting viral vectors, like retroviruses. After 3–4 weeks, small numbers of transfected cells begin to become morphologically and biochemically similar to pluripotent SCs, and are typically isolated through morphological selection, doubling time, or through a reporter gene and antibiotic selection. Although the initial mouse iPS cells did not contribute to full-term pregnancies chimeras, subsequent modification of the procedure to select iPS cells based on the reactivation of Oct4 or Nanog promoter resulted in iPS cells that more closely resembled mouse ES cells, including the ability to contribute to germlines [35]. Despite the high similarity between mouse iPS and ES cells, tumor formation in iPS cell chimeric mice was high, presumably due to the expression of c-Myc in iPS cell-derived somatic cells [36]. Subsequently, Yamanaka successfully transformed human fibroblasts into pluripotent SCs using the same four key genes: Oct3-4, Sox2, Klf4, and c-Myc with a retroviral transfection. Subsequently, Thomson and colleagues [37, 38] used Oct3-4, Sox2, Nanog, and a different gene Lin28 using a lentiviral system, improving transduction output. The viral transfection systems used to insert the genes at random locations in the host’s genome created concern for potential therapeutic applications of these iPS, because of the retroviral integration might increase the risk to form tumors [39]. To overcome these dangers, adenoviruses to transport the four sequences into the DNA of mice somatic cells have been used, resulting in cells identical to ES cells. Since the adenovirus does not combine any of its own genes with the targeted host, the increase danger of creating tumors is also eliminated [40]. Yamanaka and co-workers [41] demonstrated that reprograming can be accomplished via plasmid without any virus transfection system at all, although at very low efficiencies. Human iPS cells show morphological resemblance to hES cells, express typical human ES cell-specific cell surface antigens and genes, give rise to multiple lineages in vitro, and form teratomas when injected into immunocompromised mice. The efficiency of reprograming adult fibroblasts has been low (<0.1%) so far but, since reprogramed clones could consistently recovered and expanded with the existing gene combinations, for practical applications, the low reprograming efficiency itself is not really considered an issue, unless reprograming selects for abnormal genetic or epigenetic events that are stably propagated in the resulting iPS cell lines [42].
Recently, Jaenisch group found a very elegant way to derive human iPS from somatic cells of patients free of reprograming factors using Cre-recombinase excisable lentiviruses. The efficiency of reprogramed iPS is very high with a low number of proviral vector integration, the cells maintain a gene expression profile more similar to hES than to human iPS and can be subsequently differentiated into specific tissue [43]. This methodology could be considered in the future as alternative to ES cells created by SCNT. Somatic cells could easily be derived from the skin of a child with a malformation, reprogramed and differentiated after obtaining patient-specific ES cells. The tissue obtained will match completely with the patient and it will not be rejected; however, the tumorigenic potential remains unclear and the clinical use is subjected to further animal experiments.
