Abstract
Conjugated linoleic acid (CLA) in milk arises through microbial biohydrogenation of dietary polyunsaturated fatty acids (PUFA) in the rumen, and by the action of mammary Stearoyl-CoA desaturase (Scd). A large variation (up to tenfold) in the concentration of this fatty acid in milk has been observed, even in cows receiving the same diet. The reasons for this variation are not well understood. In this study the bovine core promoter region was isolated by a genome walking strategy from genomic DNA Genome Walker libraries and then cloned and characterized. This core promoter sequence extended approximately 600 bp upstream of the translation start site. The presence of putative transcription factor binding sites conserved in bovine, human, and mouse promoters was observed. Evidence that this promoter fragment was functional in vivo was obtained from expression studies in a mammary cell line. The promoter sequence of the scd gene was compared between cows selected for the ability to produce high fatty acid methyl esters (FAME) (2.22–2.72) in their milk, with the same promoter region of low-FAME-producing cows (0.81–1.12). However, such comparisons of the sequences of the scd promoter region of cows producing high milk CLA compared with low CLA revealed no polymorphisms in this promoter segment. Furthermore, no sequence polymorphisms were observed among the scd promoter region of Holstein Friesian, Montbeliarde, Normande, Norwegian Red, Charlois, Limousin and Kerry breeds.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Conjugated linoleic acid (CLA) is a collective term to describe one or more positional and geometric isomers of linoleic acid (cis-9, cis-12-C18:2). The cis-9, trans-11-C18:2 isomer is the predominant one in the human diet as a result of microbial biohydrogenation in the rumen. This isomer is produced in ruminants directly as an intermediate during the microbial biohydrogenation of dietary linoleic acid, and endogeneously from trans-vaccenic (trans-11-C18:1) acid in mammary tissue by the action of Stearoyl-CoA desaturase (Scd) (Griinari et al. 2000). Animal fat–containing foods, including dairy products, beef, and lamb, are rich sources of CLA (Chin et al. 1992; Fritsche and Steinhart 1998; O’Shea et al. 2000).
CLA has attracted much attention in recent years because of its many potential health benefits. Studies have shown that CLA exhibits anticarcinogenic activity in animal models (Ha et al. 1990; Belury et al. 1995; Liew et al. 1995; Ip et al. 1996) and in in vitro studies using a range of human cancer cell lines including mammary (Visonneau et al. 1997; Park et al. 2000; Miller et al. 2001), prostate (Cesano et al. 1998; Palombo et al. 2002) and colon (Miller et al. 2001; Palombo et al. 2002). Other properties of CLA include anti-atherogenic activity (Nicolosi et al. 1997; Lee et al. 1994), the ability to reduce the catabolic effects of immune stimulation (Cook et al. 1993; Millar et al. 1994), the ability to enhance growth promotion (Chin et al. 1994), and the ability to reduce body fat (Pariza et al. 1996).
During biohydrogenation of linoleic acid in the rumen, CLA is produced. This CLA is largely a transient intermediate and is involved as an intermediate in the biohydrogenation pathway to produce stearic acid. A buildup of trans-vaccenic acid occurs (Harfoot and Hazelwood 1988). Trans-vaccenic acid is also an intermediate in the biohydrogenation of other PUFA (Griinari and Bauman 1999). Several studies have shown that substantial amounts of trans-vaccenic acid (60–300 g/day) reach the duodenum of lactating cows (Wonsil et al. 1994; Kalscheui et al. 1997a, 1997b). Infusion studies using trans-vaccenic acid post-ruminally resulted in elevated cis-9, trans-11 CLA in milk-fat, indicating conversion in the mammary gland (Griinari and Bauman 1999). Several studies have found substantial Scd activity in both mammary and adipose tissue of ruminant animals (Kinsella 1972; St. John et al. 1991; Ward et al. 1998; Martin et al. 1999). Mammary Scd enzyme, which converts trans-vaccenic acid to cis-9, trans-11 CLA, is believed to be responsible for the formation of the majority of CLA in milk (Griinari and Bauman 1999).
Animal diet is a major factor affecting the milk-fat content of CLA, with dietary supplements containing oils rich in PUFA, such as linoleic and linolenic acids, being the most effective for CLA enrichment of milk. However, substantial variations in the CLA content of milk-fat (ranging from three- to ten fold) of cows on the same dietary treatment have been observed in a number of studies (Jiang et al. 1996; Stanton et al. 1997; Kelly et al. 1998a, 1998b; Lawless et al. 1998, 1999; Solomon et al. 2000; White et al. 2001; Peterson et al. 2002). The reasons for this variation are not well understood. Perhaps ruminally derived CLA is a more important contributor to milk CLA in grass-fed cows. The variation in the CLA content of milk-fat may also be due to a variation in rumen conditions leading to differences in the availability of CLA (or CLA precursors) that escape from the rumen. It might also be caused by differences in mammary Scd activity associated with either regulation of scd gene expression, differences in structure of the enzyme due to gene polymorphisms, or differences in downstream factors that would affect interaction between enzyme and substrate (e.g., phosphorylation) (Peterson et al. 2002).
