Abstract
The occurrence of deteriorated coronary flow associated with distal embolization during percutaneous coronary intervention results in impaired myocardial perfusion and worsens the clinical prognosis. This study aimed to examine the impact of optical coherence tomography (OCT)-determined coronary plaque morphology on the prediction of deteriorated coronary flow after stent implantation in acute as well as stable coronary syndromes (ACS and SAP, respectively). We studied 126 patients who underwent OCT during stenting for ACS (n = 44) and SAP (n = 82) with a de novo lesion. Angiographic deteriorated coronary flow was defined as the deterioration of TIMI flow grade after mechanical dilatation in the absence of a mechanical obstruction on angiograms. Patients could be divided into the deteriorated flow group (n = 21) and the reflow group (n = 105). Under these conditions, the presence of thin-cap fibroatheroma (TCFA) was more frequently observed in the deteriorated flow group than in the reflow group in both ACS and SAP. A multivariable logistic regression model revealed that TCFA was an independent predictor of deteriorated coronary flow (hazard ratio: 12.32; 95 % confidence interval: 3.02–50.31; p = 0.0005). These results demonstrate that TCFA detected by OCT could be a strong predictor of the occurrence of deteriorated coronary flow during stent implantation in ACS as well as SAP.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
With the increased use of percutaneous coronary intervention (PCI), there is an increase in the number of deteriorated coronary flow cases, including no-reflow or slow-flow phenomena, which are associated with poor functional recovery and adverse clinical outcomes in patients with acute as well as stable coronary syndromes (ACS and SAP, respectively) [1–3]. Moreover, transient no-reflow or filter no-reflow are related to myocardial damage and adverse outcomes [4, 5]. Therefore, prediction and prevention of deteriorated coronary flow is essential at the time of coronary interventions because this can be prevented by some distal protecting devices [6, 7].
Several studies demonstrated that in addition to clinical factors [8], pre-intervention intravascular ultrasound (IVUS) findings such as intracoronary thrombus, lipid pool, positive remodeling, attenuated plaque, and necrotic core volume [9–13] may be related to the occurrence of deteriorated coronary flow after PCI. Recently, we demonstrated that scattered necrotic core pattern, which might be equivalent to thin-cap fibroatheroma (TCFA) by virtual histology-IVUS, is highly associated with the no-reflow phenomenon during intervention for ACS [14, 15].
Optical coherence tomography (OCT), which is a high-resolutional imaging modality capable of characterizing important morphological features of atherosclerotic plaque such as TCFA or thrombus [16], has been widely used in a clinical setting. However, few data exist regarding OCT demonstration of atherosclerotic plaque, which can cause deteriorated coronary flow during PCI. Therefore, we examined the impact of OCT on the prediction of the deterioration of coronary flow during intervention in ACS as well as SAP.
Materials and methods
Study population
The study was approved by the hospital ethics committee of Ishikawa Prefectural Central Hospital. Written informed consent was obtained from all patients. We enrolled 141 consecutive patients undergoing OCT-guided PCI for a de novo lesion in the native coronary artery. ACS included acute myocardial infarction and unstable angina pectoris [17]. Acute myocardial infarction was diagnosed from ST-segment change and elevated creatine kinase (>3 times above the upper limit of normal), whereas unstable angina pectoris was defined as angina with a progressive crescendo pattern or angina at rest without an increase of creatine kinase. SAP was defined as no change in frequency, duration, or intensity of anginal symptoms before intervention [18]. Under these conditions, we studied 141 patients including 52 with ACS and 89 with SAP.
Study protocol
All patients received intravenous heparin (8000 IU) and intracoronary isosorbide dinitrate (2 mg) before diagnostic angiography and were administered intravenous heparin (2000–3000 IU) every one hour during the procedure. Platelet glycoprotein IIb/IIIa receptor inhibitors were not used because they are not available in Japan. PCI was performed using a 6 Fr guiding catheter.
