Abstract
To report and discuss four cases of renal cell carcinoma (RCC) in which preoperative investigations yielded contradictory results regarding the cranial extension of propagation of the tumor thrombus into the vena cava. An intraoperative ultrasound scan (IOU) was performed in all cases to identify the exact level of the tumor thrombus. We have performed an IOU of the vena cava in four patients with RCC propagation into the inferior vena cava. Preoperative investigations were performed in all patients and consisted of abdominal Ultrasound scan (USS), contrast enhanced CT scan and gadolinium enhanced MRI scan. Intraoperative ultrasound has identified correctly the cranial extension and the absence of tumor thrombus infiltration in all patients. The thrombus reached the suprahepatic vena cava in two cases and was confined to the infrahepatic vena cava in the remainder. Preoperative imaging investigation had failed to determine the correct cranial extension of the tumor thrombus in two patients.IOU is a very useful tool to accurately assess the precise extent of tumor thrombus and eventually the presence of vein wall infiltration. These data are of paramount importance to plan the optimal surgical approach. According to our experience this type of investigation identifies the cranial extent of a tumor thrombus inside the vena cava better than standard imaging techniques.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Several genitourinary malignancies have the propensity to invade and propagate along major venous structures. The vast majority of these tumours are renal cell carcinoma (RCC), while the remainder are transitional cell carcinoma, adrenal carcinoma, pheochromocytoma, and a variety of retroperitoneal sarcomas [1–6].
Propagation of renal cell carcinoma into the inferior vena cava is rare, accounting for 4–10% of all renal carcinomas, with 10–25% of these patients presenting an extension above the hepatic vein [7, 8]. Tumor thrombi in the vena cava is associated with several prognostic factors as local infiltration of perinephric tissue, lymph node metastases, caval infiltration and the presence of distant metastases [9]. As extensive tumor thrombi can be present without evidence of lymph node and distant metastases, an aggressive surgical approach with curative intent is justified [10]. Currently, most authors agree that the presence of the thrombus itself has no specific prognostic significance if it can be successfully removed [11]. In case of tumor thrombi in the vena cava a complete surgical excision represents the only potentially successful treatment with a 5 years survival rates ranging between 40 and 60% in case of non metastatic disease [12–14]. However, operative morbidity and mortality are significantly high and are directly related to the degree of vena caval extension and infiltration.
Given the complexity of excising tumor from vascular structures, e.g. the inferior vena cava, it is paramount to precisely identify the cranial extent of a tumor thrombus prior to surgery in order to plan the most suitable surgical approach.
The most commonly used investigations include magnetic resonance imaging (MRI), inferior Venacavography, abdominal ultrasound scanning (USS), multidetector helical Computerized tomography (MDHCT), transabdominal US and transesophaegeal US (TEUS). Each of these techniques has its own advantages and drawbacks and even in combination they may fail to detect involvement of the renal vein and the inferior vena cava as well as infiltration of the wall of the vena cava. Moreover, in case more than one of these investigations is performed on the same patient, there risk of contradictory results is present.
We report about our experience using intraoperative ultrasound (IOU) in patients with propagation of tumor thrombi into the vena cava in whom preoperative imaging studies had failed to unequivocally demonstrate the cranial extension of the thrombi.
Patients and methods
Four patients with RCC in whom preoperative imaging investigations had failed to unequivocally demonstrate the cranial extent of the tumor thrombi in the vena cava, underwent IOU at our Department during the last 3 years. Preoperative investigations for every patient consisted of helical CT, gadolinium enhanced MRI scan and USS of the abdomen. Helical CT scan images with a reconstructed slice thickness of 2 mm were acquired before and after the administration of contrast in all patients. Three-dimensional reconstructions have been used to improve the surgical planning. The MRI protocol included T1 weighted FAST sequences acquired after the administration of gadolinium. Images were displayed in coronal and transverse planes in all patients. For each patient the hospital records and all preoperative radiographic studies were reviewed. After the peritoneal cavity had been entered, sterile saline warmed to body temperature was poured into the operative field in order to create an acoustic interface between the ultrasound probe and whatever organ structure had to be examined. A Diasonics ultrasound unit with a 10 MHz linear array transducer was placed in a sterile plastic sheath filled with sterile gel. Generally the transducer was handled by the surgeon under the guidance of the ultrasonographer. The results obtained with the IOU were then compared with the real extension of the thrombus after incision of the wall of the vena cava and before removal of the tumor thrombus.
