Abstract
Chilling stress is an important limiting factor for rice growth and development. To examine the effect of spermidine (Spd) and trehalose (TH) priming in response to chilling stress of rice seed, we investigated the effects of seed priming with Spd, TH, and a mixture of Spd + TH on response and resilience of rice plants to chilling stress. Priming with Spd, TH and Spd + TH resulted in better seed vigor and seedling growth than hydro-priming and non-priming under low temperature (15 °C). In addition, Spd-, TH-, and Spd + TH-priming notably decreased malondialdehyde content but increased the proline and soluble sugar contents and antioxidant enzymes activities. Interestingly, spermine content and the expression of spermine-synthase (SPMS) gene were improved by Spd-priming. However, lower spermine content and SPMS expression was observed in the TH-primed plants. Moreover, expressions of trehalose-6-phosphate–phosphatase (TPP) genes was upregulated by TH-priming but downregulated by Spd-priming. It could be concluded from our results that (1) both Spd- and TH-priming could increase the contents of proline and soluble sugar, which were involved in osmotic adjustment, and antioxidant enzymes activities; (2) Spd-priming promoted the accumulation of endogenous spermine by upregulating SPMS1 and SPMS2, but inhibiting the expressions of TPP1 and TPP2; and (3) TH-priming increased the expressions of TPP1 and TPP2 but downregulated the expressions of SPMS1 and SPMS2. These findings suggested that seed priming with Spd and TH might enhance the chilling tolerance of rice seedling via different mechanisms.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Chilling is one of the major environmental stresses restricting plant growth and development. Rice yield reduction caused by chilling injury is a common problem worldwide. Rice varieties distributed in different regions are often subjected to varying degrees of low temperature hazards during the growing period (Cruz et al. 2013). The previous study showed that low temperature stress always delayed seed germination and caused poor growth of rice seedlings (Saddam et al. 2016). Therefore, it was necessary to develop novel methods to enhance the chilling tolerance of rice seed.
Polyamines (PAs), a class of physiologically active substances, widely exist in biological cells. Studies have shown that low temperature stress easily induced the accumulation of endogenous PAs in many plants, such as mustard (Mo and Pua 2010), watermelon (Akiyama et al. 2004), rice (Nayyar et al. 2005), wheat, barley and oats (Gondor et al. 2016). It suggested that PAs might involve in the cold tolerance of plants. PAs in higher plants mainly include putrescine (Put), spermidine (Spd) and spermine (Spm). Different kind of PAs often had different responses to chilling injury across plant species. Kushad and Yelenosky (1987) found that the resistance to low temperature stress of three citrus varieties was related to the Spd content. The exogenous application of Spd and Spm in water nutrient solution could effectively alleviate the damage of chickpea seedlings under low temperature, and the Spd had better effect (Nayyar and Chander 2004). Besides, Spd priming showed a more significant effect than Put and Spm on chilling tolerance of Lolium perenne seed (Wang et al. 2012a, b). The embryos of two maize inbred lines, Huang and Mo 17, were less susceptible to cold injury by Spd priming than Spm priming (Zheng et al. 2008). As above, in terms of improving plant cold tolerance, Spd might have more significant effect among three PAs to some extent. Zeng et al. (2016) found that Spd priming could promote effectively chilling tolerance by maintaining the integrity of cell structure and normal metabolism of endogenous hormones in indica-japonica hybrid rice seedlings. However, it was still not clear about the mechanism of Spd involving in the improvement of chilling tolerance of hybrid rice seed.
Trehalose (TH) is a non-reducing sugar composed of two glucose molecules (Wingler 2002). It was reported to act as an osmotic regulator in many plants under various stresses, such as chilling and osmotic stress (Luo et al. 2008; Elbein et al. 2003; Jang et al. 2003). Williams et al. (2015) reported that TH was widely detected in low temperature tolerant crops, suggesting TH involving in the establishment of cold tolerance in plants. However, the effect of TH priming on the cold resistance of rice seed was still unclear.
In our previous study, exogenous Spd increased the expression level of trehalose synthesis-related genes under high-temperature stress, indicating that there was a relationship between Spd and TH on stress resistance of rice seed. However, few reports focused on the combined effect of Spd and TH on rice chilling-resistance, and the mechanism involved in their combination effect remained completely unclear. In this experiment, we found that both rice seed vigor and seedling quality enhanced under chilling stress after Spd or TH single-priming. Therefore, antioxidant enzymes activities, soluble sugar content, polyamine contents, the corresponding genes expressions of Spd or TH biosynthesis-related genes were determined after priming with Spd, TH, and Spd + TH combination to acquire better understandings on the potential interaction of Spd + TH priming in rice seed chilling resistance.
Materials and Methods
Seed Priming
Spermidine (Spd) and trehalose (TH) solutions of different concentrations (0, 0.5, 1, and 2 mM) were prepared. Rice seeds were disinfected with 2% NaClO for 15 min, then rinsed with distilled water and soaked in the Spd and TH solutions for 24 h in the dark (25 °C). After soaking, the seeds were rinsed with distilled water and the seeds were dried off to their initial moisture.
