Abstract
The origin and developmental history of Fagus sylvatica forests in south-eastern Norway have been studied through pollen analysis and AMS radiocarbon-dating of peat from two small forest hollows. In this area F. sylvatica appears to have a long history, from the first occurrence of F. sylvatica pollen at ca. 9100 cal. b.p. to its local expansion ca. 1300–1200 cal. b.p. At this time a shift from a diverse landscape mosaic with many plant taxa present, including broad-leaved trees, to a less diverse landscape mosaic with Picea abies and F. sylvatica trees is interpreted from the pollen data. The long history of F. sylvatica suggests that the existing forests are not recent plantations, but implies that these forests are native. The presence of pollen indicative of anthropogenic activity combined with charcoal before the expansion of F. sylvatica, as well as comparison with data from nearby sites, suggest that the forest development was likely to be a result of human activity and climatic changes, particularly changes in moisture conditions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Fagus sylvatica is a widespread and ecologically important forest tree in much of central and western Europe. It just reaches western and south-eastern Norway where it grows in its northernmost known presumed native localities in Europe. The distribution of F. sylvatica in Norway is restricted to two distinct areas—a small population at Seim, north of Bergen in western Norway, and its main area in south-eastern Norway (Jalas and Souminen 1976). Botanists have long been interested in the origin and age of the Seim forest as a remarkable botanical outlying stand. Fægri (1954) published results of pollen analyses of a core from a small hollow in the Seim forest, but was not able to draw any clear conclusions about the origin of the forest. However, he suggested that the forest may have been planted, most likely between ca. 1450 and 950 cal. b.p.. (a.d. 500 and 1000). As the area had been occupied by the Norwegian kings during the Viking Age it has been hypothesised that beech was introduced by early settlers. However, its origin remains uncertain. Both Lid and Lid (2005) and Gjærevoll (1992) propose that F. sylvatica is native to Norway and that F. sylvatica migrated there via southern Sweden. Another hypothesis is that the establishment of F. sylvatica was a result of anthropogenic activity such as agriculture and trading connections with Denmark. On the basis of modern molecular analyses, Myking et al. (2011) argue that F. sylvatica in eastern Norway originates from Denmark and was most likely brought to Norway before and during the Viking Age.
Since the late 18th century, botanists have also carried out ecological (e.g. Økland 1988; Semb 1983; Diekmann et al. 1999) and palaeoecological research on the beech forests in south-eastern Norway. Høegh (1924) carried out the first palaeoecological study. He suggested that beech was a late arrival, probably before the Viking Age. Henningsmoen (1988) has made several palaeoecological studies of the beech forests in the Vestfold area of south-east Norway. In her 1988 study of a core from a small hollow within the beech forest in Larvik (Fig. 1), she dated the increase of F. sylvatica pollen to ca. 930 cal. b.p. (1020 ± 70 yr b.p.). In a study from a bog in Stokke (Fig. 1), Henningsmoen (1988) dated the increase in F. sylvatica pollen to ca. 1150 cal. b.p. (1230 ± 60 yr b.p.). In a study from the Kaupang area (Fig. 1), the rise in F. sylvatica pollen occurs at ca. 1400 cal. b.p. (Sørensen et al. 2007). This area lies close to the coast, and was therefore deglaciated earlier than the other sites mentioned above. In a study of a small hollow at Kapteinstjern north-west of Larvik (Fig. 1), Molinari et al. (2005) dated the establishment of F. sylvatica to 1150 cal. b.p. (a.d. 850). In another study from the same area at Årum (Fig. 1) F. sylvatica pollen has very low values (Bjune et al. 2009). In a detailed study from the Østfold area, Danielsen (1970) concluded that F. sylvatica probably did not grow spontaneously in Østfold. Many of these studies suffer from very few or noradiocarbon dates, and limited taxonomic resolution in the pollen analyses. The study presented here was designed to build on these earlier studies by providing more detailed pollen stratigraphies with robust chronologies.
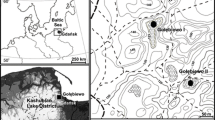
Map showing the study sites Brånakollane (1) and Bøkeskogen (2) in relation to other sites mentioned in the text: (3) Årum, Bjune et al. (2009), (4) Kapteinstjern, Molinari et al. (2005), (5) Kaupang, Sørensen et al. (2007), (6) Bøkeskogen Larvik, Henningsmoen (1988), (7) Bøkeskogen Stokke, Henningsmoen (1988), and (8) Bøkeskogen Seim, Holmboe (1908) and Fægri (1954)
Fagus sylvatica is a monoecious tree with a rather low pollen production, but produces easily recognisable pollen. It has a lifespan of about 300 years and can grow to more than 40 m high. Its shallow root system makes it very vulnerable to drought and wind throw, and its thin bark provides little protection from forest fires (Ellenberg 1988; Giesecke et al. 2007). It starts flowering at an age of about 40–60 years. The seedlings are also very vulnerable to drought and late frost in the spring. Because of their high shade tolerance, seedlings are able to regenerate under the dense canopy of a mature F. sylvatica forest and thus few, if any, other tree species can out-compete F. sylvatica (Ellenberg 1988; Bolte et al. 2007). According to Bolte et al. (2007) the climatic requirements of F. sylvatica are:
-
Precipitation of 500 mm per year, or ca. 250 mm between May and September
-
A July mean temperature of <19 °C
-
<141 frost days with a daily minimum temperature below 0 °C
-
A January mean temperature above −3 °C
-
More than 217 days with a daily mean temperature of 7 °C or more.
F. sylvatica can thus occur under a wide range of climatological conditions.
The use of small hollows as study sites for pollen analysis is a suitable method for investigating the history and development of vegetation at a local scale (e.g. Björkman and Bradshaw 1996; Overballe-Petersen and Bradshaw 2011). Many studies have shown that it is possible to detect patterns in local pollen records from small hollows that are obscured in regional pollen records from lake sediments (e.g. Bradshaw1988; Lynch 1998; Björkman 1999; Lindbladh et al. 2000). This local spatial resolution makes it possible to study succession and replacement processes within a forest, to reveal local ecological patterns, and thus to suggest conditions prior to the establishment of, for example, F. sylvatica (Bradshaw 1988). From the results from a simulation model, Poska and Pidek (2010) found that F. sylvatica pollen loading is significantly reduced at a distance of 300 m from the nearest stand, and that dispersal of F. sylvatica pollen did not exceed 1,800 m from the nearest stand. It is thus an ideal tree for local scale pollen analysis.
The aim of this study is to study the local history of the F. sylvatica forests in south-east Norway using detailed pollen analysis and AMS radiocarbon dates of peat from two small forest hollows. The primary research questions are:
- (i):
-
Are the F. sylvatica forests in south-eastern Norway recent plantations or are they native?
- (ii):
-
If they are native, under what conditions did they form?
- (iii):
-
What factors were important in their origin and subsequent development—human activity, climate change, soil changes, or a combination of these factors?
Study area
The study area belongs to the Oslo Rift geological area. The bedrock consists of syenite, granite and monzonite (Solli and Nordgulen 2007). The first site, Brånakollane, lies in an area of bare rock partly covered with thin surficial deposits and weathering materials. The second site, Bøkeskogen, lies at the top of thick moraine deposits deposited during the Younger Dryas. Mean annual precipitation is 1,000 mm, mean February temperature is −3.3 °C, and mean July temperature is 15.8 °C (www.met.no).
