Abstract
Purpose
Pretreatment measurements of systemic inflammatory response, including the Glasgow prognostic score (GPS), the neutrophil-to-lymphocyte ratio (NLR), the monocyte-to-lymphocyte ratio (MLR), the platelet-to-lymphocyte ratio (PLR) and the prognostic nutritional index (PNI) have been recognized as prognostic factors in clear cell renal cell carcinoma (CCRCC), but there is at present no study that compared these markers.
Methods
We evaluated the pretreatment GPS, NLR, MLR, PLR and PNI in 430 patients, who underwent surgery for clinically localized CCRCC (pT1-3N0M0). Associations with disease-free survival were assessed with Cox models. Discrimination was measured with the C-index, and a decision curve analysis was used to evaluate the clinical net benefit.
Results
On multivariable analyses, all measures of systemic inflammatory response were significant prognostic factors. The increase in discrimination compared with the stage, size, grade and necrosis (SSIGN) score alone was 5.8 % for the GPS, 1.1–1.4 % for the NLR, 2.9–3.4 % for the MLR, 2.0–3.3 % for the PLR and 1.4–3.0 % for the PNI. On the simultaneous multivariable analysis of all candidate measures, the final multivariable model contained the SSIGN score (HR 1.40, P < 0.001), the GPS (HR 2.32, P < 0.001) and the MLR (HR 5.78, P = 0.003) as significant variables. Adding both the GPS and the MLR increased the discrimination of the SSIGN score by 6.2 % and improved the clinical net benefit.
Conclusions
In patients with clinically localized CCRCC, the GPS and the MLR appear to be the most relevant prognostic measures of systemic inflammatory response. They may be used as an adjunct for patient counseling, tailoring management and clinical trial design.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Renal cell carcinoma (RCC) accounts for 2–3 % of adult malignancies, with clear cell RCC (CCRCC) representing the most common subtype [1]. Approximately 80 % of newly diagnosed patients present with clinically localized disease and are therefore candidates for curative management, but local or distant disease recurrence occurs in up to 20 % of patients [2]. Stratification of patients according to their risk of recurrence is crucial to discuss the management options (surgery, thermal ablation, active surveillance), to define further follow-up and to select those candidates that should be included in adjuvant clinical trials [3]. Immunohistochemical and blood-based biomarkers of angiogenesis, apoptosis, nutrition and systemic inflammatory response are currently evaluated for this purpose, as conventional prognostic factors such as stage and grade have limited accuracy [4–12].
It has been hypothesized that the tumor microenvironment triggers the release of cytokines, which subsequently alter the systemic inflammatory response [13]. Evaluation of systemic inflammatory response may be particularly relevant in CCRCC, as inflammation appears to play a critical role in the development and progression of the disease [14]. Pretreatment measurements of systemic inflammatory response such as lymphocytes, neutrophils, monocytes, thrombocytes and the C-reactive protein (CRP) represent easily measured, reproducible and inexpensive clinical routine biomarkers. Several of these measurements have been combined in a variety of scores, indices and ratios, including the Glasgow prognostic score (GPS), the prognostic nutritional index (PNI), the neutrophil-to-lymphocyte ratio (NLR), the monocyte-to-lymphocyte ratio (MLR) and the platelet-to-lymphocyte ratio (PLR). While every single marker has been associated with prognosis in RCC [5, 7, 10, 15, 16], there is at present no comparative study.
The aim of the present study was to compare the prognostic value of these pretreatment measurements of systemic inflammatory response in patients undergoing potentially curative resection for CCRCC.
Materials and methods
Study design
Following institutional review board approval, we retrospectively reviewed the renal tumor database of the Medical University of Vienna to identify 646 patients, who were treated with a radical or partial nephrectomy for clinically localized (pT1-3N0M0) unilateral RCC between 2002 and 2014. We consecutively excluded patients with subtypes other than clear cell (n = 161), those with relevant comorbidity affecting systemic inflammatory response markers (i.e., chronic liver disease, immunosuppression, cytotoxic medications, leukemia, lymphoma, autoimmune diseases and chronic inflammatory diseases, n = 31) and those with missing data on at least one of the relevant preoperative laboratory parameters within 1 month prior to surgery (n = 24). Our final cohort consisted of 430 patients, who underwent potentially curative resection of unilateral, clinically localized (pT1-3 N0 M0) CCRCC.
