Abstract
Purpose
The purpose of this study is to evaluate the accuracy of emergency Transcranial Doppler (TCD) to predict intracranial hypertension and abnormal cerebral perfusion pressure in children with severe traumatic brain injury (TBI).
Patients and methods
A descriptive and retrospective cross-sectional study was designed through data collected from medical records of children with severe TBI (Glasgow coma scale ≤8), admitted to a level I pediatric trauma center, between January 2000 and December 2005. Early TCD examination was performed upon admission, and TCD profiles were considered as altered using previously validated threshold values for diastolic velocity (<25 cm/s) and pulsatility index (>1.31) or when no-flow/backflow was detected. Invasive intracranial pressure (ICP) and cerebral perfusion pressure (CPP) monitoring were considered as the gold standard to measure intracranial hypertension (ICH). Statistical analyses compared TCD profiles to increased ICP (≥20 mmHg) and abnormal cerebral perfusion pressure (<50 mmHg) at admission.
Results
Non-invasive TCD and ICP monitoring were performed in 117 severe head-injured children. Mean age was 7.6 ± 4.4 years, with a male prevalence (71%). Median initial Glasgow coma scale was 6. TCD had 94% of sensitivity to identify ICH at admission and a negative predict value of 95% to identify normal ICP at admission. Its sensitivity to predict abnormal cerebral perfusion pressure was 80%.
Conclusions
The high sensitivity of admission TCD to predict ICH and abnormal CPP after trauma demonstrates that TCD is an excellent first-line examination to determine those children who need urgent aggressive treatment and continuous invasive ICP monitoring.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Severe traumatic brain injury (TBI) is an important cause of neurological disabilities and mortality in children [1–5], and continuous intracranial pressure (ICP) monitoring is critical to guide medical management in the pediatric intensive care unit (PICU), especially in those with the most severe injuries [5–9].
However, direct ICP measurement remains the reference method for intracranial hypertension (ICH) diagnosis and guided management [5–9], but represents an invasive surgical technique that cannot be initiated routinely at the bedside before cranial computed tomography (CCT) scan examination, and could be hazardous in patients with very critical conditions and coagulation disorders. Transcranial Doppler (TCD) examination, measuring blood flow velocity in both middle cerebral arteries, is a non-invasive method that requires training and experience to be efficient, but can be used at the bedside during the early phase of resuscitation. Its ability to detect altered cerebral hemodynamic profiles in patients with TBI and its usefulness in clinical decision making has been previously underlined [10–17]. However, clinical experience of early TCD monitoring in children with severe TBI has only been reported in short series [14, 17–19], and its reliability in predicting acutely increased ICP has not been clearly established. The aim of this study was to identify, in a series of children with severe TBI, the feasibility and reliability of TCD to predict ICH and resulting altered cerebral perfusion pressure (CPP) measured by invasive methods.
Material and methods
This is a descriptive and retrospective cross-sectional study based on the analysis of clinical data of children less than 15 years of age, presenting with a severe TBI defined by a scene Glasgow coma scale (GCS) ≤8. We reviewed the medical records of all children admitted to the emergency room of Hôpital Necker-Enfants Malades (Paris, France) between January 2000 and December 2005 (6 years). Children with non-accidental injuries and those whom TCD and invasive ICP monitoring was not performed at admission for any reason (like those with severe coagulation disorders and GCS 3 with absent brainstem reflexes or when those techniques were performed after 3 h of admission) were finally excluded from analysis.
Data were collected for age, gender, GCS, and pediatric trauma score (PTS), mechanism of accidents, and resulting injuries (multiple trauma or isolated head injury) at admission. All children were managed at the scene of the accident by a medical team of the emergency medical system (SAMU, Service d’Aide Médicale d’Urgence), transported to our trauma center, and benefited from standardized initial pre-hospital management including systematic venous access insertion, exclusive normal saline intra venous (IV) infusion, rapid sequence induction and tracheal intubation, assisted ventilation with associated continuous IV sedation, and volume expansion with colloids as needed, according to the French National Guidelines for emergency management of severe head injuries [5, 11, 20]. In emergency department and PICU, continuous IV sedation with benzodiazepine and sufentanil was maintained at least 48 h, mean arterial pressure (MAP) was adjusted within normal range for the age with fluid loading and/or vasopressor infusion as needed and assisted ventilation was carefully adapted to maintain a 35–40 mmHg end-tidal carbon dioxide.
