Abstract
Objectives
The multiple arterial-phase (AP) technique was introduced for liver MRI, but it is not really known if multiple AP MRI (MA-MRI) improves image quality and lesion detection rate on gadoxetate disodium–enhanced MRI in comparison with single AP MRI (SA-MRI). We aimed to determine the clinical usefulness of MA-MRI in comparison with SA-MRI.
Methods
Original articles reporting the percentage of adequate AP imaging and the lesion detection rate on gadoxetate disodium–enhanced MA-MRI were identified in PubMed, EMBASE, and Cochrane Library databases. The pooled percentage of adequate AP imaging and lesion detection rate were calculated using random-effects meta-analysis of single proportions. Subgroup analysis was performed to explain causes of study heterogeneity, and publication bias was evaluated using Egger’s test.
Results
Of 772 articles screened, 22 studies in 12 articles were included: 18 studies (ten MA-MRI and eight SA-MRI) suitably defined the percentage of adequate AP imaging and four (three MA-MRI and one SA-MRI) defined the lesion detection rate. MA-MRI had 16.1% higher pooled percentage of adequate AP imaging than SA-MRI (94.8% vs. 78.7%, p < 0.01). MA-MRI additionally detected 33.2% of lesions than SA-MRI (83.2% vs. 50.0%, p = 0.06). Substantial study heterogeneity was found in MA-MRI, and the definition of adequate AP imaging, lesion characteristics, and reference standards were significant factors affecting study heterogeneity (p ≤ 0.02). Significant publication bias was found in MA-MRI (p < 0.01) but not in SA-MRI studies (p = 0.87).
Conclusions
Gadoxetate disodium–enhanced MA-MRI may be more clinically useful than SA-MRI, but further study is necessary to validate this finding because of study heterogeneity and publication bias.
Key Points
• Multiple arterial-phase MRI (MA-MRI) had a 16.1% higher pooled percentage of adequate AP imaging than single arterial-phase MRI (SA-MRI) (94.8% vs. 78.7%, p < 0.01).
• MA-MRI additionally detected an extra 33.2% of lesions compared with SA-MRI (83.2% vs. 50.0%, p = 0.06).
• Substantial study heterogeneity and significant publication bias were found across MA-MRI studies.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Gadoxetate disodium (Eovist/Primovist; Bayer HealthCare) contrast agent has been widely used in liver magnetic resonance imaging (MRI) because its hepatocyte-specific uptake enables hepatobiliary-phase imaging as well as dynamic phase imaging similar to that obtained using conventional extracellular contrast agents [1, 2]. However, despite the importance of the hepatobiliary-phase images obtained on gadoxetate disodium–enhanced MRI which can provide additional lesion detection and characterization, arterial-phase (AP) imaging is of the upmost important in the diagnosis and evaluation of treatment response in hypervascular tumors such as hepatocellular carcinoma (HCC) [3,4,5]. However, the detection of arterial-phase hyperenhancement (APHE) on gadoxetate disodium–enhanced MRI can be particularly challenging because of unsatisfactory AP images, with weaker APHE due to the smaller administered volume and gadolinium content and more frequent transient severe motion artifacts in comparison with extracellular contrast agent [6, 7].
To overcome these limitations, the multiple AP technique involving the acquisition of multiple AP images with a fixed time delay was introduced [8]. The multiple AP technique provides images from more than one AP, which offers the potential for improved precise evaluation of APHE in focal hepatic lesions and a more lesion detection [2]. Although the spatial resolution and signal-to-noise ratio can be reduced because of the requirement for a higher temporal resolution to obtain multiple AP images [2], these weaknesses can be mitigated by efforts to accelerate the acquisition speed of MRI, including improvements in MRI techniques [9].
Many studies have suggested that acquisition of multiple AP images can improve the rate of adequate AP imaging and lesion detection in comparison with acquisition of a single AP image [10,11,12,13,14,15,16,17,18,19,20,21]. However, while these studies generally agree on the clinical usefulness of multiple AP MRI (MA-MRI) with gadoxetate disodium, the reported results are quite variable, and it is difficult to determine to what extent MA-MRI can improve the rate of adequate AP imaging and lesion detection in comparison with single AP MRI (SA-MRI). For example, Ichikawa et al reported that MA-MRI resulted in an additional 7% in the rate of adequate AP imaging (98% vs. 91%) compared with SA-MRI, whereas Yoon et al reported a 26% higher percentage of adequate AP imaging (90% vs. 74%) [13, 20]. Therefore, it is necessary to critically and objectively review the published literature before MA-MRI is widely adopted in clinical practice.