Management
Before surgery, a physical examination by a consultant in internal medicine and blood tests was conducted. All patients had no evidence of lymph node or distant metastasis on preoperative CT scans of the abdomen and chest X-ray or CT scan. None of the patients had preoperative systemic therapy, local radiotherapy or embolization.
A total of 247 patients (57.4 %) underwent radical nephrectomy, while 183 (42.6 %) had a partial nephrectomy. A concomitant regional lymph node dissection was performed in 72 cases (16.7 %) according to the surgeon’s discretion. Every other case was pNx but judged clinical N0. No patient received adjuvant therapy.
Because of the retrospective nature of this study, postoperative follow-up was not standardized. At our institution, risk group-based follow-up is generally recommended according to guidelines [17]. The median postoperative follow-up was 40 months (IQR 17–73), during which 46 patients (10.7 %) experienced disease recurrence. All patients were followed at our institution. Disease recurrence was defined as radiographic evidence of disease on chest X-ray, CT, MRI or bone scan. Every disease recurrence was confirmed by a physician involved in this study.
Study variables and measurements of systemic inflammation
Study variables were extracted from the database and included age, gender, T stage, Fuhrman grade, tumor size, presence of tumor necrosis, lymph node status and pretreatment laboratory values within 30 days prior to surgery. The NLR, the MLR and the PLR were calculated from the respective parameters from the differential blood count. The GPS was calculated as follows: Patients with elevated C-reactive protein serum levels (>10 mg/L) and hypoalbuminemia (<35 g/L) were allocated a score of 2, and patients with 1 or no abnormal value were allocated a score of 1 or 0, respectively. The PNI was calculated using the formula 10× serum albumin level [g/dl] + 0.005 × lymphocyte count per µl. The detection limit of the CRP assay was 0.02 mg/L. The intra-assay and interassay variability coefficients for CRP were 4.7 and 8.3 %, respectively.
Surgical specimens were processed according to standard procedures and were evaluated by a small group of experienced pathologists. Tumor size was defined by the largest diameter based on the pathological report. Pathologic stage was assigned according to the 2010 TNM classification. The stage, size, grade and necrosis (SSIGN) score was calculated [18]. The pathological evaluation was performed by a small group of experienced pathologists.
Statistical analysis
Categorical variables are presented as numbers and proportions and continuous variables as median and interquartile range (IQR). The primary study point of interest was disease-free survival (DFS), which was calculated from the date of surgery to the date of local or distant disease recurrence or last follow-up. Univariable and multivariable estimates were obtained from Cox models as hazard ratios (HR) and 95 % confidence intervals (95 % CI). The cutoffs for the PNI, the NLR, the MLR and the PLR were calculated based on the best discrimination of each inflammatory measurement on univariable Cox models. Multivariable models contained the measurement of systemic inflammation and were adjusted for standard prognostic factors. As there was a significant risk of overfit in multivariable modeling due to the low numbers of events, we summarized stage, size, grade and necrosis with the SSIGN score in a single variable. In a second set of multivariable analysis, a backward stepwise elimination process was carried out to identify the most relevant measurements of systemic inflammatory response. This process started with a full model containing the SSIGN score and all candidate measurements. By stepwise deletion of nonsignificant variables according to the likelihood ratio criteria, the final multivariable model contained only significant variables. Discrimination of Cox models was assessed with Harrell’s concordance index. Decision curve analysis was performed to determine whether models with measurements of systemic inflammation increased the net benefit over a realistic range of threshold probabilities compared with the SSIGN score alone.