Mesenchymal Stem Cells
Adult individuals equally contain stem cells but their characteristics are quite different from ES cells. Adult stem cells are considered as less proliferative, more mature with a narrower differentiation potential but a safer resource in respect of ES cells. They are virtually present in all adult tissues but, because of their implication for tissue regeneration in paediatric surgery, only mesenchymal stem cells (MSCs) will be discussed here. MSCs are multipotent SCs that can differentiate into a variety of cell types, first harvested from bone marrow via plastic adhesion, with a fibroblast-like morphology and differentiation potential into osteogenic (bone), chondrogenic (cartilage), and adipogenic (bone marrow stroma) lineages “in vitro” [44]. Some studies demonstrated that MSC can also differentiate to other cell types of mesodermal origin (skeletal muscle, smooth muscle, cardiac muscle, endothelial cells) but not solid experiments with in vivo transplantation of the progeny of a single cell could finally demonstrate terminal differentiation. To date, MSCs have been isolated in the fetus from blood, liver and bone marrow, amniotic fluid, lung, pancreas, dental pulp, and periosteum [45–50]. They have also been isolated from umbilical cord blood, Wharton’s jelly, placenta, and amniotic fluid [51–54]. The definition “mesenchymal stem cells” has been considered unclear through the years; “mesenchymal” was based on the hypothesis that multiple tissues beyond skeletal lineages, such as skeletal muscle, myocardium, smooth muscle could be generated by MSCs and secondly during embryonic organogenesis. The postnatal MSCs related tissues, are generated by a system of distinct progenitors, rather than from a common precursor. Dealing with this problem, three major criteria have been introduced to define MSCs by International Society for Cell Therapy [55]. First, cells must be plastic-adherent when maintained under standard culture conditions. When measured by flow cytometry, >95% of the cell population must express CD73 (5′-nucleotidase ecto, NT5E), CD90 (Thy-1) and CD105 (SH2 or MCAM or endoglin), LNGFR (Low affinity Nerve Growth Factor Receptor), CD166 (ALCAM adhesion protein), CD146 (P1H12), CD29, CD106 (vascular adhesion molecule-1, VCAM-1) and >98% of the cells should be negative for hematopoietic cell surface antigens: CD45, a pan-leukocyte marker; CD34, a marker of primitive hematopoietic progenitors and endothelial cells; either CD11b or CD14, markers for monocytes; either CD19 or CD79a, B-cells markers and Human Leukocyte Antigen II (HLA Class 2). Finally, to be defined as MSCs, cells should be capable to differentiate into osteoblasts, chondroblasts, and adipocytes when placed into an appropriate induction/differentiation medium. Among the MSCs collected from different tissues, there is no clear evidence of phenotypic differences in surface antigen expression. However, the success rate of MSCs isolation varies among tissues. MSCs can be isolated from only 63% of cord blood samples, while they can be easily derived from 100% of both bone marrow and adipose tissue processed [56]. Many scientific reports indicate that MSCs possess immunomodulatory properties and may play specific roles as immunomodulators in transplantation tolerance, autoimmunity, as well as fetal-maternal tolerance [57]. MSCs suppress T cell proliferation, but express different ligands that are recognized by activating NK receptors that trigger NK alloreactivity. Treatment of MSCs with IFN-gamma up-regulate expression of HLA class I molecules and decrease NK activity [58]. Recently, it has been supposed that MSCs may exert a more significant role through the release of different factors via paracrine action, rather than adopt a particular differentiated state after engraftment in target tissue [59]. In contrast with the aforementioned cells, MSCs also have a limited life span and become senescent when cultured in vitro. Several mechanisms over the progressive loss of telomeres were invoked to explain the acquisition of this phenotype and various experimental strategies have been adopted to extend MSCs life span [60–62]. Proliferation capacity of MSCs can be significantly increased by the presence of oncogenes (E6–E7) from HPV. Unexpectedly, transfected MSCs showed no signs of neoplastic transformation [63]. Nevertheless the acquisition of neoplastic features in these engineered cells could not be totally excluded and might occur. Regardless the isolation procedure, MSCs quantity obtained from primary tissues is not sufficient for any downstream application in clinical settings. In vitro expansion can affect biological properties of the cells; in fact MSCs go through very significant changes in phenotype and gene expression as a result of cell culture adaptation. Although considered a safer source, if compared to ES, the prospective clinical applications of MSCs require a meticulous examination. Some approaches aiming at improving safety have been established to evaluate the possibility of eliminating xenoproteins or xenoproducts like fetal calf serum in the feeding medium, to reduce the risk of potential viral-transmission-like unidentified zoonoses or prions and reduce immunogenicity related to serum component absorption [64]. The ability of MSCs to give rise to different lineages has been a matter of intense studies and plasticity and mechanisms of action have been studied in models of small and big animals. MSCs can differentiate beyond their traditional mesodermal lineage, at least in vitro, into both ectodermal (neurons) and endodermal (hepatocytes) nature [65–67]. However, broad abilities of MSCs are questionable and in several publications it has been demonstrated that MSCs do not undergo a proper trans-differentiation (irreversible switch of one differentiated cell into another), but rather fuse with specialized differentiated cells, thus more studies are required to a better understanding of this issue [68]. To date, MSCs have been tested on pediatric patients for several clinical indications, like inborn error of metabolism (Metachromatic leukodystrophy, Hurler syndrome, Infantile hypophosphatasemia), osteogenesis imperfecta, and GVHD [69–73]. Preliminary studies have been assessed in patients with amyotrophic lateral sclerosis and autologous MSC transplantation has also been evaluated in patients after acute myocardial infarction [74, 75]. For the engineering of mesodermal-derived tissues, MSCs certainly represent at the moment the optimal source: in the close future children with bone, smooth muscle, or cartilage defects could have their tissue loss replace using MSCs derived from their bone marrow. MSCs derived from other sources such as the placenta or the amniotic fluid have also shown to be beneficial in animal models of congenital malformations [76, 77]. While clinical applications of MSCs are progressing, basic research is carrying on big efforts to understand better cell properties and abilities through the analysis of the molecular mechanisms causing the evident clinical benefits after MSCs therapy.