After completion of diagnostic angiography, the culprit lesion was observed first by OCT (M2, LightLab Imaging, Westford, MA, USA). The culprit lesion was identified on the basis of left ventricular wall motion abnormalities, ECG findings, and angiographic lesion morphology. If a large amount of thrombus was found at the culprit lesion, thrombectomy was performed before OCT. In all patients, a conventional PCI guide wire (0.014 in.) was advanced across the lesion that was exchanged for an OCT Image wire using a microcatheter (Excelsior, Boston Scientific, Natick, MA, USA).
A continuous flushing, non-occlusive method was used for OCT image acquisition. To flush the blood in the vessel, a mixture of commercially available Dextran 40 (Low Molecular Dextran L, Otsuka Pharmaceutical Factory, Tokushima, Japan) and lactated Ringer’s solution were administered directly from the guiding catheter at a rate of 2.5–4.5 mL/s using an injector pump. The OCT Imagewire was pulled back automatically at a rate of 1 mm/s from the distal to proximal side of the culprit lesion in all cases.
Then, conventional PCI was performed with balloon dilatation before and/or after stent implantation. A filter-based distal protection device (Filtrap, Japan Lifeline, Tokyo, Japan) was used when thrombus was detected by angiogram or suitable coronary anatomy for delivery. The angiographic end point was <25 % of residual stenosis on coronary angiography. Under these conditions, 100 mg/day of aspirin plus 75 mg/day of clopidogrel or 200 mg/day of ticlopidine was administered to all patients with loading dose as needed. Creatine kinase levels were measured just after the procedure and the next day in patients with SAP, and every 6 h during the first day in patients with ACS.
Angiographic analysis
The angiographic findings in this study were assessed by an investigator blinded to the clinical data. Quantitative coronary angiography was performed off-line using an automated edge detection system (Cardiovascular Measurement System; Goodman, Nagoya, Aichi, Japan). From the quantitative angiographic images, reference diameter, minimal lumen diameter, percent diameter stenosis, and lesion length of the culprit lesion were measured. American College of Cardiology/American Heart Association lesion type was also assessed.
Coronary flow was assessed according to the thrombolysis in myocardial infarction (TIMI) criteria [19]. In this study, TIMI flow grade 3 was observed in all ACS patients just after thrombectomy. Angiographic deteriorated coronary flow or filter no-reflow [20] was defined as the deterioration of TIMI flow grade after mechanical dilatation, such as pre-dilatation or stenting compared to TIMI flow grade at initial or post thrombectomy in the absence of residual stenosis <25 %, dissection, or spasm.
Optical coherence tomography images analysis
The OCT findings in this study were assessed by an investigator blinded to the clinical and angiographic data. OCT findings, such as ruptured plaque, thrombus, calcification, TCFA, lipid-rich plaque, and lipid quadrants, were evaluated as previously reported [21]. Ruptured plaque was defined as the presence of fibrous cap discontinuity and formation of a cavity in the plaque. Thrombus was identified as a mass protruding into the vessel lumen from the surface of the vessel wall. Lipid-rich plaque was defined as lipid ≥2 quadrants. TCFA was defined as a lipid-rich plaque with fibrous cap thickness measuring <65 μm (Fig. 1). Lipid was semi-quantified as the number of involved quadrants on cross-sectional images.
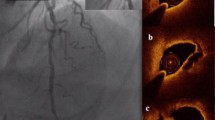
Coronary angiograms and optical coherence tomographic images in a case of acute coronary syndrome. a An initial angiography shows severe stenosis in the proximal right coronary artery (white arrow). Distal flow of the right coronary artery is well preserved. b Optical coherence tomography at the stenotic lesion (white arrow in a). c Demonstrates the presence of thrombus and a typical thin-cap fibroatheroma. d After aspiration of thrombus as much as possible, the stent is deployed dilating the proximal right coronary artery. Under these conditions, filter no-reflow is observed (black arrow)
Statistical analysis
Statistical analyses were performed with Stat View software version 5.0 (SAS Institute, Cary, NC, USA). Continuous variables are expressed as the mean ± standard deviation. Qualitative variables are presented as numbers and percentages. Continuous variables were compared using the Student’s t test or the Mann–Whitney U test, and categorical data using the Chi-square test or Fisher’s exact test, as appropriate. Multivariate logistic regression analysis was performed to identify independent predictors of deterioration of coronary flow after stenting. This model included univariate predictors with p < 0.05. If the factor was strongly correlated with others, clinically more meaningful factors were chosen (ACS, current smoking, American College of Cardiology/American Heart Association lesion type B2/C, reference diameter, thrombus, TCFA). A value of p < 0.05 was considered statistically significant.