Statistical testing was two-sided, and a P value of <0.05 was considered statistically significant. Analyses were all conducted with STATA 13 (College Station, TX, USA).
Results
Clinical and pathological characteristics
Baseline characteristics of the 430 patients are listed in Table 1. The GPS was 0 in 330 (76.8 %), 1 in 87 (20.2 %) and 2 in 13 (3.0 %) patients. The median PNI, MLR, NLR and PLR were 51.7, 0.4, 2.9 and 148.5, respectively.
Univariable survival analyses
The 4-year DFS rate (±SE) for all patients was 85.8 % (±2.6 %). As continuously coded variables, all measurements were significantly associated with DFS, and the discrimination of the single measurements ranged from 59.6 to 67.9 % (Table 2).
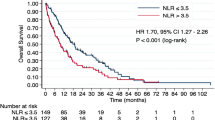
The measurements were then coded categorically according to the cut points providing the best discrimination. The optimal cut point for the PNI, the NLR, the MLR and the PLR was 48, 4.2, 0.4 and 145, respectively, with a discrimination of 58.6, 63.8, 63.9 and 59.4 %, respectively (Table 2; Fig. 1). Discriminations for continuously coded PNI, MLR and PLR were considerably higher than for the categorically coded measurement except for the NLR (Table 2). The GPS and continuously coded MLR had the best discrimination with a C-index of 72.3 and 67.9 %, respectively.
Multivariable survival analyses
On multivariable Cox models that were adjusted for the SSIGN score, GPS (P < 0.001), continuously and categorically coded PNI (P = 0.009 and P = 0.031), categorically coded NLR (P = 0.013), continuously and categorically coded MLR (P < 0.001 and P = 0.007) and categorically coded PLR (P = 0.004) were all significant prognostic factors (Table 3).
The increase in discrimination compared with the SSIGN score alone was 5.8 % for the GPS, 1.4–3.0 % for the PNI, 1.1–1.4 % for the NLR, 2.9–3.4 % for the MLR and 2.0–3.3 % for the PLR (Table 3).
On the further multivariable analysis, the SSIGN score and all measurements of systemic inflammatory response were evaluated simultaneously. By stepwise deletion of nonsignificant variables, the final multivariable model contained the SSIGN score (HR 1.40, P < 0.001), the GPS (HR 2.32, P < 0.001) and the MLR (HR 5.78, P = 0.003, Table 4). The discrimination of this model was 79.1 %, corresponding to an increase of 6.2 % compared with the SSIGN score alone.
The decision curve analysis showed that adding the GPS and the MLR to the SSIGN score was superior to the SSIGN score alone, with a great net benefit across a wide range of risk (Fig. 2).
Discussion
We analyzed and compared the prognostic value of pretreatment measurements of systemic inflammatory response in patients undergoing potentially curative surgery for CCRCC. We identified the GPS and the MLR as the most relevant prognostic measurements of systemic inflammatory response. Both variables were complementary in predicting prognosis and led to a considerable increase in both discrimination and clinical net benefit compared with a reference model.
Cancer progression and host systemic inflammatory response are closely related. Many studies support the hypothesis that pretreatment blood-based measurements of systemic inflammatory response are prognostic in patients with RCC [5, 7, 10, 15, 16, 19, 20]. As easily and reproducibly measured routine markers, they may serve as a helpful adjunct to standard prognostic factors, such as stage, size, grade and necrosis. Typically, two variables are combined in a score, an index or a ratio, which form the basis of subsequent prognostic research.
While every single measurement of systemic inflammatory response has been associated with prognosis in RCC [5, 7, 10, 15, 16], there was a lack of studies that compared these measurements. The GPS combines the CRP as a marker of inflammation and the albumin that indicates both the inflammation and the nutritional status. The largest study to date on the GPS in clinically localized CCRCC comprised 169 patients and used cancer-specific survival as the endpoint [15]. On the multivariable analysis, the GPS was a significant prognostic factor (HR 5.13, P < 0.001). The 4-year survival rate for patients with a GPS of 0, 1 and 2 was 96, 74 and 0 %, respectively. In the current study on 430 patients, we similarly identified the GPS as an independent prognostic factor, though DFS was used as the endpoint. Compared with all other measurements of systemic inflammatory response, the GPS was the measurement with the highest discrimination on both the univariable (72.3 %) and the multivariable analysis (78.7 %). On the simultaneous analysis of all measurements, the GPS remained a significant prognostic factor. Therefore, the GPS appeared to be the most relevant prognostic measurement of systemic inflammatory response. Further studies need to confirm these data.