In this systematic review and meta-analysis, we aimed to determine the clinical usefulness of gadoxetate disodium–enhanced MA-MRI in respect to obtaining adequate AP imaging and detection of hepatic lesions, making comparisons with SA-MRI.
Materials and methods
This systematic review and meta-analysis was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [22]. The following literature search, study selection, data extraction, and study quality assessment were independently conducted by two reviewers (6 years and 1 year of experience in systematic reviews and meta-analyses, respectively), and any disagreements were resolved in consensus.
Search strategy
A literature search of the PubMed, EMBASE, and Cochrane Library databases was conducted to find research articles reporting the percentage of adequate AP imaging and the lesion detection rate. The search terms included “liver,” “MRI,” and “arterial phase,” and the detailed search terms are summarized in Supplementary Table 1. The search was limited to original studies on human subjects written in English and published between January 1, 2011, and March 22, 2021.
Eligibility criteria
The inclusion criteria were (1) patients: adults (≥ 18 years old) who underwent MRI evaluation; (2) index test: gadoxetate disodium–enhanced MRI with multiple AP images; (3) comparison: gadoxetate disodium–enhanced MRI with a single AP; and (4) outcomes: proportion of adequate AP imaging and lesion detection rate.
Studies meeting any of the following criteria were excluded: (1) studies not clearly describing the percentage of adequate AP imaging or the lesion detection rate; (2) studies with a population overlapping with another included study; (3) case reports or series including fewer than ten patients; and (4) study protocols, conference abstracts/papers, reviews, guidelines, books, letters, editorials, errata, and surveys.
Data extraction and quality assessment
The following data were extracted from each eligible study: (1) study characteristics: authors, publication year, country, institution, duration of patient enrollment, and study design; (2) patient characteristics: number of patients, age, sex, and the purpose of liver MRI; (3) MRI characteristics: MRI vendor, magnetic field, type of contrast agent, free breathing image acquisition, number of multiple AP images, and specific imaging techniques for multiple AP imaging; (4) reading process: number of readers and imaging reading process used to determine representative results among the readers (average vs. consensus); (5) reference standard; and (6) outcomes: percentage of adequate AP imaging and lesion detection rate.
A tailored tool modified from the Quality Assessment of Diagnostic Accuracy Studies-2 (QUADAS-2) tool was used to assess study quality. Details of this modified QUADAS-2 tool are summarized in Supplementary Table 2. The risk of bias and applicability in regard to patient selection, index test, and image analysis were evaluated for each individual study.
Data synthesis
To evaluate the clinical usefulness of MA-MRI in comparison with SA-MRI, the results for MA-MRI and SA-MRI in all included articles were segregated and analyzed separately. The pooled percentage of adequate AP imaging and the lesion detection rate on the AP images were calculated using random-effects meta-analysis of single proportions [23]. The lesion detection rate refers to the number of lesions detected on the AP images divided by the total number of analyzed lesions. Heterogeneity was evaluated using Higgins’ inconsistency index (I2 > 50% indicating substantial heterogeneity) [24]. Publication bias was evaluated using funnel plots and Egger’s test (p < 0.05 indicating significant bias) [25].
Subgroup analyses were conducted using the following covariates: (1) geographical region of the study population (eastern vs. western); (2) study design (prospective vs. retrospective); (3) lesion characteristics (HCC or precursors vs. others); (4) magnetic field strength of the scanner; (5) multiple AP technique characteristics (free breathing imaging acquisition, number of multiple APs, and use of view sharing technique); (6) definition of adequate AP imaging (strict vs. less strict definition of adequate AP); (7) reference standard (pathology only vs. other MRI sequences); and (8) reading process (independent multiple reviewers vs. single reviewer or consensus reading). Regarding lesion characteristics, others included non-hypervascular hepatic lesions (i.e., metastasis or biliary cystadenocarcinoma) as well as HCCs. A strict definition of adequate AP imaging was applied, which required all of the following three features to be present: strong enhancement of the hepatic artery, weaker enhancement of the portal vein in comparison with the hepatic artery, and no hepatic vein enhancement [11, 24]. p-values < 0.05 were considered statistically significant. All statistical analyses were conducted using the “metafor,” “meta,” and “weightr” packages in R version 4.0.3 (R Foundation for Statistical Computing).