The promoter regions of the human (Bene et al. 2001; Zhang et al. 2001), chicken (Lefevre et al. 2001), and mouse (Ntambi et al. 1988; Kaestner et al. 1989; Mihara 1990) scd genes have been isolated, cloned, and characterized. It has been shown in these studies that there is a conserved PUFA response region in all three, and that this includes critical binding sites for sterol response element binding protein (SREBP) and Nuclear factor Y (NF-Y) transcription factors. Sequence comparison of the human and mouse promoters indicated a second region of high homology including the 5′UTR and basal/proximal promoter. It has been suggested that there are two different transcription start sites in the human promoter and that these may be dependent on tissue-specific factors (Zhang et al. 2001).
In this study the role that polymorphisms in the core promoter region of the bovine scd gene might play in influencing regulation of the scd gene was investigated. The bovine core promoter was therefore isolated from genomic DNA by a genome walking approach using primers designed to a genomic database sequence for the bovine Stearoyl-CoA desaturase gene (AF481915) that contained 162 bases of 5′UTR. Analysis of this promoter sequence identified a number of conserved potential transcription factor binding sites based on comparison with the human and mouse scd promoter regions and interrogation of the TRANSFAC 4.0 database of transcription factor sequences (Wingender et al. 2000) using the MatInspector V2.2 program (Quandt et al. 1995). Importantly, and for the first time for bovine scd, this core promoter was shown to drive transcription of the reporter gene luciferase in an in vitro mammalian culture system, confirming its presumed function. A number of animals were screened for possible polymorphisms in this promoter region. These included cows producing high milk-fat CLA as a percentage of fatty acid methyl ester (% FAME) content (2.22–2.72), compared with cows yielding a low milk-fat CLA as a percent FAME content (0.81–1.12). Additionally, sequence comparisons of the scd promoter region from a number of different breeds were made.
A longer bovine scd promoter sequence (Accession No. AY241932) is now included in the GenBank database but this was not available at the time of this study; the two promoter sequences were submitted within a very short time of each other.
Materials and methods
Construction of GenomeWalker (GW) libraries
To obtain the 5′ flanking sequence of the scd gene, GenomeWalker (GW) libraries were constructed from bovine total genomic DNA using a Universal GenomeWalker kit (Clontech, UK) according to the manufacturer’s instructions. High-quality genomic DNA was first extracted from cultured lymphoblast cells from a Holstein Friesian animal. Briefly, a cell suspension (15 ml) was centrifuged at 1200 g for 5 min and the pellets mixed and incubated overnight at 37°C in 900 μl 0.2 M EDTA, 0.5% sodium-n-lauroyl sarcosine, and 25 μl of proteinase K (20 mg/ml). Ten microliters of RNase (2 mg/ml) was added to each tube and incubated at 37°C for 1.5 h. The mix was split in two, 200 μl phenol was added and mixed and the mix was incubated for 30 min at 37°C in a rotary mixer. Chloroform (200 μl) was added and then the tubes were shaken vigorously and incubated at 37°C for 1 h in a rotary mixer. Tubes were centrifuged at 16,000 g for 15 min to form two layers. The upper layer was transferred to a fresh tube and two volumes of ice-cold 100% ethanol were added. Tubes were inverted abruptly four times, and at this point a DNA precipitate was obtained. The supernatant was decanted, 100 μl 70% ethanol was added, and the mix was incubated overnight at 21°C in a rotary mixer. Ethanol was decanted off and the pellet was allowed to air-dry. The DNA pellets were resuspended in 50 μl 10 mM Tris-HCl (pH 8.0) containing 1 mM EDTA (TE) buffer and aliquots were checked for integrity on a 0.7% w/v agarose gel containing ethidium bromide (EtBr) (2 μg/ml) gel.
The genomic DNA was digested with four restriction enzymes, DraI, StuI, EcoRV, and PvuII, to create four pools of DNA fragments. Adaptors were ligated onto both ends of these fragments to create four GW libraries: DL1 (DraI), DL2 (StuI), DL3 (EcoRV), and DL4 (PvuII).