Brånakollane (59.19740, 10.05593 (DD)) lies north of the town of Larvik (Fig. 1), in the eastern part of Norway. The area is a nature reserve with a very well developed and almost undisturbed F. sylvatica forest covering ca 19 ha. It was protected by law in 1980 and there has been no significant forestry at Brånakollane since charcoal-burning ceased with the closure of the Hagenes Ironworks in 1837. The area is hilly with large rocks and outcrops. The border of the reserve is very clear, with F. sylvatica on one side and P. abies on the other side. The dense F. sylvatica forest is surrounded by P. abies forest which is still logged. The small hollow where the core was sampled lies at 184 m a.s.l. and is situated in a dense F. sylvatica stand. The surrounding F. sylvatica trees are about 20–30 m high, with a few young P. abies trees and many young F. sylvatica trees. The ground is covered by a thick layer of old fallen leaves and the soil cover is thin. There are a few old F. sylvatica logs. The field layer is very sparse and consists of Oxalis acetosella, Anemone nemorosa, Poa sp. and ferns. P. abies logs on the ground are partly degraded. The distance to the surrounding P. abies forest is about 30 m.
The second site, Bøkeskogen (59.05761, 10.02251 (DD)), is a F. sylvatica forest in a protected landscape resembling a park inside Larvik (Fig. 1). The forest covers an area of 30 ha. Inside this forest there is one of the largest collections of burial mounds in Vestfold, the oldest one dating from Younger Iron Age/Viking Age 1100–1000 cal. b.p. (a.d. 800–950) (Bjørvik 2007). The sampling point lies in a small hollow at 64 m a.s.l. close to a ditch and with very wet soil. In addition to F. sylvatica, the main species near the sampling point are Populus tremula, Prunus padus, Sorbus aucuparia and P. abies, as well as Vaccinium myrtillus and A. nemorosa. At the other side of the ditch there is a small P. abies forest with one Betula tree. The F. sylvatica trees are rather tall and large, 25 m high and 1 m in diameter, with dense canopies and much under-storey vegetation of young saplings and small bushes of F. sylvatica. P. tremula grows where old F. sylvatica trees have fallen down and created clearings. The ground is covered with dead leaves of F. sylvatica.
Materials and methods
Field work
Overlapping cores were taken with a 50 cm long Russian peat-sampler and were stored in a cold room at 4 °C prior to sampling. The core from Brånakollane is 130 cm long and was obtained in September 2007. The core from Bøkeskogen is 124 cm long and because of the very wet and soft surface it was impossible to drive the corer down to the base. This core was collected in May 2009. The sediment layers were described according to Troels-Smith (1955) (see also Helvik 2010; Figs. 4, 5).
Laboratory work
Subsamples of 0.5 cm3 were extracted from the two cores. The samples were prepared according to standard methods (acetolysis and HF, Fægri and Iversen 1989). To permit estimation of pollen and spore concentrations, a known number of Lycopodium spore tablets were added to each sample prior to preparation (Stockmarr 1971). About 500 terrestrial pollen grains and spores were identified to the lowest taxonomic level using keys (Punt et al. 1976–95; Fægri and Iversen 1989; Beug 2004) and the modern pollen reference collection at the Department of Biology, University of Bergen. At levels 104 and 100 cm at Brånakollane. only 170 and 217 terrestrial pollen grains and spores, respectively, were counted because of low concentrations.
Charcoal fragments were identified and divided into two size classes, <60 and >60 μm. Identification was restricted to black, completely opaque and angular fragments (Clark 1988).
The pollen diagrams were drawn using TILIA (Grimm 1990) and TGView 2.0.2 (Grimm 2004). The percentages were calculated based on the sum of terrestrial pollen and spore types. The pollen diagram was zoned by optimal partitioning using the sum of-squares criterion in the program ZONE (Birks and Gordon 1985). The number of statistically significant zones was determined by comparison with the broken-stick model (Bennett 1996; Birks 1998), using the program BSTICK (Birks and Line unpublished). Plant nomenclature follows Lid and Lid (2005).
Loss-on-ignition (LOI)
Subsamples were extracted from the cores for LOI analysis at the same depths as the pollen samples. Dry weights of the samples were determined after drying overnight at 105 °C. The samples were ignited at 550 °C for six hours and then placed in a desiccator for cooling to room temperature prior to weighing (Bengtsson and Enell 1986; Heiri et al. 2001). LOI was calculated as percentages of dry weight.
Chronology
Fourteen samples of terrestrial plant macrofossils were prepared for AMS radiocarbon-dating, nine from Brånakollane and five from Bøkeskogen (Tables 1, 2). Samples of 1 cm thickness were extracted from the cores and washed through a 250 μm mesh sieve (Birks 2001). Terrestrial plant macrofossils, mainly twigs and woody fragments, were picked out from the residues, identified, and counted at 12× magnification under a stereo-microscope. The samples were analyzed by Laboratoriet for Radiologisk Datering, NTNU, Trondheim.
Calibration of the radiocarbon dates (Tables 1, 2) and age-depth modelling were done using R (R Development Core Team 2011) and routine Clam (Blaauw 2010). Calibration is based on the IntCal09 calibration curve (Reimer et al. 2009). All ages in the text have been rounded to the nearest 10 years, but it should be borne in mind that their probability ranges are larger than this (Figs. 2, 3). For Brånakollane the age-depth model was constructed using linear interpolation (Fig. 2). As the dates show that there is a clear hiatus between 101 and 104 cm at Brånakollane this was taken into account when making the age-depth model for this site (Fig. 2). The age-depth model for Bøkeskogen (Fig. 3) was constructed using a smooth spline run through randomly sampled point estimates from calibrated dates and iterating this process a thousand times. The smooth spline was weighted by the probabilities of the sampled calibrated years (Blaauw 2010). Any of the 1,000 models with age reversals were rejected in the final model.
Age-depth model for Brånakollane constructed using linear interpolation. At each date the solid line indicates the calibrated age ranges and the grey field around it represents the probability distribution. The top 10 cm is missing. The hiatus at 101–106 cm is taken into account when fitting the model. Theradiocarbon dates are given in Table 1
Age-depth model for Bøkeskogen based on a smooth spline. At each date the solid line indicates the calibrated ranges and the grey field around it represents the probability distribution. The radiocarbon dates are given in Table 2
Chronology is presented as calibrated years before present (cal. b.p.), where b.p. is a.d. 1950 (Tables 1, 2; Figs. 4, 5, 6, 7, 8).