All intended TCD examinations were performed during the resuscitation phase, and within the first 20 min following admission. All patients were, at the moment of the examination, stabilized with restored MAP, continuous IV sedation, and adapted assisted ventilation. Systolic (sV), end-diastolic (dV), and mean (mV) arterial blood flow velocities were measured in both middle cerebral arteries, using a 2-MHz continuous Doppler probe (Waki 1-TC, Atys Medical®, France), with a classical transtemporal window approach by a trained operator (senior anesthesiologist with >6 months experience in TCD). Pulsatility index (PI = sV − dV/mV) and resistivity index (RI = sV − dV/sV) were automatically calculated. TCD was considered altered in the following situations:
-
Evidence of bilateral backflow or no-flow despite careful examination with varying depth of insonation and maximum power settings.
-
End-diastolic velocity <25 cm/s or PI >1.31 as previously described [14]
Intraparenchymal pressure transducers (Codman®) were inserted as soon as possible, after CCT scan examination demonstrating cerebral injury, in the frontal region (opposite to the lesion when focal injury was present), by a trained neurosurgeon, and maintained at least 48 h. Cerebral perfusion pressure was calculated continuously as the difference between ICP and MAP measured by the means of an intra-arterial catheter. ICH was considered for any episode of ICP over 20 mmHg [21], lasting more than 10 min without concomitant external stimulation (tracheal suctioning, mobilization, physiotherapy, or nursing). Altered CPP was considered with measurements <50 mmHg with MAP in the normal range for the age [22]. MAP was considered as normal, according to age group, based on the American Heart Association criteria [23].
Statistical analysis was performed using the Statistical Package for the Social Sciences for Windows. For data analysis all quantitative variables were expressed as mean and SD. We considered, according to medical literature, the invasive ICP as the gold standard method to measure ICP. Sensitivity, specificity, positive, and negative predictive values were verified. This descriptive single-center study was performed in agreement with French law requirements (institutional review board consultation, no informed consent required, anonymous computer data bank analysis).
Results
In the study period, 315 children presented to our emergency department with severe TBI. Among them, 48 with a GCS of 3 dying within the first hours and 143 with missing data, were excluded. This let 124 severely head-injured children who benefited from early TCD examination. Initial no-flow/backflow was present and confirmed after CCT scan examination depicting severe diffuse injuries in 6% (7/124). All of them died within the first hours and were excluded from final analysis. This let 117 severely head-injured children in our final sample considered for analysis.
Mean age was 7.6 ± 4.4 years, with male prevalence (71%). Median GCS was 6, and median PTS was 4, indicating the magnitude of injury in this group of patients. Excluding children with non-accidental injuries, these patients did not differ from those where data were missing. Demographic data are presented in Table 1.
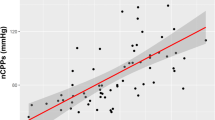
Cerebral hemodynamic profiles within the normal range were detected in 31.6% (37/117), and altered TCD profiles were identified in 68.4% (80/117) of the children. Intraparenchymal ICP transducers were inserted within the first 3 h after CCT scan examination, in this group of patients. There was no complication (infections, intracranial hemorrhage, or technical failure) related to invasive ICP monitoring. Increased ICP was noted upon transducer insertion in 27% (32/117) of the children. An alteration in initial TCD profiles was observed in 94% (30/32) of children with initially increased ICP, and 59% (50/85) of those with normal initial ICP values. Initial ICH was predicted by early TCD examination with a sensitivity of 94% and a 95% probability to identify those with normal ICP (Table 2). Regarding CPP, initial measurements were above the threshold value of 50 mmHg in 62% (72/117) of the cases. In children with decreased initial CPP, early TCD profile was considered as altered in 80% (36/45), compared to 61% (44/72) in children with normal initial CPP. Decreased initial CPP was so predicted by TCD examination with a sensitivity of 80% (Table 3).