Isolation and characterization of the scd promoter from GW libraries
To specifically isolate the scd promoter, genome walking primers pAKGW1 (5′-GCTCTCAGACACTGGGATCACTTTCTCGG G-3′) and pAKGW2 (5′-AACTGAGTGTAGAC TAGTTCCTGAGCCTGC-3′) were designed using Vector NTI software (InforMax, Inc.) to a genomic database sequence for the bovine scd gene (AF481915) and synthesized by MWG Biotech (Germany). The primary PCR was carried out using 1 μl of the GenomeWalker libraries with the gene-specific primer pAKGW1 and adaptor primer AP1 (from kit). The primary PCR products were diluted 1:50 and used as template for nested PCR with a second gene-specific primer pAKGW2 and a second adaptor primer AP2 (again from kit). The reaction final volume was 50 μl containing Taq DNA polymerase buffer (Invitrogen), 1.5 mM MgCl2, 200 μM dNTPs (Promega), 0.3 μM each primer, and 1 U Taq polymerase (Invitrogen). Immediately prior to cycling, 2.5 μl of DMSO was added to the mix. The reaction was amplified for 35 cycles for both the first and second PCR. Cycling was performed in a DNA Engine thermal cycler (MJ Research) and conditions were 95°C for 2 min, followed by 35 cycles of 95°C for 30 sec, 60°C for 30 sec, and 72°C for 3 min. This was followed by a 72°C final extension step for 7 min.
PCR products were directly cloned into the pCR2.1 vector (Invitrogen) and transformed using One Shot Top-10 chemically competent E. coli cells according to the manufacturer’s instructions (Invitrogen). Colony PCR was performed to identify clones that potentially carried the scd promoter. Small-scale preparations of plasmid DNA were made from these colonies using the procedure outlined by the manufacturer (Sigma). Sequencing of three plasmids (DL1.2, DL1.4, and DL1.8) was performed by MWG Biotech (Germany). The resulting sequences were analyzed using Vector NTI software (InforMax, Inc.). Potential transcription factor binding sites were identified using the MatInspector V2.2 program (Quandt et al. 1995) by interrogation of the TRANSFAC 4.0 database of transcription factor sequences (Wingender et al. 2000).
Luciferase reporter vector construction
pCR2.1-based plasmids containing the putative scd promoter were restriction digested at 37°C to release the fragment and create compatible ends for subcloning into the pGL3-Basic (Promega) promoterless expression vector. The pGL3-Basic vector was digested in the same manner. Ligation reactions were transformed into TAM Ultracomp chemically competent E. coli cells (Active Motif Europe), using instructions recommended by the manufacturer.
Cell culture
Chinese Hamster Ovary K1 (CHO-K1) cells obtained from ATCC (CCL-61) were cultured in Dulbecco’s Modified Eagles Medium F12 (Biowhittaker, UK) containing 10% (v/v) fetal bovine serum (Invitrogen). Human mammary MCF-7 cells (ATCC HTB22) were cultured in Eagles Minimum Essential Media (Invitrogen) containing 10% (v/v) fetal bovine serum (Invitrogen) and 1% (v/v) nonessential amino acids (Invitrogen). Cells were routinely passaged every 3–4 days by washing with phosphate buffered saline (PBS) and treating with 3 ml EDTA-trypsin (Sigma) to remove adherent cells. Cells were maintained in a humidified incubator at 37°C and 5% CO2. Cells were grown to 80% confluency and both CHO-K1 and MCF-7 cells were transiently cotransfected with luciferase reporter vectors using Fugene 6 transfection reagent (Roche Diagnostics). Six-well transfection plates were seeded at a concentration of 3 × 105 cells per well and incubated overnight at 37°C and 5% CO2. pGL3–scd experimental constructs (1 μg) were cotransfected with 25 ng of pRL–SV40 plasmid (Promega) to control for transfection efficiency. Forty-eight hours after transfection, media were removed from the wells, 300 μl Passive Lysis Buffer (Promega) was added, followed by incubation at room temperature for 10 min.
Luciferase assay
The Dual Luciferase Assay Kit (Promega) was used to measure both Renilla and Firefly luciferase expression by the reporter vectors on a Tecan Spectrafluor Plus luminometer using the Magellan software (Tecan). Mean Firefly luciferase activity values were corrected for variations in transfection efficiency using the corresponding mean Renilla luciferase figures. The corrected values were expressed as a percentage of the positive control value (pGL3–Control). Transfection values were a result of three independent transfections, with n = 6, for both cell types.
Analysis of scd promoter polymorphism incidence
A dairy herd (n = 75) had been on a ryegrass diet for a period of six months and their milk was analyzed for fatty acid methyl esters content (% FAME) using the gas liquid chromatography (GLC) method described previously (Stanton et al. 1997). FAME content was measured in milk samples from 59 animals taken at the evening milking on two occasions during the grazing season, in July and September. Nine of these 75 cows with consistently low and high milk CLA as a % FAME values over both sampling times were selected for this study. The CLA isomer measured was the cis-9, trans-11 CLA isomer. High CLA animals produced greater than 2.0% FAME (2.22–2.72) in their milk compared with low milk CLA producers of about 1.0% FAME (0.81–1.12). The CLA as a % FAME ranged from 0.81 to 2.72 as quoted in Table 1 and are the mean values of both sampling times.