Results
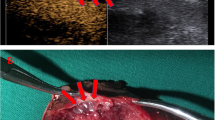
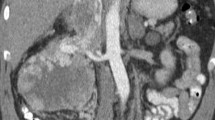
Intraoperative ultrasound correctly identified the cranial extension of the thrombus and excluded the presence of infiltration of the wall of the vena cava in all cases (Fig. 1). This information allowed the surgeon to plan the most suitable procedure. Pre-operative investigations had overestimated the cranial extension of the thrombus in two patients and had defined precisely the extension in the remaining cases (Table 1). Preoperative investigations did not demonstrate an infiltration of the wall of the vena cava by the tumor thrombus. The radiologic findings were confirmed by the histopathologic examination of the surgical specimen.
Discussion
Renal cell carcinoma has a propensity for venous invasion with 4–10% of patients having vena cava extension above the hepatic veins and the right atrium [12]. An aggressive surgical approach offers the only hope for cure in these patients without metastatic disease with 5 years survival rates of 40–68% after complete surgical excision [2–5]. The difference in survival among these studies may reflect different surgical techniques, nodal status, degree of invasion of the wall of the vena cava [15] and, possibly, the extent of the thrombus itself [14, 16, 17].
Preoperative accurate radiographic staging of tumor thrombus is of paramount importance to plan the best surgical approach, minimise morbidity and improve survival rates [18]. These investigations include Venacavography, MDHCT scan of the abdomen before and after the administration of contrast, abdominal MRI scan with T1 weighted FAST sequences acquired after the administration of gadolinium, USS and TEUS. With the introduction of MDHCT, a reconstructed slice thickness of 2 mm has become available. This technique, like MRI, provides complete staging of the abdomen including lymph node staging and loco-regional invasion. MDHCT detects thrombi in the vena cava with sensitivities of up to 64–95% [18]. The cranial extent of the thrombi is correctly diagnosed in only 33% of cases [19]. The main disadvantage of this technique is the lack of contrast medium in the vena cava in case of a tumor thrombus. In fact, in case of extensive invasion of the vena cava, the thrombus impedes contrast medium inflow from the caudal sections of the vena cava, thus prohibiting the precise delineation of the uppermost portion of the thrombus itself [18, 19]. Venacavography, which in the past was the gold standard for detecting and evaluating tumor thrombi, has the same limitations as CT since it also depends on contrast medium inflow to diagnose a tumour thrombus. This disadvantage can be overcome with catheterization and delivery of contrast medium into the superior vena cava [18]. However, this complicated procedure has its own possible complications and risks in addition to the extra burden of the patient. Even when retrograde approaches are included, inferior Venacavography may not adequately define the cranial extent of the thrombus and often offers little specific information regarding the presence or absence of thrombi in the hepatic veins. Moreover this technique is not able to distinguish between neoplastic and non neoplastic thrombus and requires the use of potentially nephrotoxic contrast agents [22].
Transabdominal US is a useful tool for preoperative evaluation of the renal vein and inferior vena cava. Its accuracy relies on the skill and experience of the ultrasonographer and on the patient’s body habitus. However, its sensitivity in detecting tumor thrombi depends on the position of the thrombus. If the tumor thrombus lies below the level of the insertion of the hepatic veins, ultrasound sensitivities are as low as 68%. If the tumor thrombus involves the intrahepatic portion of the vena cava, ultrasound sensitivity of 100% has been reported in detecting the tumor thrombus. Usually the overall sensitivity of detecting a tumor thrombus is reported from 89 to 100% [23]. However, it has been estimated that the renal vein and inferior vena cava are not completely visualised, respectively, in 12.5 and 43.5% of cases.
TEUS is also a safe, minimally invasive technique that can provide accurate information regarding the presence and extent of involvement of the vena cava, guidance for placement of a clamp on the vena cava and confirmation of complete removal of the tumor thrombus. Moreover it demonstrates an eventual migration of the tumor thrombus to the heart or the presence of a tumor thrombus embolism. These are both potentially fatal complications, and TEUS can provide the basis for an immediate intraoperative intervention [24].