Seed Germination
All primed rice seeds were subjected to a 14-day germination experiment. The primed seeds were incubated in growth chambers with a photosynthetic photon flux density of 250 mM m−2 s−1 and a photoperiod of 12 h light: 12 h dark (Ji et al. 2010). The chilling temperature was set to 15 °C. The treatments in this study were, respectively, referred to as CKn (untreated dry seeds incubated at 25 °C), CKc (untreated dry seeds incubated 15 °C), H2O (distilled water-primed seeds incubated at 15 °C), Spd0.5 (0.5 mM Spd-primed seeds incubated at 15 °C), Spd1 (1 mM Spd-primed seeds incubated at 15 °C), Spd2 (2 mM Spd-primed seeds incubated at 15 °C), TH0.5 (0.5 mM TH-primed seeds incubated at 15 °C), TH1 (1 mM TH-primed seeds incubated at 15 °C), TH2 (2 mM TH-primed seeds incubated at 15 °C), Spd0.5 + TH0.5 (0.5 mM Spd + TH-primed seeds incubated at 15 °C), Spd1 + TH1 (1 mM Spd + TH-primed seeds incubated at 15 °C), Spd2 + TH2 (2 mM Spd + TH-primed seeds incubated at 15 °C).
The number of germinated seed (the seed whose radicle visibly protruded through the seed coat and reached to the half-length of the diameter of the whole seed) was counted every day. The germination energy (GE) and germination percentage (GP) were calculated at the 5th and 14th days, respectively. After 14 days of germination, root length (RL) and shoot height (SH) were measured, and the seedling dry weight (SDW) was determined after drying at 80 °C for 48 h. Each treatment has three replications. All the measurements were based on ten randomly selected normal rice seedlings for each replication. We also calculated the Germination Index (GI) and Vigor Index (VI) following the method described by Hu et al. (2016).
Measurements of Physiological Parameters
0.3 g of fresh rice seeds was taken, and added 3 ml of phosphoric acid buffer with pH 7.8, grinded in ice bath until homogenized, and transferred to 5 ml centrifugal tube. Then, the mixture was centrifuged at 4 °C at 10,000×r min−1 for 20 min, the supernatant was the crude enzyme extract for the determination of enzyme activity.
Malondialdehyde (MDA) concentration was determined using the thiobarbital (TBA) reaction, as described by Jing et al. (2016) with some modifications. 1.5 ml of the crude enzyme solution was mixed with 2.50 ml of TBA-TCA solution, boiled in water for 15 min, rapidly cooled to room temperature, centrifuged at 1800×r min−1 for 10 min, and the supernatant was taken. The absorbance at 532 nm (OD532) and 600 nm (OD600) was measured for the mixtures, and the content of MDA was calculated. For each sample, the measurements were carried out in triplicate.
The peroxidase (POD), superoxide dismutase (SOD), catalase (CAT) and peroxidase (APX) activities were performed according to the method described by Zhu et al. (2016) with some modifications. For POD, the assay mixture consisted of 1.35 ml of 25 mM phosphate buffer (pH 7.0), 100 μl of 1.5% guaiacol, 100 μl of 100 mM H2O2, and 100 μl of enzyme extract. The increase in 470-nm absorbance was measured spectrophotometrically at 470 nm for 2 min. The enzymatic activity was calculated as nmol of guaiacol oxidized min−1 g−1 FW (fresh weight) and was expressed as nmol g−1 FW min−1. For each sample, the measurements were carried out in triplicate.
For SOD, the reaction mixture was composed of 50 mM phosphate buffer (pH 7.8), 1.22 mM riboflavin, 13 mM methionine, 56 µM NBT, 78.2 µM EDTA, and 100 µl enzyme extract. Distilled H2O was added into the mixture to bring the final volume to 3 ml. A complete reaction mixture without enzyme extract was used as a control. The reaction was initiated by placing the tubes with the mixture under 4000 Lux for 15 min. The reaction was measured spectrophotometrically at 560 nm for inhibition of the photochemical reduction of nitro blue tetrazolium (NBT). In this assay, 1 unit was defined as the amount of SOD required to inhibit the photoreduction of NBT by 50%. The specific activity of SOD was expressed as SOD g−1 protein.
For CAT, the enzyme assay contained 1.4 ml of phosphate buffer (25 mM pH 7.0 + 2 mM EDTA), 200 µl of 300 mM H2O2, and 100 µl enzyme extract in a total volume of 1.7 ml. The CAT activity was estimated by monitoring the 240-nm absorbance of the mixture for 2 min and was expressed as nmol g−1 FW min−1.
For APX, the enzyme assay contained 1.4 ml of phosphate buffer (25 mM pH 7.0 + 2 mM EDTA), 100 µl of 7.5 mM ascorbic acid, 100 µl of 300 mM H2O2, and 100 µl enzyme extract in a total volume of 1.7 ml. The APX activity was estimated by monitoring the 290-nm absorbance of the mixture for 2 min and was expressed as nmol g−1 FW min−1.
