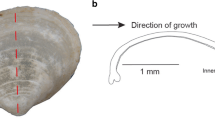
Abstract
While the elemental chemistry of marine mollusk shells contains valuable environmental information, extending these interpretations to freshwater bivalve shells may be problematic, given the wide range of aqueous chemistries that exist in freshwater environments. To better understand the significance of these records, 20 bivalves were collected from four freshwater streams to determine the geochemical relationships that exist between the bivalve shell and their sources streams. The concentrations of manganese (Mn), copper (Cu), strontium (Sr), barium (Ba), and calcium (Ca) were analyzed by laser ablation inductively coupled plasma mass spectrometry across the inner (INL) and outer nacreous layer (ONL) of each shell for comparison to stream data collected over the period of shell growth. Within an individual shell, the content of Mn, Sr, and Ba were significantly higher in the INL than the ONL, while Cu concentrations were similar. Strontium and Ba concentrations co-varied between the two layers in 1:1 relationships, while Mn displayed a preferential enrichment in the INL that increased from 2:1 to 5:1 as the Mn content of the shell decreased. Each elemental profile can be correlated between shell layers, except for the trace element Cu, which appeared to be more closely related to the organic content of the shell. These results suggest that the major element geochemistry of the shell layers differentially reflect the aqueous chemistry of the water in which a bivalve lived, but that these records are most likely overprinted by physiological processes specific to the mantle tissue from which a particular shell layer is secreted. Distribution coefficients (shell M /Ca:water M /Ca, where M = trace element of interest) were calculated using the median molar elemental concentration for each shell (by layer), and the appropriate water concentration. Bivalves from a contaminated site were excluded from this analysis because their shells were anomalous in size and color. For the remaining 15 shells, distribution coefficients (INL and ONL) were: Mn (0.50 and 0.21), Sr (0.26 and 0.17), and Ba (0.05: INL only). Inner nacreous layer values were comparable to the upper end of published estimates for freshwater bivalves and fish otoliths, while the ONL values were comparable to the lower range of values. Inclusion of shells from the contaminated site resulted in the calculation of distribution coefficients that fell outside the range of published estimates. These results suggest that exposure to pollutants may have a bearing on the biological factors that control the elemental concentration of bivalve shells in freshwater environments. While researchers generally avoid sampling shell material from the INL because of the potential for shell dissolution, the similarity in elemental profile trends observed here suggests that both the INL and ONL record accurate geochemical information, but in distinct forms. An obvious advantage of INL analysis is the relative increase in elemental concentration, but this is afforded at the expense of spatial resolution. Considered collectively, these results suggest that valuable information can be gleaned from the elemental concentration of freshwater bivalve shells when care is taken in the choice of material from which these records are extracted.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Bailey TR, Lear CH (2006) Testing the effect of carbonate saturation on the Sr/Ca of biogenic aragonite: a case study from the River Ehen, Cumbria, UK. Geochem Geophys Geosyst 7:Q03019 doi:10.1029/2005GC001084
Brand U, Wassenaar LI, Kanip J (1986) Variation in shell chemistry of terrestrial gastropods (Cerion incanum, Cerion uva, and Tudora maculata) from the Florida Keys and Bonaire. Can J Zool 64:2399–2404
Carell B, Forberg S, Grundelius E, Henrikson L, Johnels A, Lindh U, Mutvei H, Olsson M, Svardstrom K, Westermark T (1987) Can mussel shells reveal environmental history? Ambio 16:2–10