Results
Literature search
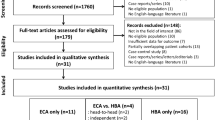
The PubMed (n = 609), EMBASE (n = 1035), and Cochrane Library (n = 17) database searches retrieved 772 articles (Fig. 1). In a first screening by titles and abstracts, 719 articles were excluded (the specific reasons for exclusion are illustrated in Fig. 1). Of the 53 articles remaining for full-text review, 12 articles were finally included in the analysis: nine articles only reporting the percentage of adequate AP image, and two only reporting the lesion detection rate, and one reporting both the two outcomes. When the results of MA-MRI and SA-MRI in all included articles were separated, among the ten articles reporting the percentage of adequate AP imaging, eight studies used a single AP acquisition and ten studies used multiple AP acquisitions. In the three articles reporting lesion detection rate, one study used a single AP acquisition and three studies used multiple AP acquisitions.
Study characteristics
The demographic characteristics of the 12 included articles are summarized in Table 1. Among the 12 articles, eight were of eastern origin [11, 13, 15,16,17,18,19,20] and four were of western origin [10, 12, 14, 21]. Three articles were prospectively conducted [14, 17, 21], and the remaining nine were retrospectively performed. The number of patients ranged from 24 to 752. MRI was performed in patients at risk for HCC in seven articles [11, 13, 15, 17,18,19,20], and patients with suspected focal liver lesions in one article [14], whereas the population characteristics were not specified in four articles.
A 3-T MRI scanner was used in 11 articles [10,11,12,13,14,15,16,17, 19,20,21], whereas one article used both 1.5-T and 3-T scanners [18] (Table 2). Only one article used a free breathing technique [19]. The number of multiple APs varied from 2 to 6. The view sharing technique was used to obtain multiple APs in six articles [10, 14,15,16, 18, 19]. Details of the MRI techniques used are summarized in Table 2.
Of the three articles that reported lesion detection rate, one article used pathology only as a reference standard [11] and two used other MRI sequences [14, 20].
Study quality results
The results of the study quality assessment using the modified QUADAS-2 are summarized in Supplementary Table 3. In the patient selection domain, four articles had an unclear risk of bias because they did not fully explain whether they avoided inappropriate exclusion or did not clearly state whether patients were consecutively included [10, 15, 18, 19]. In the index test domain, three articles had a high risk of bias because they used different multiple AP techniques in the same study [10, 11, 15]. In the image analysis domain, one article had a high risk of bias because independent image analysis was not performed [12].
Percentage of adequate AP acquisitions
In the total of 18 studies with 2371 subjects, the percentage of adequate AP imaging ranged from 56.7 to 100%, with a pooled percentage of 88.6% (95% confidence interval [CI], 83.3–92.3%) (Fig. 2). In the ten MA-MRI studies, the pooled percentage of adequate AP imaging was 94.8%, which was significantly higher than the 78.7% in the eight SA-MRI studies (94.8% [95% CI = 90.1–97.3%] vs. 78.7% [95% CI = 68.8–86.1%], p < 0.01), and provided an extra 16.1% of acquisition with adequate AP imaging. Substantial study heterogeneity was present in both groups (I2 = 82.8% in MA-MRI and 90.9% in SA-MRI). Publication bias was significant in the MA-MRI studies (p < 0.01) but not in the SA-MRI studies (p = 0.87) (Supplementary Figure 1).
In the subgroup analysis, the percentage of adequate AP imaging on MA-MRI differed significantly according to the definition of adequate AP imaging (Table 3). Studies that used a strict definition of adequate AP imaging had a significantly lower percentage of adequate AP imaging than those that used a less strict definition (91.6% vs. 98.1%, p = 0.01).
Lesion detection rate
In four studies with 221 subjects, the lesion detection rate on the AP images ranged from 50.0 to 93.9%, with a pooled detection rate of 77.3% (95% CI, 58.2–89.3%) (Fig. 3). In three MA-MRI studies, the pooled lesion detection rate was 83.2% (95% CI, 69.4–91.5%), whereas in one SA-MRI study, it was 50.0% (95% CI, 31.0–69.0%). Compared with SA-MRI, MA-MRI showed a tendency for a higher lesion detection rate, with an additional 33.2% of lesions detected, but the difference was not statistically significant (83.2% vs. 50.0%, p = 0.06). MA-MRI group showed substantial study heterogeneity (I2 = 63.5%), whereas no significant publication bias was found (p = 0.59) (Supplementary Figure 2).
In the subgroup analysis, the lesion detection rate in MA-MRI studies differed significantly according to lesion characteristics and reference standards (Table 3). Studies exclusively including HCC or precursor HCC lesions had a significantly higher lesion detection rate than those including both HCC and non-hypervascular tumors (93.9% vs. 78.3%, p = 0.02). Studies that used pathology only as the reference standard had a significantly higher lesion detection rate than those with other MRI sequences as the reference standard (93.9% vs. 78.3%, p = 0.02). Due to a lack of eligible study for SA-MRI (n = 1), subgroup analysis of SA-MRI was not available.