Amniotic Fluid Stem cells
While ES or iPS cells have the limitation of being difficult to program and could let tumor formation in vivo and MSCs are difficult to expand in vitro, it would be ideal to have a source of cells capable to overcome all the different problems. We have recently described the possibility of deriving pluripotent stem cells from the amniotic fluid. Amniotic Fluid Stem (AFS) cells represents about 1% of the whole cells in cultures of human amniocentesis specimens obtained for prenatal genetic diagnosis and can be harvested by immunoselecting the antigen c-Kit (CD117) positive population [78]. AFS cells are described as broadly multipotent SCs that can differentiate into a variety of cell types. AFS cells have been shown to differentiate to adipogenic, osteogenic, myogenic, endothelial, neurogenic, and hepatogenic lineages, inclusive of all embryonic germ layers [15]. This group of cells can be steadily expanded in cultures, has a typical doubling time of 36 h and do not need any feeder layer. Sub-confluent cells showed no evidence of spontaneous differentiation, nevertheless, under specific inducing conditions these cells are able to differentiate and if injected in vivo, showed no evidence of tumor growth in severe combined immunodeficient mice. The AFS cells are positive for a number of surface markers characteristic of mesenchymal and/or neural SCs, but not ES cells, as CD29, CD44 (hyaluronan receptor), CD73, CD90, and CD105 (endoglin). Human AFS cells are positive for stage-specific embryonic antigen (SSEA)-4, also expressed by ES cells. Moreover, more than 90% of the cells express the transcription factor Oct4, which has been associated with the maintenance of the undifferentiated state and the pluripotency of ES and EG cells [79]. Retroviral marking using a vector encoding green fluorescent protein identified differentiated positive subclones descended from a single cell. AFS cells appeared to be less plastic than ES cells, nevertheless reproducibility of the generation and differentiation of these SCs has not yet been widely reported and future studies are required to assess the potential broad use of these promising resource. In the paediatric field, however, they could play an important role for prenatally diagnosed structural defects, there is the possibility of obtaining homologous cells at the time of invasive sampling; fetal cells could be harvested, cultured, and manipulated in vitro, during the remainder of pregnancy and later used for tissue engineering of graft material that will be used for postnatal reconstruction. Moreover, they could also be stored for future use (Table 1).