Genomic DNA, for PCR amplification of the scd promoter fragment, was extracted from whole blood (200 μl) that was collected in heparinized tubes from the coccygeal vein from these nine Holstein Friesian cows and, in addition, from ten cows of different bovine breeds using a Gentra capture column (Gentra). Amplification of the bovine promoter was performed using primers D9Dfor (5′-TGATGGGG TAGTGAGGAGC-3′) and D9Drev (5′-GTTCCT GAGCCTGCTTTTGC-3′) with 1 μl of genomic DNA as template (∼200 ng), in a final volume of 50 μl containing Taq DNA polymerase buffer (Invitrogen), 1.5 mM MgCl2, 200 μM dNTPs (Promega), 0.3 μM each primer, and 1 U Taq polymerase (Invitrogen). Cycling was performed in a DNA Engine thermal cycler (MJ Research) and conditions were 95°C for 1 min, followed by 35 cycles of 93°C for 1 min, 58.5°C for 1 min, 72°C for 2 min and a final extension step of 72°C for 10 min. PCR products were purified using a PCR purification kit (Qiagen). DNA was eluted in 30 μl of PCR-grade water (Sigma).
Results and discussion
Isolation of the bovine scd promoter
Isolation of the bovine scd promoter was achieved using a genome walking strategy, which involved digestion of bovine genomic DNA with four restriction enzymes, DraI, StuI, EcoRV, and PvuII, to create pools of short DNA fragments of varying lengths. Adaptors were ligated onto the ends of these pools of DNA and PCR was employed to “walk” along the genome. Primers for PCR were designed to a bovine genomic sequence for the scd gene (AF481915) that extends approximately 160 bp upstream of the translation initiation codon. PCR products were obtained for libraries DL1 (DraI), DL2 (StuI), and DL3 (EcoRV), but not for the DL4 (PvuII) library. Library DL1 yielded a mixture of fragments but, as these were also the longest, these fragments were purified as a mixed pool and cloned into the pCR2.1 vector. Colony PCR was performed and three plasmids (DL1.2, DL1.4, and DL1.8) were sequenced. BlastN analysis of the three sequences indicated that they were all fragments of the bovine gene and the DL1.4 sequence extended upstream of the bovine genomic database sequence AF481915.
Comparison of bovine, human, and mouse scd promoters
An alignment of the DL1.4 sequence was performed with a human promoter sequence (AF320307) (Zhang et al. 2001) and a mouse promoter sequence (M21280) (Fig. 1). The bovine sequence displayed 67% identity with the human scd sequence and 59% homology with the murine promoter sequence scd1. The bovine sequence has been submitted to the GenBank database (Accession No. AJ555480). Transcription start sites have been identified in both the mouse (Ntambi et al. 1988) and human (Zhang et al. 2001) genes at 28 bp and 37 bp, respectively, from the proximal TATA box in the bovine sequence. The bovine promoter sequence presented in Fig. 1 is numbered relative to the human transcription start site and extends to −407.
Sequence alignment of the DL1.4 sequence with database entries for the human (AF320307) and murine (M21280) scd1 promoters. Numbering is indicated above sequence. Transcription factor binding sites are underlined and in bold; these sites are based on TRANSFAC 4.0 database search results and comparison of human and mouse scd gene promoters. The critical CCAAT box from human studies is overlined. Numbering is relative to the human transcription start site and is indicated by +1. The transcription start site in the mouse promoter is indicated by the symbol •. Abbreviations: RFX-1 = X-box-binding regulatory factor 1, SREBP = sterol response element binding protein, NF = nuclear factor.
Potential transcription factor binding sites were compared with those predicted for the human promoter (Zhang et al. 2001). Putative transcription factor binding sites identified using the MatInspector program (Quandt et al., 1995) included nuclear factor Y, octamer binding factor 1, and muscle-specific Mt-binding sites. Comparison of the mouse, human, and bovine sequences identified a number of conserved sequences in two particular regions of the promoters. The first area of conservation occurs between −37 to −119, where there are two conserved TATA sequences (5′-TTTAAAT-3′ and 5′-TAAAA-3′), a fat-specific element (FSE) (5′-CTGAGGAAA-3′) (−77 to −86), and binding sites for the transcription factors AP-1 (−64 to −68), NF-1 (−102 to −109), and HNF4 (−116 to −119). Analysis of the human SCD promoter has shown that for liver and hair follicles, there was a major transcription initiation site 35 nucleotides downstream from the proximal TATA box (Zhang et al. 2001). It was also shown that another transcription initiation site was present 37 nucleotides downstream from the distal TATA box (Bene et al. 2001), and it was suggested that the human SCD gene has different start sites that depend on different tissue-specific factors (Zhang et al. 2001). The TATA sequence, TTTAAAT, is somewhat unusual: This sequence, where the A in the second position is replaced by a C, T, or G, has been shown to reduce the efficiency of a promoter in in vitro studies (Conchino et al. 1983).