Discussion
Previous studies have pointed out the usefulness of TCD examination to help medical decision in patients with cerebrovascular diseases [24–27] or hydrocephalus [28–30]. This non-invasive method, even though not measuring directly ICP, can easily detect alterations of cerebral blood flow velocities reflecting ICH. Its ability to predict acute ICH in children with TBI has been sparsely advocated, but experience was still limited in this particular indication [8, 17].
All children included in the present study had severe accidental TBI resulting in coma with a median GCS of 6. Their general characteristics were similar to those previously reported in our trauma population [31]. All of them benefited from a standardized initial medical to PICU management [31] in accordance with French National Guidelines for emergency management of TBI. These recommendations include continuous ICP and CPP monitoring as the most adapted means of detecting developing ICH requiring specific therapeutic interventions in comatose children. However, it remains an invasive and expensive method that could only be used routinely in specialized units [32]. In our unit, all children except those with no cerebral blood flow, were routinely submitted from an invasive ICP, to extensive experience could maintain a null incidence of complications with intraparenchymally inserted transducers as reported in the literature [33, 34]. Regarding CPP, a threshold value of 40–45 mmHg under which children with severe TBI could experience worse outcome was cited in the literature [35, 36]. Careful cerebral pressure monitoring appears of critical interest to detect episodes of cerebral pressure alteration that could be determinant in mortality and outcome. A major point of concern could be to identify as soon as possible those children at risk of ICH and decreased CPP, who carry an increased risk of complications and bad outcome.
TCD alone could not provide reliable information regarding cerebral blood flow/metabolism coupling and should be used dynamically to detect autoregulation impairment. It has been reported as an unreliable indicator of intracranial pressure in children [37]. However, the key issue for determining within the first minutes following admission which children should be considered at risks of developing ICH and decreased CPP, needing close monitoring could be the ability to perform a non-invasive, highly reproducible examination, that could be used at the bedside once the patient has been minimally stabilized [37]. TCD allows instant measurement of blood flow velocities in major cerebral vessels that could be affected by increased external strains resulting from increased ICP [38–40]. In a previous short series of 36 children with moderate to severe TBI, we could determine that decreased end-diastolic velocity and increased PI were the most determinant factors of bad outcome with threshold values of <25 cm/s for end-dV and >1.31 for PI [14]. Moreover, negative velocity appearing as a backflow on TCD profile could be clearly associated with brain death in children with severe TBI [41]. Using these definitions of altered TCD profiles, we verified that early TCD examination upon admission could predict ICH with a 94% sensitivity and 95% negative predictive value. A major limitation of this study could be that TCD examination was performed immediately after admission and could not be precisely correlated to ICP and CPP that were only measured after CCT scan examination. It could be suspected that, in children with severe TBI, in-hospital aggressive management could result in stabilized ICH before ICP monitor insertion. As mentioned in a previous study, mean arterial pressure, fever, impaired cerebral autoregulation, PaCO2, hematocrit, or sedation could be confounders of the relationship between cerebral perfusion pressure and cerebral blood flow velocities, mainly mean blood flow velocity [16]. In our patients, end-diastolic velocity and pulsatility index were chosen as reflects of significant TCD alterations that could accurately predict bad outcome and increased risks of ICH. These variables could be more directly affected by external arterial stains than mean flow velocity and could more accurately reflect increased ICP [14].