Results
Patient characteristics and clinical results
Of 141 patients, 15 patients who could not receive OCT with satisfactory image quality were excluded: 5 had a large burden of the thrombus, 2 had a large vascular diameter, and 8 had poor image quality. Finally, 126 patients (94 males, mean age 66.8 ± 11.5 years), including 44 culprit lesions for ACS and 82 culprit lesions for SAP, were analyzed. We divided the 126 patients into two groups, the deteriorated flow group and the reflow group. ACS consisted of 13 patients in the deteriorated flow group and 31 patients in the reflow group, and SAP consisted of 8 patients in the deteriorated flow group and 74 patients in the reflow group. Patient characteristics and clinical results are summarized in Table 1.
In ACS patients, the frequency of acute myocardial infarction was significantly greater in the deteriorated flow group than in the reflow group. Peak creatine kinase levels after PCI was significantly higher in the deteriorated flow group than in the reflow group, although baseline levels were similar between both groups. In contrast, there were no significant differences in these parameters in the SAP group. When we compared the creatine kinase levels only in patients with acute myocardial infarction (deteriorated flow group 12 patients, normal reflow group 16 patients), baseline and peak creatine kinase levels were not significantly different among the groups (baseline creatine kinase; 384 ± 908 IU/L vs 573 ± 746 IU/L, p = 0.55, peak creatine kinase; 2697 ± 2510 IU/L vs 1942 ± 1987 IU/L, p = 0.38, respectively). In all patients, in addition to previous factors, the presence of current smoker, unused aspirin, unused ticlopidine or clopidogrel, and unused angiotensin-converting enzyme inhibitor or angiotensin receptor blocker were significantly greater in the deteriorated flow group than in the reflow group. On the other hand, history of myocardial infarction and interventional procedures were significantly smaller in the deteriorated flow group than in the reflow group.
Angiographic findings
Pre-procedural angiographic findings are summarized in Table 2. In patients with ACS, there was a trend toward a higher frequency of low TIMI flow grade before the procedure in the deteriorated flow group than in the reflow group. In all patients, complicated lesions were significantly greater in the deteriorated flow group than in the reflow group. The reference diameter and percent diameter stenosis were significantly greater, and minimal luminal diameter was significantly smaller in the deteriorated flow group than in the reflow group. In deteriorated coronary flow cases, all patients with a distal protection device had a TIMI flow grade 3 just after the distal protection device was removed. On the other hand, of 8 patients without a distal protection device, 2 could not finally have a TIMI flow grade 3 after the procedure. Furthermore, a hospital death occurred in one of the 2 patients and in none of the patients with a distal protection device.
Procedural results
Balloon dilation or stenting was performed in all patients. PCI was performed through the radial (n = 110), brachial (n = 13) or femoral (n = 3) approach. Procedural results are summarized in Table 3. The use of thrombectomy was significantly higher in the deteriorated flow group than in the reflow group. Stent diameter was significantly greater in the deteriorated flow group than in the reflow group.
Optical coherence tomography findings
In all patients, the presence of thrombus and TCFA were significantly greater in the deteriorated flow group than in the reflow group (Fig. 1). Minimum fibrous cap thickness was significantly smaller in the deteriorated flow group compared with the reflow group. The presence of TCFA was significantly greater in the deteriorated flow group than in the reflow group for patients with ACS and SAP (Table 4). Interestingly, multivariate analysis identified current smoker, reference diameter, and TCFA as independent predictors of deterioration of TIMI flow grade (Table 5). When we analyzed predictors of deterioration of TIMI flow grade after exclusion of patients with distal protection device, the presence of TCFA was an only independent predictor of deterioration of TIMI flow grade (hazard ratio: 6.47; 95 % confidence interval: 1.26–33.3; p = 0.02). No serious procedural complications were observed during OCT examination.