The MLR was previously described as an independent prognostic factor in RCC. Hutterer et al. [10] demonstrated that a high MLR was associated with a 2.3-fold increased risk of death from the disease. In our cohort, the MLR was a significant prognostic factor on both the univariable and the multivariable analysis and led to a considerable increase in the discrimination of 2.9 to 3.4 %. On the simultaneous analysis of all variables, the MLR, the GPS and the SSIGN score were all statistically significant. Together with the GPS, the MLR appeared to be the most significant prognostic measurement of systemic inflammatory response.
The PNI has been investigated as a prognostic index of the systemic inflammatory response in several malignancies, including colorectal carcinoma and breast cancer [21, 22]. In our previous study on RCC, the PNI was identified as an independent prognostic variable [23]. In particular, it has been shown that a lower PNI was associated with worse survival for both patients with localized and metastatic disease. In the current study, we validated this measurement as an independent prognostic variable for the particular cohort of patients with localized CCRCC. The discrimination was considerably lower than for the GPS and the MLR, and the simultaneous analysis of all measurements led to elimination of the PNI from the prognostic model. The PNI may therefore be less relevant than the GPS and the MLR for patients with localized CCRCC.
A higher NLR has been associated with worse outcomes in RCC. In a large series, Pichler et al. [24] showed that an NLR equal or higher than 3.3 was an independent prognostic factor for overall survival (P = 0.014), but not for cancer-specific survival (P = 0.15) and DFS (P = 0.18). Similarly to the current study, others identified the NLR as an independent prognostic factor for patients with localized disease [25, 26]. Although statistically significant on the multivariable analysis, the NLR led only to a small increase in discrimination and was not significant on the simultaneous analysis of all markers.
We also analyzed the prognostic role of the PLR and showed that this measurement is a significant prognostic factor. Our data confirm data from Lee et al. [16], who evaluated pretreatment PLR in 341 patients with clinically localized CCRCC. In their study, the 5-year DFS rate of patients with high versus low preoperative PLR was 89 and 96 %, respectively (P = 0.022). While Lee et al. were not able to identify the PLR as an independent prognostic factor, our data indicate that the PLR may be a significant variable on the multivariable analysis. However, the PLR led only to a moderate increase in discrimination and was eliminated in the simultaneous analysis of all measurements.
Most prognostic studies on pretreatment measurements of systemic inflammatory response in RCC focused on univariable and multivariable analyses of a single marker. If a measurement shows statistical significance on the multivariable analysis, the discrimination of the multivariable model should be further assessed with and without the measurement. We used the SSIGN score as our reference model, which is a well-established prognostic score for CCRCC that has undergone rigorous external validation by several groups [18, 27–29]. In our study, each measurement of systemic inflammatory response led to an increase in discrimination compared with the reference model. To identify the most relevant variable, all measurements were evaluated simultaneously using a backward selection process. In this analysis, the GPS and the MLR were identified as the most relevant prognostic measurements of systemic inflammatory response. Both markers led to an increase in the discrimination of the SSIGN score by 6.2 %, and their use was associated with a clinical net benefit in the decision curve analysis. Importantly, both the GPS and the MLR were complementary in predicting prognosis. This can be explained by the nature of both measurements, as they contain four different variables. Both the GPS and the MLR may therefore be used as an adjunct to the SSIGN score for patient counseling, tailoring management and the design of adjuvant clinical trials. As our findings may be specific to our cohort, further external validation is necessary.