Percentage pollen diagram showing selected pollen and spore taxa from the forest hollow at Brånakollane. The data are presented on a depth basis along with a calibrated age-scale. The group Aquatics consists of Isoetes lacustris, Myriophyllum alterniflorum, Nymphaea, Potamogeton (sect. Eupotamogeton), Sparganium-type and Typha latifolia pollen combined. The percentages are based on the sum of terrestrial pollen and spore types. The white silhouettes denote a 10× exaggeration of the percentage values. The local pollen-assemblage zones (LPAZ) are shown in the right-hand column
Summary diagram from Brånakollane showing the %values of Fagus sylvatica and Picea abies pollen and a sum of selected anthropogenic indicators, charcoal, sample scores on PCA axis I (36.7 % of the total variance) and II (16.7 % of the total variance),and estimated palynological richness. These are plotted on a depth scale, along with a calibrated age-scale
Percentage pollen diagram showing selected pollen and spore taxa from the forest hollow at Bøkeskogen. The data are presented on a depth basis along with a calibrated age-scale. The percentages are based on the sum of terrestrial pollen and spore types. The white silhouettes denote a 10× exaggeration of the percentage values. The local pollen-assemblage zones (LPAZ) are shown in the right-hand column
Summary diagram from Bøkeskogen showing the %values of Fagus sylvatica and Picea abies and a sum of selected anthropogenic indicators, charcoal, sample scores on PCA axis I (30.0 % of the total variance) and II (17.9 % of the total variance), and estimated palynological richness. These are plotted on a depth scale, along with a calibrated age-scale
A comparison of Fagus sylvatica pollen percentages at four different sites in the region: Brånakollane (this study), Bøkeskogen (this study), Kapteinstjern (Molinari et al. 2005) and Årum (Bjune et al. 2009), plotted on a common calibrated age-scale. Percentages are based on the sum of terrestrial pollen and spore types
Palynological richness
To estimate palynological richness, rarefaction analyses using the program RAREPOL 1.0 (Birks and Line 1992) were carried out using the raw counts of all terrestrial pollen and spores. Estimates of the number of expected taxa, [E(Tn)] were based on a standardized count of 170 at Brånakollane and 482 at Bøkeskogen.
Principal components analysis
To detect the hidden latent structure or major directions of variation in multivariate pollen data-sets under the assumption of linear responses of the variables to the hidden latent variable (ter Braak and Prentice 1988) principal components analysis (PCA) (Birks and Gordon 1985) was used. Preliminary detrended correspondence analysis of the two data-sets showed that both have gradient lengths of compositional turnover of <2 standard deviations, indicating that linear-based PCA is the appropriate technique to use (ter Braak and Prentice 1988). The percentage data were square-root transformed to stabilise their variances, the PCA was based on a covariance matrix between variables, and the sample scores were scaled for a Euclidian distance biplot. Computations were done using CANOCO version 4.52 (ter Braak and Šmilauer 2002).
Sample scores on PCA axes I and II were plotted stratigraphically along with the palynological richness estimates, selected pollen taxa and pollen groups, and charcoal in Figs. 6 and 7. Both PCA axes are statistically significant when compared with the broken-stick model (Legendre and Legendre 1998).
Results and reconstruction of the general vegetation development
The results are presented as stratigraphical pollen diagrams in Figs. 4 and 6 and are summarised in Figs. 5 and 7.
Brånakollane
The results from Brånakollane are described according to the local or site pollen-assemblage zones (LPAZ) suggested by optimal partitioning (Fig. 4). The two first zones BRÅ-1 and BRÅ-2 are described together as they are very short and are represented by only two samples. BRÅ-3 is divided into subzones BRÅ-3a and BRÅ-3b on the basis of visual inspection. Full details of the pollen data are given in Helvik (2010). A summary of the major changes found at Brånakollane is given in Fig. 6.
LPAZ BRÅ-1 and 2 (12500–11750 cal. yr b.p.) pioneer phase
In these zones (Fig. 4) the LOI-curve increases from 0% to 6 %. Characteristic of this initial phase are occasional pollen of many different taxa, some of them typical pioneer taxa. Alnus, Betula and Pinus sylvestris are the dominant tree types, probably a result of long-distance pollen transport or pollen from local forest stands outside the moraine. The amount of dwarf-shrub pollen is almost constant, dominated by Calluna vulgaris and Vaccinium-type. The major herb pollen types are Poaceae and Cyperaceae. However, pollen from taxa such as Artemisia, Filipendula, Melampyrum, Plantago lanceolata, Rumex acetosa-type and Sedum are also important. Although the site is today situated at 184 m a.s.l, finds of P. lanceolata may reflect the site once being close to sea level, as land-uplift has been about 160 m in this area. P. lanceolata can occur in presumed natural coastal vegetation (Birks 1973). Ferns are dominated by Dryopteris-type and Gymnocarpium dryopteris. Potamogeton (sect. Eupotamogeton), Nymphaea and Typha latifolia pollen and Botryococcus and Pediastrum algae are present, indicating shallow open-water at the site.
LPAZ BRÅ-3a (11750–2300 cal. b.p.) Betula-Pinus sylvestris forest
In this zone (Fig. 4) there is a clear hiatus representing the period from 2550–6750 cal. b.p. (at 101–105 cm). Therefore, description of this zone is divided in two; the period before (11750–6750 cal. b.p.) and the period after (6750–2300 cal. b.p.) the hiatus.
The lower part is characterized by large fluctuations in the LOI-curve (Fig. 4). It rises from less than 10–75 % at 9800 cal. b.p., and shows a minimum value of 40 % at 6800 cal. b.p. The occurrence of Hippophaë rhamnoides pollen suggests that the landscape may still have been open. The dominant tree pollen throughout is Alnus, Betula and P. sylvestris, most likely long-distance transported pollen or pollen from stands outside the immediate area. Other tree pollen present in the lower part of BRÅ-3a is Corylus avellana, Fraxinus excelsior, Populus tremula, Quercus and Salix sp., all relatively light-demanding taxa suggesting a mixed forest, but a rather open landscape as suggested by total herb pollen percentages of more than 20 %. Regular occurrences of Artemisia, Filipendula and R. acetosa -type support this reconstruction (Greisman and Gaillard 2009). From ca. 9500 cal. b.p., herbs such as Artemisia, Filipendula, Melampyrum and R. acetosa-type all show discontinuous pollen curves, suggesting less light-availability and thus a denser forest. However, Filipendula and Melampyrum pollen increases towards the top. Pteridophyte spores increase simultaneously with a decline in tree pollen percentages between 11000–9600 cal. b.p. as Dryopteris-type, Gymnocarpium dryopteris and Huperzia selago all increase.
At ca. 6800 cal. b.p. when the hiatus is observed, there is an abrupt drop in the LOI-curve simultaneously with a peak in charcoal particles of both size classes. The sediment consists of gyttja at this level. The most likely explanation for the hiatus is drying out of the site, leading to the total decomposition of organic material. Just before the hiatus, pollen or spores from aquatic plants appear, such as Isoetes lacustris, Potamogeton (sect. Eupotamogeton) and Sparganium-type, which along with the algae Botryococcus and Pediastrum suggest the presence of open water at the site, but these rapidly disappear. At the same time, the first pollen of F. sylvatica occurs with values higher than 0.5 %. Scattered pollen of P. abies also occurs in the previous zone.
The pollen composition after the hiatus is quite similar to the pollen spectra before the hiatus; LOI fluctuates, but is high. From 2400 cal. b.p. F. sylvatica shows an almost continuous pollen curve, varying between 0.2 and 0.3 %. Towards the end of this period ca. 2350 cal. b.p., there are palynological indications of clearings and openings in the forest and the total tree pollen decreases while F. excelsior, J. communis, P. tremula, Quercus and Salix sp. pollen all show small increases. An increase is seen in Cyperaceae, Filipendula, Melampyrum and R. acetosa-type pollen. The last two may indicate grazing by cattle (Hjelle 1999). Increases in Filipendula pollen and Sphagnum spores suggest wetter conditions locally.
