Abstract
Introduction
Given the large number of cerebral palsy patients who have undergone selective dorsal rhizotomy in the past two decades, it is clearly imperative that the clinical community be provided with objective and compelling evidence of the long-term sequelae of the procedure.
Materials and methods
In the early 1980s, Peacock in Cape Town shifted the site of the rhizotomy from the conus medullaris to the cauda equina, and in the past 25 years, more than 200 children have been operated on. We have studied the incidence of spinal deformities after multiple-level laminectomy and recorded a 20% incidence of isthmic spondylolysis or grade-I spondylolisthesis. We have also conducted a long-term prospective gait analysis study on a cohort of 14 ambulatory patients who were operated on in 1985.
Results
Ten years after surgery, our patients had increased ranges of motion that were within normal limits. Step length was significantly improved, although cadence was unchanged postoperatively and was significantly less than normal age-matched control subjects.
Discussion
We have recently tracked down all 14 patients from the original cohort and are currently completing a 20-year prospective follow-up analysis of their neuromuscular function and gait. Our preliminary data suggest that selective dorsal rhizotomy is not only an effective method for alleviating spasticity but it also leads to long-term functional benefits.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
With increased survival of low-birth-weight infants and increased longevity of the adult population, the number of adults with cerebral palsy (CP) is increasing. Klingbeil et al. [10] reviewed the problems of aging in several groups of individuals with disabilities, including CP. Although CP is normally described as a nonprogressive disorder, cumulative evidence over the past two decades supports the patients’ perspective of functional deterioration associated with aging. There are extra concerns regarding selective dorsal rhizotomy (SDR) and its long-term effects on the potential production of muscle weakness, the possible development of spinal deformities, and the continuing need for orthopedic intervention [2]. Given the large number of CP patients who have undergone SDR in the past two decades, particularly in the USA, it is clearly imperative that the clinical community be provided with objective and compelling evidence of the long-term sequelae of the procedure. With our long record in Cape Town, we are well placed to provide that evidence.
Materials and methods
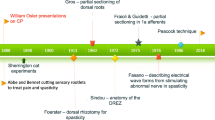
Although dorsal rhizotomy was described almost 100 years ago by the German surgeon Foerster [6], it fell into disuse for more than 50 years because of the adverse consequences of deafferentation. In the 1960s, the procedure was revised by sectioning a fraction of the rootlets [7] and then further refined into SDR by electrically stimulating the rootlets and measuring the electromyographic response before cutting the rootlets [5]. In Cape Town, Peacock and Eastman [15] shifted the site of rhizotomy from the conus medullaris region to the cauda equina and reintroduced the technique in the early 1980s for treating children with CP.
Having performed more than 100 such operations, Peacock relocated to Los Angeles in 1986, and his contribution led to the use of SDR becoming widespread in the USA [1, 12, 14, 19]. Since Peacock’s departure in 1986, SDR has continued in Cape Town, first conducted by Jonathan Peter and more recently by Graham Fieggen. In the past 25 years, more than 200 children have undergone SDR in Cape Town.
Peter et al. [17] studied the incidence of spinal deformities in children after multiple level laminectomy for SDR and concluded that spondylolysis was the only abnormality that appeared to be more common in patients who underwent the procedure compared with those who did not have SDR. They felt that it was essential to maintain an ongoing follow-up of the spines of postrhizotomy patients, both to monitor the children identified with spondylosis and spondylolisthesis and also to determine the true incidence of this condition in the whole SDR group. They also sought to determine whether these conditions increased with time. In their study, which included 163 children, the patients had five-level lumbosacral laminectomies, and 99 were X-rayed postoperatively. Of these, 20% developed incidental isthmic spondylololysis or grade-I spondylolisthesis, leading Peter et al. [16] to postulate that the laminectomy, associated lordosis and increased mobility after SDR, may all be causative factors.
In an effort to reduce the problems of lumbar instability [17], some surgeons have adopted a more limited laminectomy at the level of the conus as advocated by Park [13]. There is also some controversy as to the number of rootlets that should be sectioned, where less than 25% can lead to limited functional gains [11], but Kim et al. [9] have also suggested that more than 50% can also be problematic.
In 1985, Berman et al. [4] introduced functional assessments that were conducted preoperatively and 1 year postsurgery. Because the Gross Motor Function Measure scoring system had not yet been developed [18], they proposed their own rating scales [3]. These scales were designed to test muscle tone, voluntary movement, joint range of motion, and functional movement in certain developmental positions. The results of this study indicated positive gains after SDR, although this was dependent on the patients’ abilities before surgery [3, 4]. One of the greatest gains made was in the patients’ ability to assume and maintain certain postures, which, in turn, allowed for improved participation in activities. It was also obvious that once spasticity had been removed, the CP patients had greater freedom of movement [4].