The current study has several limitations. The retrospective inclusion of patients led to selection bias and missing data, such as performance status. The cohort was well selected, but the numbers of patients and events were fairly low. Given the relatively long median DFS time published in the ARISER trial [30], our follow-up may have been too short to fully elucidate the prognostic value of the measurements. Further, the follow-up was not standardized, although guidelines were usually followed. We did not include data on inflammatory markers within the primary tumor, and post-treatment data on the inflammatory measurements during follow-up were not available. Although pathological evaluation was performed by a small group of experienced pathologists, we did not perform slide review. Nonetheless, our data suggest an important prognostic role of the GPS and the MLR for patients with clinically localized CCRCC. Larger studies are needed to confirm our findings.
Conclusions
In patients with clinically localized CCRCC, the GPS and the MLR appear to be the most relevant prognostic measurements of systemic inflammatory response. They may be used as an adjunct for patient counseling, tailoring management and clinical trial design.
References
Shuch B, Amin A, Armstrong AJ et al (2015) Understanding pathologic variants of renal cell carcinoma: distilling therapeutic opportunities from biologic complexity. Eur Urol 67:85–97
Brookman-May SD, May M, Shariat SF et al (2013) Time to recurrence is a significant predictor of cancer-specific survival after recurrence in patients with recurrent renal cell carcinoma–results from a comprehensive multi-centre database (CORONA/SATURN-Project). BJU Int 112:909–916
Chen DYT, Uzzo RG, Viterbo R (2014) Thinking beyond surgery in the management of renal cell carcinoma: the risk to die from renal cell carcinoma and competing risks of death. World J Urol 32:607–613
Michaelson MD, Stadler WM (2013) Predictive markers in advanced renal cell carcinoma. Semin Oncol 40:459–464
Hofbauer SL, Stangl KI, de Martino M et al (2014) Pretherapeutic gamma-glutamyltransferase is an independent prognostic factor for patients with renal cell carcinoma. Br J Cancer 111:1526–1531
De Martino M, Waldert M, Haitel A et al (2014) Evaluation of ABO blood group as a prognostic marker in renal cell carcinoma (RCC). BJU Int 113:E62–E66
De Martino M, Pantuck AJ, Hofbauer S et al (2013) Prognostic impact of preoperative neutrophil-to-lymphocyte ratio in localized nonclear cell renal cell carcinoma. J Urol 190:1999–2004
De Martino M, Leitner CV, Seemann C et al (2015) Preoperative serum cholesterol is an independent prognostic factor for patients with renal cell carcinoma (RCC). BJU Int 111:E348–E353
Tan PH, Cheng L, Rioux-Leclercq N et al (2013) Renal tumors: diagnostic and prognostic biomarkers. Am J Surg Pathol 37:1518–1531
Hutterer GC, Stoeckigt C, Stojakovic T et al (2014) Low preoperative lymphocyte-monocyte ratio (LMR) represents a potentially poor prognostic factor in nonmetastatic clear cell renal cell carcinoma. Urol Oncol 32:1041–1048
Ngo TC, Wood CG, Karam JA (2014) Biomarkers of renal cell carcinoma. Urol Oncol 32:243–251
da Costa WH, Rocha RM, da Cunha IW et al (2012) CD133 immunohistochemical expression predicts progression and cancer-related death in renal cell carcinoma. World J Urol 30:553–558
Fitzgerald JP, Nayak B, Shanmugasundaram K et al (2012) Nox4 mediates renal cell carcinoma cell invasion through hypoxia-induced interleukin 6- and 8- production. PLoS One 7:e30712
De Vivar Chevez AR, Finke J, Bukowski R (2014) The role of inflammation in kidney cancer. Adv Exp Med Biol 816:197–234
Lamb GWA, Aitchison M, Ramsey S et al (2012) Clinical utility of the Glasgow Prognostic Score in patients undergoing curative nephrectomy for renal clear cell cancer: basis of new prognostic scoring systems. Br J Cancer 106:279–283