LPAZ BRÅ-3b (2300–300 cal. b.p.) broad-leaved deciduous forest and the establishment of F. sylvatica and P. abies
Throughout this zone (Fig. 4), the LOI-curve is stable around 95 %. Although the individual tree pollen types indicate large fluctuations, the total tree pollen shows small variations around its 75 % values. The total herb pollen curve fluctuates between 10 and 20 % until ca. 350 cal. b.p. when it decreases to <5 % for the rest of the zone. The dominant tree taxa are Alnus, Betula and P. sylvestris. The onset of this period is marked by a large peak in Betula pollen, probably the effect of local pollen deposition. Large fluctuations in the various tree pollen curves may indicate continuous disturbances that created gaps and openings, resulting in changes in the composition of the vegetation. Except for Ulmus and F. excelsior which decline and disappear towards the top, pollen types of other broad-leaved deciduous trees show more or less continuous pollen curves throughout the zone. The presence of Melampyrum, Plantago lanceolata, R. acetosa-type and Urtica pollen suggests some anthropogenic activity, most likely fire and grazing by cattle (Behre 1981; Hjelle 1999). Alnus pollen shows a very distinct increase in the period from 1950 to 1500 cal. b.p., simultaneous with increases in pollen and spores from several moisture-demanding taxa such as Cyperaceae, Filipendula and Dryopteris-type. Increases in pollen from light-demanding taxa such as C. avellana, J. communis and Artemisia, and in charcoal suggest clearance of the forest, as a result of natural or man-made fire or other human activity. From ca. 2200 cal. b.p. the P. abies pollen curve becomes continuous, and from ca. 1450 cal. b.p. it expands with two distinct peaks between 1350 and 800 cal. b.p. Single finds of F. sylvatica pollen occur at the same time. The F. sylvatica pollen shows a small peak at 1330 cal. b.p. and becomes continuous. This value may suggest that F. sylvatica became locally established at the site (Huntley and Birks 1983; Lindbladh et al. 2008). Just before this, there are some scattered finds of Carpinus betulus and Frangula alnus pollen (not shown on Fig. 4). These types have been connected to the establishment of F. sylvatica in southern Sweden (Greisman and Gaillard 2009). The Cyperaceae pollen curve shows some fluctuations before it peaks ca. 1000 cal. b.p.
From ca. 800 cal. b.p., there is a change in the forest composition. Light-demanding taxa such as Acer, C. avellana, F. excelsior, Populus tremula, Salix, Tilia cordata and Ulmus are shaded out and the main components of the forest are now Alnus, Betula, F. sylvatica, P. abies and Pinus sylvestris. From ca. 600 cal. b.p., there is a steep decline in the total amount of herb pollen. Herbs such as Artemisia, Filipendula, Melampyrum, Plantago lanceolata and R. acetosa-type soon disappear, suggesting a denser forest and cessation of locally anthropogenic activity. Meanwhile, increases in fern and Sphagnum spores suggest wetter conditions.
LPAZ BRÅ-4 (300 cal. b.p.—present) F. sylvatica: P. abies forest
The LOI-curve (Fig. 4) has similar values in this zone as in the previous zone. Pollen of F. sylvatica and P. abies reaches values of 20 %. This period is characterised by a new type of forest, dominated by F. sylvatica and P. abies together with Alnus. The high percentages of F. sylvatica pollen suggest a dense local F. sylvatica stand with a closed canopy, similar to the forest in the reserve today. Pollen from broad-leaved deciduous trees such as F. excelsior, P. tremula, Tilia cordata and Ulmus soon disappears and the pollen of Betula, C. avellana, P. sylvestris and Quercus decreases during this period. Small but distinct peaks in Cyperaceae, Poaceae, Rumex acetosa-type and Solidago-type may reflect anthropogenic activity close to the sampling site, most likely involving cattle-grazing.
Summary of Brånakollane
In Fig. 5 the F. sylvatica and P. abies pollen curves are discontinuous in zone BRÅ 3a, the first PCA axis (36.7 % of the total variance) shows a rather consistent trend, and the estimated palynological richness shows fluctuating high values. The charcoal curves indicate a sharp decrease in fire activity. The anthropogenic indicator taxa rise at the top of the zone. In BRÅ-3b the P. abies pollen curve becomes continuous, the F. sylvatica pollen curve is discontinuous at the start but becomes continuous when the anthropogenic indicator pollen curve ceases. In this zone sample scores on the first PCA axis show an increasing trend and by the end of the zone the estimated species richness shows a decrease. In the uppermost zone, when F. sylvatica and P. abies are well established, the PCA scores level off and estimated species richness shows a small increase. Taking all the data into account, the largest changes in the local forest vegetation occurred ca. 1350 cal. b.p. F. sylvatica and P. abies show continuous pollen curves, the anthropogenic indicator pollen types decrease, the PCA axis I scores increase, and the estimated palynological richness starts to decrease, suggesting a decrease in the local mosaic structure of the landscape. The first PCA axis thus reflects the shift from a diverse landscape with broad-leaved trees to a less diverse landscape with P. abies and F. sylvatica trees. PCA axis II (16.7 % of the total variance) appears to reflect more local changes, associated with changes in soil and site moisture.
Bøkeskogen
The results from Bøkeskogen are described according to the three local or site pollen-assemblage zones (LPAZ) and are shown in Fig. 6. LA1 is divided into two subzones based on visual inspection. Full details of the pollen data are given in Helvik (2010). A summary of changes at this site is given in Fig. 8.