The second conserved region occurs at −313 to −390 and has been designated the PUFA response region. Putative binding sites for transcription factors NF-Y (−313 to −317), NF-1 (−333 to −336), NF-Y/NF-1 (−351 to −361), SREBP (−366 to −376), and SP-1 (−386 to −390) are conserved between the three promoter sequences. The PUFA response region present in the bovine sequence is an element previously shown to mediate the downregulation of mouse scd expression in response to PUFA (Waters et al. 1997). The sterol response element binding protein (SREBP) has been shown to activate scd genes in the mouse (Shimomura et al. 1998; Tabor et al. 1999), and it has also been shown that PUFA negatively regulate SREBP mRNA and protein activation (Xu et al. 1999; Yahagi et al. 1999). This bovine promoter fragment also contains the CCAAT box at −356 to −360 previously shown to be critical for transcriptional activation in the human promoter (Zhang et al. 2001). This CCAAT element is also required for full activation of mouse scd1 and scd2 promoters and binds the NF-Y transcription factor (Tabor et al. 1999).
Transcription studies in CHO-K1 and MCF-7 cells
To determine whether this 407-bp putative promoter sequence was sufficient to direct transcription, luciferase reporter gene constructs containing the putative promoter cloned into the promoter site of the promoter-less expression vector pGL3–Basic were constructed. A plasmid (pRL–SV40) providing constitutive expression of Renilla luciferase was cotransfected to serve as transfection efficiency control. Two cell types were transfected, a human mammary cell line, MCF-7, and a Chinese hamster ovary cell line, CHO-K1. The CHO-K1 cell line is most commonly used to express mammalian genes in vitro and should contain the general transcription factors necessary for activating transcription of most genes. The human mammary cell line contains more tissue-specific transcription factors and is a more suitable system for studying regulation of a gene expressed in the mammary gland.
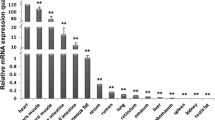
The putative promoter showed promoter activity in both CHO-K1 and MCF-7 cell lines (Fig. 2). Promoter activity was greatest in the mammary MCF-7 cell line, being six fold higher in MCF-7 cells than in CHO-K1 cells. These results indicate that the 407-bp region upstream of the proposed transcription start site is sufficient to direct transcription.
Transient transfection of mammalian cells with the putative bovine scd promoter sequence, (a) Luciferase assay following transient cotransfection of CHO-K1 cells with promoter construct. (b) Luciferase assay following transient cotransfection of MCF-7 cells with promoter construct. Luciferases activities are given as a ratio of Firefly to Renilla (pRL–SV40) values.
Promoter polymorphism screen
Animals receiving an identical dietary treatment of rye grass for a 6-month period were seen to have milk CLA as a % FAME varying from 0.81 to 2.72. The sample size selected was initially 59 Holstein Friesian cows, from which milk was analyzed for CLA content and subsequently nine cows were selected and divided into two groups of high- and low-CLA producers. There were no differences in the milk yields, feed intake, and milk-fat content of the high-versus the low-CLA animals. However, we did observe that blood glucose and nonessential fatty acids (NEFA) differed significantly between high- and low-CLA cows, with glucose being higher in the blood of the low-CLA group, whereas NEFA was lower in the low-CLA group compared with the high-CLA group. To investigate whether polymorphisms in the bovine scd promoter could explain the considerably large inter-cow variation in milk-fat CLA content, the nucleotide sequences of the promoter region of nine cows (Table 1) were analyzed to identify sequence polymorphisms which maybe related to elevation of milk-fat CLA.
In an attempt to also include an increased genetically diverse element, the promoter region of the scd gene was amplified and sequenced from ten animals of different breeds (high genetic merit Holstein Friesian, low genetic merit Holstein Friesian, Montbeliarde, Normande, Norwegian Red, Irish Friesian, Dutch Friesian, Charlois, Limousin, Kerry). No information regarding the milk CLA status of these animals was available.
The scd promoter region sequences were aligned for these 19 animals (nine animals showing high and low milk CLA as a % FAME and ten animals of varying bovine breeds) and compared to search for any polymorphisms in the area (data not shown). This alignment showed the total absence of any polymorphic sites between the bovine scd promoters of 19 animals. This high conservation may be significant in indicating that the regulation of this gene is under extremely rigid control by transcription factors and high conservation is necessary for full activation.