Determination of Proline Content
Proline content was determined using a method described by Bates et al. (1973). 100 ml of leaf samples were taken and ground in 2 ml of 3% sulfosalicylic acid. The homogenate was heated in a boiling water bath for 15 min and centrifuged at 12,000×I min−1 for 10 min. 200 μl of the supernatant was added into a mixture of 200 μl of tri-distilled water, 200 μl of glacial acetic acid, and 400 μl of acid ninhydrin. Then, the mixture was placed in the boiling water bath for 1 h. The absorbance at 520 nm was measured, and the proline content was expressed as nmol g−1 FW.
Determination of Soluble Sugar Content
The content of soluble sugar was determined by anthrone colorimetry (Oscar et al. 2010). Briefly, 0.1 g of leaves were taken. Distilled water was added into the leaf samples, and the mixtures were incubated in a water bath pot at 100 °C for 30 min. After that, the supernatant was separated, added with 5 ml distilled water and reheated for 30 min. Then, the supernatant was removed, the glass tube was cleaned, the solution was transferred to a 25-ml volumetric flask, then to a 50-ml centrifuge tube, and centrifuged at 5000×r min−1 for 15 min. Afterwards, 0.5 ml of centrifuged solution was taken, diluted, and 0.5 ml solution was transferred to a 20-ml test tube. A mixture of 1.5 ml of distilled water, 0.5 ml of anthrone, and 5 ml of concentrated sulfuric acid was added to the solution. After that, we fully vibrated the tubes and immediately put the test tubes into boiling water bath. The test tubes were taken out from boiling water bath and allowed to cool naturally in room temperature with the temperature of distilled water as a reference. Finally, we determined the absorbance at 620 nm.
Measurement of Endogenous Polyamine Content
PAs extraction from seedlings was analyzed by HPLC according to the testing method published by Huang et al. (2017). 2 g of fresh leaves from each treatment were homogenized with 2 ml of 5% (w/v) cold perchloric acid. The mixtures were kept in ice bath for 1 h and then centrifuged at 10,000 × g for 30 min at 4 °C. The supernatant was transferred and stored at − 80 °C for PAs quantification. 5 ml of the supernatant was taken and mixed with 1 ml of 2 mM NaOH solution and 10 μl of benzoyl chloride. The mixtures were incubated separately for 20 min at 37 °C. 2 ml of saturated NaCl solution and 2 ml of diethyl ether were added into the mixture, which was then centrifuged at 1500 × g for 15 min at 4 °C. From the centrifuged mixture, 1 ml of diethyl ether phase was extracted, dried by nitrogen and redissolved in 100 μl of methanol for the following test. The extracts were filtered through a 0.22 μm membrane filter and then eluted through a 6.0 mm × 150 mm, 5 mm particle size reverse-phase (C18) column (Shim-Pack CLC-ODS) at room temperature. PAs peaks were detected by an SPD-20A (Shimadzu) absorbance detector at 254 nm. The mobile phases consisted of water and methanol (35/65, v/v) at a flow rate of 1.0 ml min−1. The polyamine standards (Sigma) of Put, Spd, and Spm were used for the generation of standard curves. For each sample, the measurements were carried out in triplicate.
Real-time Quantitative Polymerase Chain Reaction (PCR)
Total RNA of each sample was extracted and 500 ng of RNA were reverse-transcribed into cDNA using the PrimeScript™ RT reagent Kit (Vazyme, China). Real-time PCR reaction was carried out with CFX96™ Real-Time PCR Detection System (Bio-Rad, Hercules, CA, USA). Primer sets were designed with the Primer5 software and the rice actin gene was used as an internal reference gene (Table 1). The 20 μl reaction system contained 1 μl of diluted cDNA, 0.6 μl of reverse and forward primers, 7.8 μl of ddH2O, and 10 μl of the AceQ qPCR SYBR Green Master Mix (Vazyme, Nanjing, China). Relative gene expression levels were calculated using the 2−ΔΔCT analytical method. All the reactions were carried out in 3 repeats and 3 independent biological replicates. The data were expressed as mean ± SD (standard deviation) after normalization.
Statistical Analysis
We used analysis of variance (ANOVA) to analyze the data in the Statistical Analysis System (SAS) followed by calculation of the least significant difference (LSD, α = 0.05). Percentage data were arc-sin-transformed prior to analysis.
Results
Seed Priming Enhanced Seed Vigor and Seedling Growth Under Chilling Stress
The seed germination was inhibited under low temperature, but improved by priming treatments (Table 2). Under the chilling stress, GP, GE, GI, and VI were significantly improved after all priming treatments (Table 2). Spd- and TH-priming had better effects than water priming. Specifically, compared with water-primed seeds, Spd1-priming improved the GI and VI by 28.88% and 65.52%, respectively; and TH1-priming improved the GI and VI by 29.92% and 87.93%, respectively. Spd1 and TH1-primed seeds recorded the highest GP and GE under chilling conditions compared with the other two concentrations (Table 2). Seeds primed with Spd1 + TH1 showed no significant differences in GP, GE, and GI compared with Spd0.5 + TH0.5 and Spd2 + TH2. However, VI was significantly higher in Spd1 + TH1-primed seeds compared with the seeds primed with other two combined concentrations.