Conclusion
In this scenario, the use of AFS and iPS [80, 81] cells could bring together researchers working for the common aim to develop new protocols to treat diseases and congenital malformations without ethical problems, although, the most up-to-date work of elegant iPS derivation is burdened with problems related to teratoma formation and possible altered epigenesis of iPS derived tissue. Regarding ES cells generation, this year a new impulse to research has been given in the USA by an executive order lifting restrictions on federal funding for stem cell research, which erased limits imposed 8 years ago by the former government. Despite the hurdles represented by cell expansion (doubling time 36–48 h), immunorejection and safety concerns, hESC-derived tissues after modification may have a promising future for transplantation thanks to the typical versatility of these cells. Adult SCs like MSCs have already been used for infusion in various clinical therapies in a relatively large number of individual, including patients of pediatric age, without any serious adverse effects [64]. Apart from the direct infusion of SCs, other applications such as in the surgical field have also been proposed. Encouraging results from the clinical application were achieved lately by the first successful transplantation of a tissue-engineered trachea built in a bioreactor, seeded with autologous cells in an adult with bronchial stenosis [82]. Adequate preclinical models together with the conclusion of ongoing clinical trials, will contribute to the establishment of SCs therapeutic potential in pediatric patients with congenital defective malformation such as oesophageal atresia that requires segmental replacement, diaphragmatic hernia, abdominal wall defects, in addition it might be possible to shift traditional surgical disease like Hirschsprung’s toward a medical setting by injection of enteric nervous system SCs, harvested from postnatal gut and transplanted into aganglionic to refill the insufficient neuronal network of the intestine wall [83]. Ultimately, SCs may protect the injured intestine in diseases such as necrotising enterocolitis (NEC), reducing the severity of bowel damage, promoting proliferation, and enhancing vascularization [2, 84], and in case of massive intestinal resection occurred after neonatal volvulus or NEC, might be in the future a valid alternative to intestinal transplantation [85, 86].
References
Placzek MR, Chung IM, Macedo HM, Ismail S, Mortera Blanco T, Lim M, Cha JM, Fauzi I, Kang Y, Yeo DC, Ma CY, Polak JM, Panoskaltsis N, Mantalaris A (2009) Stem cell bioprocessing: fundamentals and principles. J R Soc Interface 6(32):209–232
Safinia L, Datan N, Höhse M, Mantalaris A, Bismarck A (2005) Towards a methodology for the effective surface modification of porous polymer scaffolds. Biomaterials 26(36):7537–7547
Carraro A, Hsu WM, Kulig KM, Cheung WS, Miller ML, Weinberg EJ, Swart EF, Kaazempur-Mofrad M, Borenstein JT, Vacanti JP, Neville C (2008) In vitro analysis of a hepatic device with intrinsic microvascular-based channels. Biomed Microdevices 10(6):795–805
Nagy RD, Tsai BM, Wang M et al (2005) Stem cell transplantation as a therapeutic approach to organ failure. J Surg Res 129(1):152–160
Jiang Y, Jahagirdar BN, Reinhardt RL, Schwartz RE, Keene CD, Ortiz-Gonzalez XR, Reyes M, Lenvik T, Lund T, Blackstad M et al (2002) Pluripotency of mesenchymal stem cells derived from adult marrow. Nature 418:41–49
Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, Moorman MA, Simonetti DW, Craig S, Marshak DR (1999) Multilineage potential of adult human mesenchymal stem cells. Science 284(5411):143