LPAZ LA-1a (1600–900 cal. b.p.) deciduous forest and anthropogenic activity
The LOI-curve (Fig. 6) varies throughout the zone. The main tree pollen curves, both Alnus and Betula, decrease simultaneously with the LOI-curve. At the same time there is a peak in large and small charcoal particles, suggesting clearances caused by fires, either natural or man-made. The pollen curves of deciduous trees such as Quercus and Corylus avellana show high values. Scattered pollen of Acer, Fraxinus excelsior and Populus tremula also indicates some disturbance of the forest, as these tree species respond quickly and sprout in openings and gaps in the forest. They are subsequently out-competed by more competitive taxa to form a mixed deciduous forest. From the onset of this period, a typical F. sylvatica pollen curve is present with a long tail of scattered grains with low percentages (Van der Knaap et al. 2005). F. sylvatica pollen values of 0.4 % from ca. 1090 cal. b.p. and > 1.5 % at 1000 cal. b.p. suggest that the tree was locally present (Huntley and Birks 1983; Lindbladh et al. 2008). Studies from Denmark suggest that human impact may be the underlying reason for the lack of, or restricted, expansion of F. sylvatica in some areas (Odgaard 1994). The juvenile seedlings and saplings of F. sylvatica are susceptible to browsing (Boulanger et al. 2009) and this may have prevented their further expansion at this time. The P. abies pollen curve shows scattered finds at the start but becomes continuous throughout the zone. The herb pollen present suggests that the field layer was rich in species, several of which are strong indicators of anthropogenic activity. The main herb pollen type is Poaceae which has its peak simultaneously with the decrease in the tree pollen curve and the peak in charcoal particles, indicating open vegetation. The continuous presence of Hordeum-type pollen is the strongest evidence for the role of human activity in keeping the vegetation open (Lindbladh et al. 2007). Pollen from herbs such as Artemisia, Chenopodiaceae, Filipendula and Urtica are all known indicators of human activity. Ranunculus acris-type and Rumex acetosa-type are indicators of grazing by cattle (Behre 1981; Hjelle 1999). Small but significant peaks in cultural grazing indicators such as Melampyrum and R. acetosa-type are strongly suggestive of local anthropogenic impact on the forest (Molinari et al. 2005). Several herb species suggest that fire may have been an important factor in disturbing the vegetation. The charcoal curves are continuous throughout, suggesting high fire activity. Melampyrum is thought to be one of the most reliable pollen signals of burning, correlating closely with charcoal at many scales of resolution (Innes and Simmons 2000; Tinner and Lotter 2006). Melampyrum pollen percentages are low but present throughout the period. Small fires do not have a great impact on forest composition, but Potentilla-type pollen comes from several taxa, many of which can respond quickly to fire that can cause forest disturbance and an increase in available light (Delarze et al. 1992; Tinner et al. 2000; Tinner and Lotter 2006). The Salix and Cyperaceae pollen curves are rather constant and low until there is a slight rise at the top of the zone ca. 950 cal. b.p. This may be a result of the clearance of the local Alnus stand at this time. The fern curve is rather constant throughout the period, although there is a slow rise simultaneous with a rise in Alnus and Filipendula pollen that may suggest wetter conditions locally.
LPAZ LA-1b (900–400 cal. b.p.) abandonment and reforestation
The LOI-curve (Fig. 6) is rather smooth throughout this zone. Alnus, Betula and P. sylvestris pollen fluctuate, perhaps indicating disturbance and change. The transition to this period starts with steep increases in the Alnus and Betula pollen curves, which suggest that reforestation was taking place. The forest changes from a mixed deciduous forest to a mixed Betula-Pinus sylvestris forest with the establishment and expansion of P. abies, which reaches its maximum pollen value of 17 % ca. 600 cal. b.p. An increase in Betula pollen may be indicative of a post-disturbance succession (Bradshaw and Lindbladh 2005). The F. sylvatica pollen curve starts very low at 900 cal. b.p. and remains low until a small peak >2 % at ca. 500 cal. b.p.; it then starts to increase, suggesting the local expansion of F. sylvatica. Hordeum-type pollen shows only one occurrence in this zone and cultural indicators such as Artemisia, Filipendula, Melampyrum, Plantago lanceolata, Potentilla-type, Ranunculus acris-type, Rumex acetosa-type, Solidago-type and Urtica (Behre 1981; Hjelle 1999) are present but in lower amounts than in the previous zone, probably reflecting reduced anthropogenic activity. High values of Juniperus communis pollen suggest some grazing. A decrease in fern spores and pollen from Filipendula suggests drier conditions until ca. 750 cal. b.p. when conditions again became wetter. From then on, Cyperaceae pollen increases and Sphagnum shows a steep rise, simultaneous with an increase in terrestrial spores and finds of Menyanthes trifoliata pollen. About 750 cal. b.p., there is a peak in Poaceae pollen, indicative of forest clearance since total tree pollen decrease. Simultaneously, there is a peak in charcoal particles, suggesting that fire may be the reason for the clearance, and after this disturbance, light-demanding species such as F. excelsior and Quercus show increases in the pollen curves, possibly reflecting a post-disturbance succession. From ca. 700 cal. b.p., the total tree pollen percentages start to increase and the total herb pollen percentages decrease, indicating a denser forest (Broström et al. 2005; Lindbladh et al. 2007). The forest is now dominated by Pinus sylvestris, Picea abies, Betula and F. sylvatica.
LPAZ LA-2 (400 cal. b.p.—present) Fagus: Pinus forest
The LOI-curve (Fig. 6) starts at 95 % and drops to 80 % ca. 30 cal. b.p. This period is characterised by high values of Alnus, F. sylvatica and Pinus sylvestris pollen. A distinct decrease in Alnus pollen, simultaneous with a peak in large charcoal particles, may suggest forest clearances ca. 230 cal. b.p. caused by fire. F. sylvatica pollen increases from 5 to 25 %, suggesting that F. sylvatica was becoming well established and even became the dominant tree locally. The P. abies pollen curve decreases throughout the period. During the fifteenth and sixteenth centuries, two ironworks were established in the area, which required a considerable supply of charcoal. The preferred tree was P. abies and this may be the reason for the decreased pollen values of P. abies (Molinari et al. 2005). Increases in J. communis and dwarf-shrub pollen may indicate a reduction in browsing intensity (Molinari et al. 2005). Increases in Cyperaceae and Poaceae pollen suggest more open vegetation. Small but distinct increases in Achillea-type, Filipendula, Potentilla-type, Ranunculus acris-type, R. acetosa-type and Urtica pollen indicate that there was still some anthropogenic activity locally. Wetter conditions may be reflected by increases in the Sphagnum spore curve and the occurrence of Menyanthes trifoliata pollen.
Summary of Bøkeskogen
Compared with the record from Brånakollane, this record (Fig. 8) only reflects a limited time from ca. 1600 cal. b.p. to the present-day, although the same trends can be observed. The F. sylvatica and P. abies pollen curves are present discontinuously from the start of the sequence. Anthropogenic indicator pollen is present throughout the record, but decreases simultaneously with the charcoal curves. The charcoal curves indicate high fire activity in the first zone, LA-1a, but show an abrupt fall at the end of the zone. Scores on PCA axis I (30.0 % of total variance) show an increasing trend throughout the record and the estimated palynological richness curve, despite short-term fluctuations, shows a decreasing trend through the record. The interpretations of the PCA axis I scores and the estimated palynological richness are similar to those at Brånakollane, namely a shift towards oligocratic trees in LA-1b and LA-2 and a decrease in the local mosaic structure of the landscape as P. abies and F. sylvatica expanded in LA-2.
Discussion
The Holocene spread of F. sylvatica, as the last of the major European trees, started about 9,000 years ago. The spread is not simultaneous through different part of Europe and does not show a continuous “moving wave” pattern, as there are long delays in different regions at different times (Küster 1997; Magri et al. 2006). Much research has been carried out to establish the major driving forces for the migration of F. sylvatica (e.g. Bradshaw et al. 2010). Climate, natural disturbance, and/or disturbance caused by human activity have all been suggested as possible forcing factors.