Twenty years ago, the Cape Town team, led by Vaughan, was the first group to study the effects of SDR with gait analysis. In their original study, 14 ambulatory patients were involved in a gait analysis follow-up study 1 year after surgery [22, 23]. The study was then repeated on a cohort of 11 patients 2 years later as a 3-year follow-up study [21] and also 10 years postoperatively [20, 24]. The parameters they used to substantiate their findings were joint kinematics (hip and knee ranges of motion and hip and knee midrange values) and temporal-distance parameters (step length, cadence, and velocity). To accommodate musculoskeletal growth, the temporal-distance parameters were normalized to yield dimensionless numbers according to the method of Hof [8].
Results
Figure 1 presents the ranges of motion and midrange (i.e., the angle of the knee when the leg is at its midpoint in stance phase) values for the knee and hip joints. The range of motion at the knee and hip increased as a result of surgery, and by 10 years, there was no statistical difference from normal ranges. Concomitant with the increased ranges of motion, there was also the deleterious increase in midrange values immediately after surgery. This was of some concern because the midrange values represent a measure of strength and control exhibited by a patient during locomotion. The greater the midrange value, the more flexed the posture of the individual, and this resulted in an exaggerated crouch gait. However, these midrange values decreased over time, so that by 10 years postoperatively, there was no variance from normal values. Thus, 10 years after surgery, patients not only had increased ranges of motion that were within normal limits, but they also used that functional movement at approximately a normal midrange point [20].
The mean data for the normalized temporal-distance parameters of the 10-year follow-up study are presented in Fig. 2 [20]. The dimensionless cadence (step frequency) was unchanged postoperatively and was always significantly less than normal (Fig. 2a). Because cadence is likely to be centrally mediated and SDR attacks the problem at the level of the spinal cord, it is not surprising that the procedure has a limited influence on this parameter [24]. Figure 2 also shows the benefits of SDR in significantly improved step length as a natural consequence of the release of spasticity and the increase in knee range of motion and hip range of motion. Ten years after surgery, there is a small deterioration, although this may have resulted from the fact that 5 of the 11 subjects were videotaped in a confined room that was not conducive to attaining a standard gait. Velocity is the product of cadence and step length and shows a trend that is comparable to step length after surgery because cadence does not change postoperatively [20].
Dimensionless temporal-distance parameters: a cadence or step frequency, b step length, and c velocity, for 11 subjects before selective dorsal rhizotomy, and at 1, 3, and 10 years after surgery compared with normal controls [20, 24]. Note that the parameters have all been normalized according to Hof [8]
Discussion
In line with our original functional assessment and gait analysis studies performed in 1985, our Cape Town research group has recently conducted a 20-year prospective follow-up study. We have repeated the tests used in our previous functional [3, 4] and gait [20, 21, 23] studies, and the data are currently being analyzed. We were able to track down the original cohort of 14 spastic diplegic CP patients who underwent SDR by Peacock. The mean age of these 14 patients in 2005 was 27.9 years (range 22.1–33.0) with a gender distribution of six women and eight men.
Besides participating in the functional assessment and gait studies, we requested the 14 patients to complete a questionnaire regarding details about their past and present life, health status, and their outlook on SDR. Twelve of the cohort had a positive outlook on SDR, while two had mixed feelings. Both of them were suffering from muscle weakness postoperatively, which resulted in one of them becoming wheelchair bound. Although all patients received physical therapy at school preoperatively, more than half of the group needed tendon lengthening before SDR. After rhizotomy, another four underwent this orthopedic procedure, while half of them required osteotomy. More than half of the cohort was suffering from some back pain, while two of the patients walked with a crutch and cane. In answer to the question “Do you need help with activities of daily living?” all 14 patients answered “No.” Only two of them were not employed or studying at college. We established that besides medical intervention, it is very important that the patients have a good understanding of their specific motor problems and that they are supported by family and friends who help to motivate them.
From this series of follow-up studies in Cape Town, it has been shown that SDR is an effective method for alleviating spasticity, and the team has provided evidence to show that functional benefits, as measured by improved gait, can also be obtained [20]. Our Cape Town research group would appear to be the only authors to have published long-term (greater than 5 years) gait analysis studies on the efficacy of SDR.