Discussion
Several studies suggested that pre-intervention IVUS findings such as intracoronary thrombus, lipid pool, positive remodeling, and attenuated plaque were related to the occurrence of deteriorated coronary flow such as no-reflow or slow-flow phenomena after PCI [9–12]. A virtual histology-IVUS paper reported that necrotic core volume was also related to the no-reflow phenomenon [13]. However, a study reported that patients with the no-reflow phenomenon had more fibrotic or fibro-fatty volumes, but not necrotic core volumes [22]. Therefore, there still exist controversies regarding the association between plaque components detected by virtual histology-IVUS and no-reflow or slow-flow phenomena after PCI.
The OCT is a high-resolutional imaging modality capable of characterizing important morphological features of atherosclerotic plaque, such as the minimal fibrous cap thickness or thrombus which cannot be identified in IVUS. The present study showed a significant relationship between TCFA observed by OCT and deterioration of coronary flow in both ACS and SAP patients. In addition, minimum fibrous cap thickness was significantly smaller in the deteriorated flow group compared with the reflow group in our study. From these findings, we speculate that TCFA contains a large lipid-rich plaque covered with a thinner fibrous cap that can easily be disrupted, and once TCFA is destroyed mechanically by balloon dilatation or stenting, a large amount of debris is occluded in the distal end of the vessel. In humans, the no-reflow phenomenon has a multifocal pathogenesis. In addition to angioplasty-induced mechanical embolization, an inflammatory response induced by neutrophils and platelets that occurs at the time of reperfusion may also exacerbate coronary flow [23]. This inflammatory process may affect the result of the present study less because TIMI flow grade 3 was observed in all patients just after thrombectomy.
Some studies demonstrated that patients with an intracoronary thrombus are prone to distal embolization after PCI [24]. Indeed, previous IVUS studies showed that intracoronary thrombus was related to the no-reflow phenomenon after PCI [9]. In our study, although thrombectomy was performed to remove thrombus as much as possible before OCT procedure to minimize the effect of thrombus, residual thrombus was observed in many ACS patients because OCT can easily detect thrombus which might not be detected by IVUS and angiogram. Multivariate analysis revealed that the presence of residual thrombus was not an independent predictor of deteriorated coronary flow. Therefore, TCFA may be a more important finding than thrombus to identify the lesion developing deterioration of coronary flow.
Multivariate analysis also demonstrated that deterioration of coronary flow was predicted by reference diameter. In the present study, reference diameter, percent diameter stenosis, and minimal luminal diameter were strongly correlated with each other. Large reference diameter, great percent diameter stenosis, and small minimal luminal diameter in the deteriorated coronary flow group suggest a large plaque burden or positive remodeling which generally includes lipid-rich plaque [11]. Therefore, the mechanical destruction of lipid-rich plaques in the large plaque burden or positive remodeling by PCI may provoke a large amount of debris and cause deteriorated coronary flow, as shown in previous IVUS and computed tomographic angiography studies [25, 26].
The proportion of deteriorated coronary flow in this study was higher than that reported previously [27]. This may be due to including the patients who showed transient flow deterioration with filter no-reflow in the deteriorated flow group in the present study. Previous studies demonstrated that the occurrence of filter no-reflow during PCI was closely related to post-procedural myocardial damage [5]. These results suggest that filter no-reflow could be equivalent to persistent no-reflow phenomenon in terms of clinical events although it is still controversial whether distal protection devices can preserve distal coronary flow during mechanical dilatation [28]. Moreover, in this study the presence of TCFA was an only independent predictor of deteriorated flow even after exclusion of patients with distal protection device, which was similar to an overall result containing patients with distal protection device. Therefore, TCFA may be associated with deteriorated coronary flow during PCI regardless of the use of filter device.
Clinical implications
With the increased use of PCI, we frequently experience no-reflow phenomenon in patients with ACS as well as SAP. The present study revealed that TCFA was an independent predictor of deteriorated coronary flow in both patients. Therefore, a careful interventional procedure is needed in cases with TCFA in culprit lesion regardless of the stability of the disease [29].