Similar changes were found in seedling quality (Table 3). Compared with water-primed seeds, priming with Spd1 improved the full length (FL) and SDW by 60.03% and 40%, respectively; and priming with TH1 improved the FL and SDW by 64.36% and 60.00%, respectively. The results also showed that priming with Spd1 and TH1 produced the highest seedling quality under normal and chilling conditions compared with the other two concentrations (Table 3). Priming with Spd1 + TH1 had no significant effects on root length, but the treatment significantly increased the SH, FL, and SDW compared with the other two concentrations.
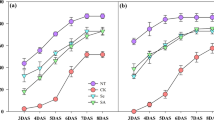
According to the effects of priming treatments on seed vigor and seedling growth (Fig. 1), unprimed seeds under normal temperature (CKn), unprimed seeds under low temperature (CKc), water-, Spd1-, TH1-, and Spd1 + TH1- primed seeds were chosen for the following experiments.
Effect of seed priming with Spd1, TH1 and Spd1 + TH1 on rice seedling growth under chilling stress (15 °C). CKn: normal temperature control (25 °C); CKc: low temperature control (15 °C); H2O: water priming + low temperature; Spd1: 1 mM Spd priming + low temperature; TH1: 1 mM TH priming + low temperature; Spd1 + TH1: 1 mM Spd + 1 mM TH priming + low temperature
Seed Priming Decreased MDA Content and Increased Proline and Soluble Sugar Contents Under Chilling Stress
Compared with the CKc seeds, water priming significantly decreased MDA content by 8.33%. Furthermore, Spd1-, TH1-, and Spd1 + TH1-priming decreased MDA content by 40.63%, 44.80%, and 55.99%, respectively (Fig. 2a).
Effects of Spd1, TH1 and Spd1 + TH1 on MDA (a), proline (b) and soluble sugar (c) content in rice seedlings under chilling stress. The bars mean standard deviation and different letters indicate significant differences between treatments (α = 0.05, LSD). The treatment methods were the same as those in the Fig. 1 notes
The proline and soluble sugar contents after CKc treatment increased significantly compared with those in CKn seeds (Fig. 2b, c). Water priming enhanced the accumulation of proline and soluble sugar. Compared with the CKc treatment, Spd1-, TH1-, and Spd1 + TH1-priming increased the proline content by 27.22%, 13.99%, and 29.01%, respectively and increased the soluble sugar content by 34.44%, 73.69%, and 51.98%, respectively.
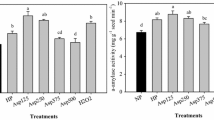
Seed Priming Stimulated Antioxidant Enzymes Activities in Response to Chilling Stress
The priming treatments elevated the activities of antioxidant enzymes, including CAT, POD, and APX (Fig. 3). Compared with water priming, Spd1-, TH1-, and Spd1 + TH1-priming significantly increased the CAT activity under chilling stress by 50.53%, 30.42%, and 27.33%, respectively. Furthermore, Spd1-, TH1-, and Spd1 + TH1-priming significantly improved the POD activity irrespective of chilling stress by 62.21%, 67.89%, and 40.33%, respectively, compared with the water priming. Similarly, APX activities in Spd1-, TH1-, and Spd1 + TH1-primed seeds were significantly improved under chilling stress by 6.44%, 83.44%, and 78.86%, respectively, compared with that of water-primed seeds. In contrast, SOD decreased significantly after Spd1-, TH1-, and Spd1 + TH1-priming treatments under chilling stress compared with water priming (Fig. 3).
Effects of seed priming with Spd1, TH1 and Spd1 + TH1 on antioxidant enzyme activities of rice seedling under chilling stress. The bars mean standard deviation and different letters indicate significant differences between treatments (α = 0.05, LSD). The treatment methods were the same as those in Fig. 1 notes
Seed Priming Regulated Polyamine Metabolism in Response to Chilling Stress
Put and Spd contents in the seedlings significantly increased under chilling stress compared with those in CKn seedlings (Fig. 4a, b). Spd1- and Spd1 + TH1-priming significantly increased the content of Spm under chilling stress compared with unprimed seeds. However, TH1-priming showed no significant effects (Fig. 4c). Compared with CKc, priming with water, Spd1, TH1, and Spd1 + TH1 resulted in a significant decrease in the Put content under chilling stress, but there was no significant difference in Put content among seedlings underwent these treatments. The Spd content in seedlings primed with water is significantly lower than that of CKc seedlings, but is significantly higher than those in seedlings primed with Spd1, TH1, and Spd1 + TH1. Priming with TH and Spd + TH substantially decreased Spd content. However, Spm content increased substantially under chilling stress across the priming treatments. Particularly, after Spd treatment, the Spm content in seedlings reached the highest level. Interestingly, Spd + TH-priming increased the Spm content to a greater extent than TH-priming, but to a lesser extent than Spd-priming (Fig. 4).