Abdominal MRI scan with T1 weighted FAST images acquired in transversal and coronal planes after the administration of gadolinium, has been shown to be accurate for diagnosing the presence of tumor thrombi in the inferior vena cava. The ability of MRI to accurately delineate the cranial extension of a thrombus into the vena cava is superior to that of Venacavography. In addition, it has the advantage of being non invasive and of distinguishing between tumor thrombus and clot. MRI has been reported to have a sensitivity of up to 100% in detecting caval thrombi. This technique is thought to be superior to helical CT and Venacavography because of its ability to chose additional imaging planes with optimal spatial resolution, e.g. sagittal and coronal imaging planes [25]. MRI has intrinsic contrast superiority to CT and does not need to rely on contrast medium to differentiate tumour thrombus from blood [18, 25]. The value of information obtained with this technique can be limited by motion and flow artefact. In fact, turbulent blood flow may create a signal artefact that may falsely suggest or even mask the presence of intravascular thrombus [26]. However, recently some authors reported that MDHCT scan was comparable with MRI in determining the cranial extent of tumour thrombi [27].
Since these preoperative imaging investigations may yield equivocal results and the pre-operative correct identification of the cranial extension of the tumor thrombus is paramount for the surgeon to plan the most suitable approach, intraoperative imaging modalities may play a very important role.
IOU was first used in urologic surgery in 1961 to facilitate the localisation of renal stones during open nephrolithotomy. Long et al. reported on eight patients considered to be at risk for inferior vena cava involvement by tumor and for whom intraoperative ultrasound was obtained to clarify the presence or extent of thrombus [1]. In 1994, the effectiveness of IOU for ruling out the presence of renal vein and inferior vena cava tumor thrombus involvement in patients with RCC was demonstrated in five cases [26].
With the aid of the ultrasound probe, in the four patients of our series, we have performed a real time scan of the vena cava to establish the exact location of the thrombus underneath the vein wall. IOU provided excellent dynamic images of the thrombus and was successful in ruling out the limits of the thrombus and in excluding the presence of infiltration thus allowing the surgeon to plan the most suitable operative procedure. Therefore, we think that surgeons should be encouraged to use this technique in those cases in which preoperative investigation have demonstrated the involvement of the vena cava and when the cranial extension of the tumor thrombus has to be delineated.
References
Long JP, Choyke PL, Shawker TA, Robertson CA, Pass HI, Walther MM, Linehan WM (1993) Intraoperative ultrasound in the evaluation of tumour involvement of the inferior vena cava. J Urol 150:13–17
Goldfarb DA, Lorig R, Zelch M, Patrone P, Bukowski RM, Pontes JE (1990) Right renal mass with vena caval thrombus. J Urol 143:574–577
Dunnick NR, Doppman JL, Geelhoed GW (1980) Intravenous extension of endocrine tumours. AJR Am J Roentgenol 135(3):471–476
Sullivan M, Boileau M, Hodges C (1978) Adrenal cortical carcinoma. J Urol 120:660–665
Hoffman JC, Weiner SN, Koenigsberg M, Morehouse HT, Smith T (1983) Pheochromocytoma invasion of the inferior vena cava: sonographic evaluation. Radiology 149:793–795
Pussel SJ, Cosgrove DO (1981) Ultrasound features of tumour thrombus in the IVC in retroperitoneal tumours. Br J Rad 54:866–869
Sigman DB, Hasnain JU, Del Pizzo JJ, Sklar GN (1999) Real-time transesophageal echocardiography for intraoperative surveillance of patients with renal cell carcinoma and vana caval extension undergoing radical nephrectomy. J Urol 161:36–38