In our results, all patients with a distal protection device had TIMI flow grade 3 just after removal of the distal protection device, suggesting that a large amount of debris was captured by the filter device. However, it is still controversial whether distal protection devices can preserve distal coronary flow during mechanical dilatation [28], Additional studies with a larger number of patients are needed to confirm and clarify our results.
Study limitations
There remain several limitations in the present study. First, this study was an observation study from a single center. However, the present data suggest the presence of TCFA by OCT could indicate deteriorated coronary flow during PCI, although further studies with a larger number of patients and prospective are necessary to assess clinical outcomes. Second, thrombectomy prior to OCT was not performed for all patients, which may have biased the results. Also, the presence of residual thrombus at a culprit site may disturb the assessment by OCT. Although we removed thrombus as much as possible before and after OCT observation, minimizing the effect of residual thrombus on the present observation, there remains a possibility that the residual thrombus might affect the occurrence of deteriorated coronary flow. Third, distal protection device was not used for all patients, which may have biased the results. However, when we analyzed predictors of deterioration of TIMI flow grade after exclusion of patients with distal protection device, the presence of TCFA was an only independent predictor of deterioration of TIMI flow grade. Therefore, the impact of distal protection device on the results is minimal. Finally, this study was performed by OCT with time-domain system which has a disadvantage about image quality in comparison with the recent system with frequency-domain version [30]. However, fundamental performance such as axial and lateral resolutions was found to be same among the systems. Therefore, the present observation can provide the information even in the case examined by frequency-domain OCT.
Conclusions
The present study demonstrated that OCT might be useful for identifying culprit plaques at high risk for developing deterioration of coronary flow. Further studies are necessary to clarify whether the use of distal protection based on the present findings can predict clinical outcomes.
References
Ndrepepa G, Tiroch K, Fusaro M, Keta D, Seyfarth M, Byrne RA, Pache J, Alger P, Mehilli J, Schömig A, Kastrati A (2010) 5 year prognostic value of no-reflow phenomenon after percutaneous coronary intervention in patients with acute myocardial infarction. J Am Coll Cardiol 55:2383–2389
Henriques J, Zijlstra F, Ottervanger J, de Boer M, van’t Hof A, Hoorntje J, Suryapranata H (2002) Incidence and clinical significance of distal embolization during primary angioplasty for acute myocardial infarction. Eur Heart J 23:1112–1117
Kimura S, Kakuta T, Yonetsu T, Suzuki A, Iesaka Y, Fujiwara H, Isobe M (2009) Clinical significance of echo signal attenuation on intravascular ultrasound in patients with coronary artery disease. Circ Cardiovasc Interv 2:444–454
Chan W, Stub D, Clark DJ, Ajani AE, Andrianopoulos N, Brennan AL, New G, Black A, Shaw JA, Reid CM, Dart AM, Duffy SJ (2012) Usefulness of transient and persistent no reflow to predict adverse clinical outcomes following percutaneous coronary intervention. Am J Cardiol 109:478–485
Porto I, Belloni F, Niccoli G, Larosa C, Leone AM, Burzotta F, Trani C, De Maria GL, Hamilton-Craig C, Crea F (2011) Filter no-reflow during percutaneous coronary intervention of saphenous vein grafts: incidence, predictors and effect of the type of protection device. EuroIntervention 7:955–961