Effects of priming with Spd1, TH1 and Spd1 + TH1 on endogenous polyamines content (a) and polyamine synthesis-related genes (b) in rice seedlings under chilling stress. The bars mean standard deviation and different letters indicate significant differences between treatments (α = 0.05, LSD). The treatment methods were the same as those in the Fig. 1 notes
The results showed that ADC2, SAMDC, SPMS1, and SPMS2 were downregulated, while SPDS was upregulated under chilling stress compared with the normal condition (Fig. 4). However, compared with CKc seeds, Spd1-primed seeds showed increased SPMS1 and SPMS2 expressions under chilling stress, while TH1-priming resulted in decreased expressions of the genes (Fig. 4). Interestingly, Spd1 + TH1-priming generated a higher SPMS2 expression than TH-priming, but a lower SPMS2 expression than Spd-priming (Fig. 4).
Seed Priming Regulated the Expression Levels of Trehalose-6-phosphate Phosphatase Genes in Response to Chilling Stress
OsTPP1 and OsTPP2 were upregulated under chilling stress compared with the normal condition (Fig. 6). TH1-priming significantly improved the OsTPP1 and OsTPP2 expressions under chilling stress compared with unprimed seeds. However, Spd1-priming decreased the expressions of the genes (Fig. 6). Interestingly, the expression of OsTPP2 in Spd1 + TH1-primed seeds is higher than that of Spd1-primed seeds, but lower than that of TH1-primed seeds (Fig. 5).
Effects of priming with Spd1, TH1 and Spd1 + TH1 on trehalose synthesis-related genes OsTPP1 and OsTPP2 in rice seedlings under low temperature stress. The expression level of different genes was measured by RT-qPCR analysis; the expression level of CKn was regarded as 1.0. The treatment methods were the same as those in the Fig. 1 notes
Discussion
It is well known that rice originates from the tropics and subtropics and is extremely sensitive to chilling stress (Howarth and Ougham 2010). In this study, 15 °C was applied as chilling temperature according to the study of Ji et al. (2010), and results showed that seed vigor and seedling quality decreased substantially under chilling stress (Tables 2, 3). These results were consistent with Saddam et al. (2016), who found that rice seeds exposed to chilling stress (18 °C for 14 days) had considerably decreased germination speed, germination rate, and seedling growth. However, Spd-, TH-, and Spd + TH-priming alleviated the damage of chilling stress to rice seed vigor and seedling quality. Spd and TH treatments were effective under chilling stress, which was consistent with previous studies (Diao et al. 2015, 2016; Zhang et al. 2018; Kosar et al. 2018). In contrast, combined priming of Spd + TH did not further improve seed germination compared with separate Spd- and TH-priming.
Previous study indicated that there was a positive correlation between proline content and plant cold tolerance (Chen et al. 2010). Besides, the soluble sugar content of several plants, including cucumber (Wan et al. 2015) and Arabidopsis (Moustakas et al. 2011) increased under low temperature. In our study, Spd-priming significantly reduced MDA content but increased proline and soluble sugar contents in rice seedlings under chilling stress (Fig. 2b, c), which was in accord with the results of Diao et al. (2015) and Nahar et al. (2015). The effects of TH-priming were similar to those of Spd-priming. Similar observations were reported by Xie et al. (2015), who found that exogenous TH helped to increase the contents of soluble protein and soluble sugar in wheat seedlings, improve the antioxidant capacity, reduce the degree of membrane lipid peroxidation injury, and therefore, enhanced the cold resistance in wheat. Moreover, the soluble sugar content in TH-primed seedlings was significantly higher than that in Spd-primed seedlings, but the effects of TH- and Spd-priming on proline content were just the opposite. It might be due to the fact that TH participates in sugar metabolism, while proline and Spd have the same synthetic precursor. The MDA content, as an index of oxidative damage degree, was consequently analyzed, and the results showed that priming treatments substantially down-regulated MDA accumulation during seed germination under chilling stress (Fig. 2a). The proline, soluble sugar, and MDA contents of Spd + TH-primed seedlings were just between those in seedlings primed with TH and Spd separately.
To adapt various adverse conditions, the antioxidant defense system had been formed internally in the plants, which removed the excessive reactive oxygen species through enzymatic and non-enzymatic mechanisms. The enzymatic defense system includes SOD, CAT, POD, and APX. In our study, Spd-, TH-, and Spd + TH-priming significantly improved CAT, POD, and APX activities in rice seedlings under chilling stress, which was consistent with Li et al. (2017), who found an upregulation of antioxidant enzymes activities after priming treatment. In our study, the activities of SOD in Spd- and TH-primed seedlings were significantly decreased under chilling stress (Fig. 3). The difference in the response of SOD activity to Spd- and TH-priming between different studies might be attributed to differences in plant individuals and chilling treatments. It was suggested that Spd and TH participate in the establishment of the plant’s cold resistance through osmotic regulation and removal of reactive oxygen species.