McKinney-Freeman SL, Jackson KA, Camargo FD, Ferrari G, Mavilio F, Goodell MA (2002) Muscle-derived hematopoietic stem cells are hematopoietic in origin. Proc Natl Acad Sci USA 99:1341–1346
Schwartz RE, Reyes M, Koodie L, Jiang Y, Blackstad M, Lund T, Lenvik T, Johnson S, Hu WS, Verfaillie CM (2002) Multipotent adult progenitor cells from bone marrow differentiate into functional hepatocyte-like cells. J Clin Invest 109(10):1291–1302
LaBarge MA, Blau HM (2002) Biological progression from adult bone marrow to mononucleate muscle stem cell to multinucleate muscle fiber in response to injury. Cell 111:589–601
Ferrari G, Cusella-De Angelis G, Coletta M, Paolucci E, Stornaiuolo A, Cossu G, Mavilio F (1998) Muscle regeneration by bone marrow-derived myogenic progenitors. Science 279(5356):1528–1530 (erratum in: Science 1998 Aug 14;281(5379):923)
Shamblott MJ, Axelman J, Littlefield JW, Blumenthal PD, Huggins GR, Cui Y, Cheng L, Gearhart JD (2001) Human embryonic germ cell derivatives express a broad range of developmentally distinct markers and proliferate extensively in vitro. PNAS 98(1):113–118
Kofidis T, de Bruin JL, Hoyt G, Ho Y, Tanaka M, Yamane T, Lebl DR, Swijnenburg RJ, Chang CP, Quertermous T, Robbins RC (2005) Myocardial restoration with embryonic stem cell bioartificial tissue transplantation. J Heart Lung Transplant 24(6):737–744
Thomson JA, Itskovitz-Eldor J, Shapiro SS, Waknitz MA, Sweier JJ, Marshall VS, Jones JM (1998) Embryonic stem cell lines derived from human blastocysts. Science 282(5391):1145
Markel TA, Crisostomo PR, Lahm T, Novotny NM, Rescorla FJ, Tector J, Meldrum DR (2008) Stem cells as a potential future treatment of pediatric intestinal disorders. J Pediatr Surg 43(11):1953–1963
De Coppi P, Bartsch G Jr, Siddiqui MM, Xu T, Santos CC, Perin L, Mostoslavsky G, Serre AC, Snyder EY, Yoo JJ, Furth ME, Soker S, Atala A (2007) Isolation of amniotic stem cell lines with potential for therapy. Nat Biotechnol 25(1):100–106
Takahashi K, Yamanaka S (2006) Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126:663–676
Odorico JS, Kaufman DS, Thomson JA (2001) Multilineage differentiation from human embryonic stem cell lines. Stem Cells 19(3):193–204
Amit M, Shariki C, Margulets V et al (2004) Feeder layer and serum-free culture of human embryonic stem cells. Biol Reprod 70(3):837–845
Richards M, Fong CY, Chan WK et al (2002) Human feeders support prolonged undifferentiated growth of human inner cell masses and embryonic stem cells. Nat Biotechnol 20(9):933–936
Evans MJ, Kaufman MH (1981) Establishment in culture of pluripotential cells from mouse embryos. Nature 292:154–156
Martin GR (1981) Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. Proc Natl Acad Sci USA 78:7634–7638
Fong H, Hohenstein KA, Donovan PJ (2008) Regulation of self-renewal and pluripotency by Sox2 in human embryonic stem cells. Stem Cells 26(8):1931–1938 (Epub 2008 Apr 3)
Lott JP, Savulescu J (2007) Towards a global human embryonic stem cell bank. Am J Bioeth 7(8):37–44
Hipp J, Atala A (2008) Sources of stem cells for regenerative medicine. Stem Cell Rev 4(1):3–11 Review
Klimanskaya I, Chung Y, Becker S, Lu SJ, Lanza R (2006) Human embryonic stem cell lines derived from single blastomeres. Nature 444(7118):481–485 (epub 2006 Aug 23. Erratum in: Nature. 2006 Nov 23;444(7118):512. Nature. 2007 Mar 15;446(7133):342)
Chung Y, Klimanskaya I, Becker S, Marh J, Lu SJ, Johnson J, Meisner L, Lanza R (2006) Embryonic and extraembryonic stem cell lines derived from single mouse blastomeres. Nature 439(7073):216–219 (epub 2005 Oct 16)