Conclusions
In conclusion we have isolated and partially characterized the bovine scd promoter. This promoter fragment showed 59% and 67% similarity to both the mouse and human sequences, respectively. Differences in the CLA content of milk from these animals were not a consequence of polymorphisms within the core promoter of this gene. The scd promoter sequence exhibits remarkable sequence conservation not just across high-and low-CLA yielding Holstein Friesians but also across ten different breeds. In contrast, considerable differences were seen between human and mouse sequences which included variation in nonbinding regions and binding regions for AP-1, SP-1, and NF-KB transcription factors. Consequently, the observed variation in the levels of milk CLA produced in the Holstein Friesian animals may be explained by differences in ruminant synthesis of CLA or CLA precursors, polymorphisms in the coding sequences of the bovine scd gene, or differences in the regulatory proteins themselves, an area that requires further investigation.
References
MA Belury (1995) ArticleTitleConjugated dienoic linoleate: a polyunsaturated fatty acid with unique chemoprotective properties Nutr Rev 53 83–89
H Bene D Lasky JM Ntambi (200l) ArticleTitleCloning and characterisation of the human Stearoyl-CoA desaturase gene promoter: Transcriptional activation by sterol regulatory element binding protein and repression by polyunsaturated fatty acids and cholesterol Biochem. Biophys Res Commun 284 1194–1198
A Cesano S Vissonneau JA Scimeca D Kritchevsky D Santoli (1998) ArticleTitleOpposite effects of linoleic acid and conjugated linoleic acid on human prostate cancer in SCID mice Anticancer Res 18 1429–1434
SF Chin W Liu JM Storkson YL Ha MW Pariza (1992) ArticleTitleDietary sources of conjugated dienoic isomers of linoleic acid, a newly recognised class of anticarcinogens J Food Comp Anal 5 185–197
SF Chin Storkson KJ Albright ME Cook MW Pariza (1994) ArticleTitleConjugated linoleic acid is a growth-factor for rats as shown by enhanced weight gain and improved feed efficiency J Nutr 124 2344–2349
M Conchino RA Goldman MH Caruthers R Weinmann (1983) ArticleTitlePoint mutations of the adenovirus major late promoter with different transcriptional efficiencies in vitro J Biol Chem 258 8493–8496
ME Cook CC Miller Y Park MW Pariza (1993) ArticleTitleImmune modulation by altered nutrient metabolism: nutritional control of immune-induced growth depression Poult Sci 72 1301–1305
J Fritsche H Steinhart (1998) ArticleTitleAmounts of conjugated linoleic acid (CLA) in German foods and evaluation of dairy intake Z Lebensm Unters Forsch A 206 77–82
JM Griinari DE Bauman (1999) Biosynthesis of conjugated linoleic acid and its incorporation into meat and milk in ruminants MP Yurawecz MM Mossaba JKG Kramer MW Pariza GJ Nelson (Eds) Advances in conjugated linoleic acids research AOCS Press Champaign, IL 180–200
JM Griinari BA Corl SE Lacy PY Chouinard KV Nurmela et al. (2000) ArticleTitleConjugated linoleic acid is synthesised endogenously in lactating cows by Delta-9 desaturase J Nutr 130 2285–2291
YC Ha JM Storkson MW Pariza (1990) ArticleTitleInhibition of benzo(a)pyrene-induced mouse forestomach neoplasia by conjugated dienoic derivatives of linoleic acid Cancer Res 50 1097–1101
CG Harfoot GP Hazlewood (1988) Lipid metabolism in the rumen PN Hobson (Eds) The rumen microbial ecosystem Elsevier Applied Science Publishers London 285–322
C Ip SP Briggs AD Haegele HJ Thompson J Storkson et al. (1996) ArticleTitleThe efficacy of conjugated linoleic acid in mammary cancer prevention is independent of the level or type of fat in the diet Carcinogenesis 17 1045–1050
J Jiang L Bjoerck R Fonden M Emanuelson (1996) ArticleTitleOccurrence of conjugated cis-9, trans-11-octadecadienoic acid in bovine milk: Effects of feed and dietary regimen J Dairy Sci 79 438–445
KH Kaestner JM Ntambi TJ Kelly SuffixJr MD Lane (1989) ArticleTitleDifferentiation-induced gene expression in 3T3-L1 preadipocytes. A second differentially expressed gene encoding Stearoyl-CoA desaturase J Biol Chem 264 14755–14761