Umeda H, Katoh T, Iwase M, Izawa H, Nagata K, Watanabe K, Okada T, Yamada T, Tani T, Matsushita T, Murakami Y, Okamoto M, Shimizu T, Murohara T, Yokota M (2006) The distal protection during primary percutaneous coronary intervention alleviates the adverse effects of large thrombus burden on myocardial reperfusion. Circ J 70:232–238
Mizote I, Ueda Y, Ohtani T, Shimizu M, Takeda Y, Oka T, Tsujimoto M, Hirayama A, Hori M, Kodama K (2005) Distal protection improved reperfusion and reduced left ventricular dysfunction in patients with acute myocardial infarction who had angioscopically defined ruptured plaque. Circulation 112:1001–1007
Yundai C, Changhua W, Xinchun Y, Lefeng W, Zhijun S, Hongbin L, Lian C (2012) Independent no-reflow predictors in female patients with ST-elevation acute myocardial infarction treated with primary percutaneous coronary intervention. Heart Vessels 27:243–249
Fukuda D, Tanaka A, Shimada K, Nishida Y, Kawarabayashi T, Yoshikawa J (2003) Predicting angiographic distal embolization following percutaneous coronary intervention in patients with acute myocardial infarction. Am J Cardiol 93:403–407
Tanaka A, Kawarabayashi T, Nishibori Y, Sano T, Nishida Y, Fukuda D, Shimada K, Yoshikawa J (2002) No-reflow phenomenon and lesion morphology in patients with acute myocardial infarction. Circulation 105:2148–2152
Watanabe T, Nanto S, Uematsu M, Ohara T, Morozumi T, Kotani Ji, Nishio M, Awata M, Nagata S, Hori M (2003) Prediction of no-reflow phenomenon after successful percutaneous coronary intervention in patients with acute myocardial infarction -intravascular ultrasound findings-. Circ J 67:667–671
Lee SY, Mintz GS, Kim SY, Hong YJ, Kim SW, Okabe T, Pichard AD, Satler LF, Kent KM, Suddath WO, Waksman R, Weissman NJ (2009) Attenuated plaque detected by intravascular ultrasound: clinical, angiographic, and morphologic features and post-percutaneous coronary intervention complications in patients with acute coronary syndromes. J Am Coll Cardiol Intv 2:65–72
Hong YJ, Jeong MH, Choi YH, Ko JS, Lee MG, Kang WY, Lee SE, Kim SH, Park KH, Sim DS, Yoon NS, Youn HJ, Kim KH, Park HW, Kim JH, Ahn Y, Cho JG, Park JC, Kang JC (2011) Impact of plaque components on no-reflow phenomenon after stent deployment in patients with acute coronary syndrome: a virtual histology-intravascular ultrasound analysis. Eur Heart J 32:2059–2066
Sakata K, Kawashiri MA, Ino H, Matsubara T, Uno Y, Yasuda T, Miwa K, Kanaya H, Yamagishi M (2012) Intravascular ultrasound appearance of scattered necrotic core as an index for deterioration of coronary flow during intervention in acute coronary syndrome. Heart Vessels 5:443–452
Sakata K, Namura M, Takagi T, Tama N, Inoki I, Terai H, Horita Y, Ikeda M, Yamagishi M (2014) Repeated occurrence of slow flow phenomenon during and late after sirolimus-eluting stent implantation. Heart Vessels. doi:10.1007/s00380-014-0477-8
Kubo T, Imanishi T, Takarada S, Kuroi A, Ueno S, Yamano T, Tanimoto T, Matsuo Y, Masho T, Kitabata H, Tsuda K, Tomobuchi Y, Akasaka T (2007) Assessment of culprit lesion morphology in acute myocardial infarction: ability of optical coherence tomography compared with intravascular ultrasound and coronary angioscopy. J Am Coll Cardiol 50:933–939
Hiro T, Kimura T, Morimoto T, Miyauchi K, Nakagawa Y, Yamagishi M, Ozaki Y, Kimura K, Saito S, Yamaguchi T, Daida H, Matsuzaki M (2009) Effect of intensive statin therapy on regression of coronary atherosclerosis in patients with acute coronary syndrome: a multicenter randomized trial evaluated by volumetric intravascular ultrasound using pitavastatin versus atorvastatin (japan-acs [japan assessment of pitavastatin and atorvastatin in acute coronary syndrome] study). J Am Coll Cardiol 54:293–302
Lee T, Yonetsu T, Koura K, Hishikari K, Murai T, Iwai T, Takagi T, Iesaka Y, Fujiwara H, Isobe M, Kakuta T (2011) Impact of coronary plaque morphology assessed by optical coherence tomography on cardiac troponin elevation in patients with elective stent implantation. Circ Cardiovasc Interv 4:378–386