Deb KD, Sarda K (2008) Human embryonic stem cells: preclinical perspectives. J Transl Med 6:7
Briggs R, King TJ (1952) The transplantation of living nuclei from blastula cells into enucleated frog’s eggs. Proc Natl Acad Sci USA 38:455–463
Gurdon JB, Laskey RA (1970) The transplantation of nuclei from single cultured cells into enucleate frogs’eggs. J Embryol Exp Morphol 24(2):227–248
Campbell KH, McWhir J, Ritchie WA, Wilmut I (1996) Sheep cloned by nuclear transfer from a cultured cell line. Nature 380(6569):64–66
Franco D, Moreno N, Ruiz-Lozano P (2007) Non-resident stem cell populations in regenerative cardiac medicine. Cell Mol Life Sci 64(6):683–691
Dalgetty DM, Medine CN, Iredale JP, Hay DC (2009) Progress and future challenges in stem cell-derived liver technologies. Am J Physiol Gastrointest Liver Physiol 297(2):G241–G248
Anglani F, Forino M, Del Prete D, Tosetto E, Torregrossa R, D’Angelo A (2004) In search of adult renal stem cells. J Cell Mol Med 8(4):474–487
Lerou PH, Yabuuchi A, Huo H, Takeuchi A, Shea J, Cimini T, Ince TA, Ginsburg E, Racowsky C, Daley GQ (2008) Human embryonic stem cell derivation from poor-quality embryos. Nat Biotechnol 26(2):212–214 (epub 2008 Jan 27)
Wernig M, Meissner A, Foreman R, Brambrink T, Ku M, Hochedlinger K, Bernstein BE, Jaenisch R (2007) In vitro reprogramming of fibroblasts into a pluripotent ES-cell-like state. Nature 448(7151):318–324 (epub 2007 Jun 6)
Maherali N, Sridharan R, Xie W, Utikal J, Eminli S, Arnold K, Stadtfeld M, Yachechko R, Tchieu J, Jaenisch R, Plath K, Hochedlinger K (2007) Directly reprogrammed fibroblasts show global epigenetic remodeling and widespread tissue contribution. Cell Stem Cell 1(1):55–70
Yu J, Vodyanik MA, Smuga-Otto K, Antosiewicz-Bourget J, Frane JL, Tian S, Nie J, Jonsdottir GA, Ruotti V, Stewart R, Slukvin II, Thomson JA (2007) Induced pluripotent stem cell lines derived from human somatic cells. Science 318(5858):1917–1920
Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K, Yamanaka S (2007) Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131(5):861–872
Okita K, Ichisaka T, Yamanaka S (2007) Generation of germline-competent induced pluripotent stem cells. Nature 448(7151):313–317
Meissner A, Wernig M, Jaenisch R (2007) Direct reprogramming of genetically unmodified fibroblasts into pluripotent stem cells. Nat Biotechnol 25(10):1177–1181
Okita K, Nakagawa M, Hyenjong H, Ichisaka T, Yamanaka S (2008) Generation of mouse induced pluripotent stem cells without viral vectors. Science 322(5903):949–953
Yu J, Thomson JA (2008) Pluripotent stem cell lines. Genes Dev 22(15):1987–1997 Review
Soldner F, Hockemeyer D, Beard C, Gao Q, Bell GW, Cook EG, Hargus G, Blak A, Cooper O, Mitalipova M, Isacson O, Jaenisch R (2009) Parkinson’s disease patient-derived induced pluripotent stem cells free of viral reprogramming factors. Cell 136(5):964–977
Friedenstein AJ, Kulagina NN, Panasuk AF, Rudakowa SF (1974) Precursors for fibroblasts in different populations of hematopoietic cells as detected by the in vitro colony assay method. Exp Hematol 2:83–92
Campagnoli C, Roberts IAG (2001) Identification of mesenchymal stem/progenitor cells in human first-trimester fetal blood, liver, and bone marrow. Blood 98:2396–2402
In ‘t Anker PS, Scherjon SA (2003) Amniotic fluid as a novel source of mesenchymal stem cells for therapeutic transplantation. Blood 102:1548–1549
Tsai MS, Lee JL (2004) Isolation of human multipotent mesenchymal stem cells from second-trimester amniotic fluid using a novel two-stage culture protocol. Hum Reprod 19:1450–1456
Fan CG, Thang FW, Zhang Q (2005) Characterization and neural differentiation of fetal lung mesenchymal stem cells. Cell Transplant 14:311–321