KF Kalscheur BB Teter LT Piperovay RA Erdman (1997a) ArticleTitleEffect of dietary forage concentration and buffer addition on duodenal flow of trans-C18:1 fatty acids and milk fat production in dairy cows J Dairy Sci 80 2104–2114
KF Kalscheur BB Teter LS Piperova RA Erdman (1997b) ArticleTitleEffect of fat source on duodenal flow of trans- C18:1 fatty acids and milk fat production in dairy cows J Dairy Sci 80 2115–2126
ML Kelly JR Berry DA Dwyer JM Griinari PY Chouinard et al. (1998a) ArticleTitleDietary fatty acid sources affect conjugated linoleic acid concentrations in milk from lactating dairy cows J Nutr 128 881–885
ML Kelly ES Kolver DE Bauman ME Amburgh ParticleVan LD Muller (1998b) ArticleTitleEffect of intake of pasture on concentrations of conjugated linoleic acid in milk of lactating cows J Dairy Sci 81 1630–1636
JE Kinsella (1972) ArticleTitleJ Stearoyl-CoA as a precursor of oleic acid and glycerolipids in mammary microsomes from lactating bovine: possible regulatory step in milk triglycerides synthesis Lipids 7 349–355
ML Lawless JJ Murphy D Harrington R Devery C Stanton (1998) ArticleTitleElevation of conjugated cis-9, trans-11-octadecadienoic acids in bovine milk because of dietary supplementation J Dairy Sci 81 3259–3267
F Lawless JJ Murphy D Darrington R Devery C Stanton (1999) ArticleTitleInfluence of breed on bovine milk cis-9, trans-11 conjugated linoleic acid content Livestock Prod Sci 62 43–49
KN Lee D Kritchevsky MW Pariza (1994) ArticleTitleConjugated linoleic acid and atherosclerosis in rabbits Atherosclerosis 108 19–25
P Lefevre E Tripon C Plumelet M Douaire C Diot (2001) ArticleTitleEffects of polyunsaturated fatty acids and clofibrate on chicken Stearoyl-CoA desaturase 1 gene expression Biochem Biophys Res Common 280 25–31
C Liew HAJ Schut SF Chin MW Pariza RH Dashwood (1995) ArticleTitleProtection of conjugated linoleic acids against 2-amino-3-methylimidazo [4,5-f] quinoline-induced colon carcinogenesis in the f344 ra—a study of inhibitory mechanisms Carcinogenesis 16 3037–3043
GS Martin DK Lunt KG Britian SB Smith (1999) ArticleTitlePostnatal development of Stearoyl coenzyme-A desaturase gene expression and adiposity in bovine subcutaneous adipose tissue L Anim Sci 77 630–636
K Mihara (1990) ArticleTitleStructure and regulation of rat liver microsomal Stearoyl-CoA desaturase gene L Biochem (Tokyo) 108 1022–1029
CC Millar Y Park MW Pariza ME Cook (1994) ArticleTitleFeeding conjugated linoleic acid to animals partially overcomes catabolic responses to endotoxin injection Biochem Biophys Res Commun 198 1107–1112
A Miller C Stanton Devery (2001) ArticleTitleModulation of arachidonic acid distribution by conjugated linoleic acid isomers and linoleic acid in MCF-7 and SW480 cancer cells Lipids 36 1161–1168
RJ Nicolosi EJ Rogers D Kritchevsky JA Scimeca PJ Huth (1997) ArticleTitleDietary conjugated linoleic acid reduces plasma lipoproteins and early aortic atherosclerosis in hypercholesterolemic hamsters Artery 22 266–277
JM Ntambi SA Buhrow KH Kaestner RJ Christy E Sibley et al. (1988) ArticleTitleDifferentiation-induced gene expression in 3T3-L1 preadipocytes A second differentially expressed gene encoding Stearoyl-CoA desaturase. J Biol Chem 263 17291–17300
M O’Shea R Devery F Lawless J Murphy C Stanton (2000) ArticleTitleMilk fat conjugated linoleic acid (CLA) inhibits growth of human mammary MCF-7 cancer cells Anticancer Res 20 3591–3601
JD Palombo AJ Ganguly BR Bistrian MP Menard (2002) ArticleTitleThe antiproliferative effects of biologically active isomers of conjugated linoleic acid on human colorectal and prostatic cancer cells Cancer Lett 177 163–172
MW Pariza Y Park M Cook K Albright W Liu (1996) ArticleTitleConjugated linoleic acid (CLA) reduces body fat FASEB J 10 3227
Y Park KGD Allen TD Shultz (2000) ArticleTitleModulation of MCF-7 breast cancer cell signal transduction by linoleic acid and conjugated linoleic acid in culture Anticancer Res 20 669–676
DG Peterson JA Kelsey DE Bauman (2002) ArticleTitleAnalysis of variation in cis-9, trans-11 conjugated linoleic acid in milk fat of dairy cows J Dairy Sci 85 2164– 2172