The TIMI III, Investigators B (1994) Effects of tissue plasminogen activator and a comparison of early invasive and conservative strategies in unstable angina and non-q-wave myocardial infarction. Results of the TIMI III B trial. Thrombolysis in myocardial ischemia. Circulation 89:1545–1556
Porto I, Choudhury RP, Pillay P, Burzotta F, Trani C, Niccoli G, Blackman DJ, Channon KM, Banning AP (2006) Filter no reflow during percutaneous coronary interventions using the filterwire distal protection device. Int J Cardiol 109:53–58
Yabushita H, Bouma BE, Houser SL, Aretz HT, Jang IK, Schlendorf KH, Kauffman CR, Shishkov M, Kang DH, Halpern EF, Tearney GJ (2002) Characterization of human atherosclerosis by optical coherence tomography. Circulation 106:1640–1645
Bae JH, Kwon TG, Hyun DW, Rihal CS, Lerman A (2008) Predictors of slow flow during primary percutaneous coronary intervention: an intravascular ultrasound-virtual histology study. Heart 94:1559–1564
Jaffe R, Charron T, Puley G, Dick A, Strauss BH (2008) Microvascular obstruction and the no-reflow phenomenon after percutaneous coronary intervention. Circulation 117:3152–3156
Singh M, Berger P, Ting H, Rihal C, Wilson S, Lennon R, Reeder G, Bresnahan J, Holmes J (2001) Influence of coronary thrombus on outcome of percutaneous coronary angioplasty in the current era (the mayo clinic experience). Am J Cardiol 88:1091–1096
Kusama I, Hibi K, Kosuge M, Nozawa N, Ozaki H, Yano H, Sumita S, Tsukahara K, Okuda J, Ebina T, Umemura S, Kimura K (2007) Impact of plaque rupture on infarct size in st-segment elevation anterior acute myocardial infarction. J Am Coll Cardiol 50:1230–1237
Kodama T, Kondo T, Oida A, Fujimoto S, Narula J (2012) Computed tomographic angiography-verified plaque characteristics and slow-flow phenomenon during percutaneous coronary intervention. JACC Cardiovasc Interv 5:636–643
McEntegart MB, Kirtane AJ, Cristea E, Brener S, Mehran R, Fahy M, Moses JW, Stone GW (2012) Intraprocedural thrombotic events during percutaneous coronary intervention in patients with non-st-segment elevation acute coronary syndromes are associated with adverse outcomes: analysis from the acuity (acute catheterization and urgent intervention triage strategy) trial. J Am Coll Cardiol 59:1745–1751
De Luca G, Suryapranata H, Stone GW, Antoniucci D, Neumann FJ, Chiariello M (2007) Adjunctive mechanical devices to prevent distal embolization in patients undergoing mechanical revascularization for acute myocardial infarction: a meta-analysis of randomized trials. Am Heart J 153:343–353
Lee T, Kakuta T, Yonetsu T, Takahashi K, Yamamoto G, Iesaka Y, Fujiwara H, Isobe M (2011) Assessment of echo-attenuated plaque by optical coherence tomography and its impact on post-procedural creatine kinase-myocardial band elevation in elective stent implantation. J Am Coll Cardiol Intv 4:483–491
Takarada S, Imanishi T, Liu Y, Ikejima H, Tsujioka H, Kuroi A, Ishibashi K, Komukai K, Tanimoto T, Ino Y, Kitabata H, Kubo T, Nakamura N, Hirata K, Tanaka A, Mizukoshi M, Akasaka T (2010) Advantage of next-generation frequency-domain optical coherence tomography compared with conventional time-domain system in the assessment of coronary lesion. Catheter Cardiovasc Interv 75:202–206
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gamou, T., Sakata, K., Matsubara, T. et al. Impact of thin-cap fibroatheroma on predicting deteriorated coronary flow during interventional procedures in acute as well as stable coronary syndromes: insights from optical coherence tomography analysis. Heart Vessels 30, 719–727 (2015). https://doi.org/10.1007/s00380-014-0542-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00380-014-0542-3