Several studies of small hollows have been used to study the immigration and local establishment of F. sylvatica in Denmark and southern Sweden (Lindbladh and Bradshaw 1998; Björkman 1999; Bradshaw and Lindbladh 2005). F. sylvatica arrived in an already forested landscape, but managed to become an important tree in the woodlands of northern Europe where it mainly replaced Quercus (Küster 1997). It spread from central Europe into the southern part of Scandinavia and reached its maximum distribution at about 930 cal. b.p. (1000 b.p.). In Denmark the spread of F. sylvatica shows a clear pattern, beginning about 3200 cal. b.p. (3000 b.p.) in the east, then extending to the west and north where it reached its maximum distribution at about 500 b.p. (Bradshaw and Holmquist 1999; Fritzbøger and Odgaard 2010). The spread of F. sylvatica is probably linked to anthropogenic activity and disturbance by fire. Site conditions such as water availability, soil texture, nutrient availability, microclimate and disturbance can also be important rate-limiting factors and may explain the patchy spreading dynamics of F. sylvatica (Bradshaw and Lindbladh 2005). Studies of palaeo-records show that F. sylvatica values are low at levels where there is evidence for local fire. Mean charcoal concentrations are higher before the establishment of F. sylvatica than after (Bradshaw and Lindbladh 2005). This may suggest that F. sylvatica was able to establish after fire had created disturbance and gaps. Since F. sylvatica is vulnerable to fire, this may also indicate a lower fire frequency after the establishment of F. sylvatica.
The first occurrences of F. sylvatica pollen at Brånakollane are recorded at ca. 9100 cal. b.p. with a percentage of only 0.6 % (Fig. 8). From this time the pollen curve remains discontinuous, and is most likely a result of long-distance pollen transport, but Björkman (1999) suggests that pollen percentages >0.5 % may indicate small stands in the region. The F. sylvatica pollen curve is discontinuous until ca. 1350 cal. b.p. when it reaches a pollen value of 1.9 % suggesting local establishment (Huntley and Birks 1983; Lindbladh et al. 2008). This is in accordance with results from other studies in the area (Henningsmoen 1988; Molinari et al. 2005; Sørensen et al. 2007; Bjune et al. 2009) and suggests that the establishment of F. sylvatica was broadly synchronous within south-east Norway (Fig. 8). P. abies was also established at the site at this time. Variation in the pollen values for Alnus, Corylus avellana and Pinus sylvestris and occasional Acer pollen can be observed in the period before F. sylvatica pollen increases. These may suggest clearances and local successions of trees at or near the site. Simultaneously an increase in Filipendula and Cyperaceae pollen and fern spores and the presence of scattered aquatic-plant pollen types may suggest wetter conditions. Increases in large and small charcoal particles may indicate local fires (Innes and Simmons 2000). Such fires may have resulted in an increase of Corylus avellana, Pinus sylvestris, Calluna vulgaris, Melampyrum and Potentilla-type, all of which are regarded as positive pyrophytes or, at least, as being competitively favoured by fire (Innes and Simmons 2000). Due to the wet climatic conditions at this time (Seppä et al. 2009) the fires were most likely man-made. Pollen from anthropogenic indicator species decreases, suggesting less human activity in the immediate surroundings of the site (Fig. 8).
At the Bøkeskogen site in Larvik (Fig. 7), F. sylvatica pollen is present from ca. 1600 cal. b.p. (0.4 %). Similar to Brånakollane, the low and discontinuous pollen percentages suggest the presence of scattered trees in the area or may be the result of long-distance pollen transport (Fig. 8). This is in accordance with Henningsmoen (1988) where the F. sylvatica pollen curve is present but discontinuous from the bottom of the core at 190 cm depth. At ca. 950 cal. b.p. F. sylvatica reaches pollen percentages that are high enough to indicate local establishment at this site (Huntley and Birks 1983; Lindbladh et al. 2008). The values of the charcoal particle curves suggest more or less continuous fire activity until ca. 900 cal. b.p. (Fig. 7). Simultaneously continuous curves of pollen from anthropogenic indicators such as Hordeum-type are observed, suggesting human activity in the area. In the period preceding ca. 1000 cal. b.p. several small peaks in large charcoal particles occur simultaneously with fluctuations in the LOI-curve, indicating fire as a disturbance factor, making conditions suitable for F. sylvatica to establish. Clearance of Alnus may have facilitated this. During the next 600 years the conditions at the site became drier, the forest denser, and human activity had changed, perhaps with a stronger pressure from grazing cattle. All this may have contributed to the failure of F. sylvatica to expand. The charcoal curve suggests reduced fire activity. Competition from P. abies may also be a critical factor. This may have been exacerbated by a drier climate during the Medieval Warm Period or by grazing by cattle, or, more likely, by a combination of both (Odgaard 1994; Bolte et al. 2007). At 500 cal. b.p. it is likely that the clearance of Alnus and a wetter climate made it possible for F. sylvatica to expand and become the dominant tree at this site (Fig. 7).
Based on the data from these two forest hollows it is most likely that the establishment of F. sylvatica is a result of succession after disturbance by fire and human activities at the two localities studied. At Brånakollane small changes in moisture can be inferred from the pollen diagram simultaneous with the establishment and expansion of F. sylvatica, as reflected by an increase in fern spores and Sphagnum spores in the pollen diagram. In contrast to the establishment of beech in Denmark and southern Sweden, the establishment in Norway coincides with the establishment of P. abies. Competition between F. sylvatica and P. abies may thus have occurred. F. sylvatica is more vulnerable to drought than P. abies (Bolte et al. 2007). This may be a reason why F. sylvatica established after P. abies at Brånakollane. The location of the forest on top of a hill may have benefited P. abies in competition with F. sylvatica. Because of drier climatic conditions F. sylvatica failed to expand in competition with P. abies which expanded simultaneously. Even though F. sylvatica is known to be a strong competitor in forests, it did not manage to be the dominant tree in the forests in south-eastern Norway, as it did in Denmark (Bradshaw and Holmquist 1999) and southern Sweden (Lindbladh et al. 2000). Competition from P. abies may be the reason for this. In this region F. sylvatica is close to its northernmost limits of its natural distribution. This may cause climatic stress which can intensify competition with P. abies. Severe frost during winter and late frost in spring may be critical factors in the regeneration of F. sylvatica (Bolte et al. 2007). In Denmark it was F. sylvatica that out-competed the broad-leaved deciduous forest ca. 3200–2600 cal. b.p. (3000–2500 b.p.) (Bradshaw and Holmquist 1999) and in southern Sweden ca. 1400–900 cal. yr b.p. (1500–1000 b.p.) (Björse and Bradshaw 1998), but in Norway it was F. sylvatica and P. abies that caused this change.
At Bøkeskogen it is possible that F. sylvatica was present in the surroundings from the beginning of the record but it did not become established until the amount of anthropogenic indicator pollen decreased. Seedlings of F. sylvatica are vulnerable to grazing (Odgaard 1994) and pressure from cattle-grazing may have hindered the establishment and expansion of F. sylvatica in Bøkeskogen. The moisture conditions at the site seem to be stable in this period and may therefore not have influenced the establishment of F. sylvatica here. Björkman (1999) suggests that to evaluate whether climatic or anthropogenic causes are the driving forces in establishment of F. sylvatica in a region, it is necessary to see if there is coherence in the time of establishment at the different sites. His study from southern Sweden showed large variations in the time of establishment of F. sylvatica and he proposed therefore that climatic change had not been the limiting factor. As shown in Fig. 8 the establishment of F. sylvatica in south-eastern Norway can be dated to ca. 1300 cal. b.p. In addition the study of Sørensen et al. (2007) dates the establishment of F. sylvatica to ca. 1400 cal. b.p. This suggests that the time period for F. sylvatica establishment was ca. 100–200 years. This narrow interval suggests that climatic conditions may have been an important factor, or at least a contributory factor together with human activity, for the establishment of F. sylvatica in the Larvik region of south-eastern Norway. This contrasts with results from many studies in southern Sweden (e.g. Björse and Bradshaw 1998; Björkman 1999; Lindbladh et al. 2000, 2007) that emphasize anthropogenic activities as the main driving force for the establishment and expansion of F. sylvatica.