Regarding CPP, that could be a major factor of bad outcome, altered TCD predicted initial decreased CPP with an 80% sensitivity and 76% probability to identify those with normal CPP (negative predict value). More than trying to correlate TCD variables with measured ICP and CPP, the aim of this study was to identify upon admission before CCT scan examination can be performed, children at risk of increased ICP. Initial TCD might be considered as a valuable tool to identify children at risk of developing ICH and decreased CPP at admission. These results confirm the benefit of using TCD in children with increased ICP reported by others [10, 14, 17, 18]. It could be argued that TCD examination and ICP monitoring were not performed within the first 3 h in all children admitted to our trauma center during the study period. This may introduce a potential bias of selecting patients according to the severity of the initial presentation. As previously mentioned, the group of patients included in the study did not differ significantly from our general trauma population, invasive ICP monitoring was not used in this population in 20% of children with an admission GCS of 3 who died within the first 6 h in 90% of the cases. Early TCD examination was performed by physicians with extensive experience of this method, and moreover, in patients who could be minimally stabilized during the early phase of resuscitation, eliminating the confounding factor of altered cerebral blood flow velocity related to severe hypotension, profound anemia, or hypo/hypercapnia. Finally, all the intended TCD examination could be performed successfully within the first 20 min after admission without major technical difficulties and none depicted a “no-flow” profile that could be reported to the inability of the operator to identify middle cerebral artery. To our knowledge, the ability of first-line TCD, performed within minutes in the resuscitation room, before CT scan, and ICP transducer insertion, to identify patients at risk of initial increased ICP, has not been reported previously.
We can conclude that TCD examination is a safe, reproducible, and reliable method to identified children at increased risk of ICH and decreased CPP after severe TBI, and its use should be encouraged in PICU. With 94% sensitivity, TCD demonstrate the possibility of screening patients with high levels of ICP, and the negative predictive value of 95% demonstrates its ability to identify patients with normal ICP. It could be used as a first-line non-invasive method and unlike invasive ICP monitoring, it could be performed within the first minutes following admission before CCT scan examination. Other studies must be performed to verify the possibility of its use as an alternative to systematic invasive ICP monitoring.
References
Anderson VA, Catroppa C, Haritou F, Morse S, Pentland L, Rosenfeld J, Stargatt R (2001) Predictors of acute child and family outcome following traumatic brain injury in children. Pediatr Neurosurg 34:138–348
Chiaretti A, Piastra M, Pulitano S, Pietrini D, De Rosa G, Barbaro R, Di Rocco C (2002) Prognostic factors and outcome of children with severe head injury: an 8-year experience. Childs Nerv Syst 18:129–136
Melo JRT, Silva RA, Moreira ED Jr (2004) Characteristics of patients with head injury at Salvador City (Bahia-Brazil). Arq Neuropsiquiatr 62:711–714
Melo JRT, de Santana DL, Pereira JL, Ribeiro TF (2006) Traumatic brain injury in children and adolescents at Salvador City, Bahia, Brazil. Arq Neuropsiquiatr 64:994–996
Meyer P, Legros C, Orliaguet G (1999) Critical care management of neurotrauma in children: new trends and perspectives. Childs Nerv Syst 15:732–739
Orliaguet GA, Meyer PG, Blanot S, Jarreau MM, Charron B, Buisson C, Carli PA (1998) Predictive factors of outcome in severely traumatized children. Anesth Analg 87:537–542