Hatcher PA, Anderson EE, Paulson DF, Carson CC, Robertson JE (1991) Surgical prognosis and management of renal cell carcinoma invading the inferior vena cava. J Urol 145:23–24
Novick AC, Campbell SC (2002) Renal tumors. In: Walsh PC, Retik AB, Vaughan Ed, vein AJ (eds) Campbell’s urology, 8th edn, vol 4, Chap 75. WB Saunders, Philadelphia, pp 2672–2719
Staheler G, Brkovic D (2000) The role of radical surgery for renal cell carcinoma with extension into the vena cava. J Urol 163(6):1671–1675
Bachmann A, Seitz M, Graser A, Reiser MF, Schafers HJ, Lohe F, Jauch KW, Stief CG (2005) Tumor nephrectomy with vena cava thrombus. BJU Int 95(9):1373–1384
Libertino J, Zinman L, Watkins E (1987) Long-term results of resection of renal cell cancer with extension into inferior vena cava. J Urol 137(1):21–24
Neves RJ, Zincke H (1987) Surgical treatment of renal cancer with vena cava extension. Br J Urol 59:390–395
Novick AC, Kaye MC, Cosgrove DM, Angermeier K, Pontes JE, Montie JE, Streem SB, Klein E, Stewart R, Goormastic M (1990) Experience with cardiopulmunary bypass and deep hypothermic circulatory arrest in the management of retroperitoneal tumours with large vena caval thrombi. Ann Surg 212:472–476
Skinner DG, Pritchett TR, Lieskovsky G, Boyd SD, Stiles QR (1989) Vena caval involvement by renal cell carcinoma. Surgical resection provides meaningful long-term survival. Ann Surg 210:387–392
Hatcher PA, Anderson EE, Paulson DF, Carson CC, Robertson JE (1991) Surgical management and prognosis of renal cell carcinoma invading the vena cava. J Urol 145:20–23
Belis JA, Kandzari SJ (1990) Five-year survival following excision of renal cell carcinoma extending into inferior vena cava. Urology 35:228–230
Hallscheidt PJ, Fink C, Haferkamp A, Bock M, Luburic A, Zuna I, Noeldge G, Kauffmann G (2005) Preoperative staging of renal cell carcinoma with inferior vena cava thrombus using multidetector CT and MRI. J Comput Assist Tomogr 29(1):64–68
Goldfarb DA, Novick AC, Lorig R, Bretan PN, Montie JE, Pontes JE, Streem SB, Siegel SW (1990) Magnetic resonance imaging for assessment of vena caval tumor thrombi: a comparative study with venacavography and computerized tomography scanning. J Urol 144:1103–1104
Sosa RE, Muecke EC, Vaughan ED Jr, McCarron JP Jr (1984) Renal cell carcinoma extending into the inferior vena cava: the prognostic significance of the level of vena caval involvement. J Urol 132:1097–1100
Siminovitch JMP, Montie JE, Straffon RA (1982) Inferior venacavography in the preoperative assessment of renal adenocarcinoma. J Urol 128:908–909
Hietala SO, Ekelund L, Ljungberg B (1988) Venous invasion in renal cell carcinoma: a correlative clinical and radiological study. Urol Radiol 9:210–216
Schwerk WB, Schwerk WN, Rodeck G (1985) Venous renal tumor extension: a prospective US evaluation. Radiology 156:491–495
Sigman DB, Hasnain JU, Del Pizzo JJ, Sklar GN (1999) Real-time transesophageal echocardiography for intraoperative surveillance of patients with renal cell carcinoma and vena caval extension undergoing radical nephrectomy. J Urol 161(1):36–8
Choyke PL, Walther MM, Wagner JR, Rayford W, Lyne JC, Linehan WM (1997) Renal cancer: preoperative evaluation with dual phase three dimensional MR angiography. Radiology 205:767–771
Harris DD, Ruckle HC, Gaskill DM, Wang Y, Hadley HR (1994) Intraoperative ultrasound: determination of the presence and extent of vena caval tumour thrombus. Urology 44:189–193
Konety BR; Lawrentschuk N, Gani J, Riordan R, Esler S, Bolton DM (2006) Multidetector computed tomography vs magnetic resonance imaging for defining the upper limit of tumour thrombus in renal cell carcinoma: a study and review. Urol Oncol 24(1):84–85
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Trombetta, C., Liguori, G., Bucci, S. et al. Evaluation of tumor thrombi in the inferior vena cava with intraoperative ultrasound. World J Urol 25, 381–384 (2007). https://doi.org/10.1007/s00345-007-0191-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-007-0191-6