Carré M, Bentaleb I, Bruguier O, Ordinola E, Barrett NT, Fontugne M (2006) Calcification rate influence on trace element concentrations in aragonitic bivalve shells: evidences and mechanisms. Geochim Cosmochim Acta 70:4906–4920
Crenshaw MA (1972) The soluble matrix from Mercenaria mercenaria shell. In: Proc Int Symp Probleme der Biomineralisation, Int Symp Problems in Biomineralization, 1970, Mainz, vol. 6, pp 6–11
Crenshaw MA (1980) Mechanisms of shell formation and dissolution. In: Rhoads DC, Lutz R (eds) Skeletal growth of aquatic organisms. Topics in geobiology. Plenum, New York, pp 115–132
Crossgrove JS, Yokel RA (2004) Manganese distribution across the blood–brain barrier IV. Evidence for brain influx through store-operated calcium channels. NeuroToxicol 26:297–307
de Vries MC, Gillanders BM, Elsdon TS (2005) Facilitation of barium uptake into fish otoliths: influence of strontium concentration and salinity. Geochim Cosmochim Acta 69:4061–4072
Dunca E, Schöne BR, Mutvei H (2005) Freshwater bivalves tell of past climates: but how clearly do shells from polluted rivers speak? Palaeogeogr Palaeoclimatol Palaeoecol 228:43–57
EPD (1997–2004) Savannah River Site Environmental Data for 1996–2003. Environmental Monitoring Section, Environmental Protection Department Rep WSRC-TR-97-0077. Westinghouse Savannah River Company, Aiken
Faure G, Crocket JH, Hurley PM (1967) Some aspects of geochemistry of strontium and calcium in Hudson Bay and Great Lakes. Geochim Cosmochim Acta 31:451
Ferrier-Pagès C, Boisson F, Allemand D, Tambutté E (2002) Kinetics of strontium uptake in the scleractinian coral Stylophora pistillata. Mar Ecol Prog Ser 245:93–100
Gaetani GA, Cohen AL (2006) Element partitioning during precipitation of aragonite from seawater: a framework for understanding paleoproxies. Geochim Cosmochim Acta 70:4617–4634
Gillikin DP, Lorrain A, Navez J, Taylor JW, Keppens E, Baeyens W, Dehairs F (2005) Strong biological controls on Sr/Ca ratios in aragonitic marine bivalve shells. Geochem Geophys Geosyst 6:Q05009 doi:10.1029/2004GC000874
Gillikin DP, Dehairs F, Lorrain A, Steenmans D, Baeyens W, Andre L (2006) Barium uptake into the shells of the common mussel (Mytilus edulis) and the potential for estuarine paleo-chemistry reconstruction. Geochim Cosmochim Acta 70:395–407
González DA, Ostuni MA, Lacapère J-J, Alonso GL (2006) Stoichiometry of ATP and metal cofactor interaction with the sarcoplasmic reticulum Ca2+–ATPase: a binding model accounting for radioisotopic and fluorescence results. Biophys Chem 124:27–34
Hart SR, Blusztajn J (1998) Clams as recorders of ocean ridge volcanism and hydrothermal vent field activity. Science 280:883–886
Kinsman D, Holland HD (1969) The co-precipitation of cations with CaCO3. IV. The co-precipitation of Sr 2+ with aragonite between 16° and 96°C. Geochim Cosmochim Acta 33:1–17
Klein RT, Lohmann KC, Thayer CW (1996) Sr/Ca and 13C/12C ratios in skeletal calcite of Mytilus trossulus: covariation with metabolic rate, salinity, and carbon isotopic composition of seawater. Geochim Cosmochim Acta 60:4207–4221
Langlet D, Alleman LY, Plisnier P-D, Hughes H, André L (2007) Manganese content records seasonal upwelling in Lake Tanganyika mussels. Biogeosciences 4:195–203
Lazareth CE, Vander Putten E, Andre L, Dehairs F (2003) High-resolution trace element profiles in shells of the mangrove bivalve Isognomon ephippium: a record of environmental spatio-temporal variations? Estuar Coast Shelf Sci 57:1103–1114
Lea DW, Spero HJ (1992) Experimental determination of barium uptake in shells of the planktonic foraminifera Orbulina universa at 22 degrees C. Geochim Cosmochim Acta 56:2673–2680
Lorens RB (1981) Sr, Cd, Mn and Co distribution coefficients in calcite as a function of calcite precipitation rate. Geochim Cosmochim Acta 45:553–561
Lorens RB, Bender ML (1980) The impact of solution chemistry on Mytilus edulis calcite and aragonite. Geochim Cosmochim Acta 44:1265–1278