Lee JH, Soo AH, Se JK, Il Kim S (2014) Abstracts: prognostic significance of preoperative neutrophil-to-lymphocyte ratio and platelet-to-lymphocyte ratio in patients with non-metastatic clear cell renal cell carcinoma. J Urol 191(4):e704
Neuzillet Y, Tillou X, Mathieu R et al (2011) Renal cell carcinoma (RCC) in patients with end-stage renal disease exhibits many favourable clinical, pathologic, and outcome features compared with RCC in the general population. Eur Urol 60:366–373
Frank I, Blute ML, Cheville JC et al (2002) An outcome prediction model for patients with clear cell renal cell carcinoma treated with radical nephrectomy based on tumor stage, size, grade and necrosis: the SSIGN score. J Urol 168:2395–2400
Wald G, Barnes KT, Bing MT et al (2014) Minimal changes in the systemic immune response after nephrectomy of localized renal masses. Urol Oncol 32:589–600
Brookman-May S, May M, Ficarra V et al (2013) Does preoperative platelet count and thrombocytosis play a prognostic role in patients undergoing nephrectomy for renal cell carcinoma? Results of a comprehensive retrospective series. World J Urol 31:1309–1316
Neal CP, Cairns V, Jones MJ et al (2015) Prognostic performance of inflammation-based prognostic indices in patients with resectable colorectal liver metastases. Med Oncol 32:590
Yang Z, Zhang B, Hou L et al (2014) Pre-operative prognostic nutritional index predicts the outcomes for triple-negative breast cancer. Tumour Biol 35:12165–12171
Hofbauer S, Pantuck A, de Martino M et al (2015) The preoperative prognostic nutritional index is an independent predictor of survival in patients with renal cell carcinoma. Urol Oncol 33:e1–e7
Pichler M, Hutterer GC, Stoeckigt C et al (2013) Validation of the pre-treatment neutrophil-lymphocyte ratio as a prognostic factor in a large European cohort of renal cell carcinoma patients. Br J Cancer 108:901–907
Viers BR, Houston Thompson R, Boorjian SA et al (2014) Preoperative neutrophil-lymphocyte ratio predicts death among patients with localized clear cell renal carcinoma undergoing nephrectomy. Urol Oncol 32:1277–1284
Ohno Y, Nakashima J, Ohori M et al (2010) Pretreatment neutrophil-to-lymphocyte ratio as an independent predictor of recurrence in patients with nonmetastatic renal cell carcinoma. J Urol 184:873–878
Ficarra V, Martignoni G, Lohse C et al (2006) External validation of the Mayo Clinic Stage, Size, Grade and Necrosis (SSIGN) score to predict cancer specific survival using a European series of conventional renal cell carcinoma. J Urol 175:1235–1239
Volpe A, Patard JJ (2010) Prognostic factors in renal cell carcinoma. World J Urol 28:319–327
Ficarra V, Galfano A, Novara G et al (2008) Risk stratification and prognostication of renal cell carcinoma. World J Urol 26:115–125
Belldegrun AS, Fall B, Bevan P et al (2013) ARISER: a randomized double blind phase III study to evaluate adjuvant cG250 treatment versus placebo in patients with high-risk ccRCC—results and implications for adjuvant clinical trials. J Clin Oncol 31:4507
Acknowledgments
IL is supported by the development fund of the CHUV-University Hospital. TK is supported by funds of the Oesterreichische Nationalbank (Anniversary Fund, Project Number: 15362).
Conflict of interest
The authors declare no conflict of interest.
Ethical standard
This study has been approved by the appropriate ethics committee, and all patients included gave their informed consent prior to their inclusion in the study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lucca, I., de Martino, M., Hofbauer, S.L. et al. Comparison of the prognostic value of pretreatment measurements of systemic inflammatory response in patients undergoing curative resection of clear cell renal cell carcinoma. World J Urol 33, 2045–2052 (2015). https://doi.org/10.1007/s00345-015-1559-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-015-1559-7