Endogenous polyamine content was believed to be related to chilling tolerance in plants. In response to chilling stress, the contents of endogenous polyamines in different plants changed with similar trends, but the contents of Put, Spd, and Spm changed in different ways (Cuevas et al. 2008; Shen et al. 2000). After 24 h of low temperature treatment, the free Put content in Arabidopsis seedlings increased significantly, but there was no significant change as for Spd content (Cuevas et al. 2008). Shen et al. (2000) found that the Spd content in chilling-tolerant cucumbers increased significantly under low temperature, while that in chilling-sensitive ones did not change significantly; while the contents of Put and Spm in two types of varieties did not change significantly. In our study, Spm content significantly increased under chilling stress, especially after Spd-priming (Fig. 4). In addition, the polyamine synthesis-related genes include arginine decarboxylase (ADC), ornithine decarboxylase (ODC), S-adenosylmethionine decarboxylase (SAMDC), spermidine synthase (SPDS), and spermine synthase (SPMS). The similar results also had been found by Imai et al. (2004) that low temperature significantly induced the expression of SPMS1 in rice roots. Kasukabe et al. (2004, 2006) reported that upregulating SPDS gene of black-seeded pumpkin improved the cold tolerance of Arabidopsis thaliana and sweet potato. Under NaCl, drought and abscisic acid (ABA) treatments, the expression of AtSPMS, Put and Spm contents were all induced (Urano et al. 2003). Interestingly, SPMS expression of Spd + TH-primed seedlings in our study was higher than that of TH-primed ones, but lower than that of Spd-primed ones. It might be due to that TH inhibited the expression of polyamine biosynthesis genes, leading to decreased polyamine content.
The transcriptional levels of TH biosynthetic genes tend to increase under chilling stress. The expressions of several members of the TPP gene family were related to cold, salt, and ABA; and their expressions change in different organs and during different developmental stages (Elena et al. 2007). In rice plants, OsTPP1 and OsTPP2 all had TPP enzyme activity. In the roots and shoots of rice seedlings, OsTPP1 and OsTPP2 could be induced instantaneously by low temperature stress, drought, and exogenous ABA (Pramanik and Imai 2005). The expression of OsTPP1 was upregulated rapidly by salt and ABA treatments, but was upregulated more slowly by cold stress (Ge et al. 2008), which is partially consistent with our findings (Fig. 5). Moreover, the OsTPP2 expression induced by Spd + TH-priming is higher than that induced by Spd-priming but lower than that induced by TH-priming (Fig. 5). Based on these results, it can be inferred that the Spd and TH may have antagonistic relationship and unable to enhance the cold tolerance of plants.
In this study, the Spd- and TH-priming significantly improved rice seed germination and seedling growth under chilling stress. The improved chilling tolerance was closely related with the increased contents of osmotic adjustment-related substances, the activities of antioxidant enzymes, PAs contents, and the expressions of PAs biosynthesis-related genes and trehalose-6-phosphate–phosphatase genes. The levels of endogenous Spm induced by Spd-priming and TPP genes induced by TH-priming might be the major factors responsible for chilling tolerance in rice (Fig. 6), which still needs further study.
Schematic presentation of main metabolic pathways regulated by spermidine and trehalose in rice seedling exposed to chilling stress. Both of spermidine and trehalose priming could increase osmotic adjustment substances (such as proline and soluble sugar) contents, antioxidant enzymes activities. The spermidine priming promoted the accumulation of endogenous spermine content by upregulating SPMS1 and SPMS2, but inhibited the expression of TPP1 and TPP2. On the contrary, trehalose priming increased the expression of TPP1 and TPP2 but downregulated SPMS1 and SPMS2 expression levels. Therefore, it was supposed that seed priming with spermidine and trehalose might enhance the chilling tolerance of rice seedling via different mechanisms. “→” indicated enhanced effect, “” indicated inhibited effect, “” indicated possible effect
Reference
Akiyama T, Pillai MA, Sentoku N (2004) Cloning, characterization and expression of OsGLN2, a rice endo-1, 3-β-glucanase gene regulated developmentally in flowers and hormonally in germinating seeds. Planta 220(1):129–139
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39(1):205–207
Chen WP, Li PH (2010) Membrane stabilization by abscisic acid under cold aids proline in alleviating chilling injury in maize (Zea mays L.) cultured cells. Plant Cell Environ 25(8):955–962