Orliaguet GA, Meyer PG, Baugnon T (2008) Management of critically ill children with traumatic brain injury. Paediatr Anaesth 18:455–461
Ricard-Hibon A, Marty J (2000) Management of severe head-injured patients in the first 24 h. Resuscitation and initial diagnostic strategy. Ann Fr Anesth Rèanim 19:286–295
Ter MA, Dube L, Guilleux AM, Wehrmann N, Ursino M, Beydon L (2002) Changes in intracranial pressure and cerebral autoregulation in patients with severe traumatic brain injury. Crit Care Med 30:1616–1622
Bellner J, Romner B, Reinstrup P, Kristiansson KA, Ryding E, Brandt L (2005) Transcranial Doppler reflects intracranial pressure. Lakartidningen 102:840–841
Meyer PG, Ducrocq S, Carli P (2001) Pediatric neurologic emergencies. Curr Opin Crit Care 7:81–87
Ng SC, Poon WS, Chan MT (2006) Cerebral hemisphere asymmetry in cerebrovascular regulation in ventilated traumatic brain injury. Acta Neurochir Suppl 96:21–23
Safin AM, Mandorskii SV, Parfenov AL, Oshorov AV (2007) Cerebral circulatory disorders in varying brain injury, as evidenced by transcranial Doppler study. Zh Vopr Neirokhir Im N N Burdenko 2:16–20
Trabold F, Meyer PG, Blanot S, Carli PA, Orliaguet GA (2004) The prognostic value of transcranial Doppler studies in children with moderate and severe head injury. Intensive Care Med 30:108–112
Visocchi M (2008) Hyper flow and intracranial hypertension in diffuse axonal injury: an update to gennarelli doctrine. Acta Neurochir Suppl 101:137–140
Philip S, Chaiwat O, Udomphorn Y, Moore A, Zimmerman JJ, Armstead W, Vavilala MS (2009) Variation in cerebral blood flow velocity with cerebral perfusion pressure >40 mm Hg in 42 children with severe traumatic brain injury. Crit Care Med 37:2973–2978
Vavilala MS, Lee LA, Boddu K, Visco E, Newell DW, Zimmerman JJ, Lam AM (2004) Cerebral autoregulation in pediatric traumatic brain injury. Pediatr Crit Care Med 5:257–263
Meyer PG, Ducrocq S, Rackelbom T, Orliaguet G, Renier D, Carli P (2005) Surgical evacuation of acute subdural hematoma improves cerebral hemodynamics in children: a transcranial Doppler evaluation. Childs Nerv Syst 21:133–137
Udomphorn Y, Armstead WM, Vavilala MS (2008) Cerebral blood flow and autoregulation after pediatric traumatic brain injury. Pediatr Neurol 38:225–234
Trabold F, Meyer P, Orliaguet G (2002) Severe head injuries in the young child: early management. Ann Fr Anesth Rèanim 21:141–147
Grinkeviciute DE, Kevalas R, Matukevicius A, Ragaisis V, Tamasauskas A (2008) Significance of intracranial pressure and cerebral perfusion pressure in severe pediatric traumatic brain injury. Medicina (Kaunas) 44:119–125
Nordstrom CH, Reinstrup P, Xu W, Gardenfors A, Ungerstedt U (2003) Assessment of the lower limit for cerebral perfusion pressure in severe head injuries by bedside monitoring of regional energy metabolism. Anesthesiology 98:809–814
Bardella IJ (1999) Pediatric advanced life support: a review of the AHA recommendations. American Heart Association. Am Fam Physician 60:1743–1750
Sviri GE, Newell DW, Lewis DH, Douville C, Ghodke B, Chowdhary M, Lam AM, Haynor D, Zaaroor M, Britz GW (2006) Impact of basilar artery vasospasm on outcome in patients with severe cerebral vasospasm after aneurysmal subarachnoid hemorrhage. Stroke 37:2738–2743
Takahashi K, Hayashi S, Ootani T, Sera M, Negishi M (2008) Dural arteriovenous fistula manifesting as subarachnoid hemorrhage at the craniocervical junction. A case report. No Shinkei Geka 36:901–906
Jesus PA, Vieira-de-Melo RM, Reis FJ, Viana LC, Lacerda A, Dias JS, Oliveira-Filho J (2006) Cognitive dysfunction in congestive heart failure: transcranial Doppler evidence of microembolic etiology. Arq Neuropsiquiatr 64:207–210
Oliveira-Filho J, Ezzeddine MA, Segal AZ, Buonanno FS, Chang Y, Ogilvy CS, Rordorf G, Schwamm LH, Koroshetz WJ, McDonald CT (2001) Fever in subarachnoid hemorrhage: relationship to vasospasm and outcome. Neurology 56:1299–1304