K Quandt K Frech H Karas E Wingender T Werner (1995) ArticleTitleMatInd and Matinspector—new fast and versatile tools for detection of consensus matches in nucleotide sequence data Nucleic Acids Res 23 4878–4884
I Shimomura H Shimano BS Korn Y Bashmakov JD Horton (1998) ArticleTitleNuclear sterol regulatory element-binding proteins activate genes responsible for the entire program of unsaturated fatty acid biosynthesis in transgenic mouse liver J Biol Chem 273 35299–35306
R Solomon LE Chase D Ben–Ghedalia DE Bauman (2000) ArticleTitleThe effect of non-structural carbohydrate and addition of full fat extruded soybeans on the concentration of conjugated linoleic acid in the milk fat of dairy cows J Dairy Sci 83 1322–1329
LC John DK Lunt SB Smith (1999) ArticleTitleFatty acid elongation and desaturation enzyme activities of bovine liver and subcutaneous adipose tissue microsomes J Anim Sci 69 1064–1073
C Stanton F Lawless G Kjellmer D Harrington R Devery et al. (1997) ArticleTitleDietary influences on bovine milk cis-9, trans-11 conjugated linoleic acid content J Food Sci 62 1083–1086
DE Tabor JB Kim BM Spiegahnan PA Edwards (1999) ArticleTitleIdentification of conserved cis-elements and transcription factors required for sterol-regulated transcription of Stearoyl-CoA desaturase 1 and 2 J Biol Chem 274 20603–20610
S Visonneau A Cesano SA Tepper JA Scimeca D Santoli et al. (1997) ArticleTitleConjugated linoleic acid suppresses the growth of human breast adenocarcinoma cells in SCID mice Anticancer Res 17 969–974
RJ Ward MT Travers S Richards RG Vermm AM Salter et al. (1998) ArticleTitleStearoyl-CoA desaturase mRNA is transcribed from a single gene in the ovine genome Biochim Biophys Acta 1391 145–156
KM Waters CW Miller JM Ntambi (1997) ArticleTitleLocalisation of a polyunsatnrated fatty acid response region of Stearoyl-CoA desaurase gene 1 Biochim Biophys Acta 1349 83–42
SL White JA Bertrand MR Wade SP Washburn JT Green SuffixJr et al. (2001) ArticleTitleComparison of fatty acid content of milk from Jersey and Holstein cows consuming pasture or a total mixed ration J Dairy Sci 84 2295–2301
E Wingender X Chen R Hehl H Karas I Liebich et al. (2000) ArticleTitleTRANSFAC: an integrated system for gene expression regulation Nucleic Acids Res 28 316– 319
BJ Wonsil JH Herbein BA Watkins (1994) ArticleTitleDietary and ruminally derived trans-C18:1 fatty acids alter bovine milk lipids J Nutr 124 556–565
J Xu MT Nakamura HP Cho SD Clarke (1999) ArticleTitleSterol regulatory element-binding protein-1 expression is suppressed by dietary polyunsaturated fatty acids. A mechanism for the coordinate suppression of lipogenic genes by polyunsaturated fats J Biol Chem 274 23577–25383
N Yahagi H Shimano AH Hasty M Amemiya–Kudo H Okazaki et al. (1999) ArticleTitleA crucial role of sterol regulatory element-binding protein-1 in the regulation of lipogenic gene expression by polyunsaturated fatty acids J Biol Chem 274 35840–35844
L Zhang GE Lan K Stenn SM Prouty (2001) ArticleTitleIsolation and characterisation of the human Stearoyl-CoA desaturase gene promoter: requirement of a conserved CCAAT cis-element Biochem J 357 183–193
Acknowledgments
This work was supported by a Teagasc Walsh Fellowship. The human mammary cell line, MCF-7, was a gift from Dr. C Curran, University College Hospital, Galway.
Author information
Authors and Affiliations
Corresponding author
Additional information
Abbreviations: CLA= conjugated linoleic acid, Scd= Stearoyl-CoA desaturase, PCR= polymerase chain reaction, SNPs= single nucleotide polymorphisms, PUFA= polyunsaturated fatty acids, FAME= fatty acid methyl esters.
The nucleotide sequence data reported in this article have been submitted to GenBank and has been assigned the accession number AJ555480.
Rights and permissions
About this article
Cite this article
Keating, A.F., Stanton, C., Murphy, J.J. et al. Isolation and characterization of the bovine Stearoyl-CoAdesaturase promoter and analysis of polymorphisms in the promoter region in dairy cows. Mamm Genome 16, 184–193 (2005). https://doi.org/10.1007/s00335-004-2325-0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s00335-004-2325-0