Cruz RPD, Sperotto RA, Cargnelutti D, Adamski JM, Freitasterra TD, Fett JP (2013) Avoiding damage and achieving cold tolerance in rice plants. Food Energy Secur 2(2):96–119
Cuevas JC, Lópezcobollo R, Alcázar R (2008) Putrescine is involved in Arabidopsis freezing tolerance and cold acclimation by regulating abscisic acid levels in response to low temperature. Plant Physiol 148(2):1094–1105
Diao Q, Song Y, Qi H (2015) Exogenous spermidine enhances chilling tolerance of tomato (Solanum lycopersicum L.) seedlings via involvement in polyamines metabolism and physiological parameter levels. Acta Physiol Plant 37(11):1–15
Diao Q, Song Y, Shi D, Qi H (2016) Nitric oxide induced by polyamines involves antioxidant systems against chilling stress in tomato (Lycopersicon esculentum mill.) seedling. J Zhejiang Univ Sci B 17(12):916–930
Elbein AD, Pan YT, Pastuszak I, Carroll D (2003) New insights on trehalose: a multifunctional molecule. Glycobiology 13(4):17R–27R
Elena BG, Rolland F, Sheen J, Thevelein JM (2007) A central integrator of transcription networks in plant stress and energy signalling. Nature 448(7156):938–942
Ge LF, Chao DY, Shi M, Zhu MZ, Gao JP, Lin HX (2008) Overexpression of the trehalose-6-phosphate phosphatase gene OsTPP1 confers stress tolerance in rice and results in the activation of stress responsive genes. Planta 228(1):191–201
Gondor OK, Szalai G, Kovács V, Janda T, Pál M (2016) Relationship between polyamines and other cold-induced response mechanisms in different cereal species. J Agron Crop Sci 202(3):217–230
Howarth CJ, Ougham HJ (2010) Gene expression under temperature stress. New Phytol 125(1):1–26
Hu Q, Fu Y, Guan Y, Lin C, Cao D, Hu W (2016) Inhibitory effect of chemical combinations on seed germination and pre-harvest sprouting in hybrid rice. Plant Growth Regul 80:281–289
Huang Y, Lin C, He F, Li Z, Guan Y, Hu Q (2017) Exogenous spermidine improves seed germination of sweet corn via involvement in phytohormone interactions, H2O2 and relevant gene expression. BMC Plant Biol 17:1–17
Imai R, Ali A, Pramanik HR, Nakaminami K, Sentoku N, Kato H (2004) A distinctive class of spermidine synthase is involved in chilling response in rice. J Plant Physiol 161(7):883–886
Jang IC, Oh SJ, Seo JS, Choi WB, Song IS, Kim CH, Kim YS, Seo HS, Choi WD, Nahm BH, Kim JK (2003) Expression of a bifunctional fusion of the escherichia coli genes for trehalose-6-phosphate synthase and trehalose-6-phosphate phosphatase in transgenic rice plants increases trehalose accumulation and abiotic stress tolerance without stunting growth. Plant Physiol 131(2):516–524
Ji SL, Jiang L, Wang YH, Zhang WW, Liu X, Liu SJ (2010) Quantitative trait loci mapping and stability for low temperature germination ability of rice. Plant Breeding 128(4):387–392
Jing C, Siqi P, Xiulan Z (2016) Effects of exogenous auxins on growth and antioxidation system in maize seedlings under cadmium stress. Plant Physiol J 29:45–59
Kasukabe Y, He L, Nada K, Misawa S, Ihara I, Tachibana S (2004) Overexpression of spermidine synthase enhances tolerance to multiple environmental stresses and up-regulates the expression of various stress-regulated genes in transgenic Arabidopsis thaliana. Plant Cell Physiol 45(6):712–722
Kasukabe Y, He L, Watakabe Y, Otani M, Shimada T, Tachibana S (2006) Improvement of environmental stress tolerance of sweet potato by introduction of genes for spermidine synthase. Plant Biotechnol 23:75–83
Kosar F, Akram NA, Ashraf M, Sadiq M, Al-Qurainy F (2018) Trehalose-induced improvement in growth, photosynthetic characteristics and levels of some key osmoprotectants in sunflower (Helianthus annuus L.) under drought stress. Pak J Bot 50(3):955–961
Kushad MM, Yelenosky G (1987) Evaluation of polyamine and proline levels during low temperature acclimation of citrus. Plant Physiol 84(3):692–695
Li Z, Xu JG, Gao Y, Wang C, Guo GY, Guan YJ, Hu J (2017) The synergistic priming effect of exogenous salicylic acid and H2O2 on chilling tolerance enhancement during maize (Zea mays L.) seed germination. Front Plant Sci 8:1153
Luo Y, Li WM, Wang W (2008) Trehalose: protector of antioxidant enzymes or reactive oxygen species scavenger under heat stress. Environ Exp Bot 63(1–3):378–384
Mo H, Pua EC (2010) Up-regulation of arginine decarboxylase gene expression and accumulation of polyamines in mustard (Brassica juncea) in response to stress. Physiol Plant 114(3):439–449
Moustakas M, Sperdouli I, Kouna T, Antonopoulou CI, Therios I (2011) Exogenous proline induces soluble sugar accumulation and alleviates drought stress effects on photosystem ii functioning of Arabidopsis thaliana leaves. Plant Growth Regul 65(2):315–325
Nahar K, Hasanuzzaman M, Alam MM, Fujita M (2015) Exogenous spermidine alleviates low temperature injury in mung bean (Vigna radiate L.) seedlings by modulating ascorbate-glutathione and glyoxalase pathway. Int J Mol Sci 16(12):30117–30132