References
Abbott R, Forem SL, Johann M (1989) Selective posterior rhizotomy for the treatment of spasticity: a review. Child’s Nerv Syst 5:337–346
Aiona MD, Sussman MD (2004) Treatment of spastic diplegia in patients with cerebral palsy: Part II. J Pediatr Orthop B 13:S13–S38
Berman B (1989) Selective posterior rhizotomy: does it do any good? Neurosurg State Art Rev 4:431–444
Berman B, Vaughan CL, Peacock WJ (1990) The effect of rhizotomy on movement in patients with cerebral palsy. Am J Occup Ther 44:511–516
Fasano VA, Barolat-Romana G, Ivaldi A, Sguazzi A (1976) La radicotomie postérieure fonctionnelle dans le traitement de la spasticité cérébrale. Neurochirurgie 22:23–34
Foerster O (1913) On the indications and results of the excision of posterior nerve roots in men. Surg Gynecol Obstet 6:463–474
Gros C, Ouaknine G, Vlahovitch B, Frerebeau P (1967) La radicotomie sélective postérieure dans le traitment neuro-chirurgical de l’hypertonie pyramidale. Neurochirurgie 13:505–518
Hof AL (1996) Scaling gait data to body size. Gait Posture 4:222–223
Kim DS, Choi JU, Yang KH, Park CI, Park ES (2002) Selective posterior rhizotomy for lower extremity spasticity: how much and which of the posterior rootlets should be cut? Surg Neurol 57:87–93
Klingbeil H, Baer HR, Wilson PE (2004) Aging with a disability. Arch Phys Med Rehabil 85:S68–S73
McLaughlin JF, Bjornson KF, Astley SJ, Hays RM, Hoffinger SA, Armantrout EA, Roberts TS (1994) The role of selective dorsal rhizotomy in cerebral palsy: critical evaluation of a prospective clinical series. Dev Med Child Neurol 36:755–769
Oppenheim WL (1990) Selective posterior rhizotomy for spastic cerebral palsy. A review. Clin Orthop Relat Res Apr(253):20–29
Park TS (2000) Selective dorsal rhizotomy: an excellent therapeutic option for spastic cerebral palsy. Clin Neurosurg 47:422–439
Park TS, Owen JH (1992) Surgical management of spastic diplegia in cerebral palsy. N Engl J Med 326:745–749
Peacock WJ, Eastman RW (1981) The neurosurgical management of spasticity. S Afr Med J 60:849–850
Peter JC, Hoffman EB, Arens LJ, Peacock WJ (1990) Incidence of spinal deformity in children after multiple level laminectomy for selective posterior rhizotomy. Child’s Nerv Syst 6:30–32
Peter JC, Hoffman EB, Arens LJ (1993) Spondylolysis and spondylolisthesis after five-level lumbosacral laminectomy for selective posterior rhizotomy in cerebral palsy. Child’s Nerv Syst 9:285–287
Russell D, Rosenbaum P, Cadman D, Gowland C, Hardy S, Jarvis S (1989) The Gross Motor Function Measure: a means to evaluate the effects of physical therapy. Dev Med Child Neurol 31:341–352
Steinbok P (2001) Outcomes after selective dorsal rhizotomy for spastic cerebral palsy. Child’s Nerv Syst 17:1–18
Subramanian N, Vaughan CL, Peter JC, Arens LJ (1998) Gait before and 10 years after rhizotomy in children with cerebral palsy spasticity. J Neurosurg 88:1014–1019
Vaughan CL, Berman B, Peacock WJ (1991) Cerebral palsy and rhizotomy. A 3-year follow-up evaluation with gait analysis. J Neurosurg 74:178–184
Vaughan CL, Berman B, Peacock WJ, Eldridge NE (1989) Gait analysis and rhizotomy: past experience and future considerations. Neurosurg State Art Rev 4:445–458
Vaughan CL, Berman B, Staudt LA, Peacock WJ (1988) Gait analysis of cerebral palsy children before and after rhizotomy. Pediatr Neurosci 14:297–300
Vaughan CL, Subramanian N, Busse ME (1998) Selective dorsal rhizotomy as a treatment option for children with spastic cerebral palsy. Gait Posture 8:43–59
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Langerak, N.G., Lamberts, R.P., Fieggen, A.G. et al. Selective dorsal rhizotomy: long-term experience from Cape Town. Childs Nerv Syst 23, 1003–1006 (2007). https://doi.org/10.1007/s00381-007-0383-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-007-0383-9