Lutz RA, Clark GR (1984) Seasonal and geographic variation in the shell microstructure of a salt marsh bivalve (Geukensia demissa (Dillwyn)). J Mar Res 42:943–956
Markich SJ, Jeffree RA, Burke PT (2002) Freshwater bivalve shells as archival indicators of metal pollution from a copper-uranium mine in tropical northern Australia. Environ Sci Technol 36:821–832
Mutvei H, Westermark T (2001) How environmental information can be obtained from Naiad shells. In: Bauer GG, Waechtler K (eds) Ecology and evolution of the freshwater mussels Unionoida. Springer, Berlin, pp 367–379
Neves RJ, Moyer SN (1988) Evaluation of techniques for age-determination of fresh-water mussels (Unionidae). Am Malacol Bull 6:179–188
Nystrom J, Dunca E, Mutvei H, Lindh U (1996) Environmental history as reflected by freshwater pearl mussels in the River Vramsan, southern Sweden. Ambio 25:350–355
Ravera O, Beone GM, Trincherini PR, Riccardi N (2007) Seasonal variations in metal content of two Unio pictorum mancus (Mollusca, Unionidae) populations from two lakes of different trophic state. J Limnol 66:28–39
Rosenberg GD, Hughes WW (1991) A metabolic model for the determination of shell composition in the bivalve mollusk, Mytilus edulis. Lethaia 24:83–96
Rosenthal Y, Katz A (1989) The applicability of trace elements in fresh water shells for paleogeochemical studies. Chem Geol 78:65–76
Sokal RR, Rohlf FJ (1995) Biometry. Freeman, New York
Stecher HA, Krantz DE, Lord CJ, Luther GW, Bock KW (1996) Profiles of strontium and barium in Mercenaria mercenaria and Spisula solidissima shells. Geochim Cosmochim Acta 60(18):3445–3456
Surge D, Walker KJ (1996) Geochemical variation in microstructural shell layers of the southern quahog (Mercenaria campechiensis): implications for reconstructing seasonality. Palaeogeogr Palaeoclimatol Palaeoecol 237:182–190
Swann CP, Hansen KM, Price K, Lutz R (1991) Application of PIXE in the study of shellfish. Nuclear Instruments Methods Phys Res B Beam Interactions with Materials and Atoms 56/7:683–686
Tevesz MJS, Carter JG (1980) Environmental relationships of shell form and structure of Unionacean bivalves. In: Rhoads DC, Lutz R (eds) Skeletal growth of aquatic organisms. Topics in geobiology. Plenum, New York, pp 295–322
Toland H, Perkins B, Pearce N, Keenan F, Leng MJ (2000) A study of sclerochronology by laser ablation ICP-MS. J Anal Atom Spectrom 15:1143–1148
Vander Putten E, Dehairs F, Keppens E, Baeyens W (2000) High resolution distribution of trace elements in the calcite shell layer of modern Mytilus edulis: environmental and biological controls. Geochim Cosmochim Acta 64:997–1011
Verdegaal S (2002) The shell chemistry of Unio crassus batavus as tool for reconstructing the evolution of the Rhine–Meuse delta and its use as indicator for river water composition. Vrije Universiteit, Amsterdam
Wang Y, Xu H (2001) Prediction of trace metal partitioning between minerals and aqueous solutions: a linear free energy correlation approach. Geochim Cosmochim Acta 65:1529–1543
Wells BK, Rieman BE, Clayton JL, Horan DL, Jones CM (2003) Relationships between water, otolith, and scale chemistries of west slope cutthroat trout from the Coeur d’Alene River, Idaho: the potential application of hard-part chemistry to describe movements in freshwater. Trans Am Fish Soc 132:409–424
Wheeler AP (1992) Mechanisms of molluscan shell formation. In: Bonucci E (ed) Calcification in biological systems. CRC, Boca Raton, pp 179–216
Wilbur KM, Saleuddin ASM (1983) Shell formation. In: Saleuddin ASM, Wilbur KM (eds) The Mollusca. Academic, New York, pp 235–287
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Carroll, M., Romanek, C.S. Shell layer variation in trace element concentration for the freshwater bivalve Elliptio complanata . Geo-Mar Lett 28, 369–381 (2008). https://doi.org/10.1007/s00367-008-0117-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00367-008-0117-3