Nayyar H, Chander S (2004) Protective effects of polyamines against oxidative stress induced by water and cold stress in chickpea. J Agron Crop Sci 190(5):355–365
Nayyar H, Kaur S, Kumar S, Singh KJ, Dhir KK (2005) Involvement of polyamines in the contrasting sensitivity of chickpea (Cicer arietinum L.) and soybean (Glycine max (L.) merrill.) to water deficit stress. Bot Bull Acad Sin 46(4):333–338
Oscar G, María B, María T, María I, Merodio C (2010) The effects of high co 2 levels on anthocyanin composition, antioxidant activity and soluble sugar content of strawberries stored at low non-freezing temperature. Food Chem 122(3):673–678
Pramanik MH, Imai R (2005) Functional identification of a trehalose 6-phosphate phosphatase gene that is involved in transient induction of trehalose biosynthesis during chilling stress in rice. Plant Mol Biol 58(6):751–762
Saddam H, Fahad K, Hussain HA, Lixiao N (2016) Physiological and biochemical mechanisms of seed priming-induced chilling tolerance in rice cultivars. Front Plant Sci 7:116
Shen W, Nada K, Tachibana S (2000) Involvement of polyamines in the chilling tolerance of cucumber cultivars. Plant Physiol 124(1):431–439
Urano K, Yoshiba Y, Nanjo T, Igarashi Y, Seki M, Sekiguchi F, Yamaguchi-Shinozaki K, Shinozaki K (2003) Characterization of Arabidopsis genes involved in biosynthesis of polyamines in abiotic stress responses and developmental stages. Plant Cell Environ 26:1917–1926
Wan YY, Zhang Y, Zhang L, Zhou ZQ, Li X, Shi Q (2015) Caffeic acid protects cucumber against chilling stress by regulating antioxidant enzyme activity and proline and soluble sugar contents. Acta Physiol Plant 37(1):1706
Wang Y, Wang P, Jiahai WU, Zhao L, Bentian MO, Jun LI (2012a) Effect of polyamine priming on chilling tolerance of lolium perenne during seed imbibition. Agric Sci Technol 13(9):1859–1863
Wang Y, Wang P, Jiahai WU, Zhao L, Bentian MO, Jun LI (2012b) Effect of polyamine priming on chilling tolerance of lolium perenne during seed imbibition. Agric Sci Technol 13(9):1859–1863
Williams B, Njaci I, Moghaddam L, Long H, Dickman MB, Zhang X (2015) Trehalose accumulation triggers autophagy during plant desiccation. PLoS Genet 11(12):1–17
Wingler A (2002) The function of trehalose biosynthesis in plants. Phytochemistry 60(5):437–440
Xie DW, Wang XN, Fu LS, Sun J, Li ZF, Zheng W (2015) Effect of exogenous trehalose on germ length and seedling freeze resistance of winter wheat under cold stress. J Triticeae Crops 32(2):215–223
Zeng YH, Zhang YP, Xiang J, Wu H, Chen HZ, Zhang YK, Zhu DF (2016) Effects of chilling tolerance induced by spermidine pretreatment on antioxidative activity, endogenous hormones and ultrastructure of indica-japonica hybrid rice seedlings. Agric Sci Technol 15(2):295–308
Zhang YP, Zhang HM, Gu HF, Zhang WX, Fan HW, Diao QN (2018) Effect of spermidine on photosynthesis, antioxidant characteristics and related gene expression in melon under chilling stress. Northwest Bot J 38(04):671–681
Zheng YY, Cao DD, Zhang S, Guan YJ, Hu J (2008) Effect of polyamines on chilling tolerance in seed imbibition and seed germination in maize. Acta Agron Sin 34(2):261–267
Zhu L, Cao D, Hu Q, Guan Y, Hu W, Nawaz A (2016) Physiological changes and sHSPs genes relative transcription in relation to the acquisition of seed germination during maturation of hybrid rice seed. J Sci Food Agric 96:1764–1771
Acknowledgements
We greatly appreciate Zhejiang Agricultural Seed Industry co. LTD for providing the seed materials.
Funding
This research was supported by National Key R & D Program of China(No. 2018YFD0100806, 2018YFD0100902), Zhejiang Provincial Natural Science Foundation (LQ19C130004), Key Research and Development Project of Zhejiang Province (No. 2019C02011, 2019C02004), the Fundamental Research Funds for the Central Universities (No. 2019QNA6015), National Natural Science Foundation of China (No. 31671774), Science and Technology Program of Zhejiang Province SannongLiufang Cooperation (No. 2018SNLF004), Dabeinong Funds for Discipline Development and Talent Training in Zhejiang University and Jiangsu Collaborative Innovation Center for Modern Crop Production, P. R. China.
Author information
Authors and Affiliations
Contributions
YYF designed and performed most of the experiments; ZHZ, JL and MC assisted with the experiments; YYF analyzed the data and wrote the article with contributions from all of the authors; WMH provided the seed materials; RHP and JH polished the manuscript; YJG analyzed the data and revised the whole manuscript;
Corresponding author
Ethics declarations
Conflict of interest
There is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fu, Y., Zhang, Z., Liu, J. et al. Seed Priming with Spermidine and Trehalose Enhances Chilling Tolerance of Rice via Different Mechanisms. J Plant Growth Regul 39, 669–679 (2020). https://doi.org/10.1007/s00344-019-10009-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-019-10009-y