Conclusions
Studies of the two localities, Brånakollane and Bøkeskogen, show that F. sylvatica has a long history in this region of south-eastern Norway, from the first occurrence of F. sylvatica pollen ca. 9100 cal. b.p. to its local expansion ca. 1300–1200 cal. b.p. Such a long history strongly suggests that these F. sylvatica forests are not recent plantations. To decide if they are “natural” forests requires further detailed ecological and demographic investigations. Our pollen-analytical data suggest that the forests are native, and thus that F. sylvatica can be considered a native tree in Norway, at least in south-eastern Norway. The results from these two localities suggest that the F. sylvatica establishment was likely to have been a result of human activity and climatic changes, particularly changes in moisture conditions. This is in contrast to studies in southern Sweden where climatic factors appear to have been unimportant or to have played a minor role in the establishment of beech.
References
Behre KE (1981) The interpretation of anthropogenic indicators in pollen diagrams. Pollen Spores 23:225–245
Bengtsson L, Enell M (1986) Chemical analysis. In: Berglund BE (ed) Handbook of holocene palaeoecology and palaeohydrology. Wiley, Chichester, pp 423–452
Bennett KD (1996) Determination of the number of the zones in a biostratigraphical sequence. New Phytol 132:155–170
Beug HJ (2004) Leitfaden der Pollenbestimmung für Mitteleuropa und angrenzende Gebiete. Pfeil, München
Birks HH (2001) Plant macrofossils. In: Smol JP, Birks HJB, Last WM (eds) Tracking environmental change using lake sediments, vol 3., Terrestrial, algal, and siliceous indicatorsKluwer, Dordrecht, pp 49–74
Birks HJB (1973) Past and present vegetation of the Isle of Skye—A palaeoecological study. Cambridge University Press, Cambridge
Birks HJB (1998) Numerical tools in paleolimnology—progress, potentialities, and problems. J Paleolimnol 20:307–332
Birks HJB, Gordon AD (1985) Numerical methods in Quaternary pollen analysis. Academic, London
Birks HJB, Line JM (1992) The use of rarefaction analysis for estimating palynological richness from Quaternary pollen-analytical data. Holocene 2:1–10
Björkman L (1999) The establishment of Fagus sylvatica at the stand-scale in southern Sweden. Holocene 9:237–245
Björkman L, Bradshaw R (1996) The immigration of Fagus sylvatica L. and Picea abies (L.) Karst. into a natural forest stand in southern Sweden during the last 2000 years. J Biogeogr 23:235–244
Björse G, Bradshaw R (1998) 2000 years of forest dynamics in southern Sweden: suggestions for forest management. For Ecol Manag 104:15–26
Bjørvik T (2007) Kulturminner i Larvik-distriktet. Østlands-posten, Larvik. ISBN 978-82-990835-7-7
Bjune AE, Ohlson M, Birks HJB, Bradshaw RHW (2009) The development and local stand-scale dynamics of a Picea abies forest in southeastern Norway. Holocene 19:1,073–1,084
Blaauw M (2010) Methods and code for ‘classical’ age-modelling of radiocarbon sequences. Quat Geochronol 5:512–518
Bolte A, Czajkowski T, Kopa T (2007) The north-eastern distribution range of European beech—a review. Forestry 80:413–429
Boulanger V, Baltzinger C, Saïd S, Ballon P, Picard J-F, Dupouey J-L (2009) Ranking temperate woody species along a gradient of browsing by deer. For Ecol Manag 258:1,397–1,406
Bradshaw RHW (1988) Spatially-precise studies of forest dynamics. In: Huntley B, Webb T (eds) Vegetation history, Kluwer, Dordrecht, pp 725–751
Bradshaw RHW, Holmquist BH (1999) Danish forest development during the last 3000 years reconstructed from regional pollen data. Ecography 22:53–62
Bradshaw RHW, Lindbladh M (2005) Regional spread and stand-scale establishment of Fagus sylvatica and Picea abies in Scandinavia. Ecol 86:1,679–1,686
Bradshaw RHW, Kito N, Giesecke T (2010) Factors influencing the Holocene history of Fagus. For Ecol Manag 259:2,204–2,212
Broström A, Sugita S, Gaillard MJ, Pilesjö P (2005) Estimating the spatial scale of pollen dispersal in the cultural landscape of southern Sweden. Holocene 15:252–262
Clark JS (1988) Particle motion and theory of charcoal analysis: source area, transport, deposition, and sampling. Quat Res 30:67–80
Danielsen A (1970) Pollen-analytical late Quaternary studies in the Ra district of Østfold, southeastern Norway. Årbok for Universitetet i Bergen 1969:14
Delarze R, Caldelari D, Hainard P (1992) Effects of fire on forest dynamics in southern Switzerland. J Veg Sci 3:55–60
Diekmann M, Eilertsen O, Fremstad E, Lawesson J, Aude E (1999) Beech forest communities in the Nordic countries—a multivariate analysis. Plant Ecol 149:203–220
Ellenberg H (1988) Vegetation ecology of central Europe. Cambridge University Press, Cambridge
Fægri K (1954) On age and origin of the beech forest (Fagus sylvatica L) at Lygrefjorden, near Bergen (Norway). Danmarks Geologiske Undersøkelser Rekke II 80:230–249
Fægri K, Iversen J (1989) Textbook of pollen analysis (4th edn). Wiley, Chichester
Fritzbøger B, Odgaard BV (2010) Skovenes historie. In: Møller PF (ed) Naturen i Danmark. Skovene, Gyldendal, pp 55–88
Giesecke T, Hickler T, Kunkel T, Sykes MT, Bradshaw RHW (2007) Towards an understanding of the Holocene distribution of Fagus sylvatica L. J Biogeogr 34:118–131
Gjærevoll O (1992) Plantegeografi. Tapir Forlag, Trondheim
Greisman A, Gaillard MJ (2009) The role of climate variability and fire in early and mid-Holocene forest dynamics of southern Sweden. J Quat Sci 24:593–611
Grimm EC (1990) TILIA and TILIA.GRAPH. PC spreadsheet and graphic software for pollen data. INQUA. Working Group on Data-Handling Methods. Newsletter 4:5–7
Grimm EC (2004) TGView version 2.0.2. Illinois State Museum
Heiri O, Lotter AF, Lemcke G (2001) Loss on ignition as a method for estimating organic and carbonate content in sediments: reproducibility and comparability of results. J Paleolimnol 25:101–110
Helvik I (2010) The beech (Fagus sylvatica) forests in forests in the Larvik region, south-east Norway—their origin and history. MSc thesis, University of Bergen
Henningsmoen K (1988) Bøkeskogen i Larvik—en pollenanalytisk undersøkelse. Blyttia 46:203–207
Hjelle KL (1999) Use of modern pollen samples and estimated pollen representation factors as aids in the interpretation of cultural activity in local pollen diagrams. Nor Archaeol Rev 32:19–39