Waddington RJ, Youde SJ, Lee CP, Sloan AJ (2009) Isolation of distinct progenitor stem cell populations from dental pulp. Cells Tissues Organs 189(1–4):268–274 (epub 2008 Aug 14)
Eyckmans J, Luyten FP (2006) Species specificity of ectopic bone formation using periosteum-derived mesenchymal progenitor cells. Tissue Eng 12(8):2203–2213
Erices A, Conget P (2000) Mesenchymal progenitor cells in human umbilical cord blood. Br J Haematol 109:235–242
Romanov YA, Svintsitskaya VA (2003) Searching for alternative sources of postnatal human mesenchymal stem cells: candidate MSC-like cells from umbilical cord. Stem Cells 21:105–110
Igura K, Takahashi K, Mitsuru A, Yamaguchi S (2004) Isolation and characterization of mesenchymal progenitor cells from chorionic villi of human placenta. Cytotherapy 6:543–553
Perin L, Sedrakyan S, Da Sacco S, De Filippo R (2008) Characterization of human amniotic fluid stem cells and their pluripotential capability. Methods Cell Biol 86:85–99
Horwitz EM, Blanc KL (2005) Clarification of the nomenclature for MSC: The International Society for Cellular Therapy position statement. Cytotherapy 7:393–395
Kern S, Eichler H (2006) Comparative analysis of mesenchymal stem cells from bone marrow, umbilical cord blood, or adipose tissue. Stem Cells 24:1294–1301
Le Blanc K (2003) Immunomodulatory effects of fetal and adult mesenchymal stem cells. Cytotherapy 5(6):485–489
Karlsson H, Samarasinghe S, Ball LM, Sundberg B, Lankester AC, Dazzi F, Uzunel M, Rao K, Veys P, Le Blanc K, Ringdén O, Amrolia PJ (2008) Mesenchymal stem cells exert differential effects on alloantigen and virus-specific T-cell responses. Blood 112(3):532–541 (epub 2008 Apr 28)
Crisostomo PR, Markel TA, Wang Y, Meldrum DR (2008) Surgically relevant aspects of stem cell paracrine effects. Surgery 143(5):577–581 (epub 2008 Jan 30)
Takeda Y, Mori T, Imabayashi H, Kiyono T, Gojo S, Miyoshi S, Hida N, Ita M, Segawa K, Ogawa S, Sakamoto M, Nakamura S, Umezawa A (2004) Can the life span of human marrow stromal cells be prolonged by bmi-1, E6, E7, and/or telomerase without affecting cardiomyogenic differentiation? J Gene Med 6:833–845
Ksiazek K (2009) A comprehensive review on mesenchymal stem cell growth and senescence. Rejuvenation Res 12(2):105–116
Terai M, Uyama T, Sugiki T, Li XK, Umezawa A, Kiyono T (2005) Immortalization of human fetal cells: the life span of umbilical cord blood-derived cells can be prolonged without manipulating p16INK4a/RB braking pathway. Mol Biol Cell 16:1491–1499
Takeuchi M, Takeuchi K, Kohara A, Satoh M, Shioda S, Ozawa Y, Ohtani A, Morita K, Hirano T, Terai M, Umezawa A, Mizusawa H (2007) Chromosomal instability in human mesenchymal stem cells immortalized with human papilloma virus E6, E7, and hTERT genes. In Vitro Cell Dev Biol Anim 43(3–4):129–138 (epub 2007 May 21)
Pelagiadis I, Dimitriou H, Kalmanti M (2008) Biologic characteristics of mesenchymal stromal cells and their clinical applications in pediatric patients. J Pediatr Hematol Oncol 30(4):301–309
Sanchez-Ramos J, Song S, Cardozo-Pelaez F et al (2000) Adult bone marrow stromal cells differentiate into neural cells in vitro. Exp Neurol 164:247–256
Hong SH, Gang EJ, Jeong JA et al (2005) In vitro differentiation of human umbilical cord blood-derived mesenchymal stem cells into hepatocyte-like cells. Biochem Biophys Res Commun 330:1153–1161
Sugaya K (2003) Potential use of stem cells in neuroreplacement therapies for neurodegenerative diseases. Int Rev Cytol 228:1–30
Dimmeler S, Zeiher AM (2009) Cell therapy of acute myocardial infarction: open questions. Cardiology 113(3):155–160 (epub 2008 Dec 22)