Czosnyka M, Czosnyka Z, Keong N, Lavinio A, Smielewski P, Momjian S, Schmidt EA, Petrella G, Owler B, Pickard JD (2007) Pulse pressure waveform in hydrocephalus: what it is and what it isn’t. Neurosurg Focus 22:E2
Riggo JD, Kolarovszki B, Richterova R, Kolarovszka H, Sutovsky J, Durdikc P (2007) Measurement of the blood flow velocity in the pericallosal artery of children with hydrocephalus by transcranial Doppler ultrasonography—preliminary results. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub 151:285–289
Rodriguez-Nunez A, Somoza-Martin M, Gomez-Lado C, Eiris-Punal J, Camina-Darriba F, Rodriguez-Segade S, Castro-Gago M (2008) Therapeutic criteria in communicating childhood hydrocephalus. J Neurosurg Sci 52:17–21
Ducrocq SC, Meyer PG, Orliaguet GA, Blanot S, Laurent-Vannier A, Renier D, Carli PA (2006) Epidemiology and early predictive factors of mortality and outcome in children with traumatic severe brain injury: experience of a French pediatric trauma center. Pediatr Crit Care Med 7:461–467
Javouhey E, Guerin AC, Martin JL, Floret D, Chiron M (2009) Management of severely injured children in road accidents in France: impact of the acute care organization on the outcome. Pediatr Crit Care Med 10:472–478
Bekar A, Taskapilioglu O, Yilmazlar S, Ender K, Aksoy K (2008) Is supratentorial pressure difference clinically relevant? Analysis of 55 consecutive cases by bilateral intracranial pressure monitoring. Neurol Res 30:465–470
Bekar A, Dogan S, Abas F, Caner B, Korfali G, Kocaeli H, Yilmazlar S, Korfali E (2009) Risk factors and complications of intracranial pressure monitoring with a fiberoptic device. J Clin Neurosci 16:236–240
Chambers IR, Treadwell L, Mendelow AD (2001) Determination of threshold levels of cerebral perfusion pressure and intracranial pressure in severe head injury by using receiver-operating characteristic curves: an observational study in 291 patients. J Neurosurg 94:412–416
Chambers IR, Jones PA, Lo TY, Forsyth RJ, Fulton B, Andrews PJ, Mendelow AD, Minns RA (2006) Critical thresholds of intracranial pressure and cerebral perfusion pressure related to age in paediatric head injury. J Neurol Neurosurg Psychiatry 77:234–240
Figaji AA, Zwane E, Fieggen AG, Siesjo P, Peter JC (2009) Transcranial Doppler pulsatility index is not a reliable indicator of intracranial pressure in children with severe traumatic brain injury. Surg Neurol 72:389–394
Visocchi M, Chiaretti A, Genovese O, Di Rocco F (2007) Haemodynamic patterns in children with posttraumatic diffuse brain swelling. A preliminary study in 6 cases with neuroradiological features consistent with diffuse axonal injury. Acta Neurochir (Wien) 149:347–356
Murillo-Cabezas F, Arteta-Arteta D, Flores-Cordero JM, Munoz-Sanchez MA, Rincon-Ferrari MD, Rivera-Fernandez MV, Alacron-Cruz JC (2002) The usefulness of transcranial Doppler ultrasonography in the early phase of head injury. Neurocirugia (Astur) 13:196–208
Muttaqin Z, Uozumi T, Kuwabara S, Arita K, Kurisu K, Ohba S, Kohno Ogasawara H, Ohtani M, Mikami T (1993) Hyperaemia prior to acute cerebral swelling in severe head injuries: the role of transcranial Doppler monitoring. Acta Neurochir (Wien) 123:76–81
Hindy-Francois C, Orliaguet G, Meyer P, Carli P, Blanot S, Hertz-Pannier L, Brunelle F (2009) Pediatric brain death diagnosis in the view of organ donation in France. Transplantation 87:616–617
Acknowledgement
CAPES-Brazil (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior) funded Jose Roberto Tude Melo’s studies abroad.
Conflict of Interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Melo, J.R.T., Di Rocco, F., Blanot, S. et al. Transcranial Doppler can predict intracranial hypertension in children with severe traumatic brain injuries. Childs Nerv Syst 27, 979–984 (2011). https://doi.org/10.1007/s00381-010-1367-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-010-1367-8