Høegh OA (1924) Bøken i Norge. Tidsskr Skogbruk 2:61–254
Holmboe J (1908) Bøkeskogen ved Lygrefjorden i Nordhordaland. Bergen Museums Årbok 13:33–52
Huntley B, Birks HJB (1983) An atlas of past and present pollen maps for Europe: 0–13,000 years ago. Cambridge University Press, Cambridge
Innes JB, Simmons IG (2000) Mid-Holocene charcoal stratigraphy, fire history and palaeoecology at North Gill, North York Moors, UK. Palaeogeogr Palaeoclimatol Palaeoecol 164:151–165
Jalas J, Souminen J (1976) Atlas Florae Europaeae. 3. Salicaceae to Balanophoraceae. The Committee for Mapping the Flora of Europe and Societas Biologica Fennica Vanamo, Helsinki
Küster H (1997) The role of farming in the post glacial expansion of beech and hornbeam in the oak woodlands of central Europe. Holocene 7:239–242
Legendre P, Legendre L (1998) Numerical ecology. Elsevier, Amsterdam
Lid J, Lid DT (2005) Norsk Flora. Det Norske Samlaget, Oslo
Lindbladh M, Bradshaw R (1998) The origin of present forest composition and pattern in southern Sweden. J Biogeogr 25:463–477
Lindbladh M, Bradshaw R, Holmquist BH (2000) Pattern and process in South Swedish forests during the last 3000 years, sensed at stand and regional scales. J Ecol 88:113–128
Lindbladh M, Brunet J, Hannon G, Niklasson M, Eliasson P, Eriksson G, Ekstand A (2007) Forest history as a basis for ecosystem restoration—a multiple case study in a south Swedish temperate landscape. Restor Ecol 15:284–295
Lindbladh M, Niklasson M, Karlsson M, Björkman L, Churski M (2008) Close anthropogenic control of Fagus sylvatica establishment and expansion in a Swedish protected landscape—implication for forest history and conservation. J Biogeogr 35:682–697
Lynch EA (1998) Origin of a park-forest vegetation mosaic in the Wind River Range, Wyoming. Ecol 79:1,320–1,338
Magri D, Vendramin GG, Compe B, Dupanloup I, Geburek T, Gömöry D, Latalowa M, Litt T, Paule L, Roure JM, Tantau I, Van der Knaap WO, Petit RJ, De Beaulieu J-L (2006) A new scenario for the Quaternary history of European beech populations: palaeobotanical evidence and genetic consequenses. New Phytol 171:199–221
Molinari C, Bradshaw RHW, Risbøl O, Lie M, Ohlson M (2005) Long-term vegetational history of a Picea abies stand in south-eastern Norway: implications for the conservation of biological values. Biol Conserv 126:155–165
Myking T, Yakovlev I, Ersland GA (2011) Nuclear genetic markers suggest Danish origin of East-Norwegian beech (Fagus sylvatica L.) populations established 650–800 AD. Tree Genet Genomes 7:587–596
Odgaard BV (1994) Holocene vegetation history of northern west Jutland, Denmark. Opera Bot 123:1–171
Økland T (1988) An ecological approach to the investigation of beech forest in Vestfold, SE Norway. Nord J Bot 8:375–407
Overballe-Petersen MV, Bradshaw RHW (2011) The selection of small hollows for pollen analysis in boreal and temperate forest regions. Palynology 35:146–153
Poska A, Pidek IA (2010) Pollen dispersal and deposition characteristics of Abies alba, Fagus sylvatica and Pinus sylvestris, Roztocze region (SE Poland). Veget Hist Archaeobot 19:91–101
Punt W (eds) et al (1976–95) The North-west European pollen flora, vols. I–V. Elsevier, Amsterdam
R Development Core Team (2011) R: a language and environment for statistical computing. R foundation for statistical computing. Vienna, Austria. www.R-project.org
Reimer PJ, Baillie MGL, Bard E, Bayliss A, Beck JW, Blackwell PG, Bronk RC, Buck CE, Burr GS, Edwards RL, Friedrich M, Grootes PM, Guilderson TP, Hajdas TP, Heaton TJ, Hogg AG, Hughen KA, Kaiser KF, Kromer B, McCormac G, Manning S, Reimer RW, Richards DA, Southon JR, Talamo S, Turney CSM, Van der Plicht J, Weyhenmeyer CE (2009) IntCal09 and Marine09 radiocarbon age calibration curves, 0–50,000 years cal b.p. Radiocarbon 51:1,111–1,150
Semb G (1983) Jordundersøkelser i norske bøke- og eikeskoger. Jord Myr 2:25–70
Seppä H, Bjune AE, Telford RJ, Birks HJB, Veski S (2009) Last nine-thousand years of temperature variability in Northern Europe. Climates of the Past 5:523–535
Solli A, Nordgulen Ø (2007) Berggrunnskart over Norge og kaledoniene i Sverige og Finland-M1:2 000 000 Norges geologiske undersøkelse, Trondheim
Sørensen R, Henningsmoen KE, Høeg HI, Stabell B, Bukholm K (2007) Geology, soils, vegetation and sea-levels in the Kaupang area. In: Skre D (ed) Kaupang in Skiringssal Kaupang excavation project publication series, I Norske Oldfunn 22. Aarhus University Press, Aarhus, pp 251–272
Stockmarr J (1971) Tablets with spores used in absolute pollen analysis. Pollen Spores 13:615–621
Ter Braak CJF, Prentice IC (1988) A theory of gradient analysis. Adv Ecol Res 18:271–317
Ter Braak CJF, Šmilauer P (2002) CANOCO reference manual and CanoDraw for Windows user`Guide: Software for canonical community ordination (version 4,5). Microcomputer Power, Ithaca
Tinner W, Lotter AF (2006) Holocene expansions of Fagus sylvatica and Abies alba in Central Europe: where are we after eight decades of debate? Quat Sci Rev 23:526–549
Tinner W, Conedera M, Gobet E, Hubschmid P, Wehrli M, Ammann B (2000) A palaeoecological attempt to classify fire sensitivity of trees in the southern Alps. Holocene 10:565–574
Troels-Smith J (1955) Karakterisering af løse jordarter. Danmarks Geologiske Undersøkelser series IV 3
Van der Knaap WO, Van Leeuwen JFN, Finsinger W, Gobet E, Pini R, Schweizer A, Valsecchi V, Amman B (2005) Migration and population expansion of Abies, Fagus, Picea and Quercus since 15000 years in and across the Alps, based on pollen-percentage threshold values. Quat Sci Rev 24:645–680
Acknowledgments
We are grateful to Mikael Ohlson for providing the core and the material from Brånakollane, and to Kari Henningsmoen for access to unpublished work and the two anonymous reviewers for their comments. This is publication no. A 401 from the Bjerknes Centre for Climate Research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M.-J. Gaillard.
Rights and permissions
About this article
Cite this article
Bjune, A.E., Helvik, I. & Birks, H.J.B. The Fagus sylvatica forests in the Larvik region, south-eastern Norway: their origin and history. Veget Hist Archaeobot 22, 215–229 (2013). https://doi.org/10.1007/s00334-012-0371-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00334-012-0371-1