Koc ON, Gerson SC, Lazarus HM et al (2002) Allogenic mesenchymal stem cell infusion for treatment of metachromatic leucodystrophy (MLD) and Hurler syndrome (MPS-IH). Bone Marrow Transplant 30:215–222
Whyte MP, Kurtzberg J, McAlister WH et al (2003) Marrow cell transplantation for infantile hypophosphatasia. J Bone Miner Res 18:624–636
Horwitz EM, Prockop DJ, Fitzpatrick LA et al (1999) Transplantability and therapeutic effects of bone marrow-derived mesenchymal cells in children with osteogenesis imperfecta. Nat Med 5:309
Horwitz EM, Gordon PL, Koo WK et al (2002) Isolated allogeneic bone marrow-derived mesenchymal cells engraft and stimulate growth in children with osteogenesis imperfecta: implications for cell therapy of bone. Proc Natl Acad Sci USA 99:8932
Le Blanc K, Gotherstrom C, Ringden O et al (2005) Fetal mesenchymal stem-cell engraftment in bone after in utero transplantation in a patient with severe osteogenesis imperfecta. Transplantation 79:1607–1614
Mazzini L, Mareschi K, Ferrero I, Vassallo E, Oliveri G, Nasuelli N, Oggioni G, Testa L, Fagioli F (2008) Stem cell treatment in amyotrophic lateral sclerosis. J Neurol Sci 265(1):78–83
Burt RK, Loh Y, Pearce W, Beohar N, Barr WG, Craig R, Wen Y, Rapp JA, Kessler J (2008) Clinical applications of blood-derived and marrow-derived stem cells for nonmalignant diseases. JAMA 299(8):925–936
Fuchs JR, Hannouche D, Terada S et al (2005) Cartilage engineering from ovine umbilical cord blood mesenchymal progenitor cells. Stem Cells 23:958–964
Kunisaki SM, Freedman DA, Fauza DO (2006) Fetal tracheal reconstruction with cartilaginous grafts engineered from mesenchymal amniocytes. J Pediatr Surg 41:675–682
Zsebo KM et al (1990) Stem cell factor is encoded at the Sl locus of the mouse and is the ligand for the c-kit tyrosine kinase receptor. Cell 63:213–224
Pan GJ, Chang ZY, Scholer HR, Pei D (2002) Stem cell pluripotency and transcription factor Oct4. Cell Res 12:321–329
Takahashi K, Okita K, Nakagawa M, Yamanaka S (2007) Induction of pluripotent stem cells from fibroblast cultures. Nat Protoc 2(12):3081–3089
Park IH, Lerou PH, Zhao R, Huo H, Daley GQ (2008) Generation of human-induced pluripotent stem cells. Nat Protoc 3(7):1180–1186
Macchiarini P, Jungebluth P, Go T, Asnaghi MA, LE Rees, Cogan TA, Dodson A, Martorell J, Bellini S, Parnigotto PP, Dickinson SC, Hollander AP, Mantero S, Conconi MT, Birchall MA (2008) Clinical transplantation of a tissue-engineered airway. Lancet 372(9655):2023–2030 (epub 2008 Nov 18)
Thapar N (2009) New frontiers in the treatment of Hirschsprung disease. J Pediatr Gastroenterol Nutr 48(Suppl 2):S92–S94
Zani A, Cananzi M, Eaton S, Pierro A, De Coppi P (2009) Stem cells as a potential treatment of necrotizing enterocolitis. J Pediatr Surg 44(3):659–660 Comment on: J Pediatr Surg. 2008 Nov;43(11):1953–63
Javaid-Ur-Rehman, Waseem T (2008) Intestinal tissue engineering: where do we stand? Surg Today 38:484–486
Grikscheit TC, Siddique A, Ochoa ER, Srinivasan A, Alsberg E, Hodin RA, Vacanti JP (2004) Tissue-engineered small intestine improves recovery after massive small bowel resection. Ann Surg 240(5):748–754
Ware CB, Nelson AM, Blau CA (2006) A comparison of NIH-approved human ESC lines. Stem Cells 24(12):2677–2684
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pozzobon, M., Ghionzoli, M. & De Coppi, P. ES, iPS, MSC, and AFS cells. Stem cells exploitation for Pediatric Surgery: current research and perspective. Pediatr Surg Int 26, 3–10 (2010). https://doi.org/10.1007/s00383-009-2478-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-009-2478-8