Discussion
Our meta-analysis found that MA-MRI had a significantly higher percentage of adequate AP imaging than SA-MRI (94.8% vs. 78.7%, p < 0.01). In addition, although it was not statistically significant, there was a tendency for the lesion detection rate to be higher on MA-MRI than on SA-MRI (83.2% vs. 50.0%, p = 0.06). However, as the reported results varied across the studies according to the definition of adequate AP imaging lesion characteristics, and reference standards, careful interpretation of the findings is needed.
The higher percentage of adequate AP imaging and the higher lesion detection rate on MA-MRI compared with SA-MRI may be associated with technical advances in liver MRI. Our results are in concordance with those in previous studies, which reported improvements of MA-MRI in obtaining adequate AP with reduced artifact [11]. Recent techniques for shortening the MRI acquisition time, such as compressed sensing (acquiring less data through undersampling of k-space) and parallel imaging (simultaneous data acquisition via multiple receiver coil elements of phase array coils), allow multiple AP imaging acquisitions with preservation of image quality [9], thereby enabling acquisition and selection of the most appropriate AP images and improving the lesion detection. As there is more chance of observing the dynamic enhancement pattern of the tumor when multiple AP images are acquired [17], MA-MRI should provide more valuable information for characterizing focal hepatic lesions. Therefore, in the characterization of focal hepatic lesion in patients at risk for HCC, MA-MRI may be more clinically useful than SA-MRI, as well as providing an increased probability of obtaining an adequate AP acquisition.
However, the percentage of adequate AP imaging on MA-MRI differed according to its definition. Unlike the studies using a strict definition of adequate AP imaging (all three of the following features present: strong enhancement of the hepatic artery, weaker enhancement of the portal vein than the hepatic artery, and no hepatic vein enhancement), those using a less strict definition of adequate AP imaging may have considered an AP image to be adequate when only two or more features were present. Because the performance of an index test can differ according to the pre-specified cut-off value or definition of positive result [26], the different definitions of adequate AP imaging provide a reasonable explanation for the cause of different percentages of adequateness. Therefore, we consider the value of 91.6% for studies that used a strict definition to be a reliable estimate of the percentage of adequate AP on MA-MRI with gadoxetate disodium.
Regarding lesion characteristics, studies exclusively including HCC or its precursors had a significantly higher lesion detection rate on MA-MRI than those including various lesions (93.9% vs. 78.3%). Because all included studies analyzed the lesion detection rate on only AP images in comparison with reference standards, the lesion detection rate may be related to the hypervascularity of the analyzed lesions. In other words, the higher lesion detection rate in studies including HCC or its precursors may be due to the hypervascular presentation of most HCCs, whereas the lower lesion detection rate in other studies may be because they analyzed heterogeneous and non-hypervascular lesions (metastasis and biliary cystadenocarcinoma) [14, 20], which are usually difficult to detect on AP images. Therefore, lesion detection rates on MA-MRI should be interpreted with consideration of the characteristics of the analyzed lesions.
In addition, lesion detection rate was also significantly different according to the type of reference standard. Generally, considering that the diagnostic performance of an index test is evaluated against a reference standard for the disease of interest, the performance of an index test can vary according to the characteristics of the participants, setting, tests, and reference standard [27]. However, our result should be cautiously interpreted because all the studies including HCC or its precursors used pathology only as the reference standard which served as another source of heterogeneity. By contrast, all the studies including both HCC and non-hypervascular tumors used other MRI sequences as the reference standard. Therefore, given the limitation due to small number of eligible studies, further studies including a larger number of studies are needed to clearly evaluate the effect of the reference standard on the diagnostic performance of MA-MRI.
Our study has several limitations. First, substantial study heterogeneity was noted. Although study heterogeneity is inevitable because of the various study designs, patient characteristics, and MRI characteristics, we tried to explain the causes of study heterogeneity through robust subgroup analyses and detected several associated factors with plausible explanations. Second, significant publication bias was found in studies reporting MA-MRI. Studies with significant results are more likely to be published than those without significant results, which might have led to an upward bias in the meta-analytic summary estimates [28], and careful interpretation is necessary when the findings are applied to clinical practice. Third, various MRI techniques were used to obtain multiple AP images across the individual studies. Although we performed subgroup analyses according to the use of free breathing imaging acquisition and view sharing technique, we could not investigate the effect of more specific or other multiple AP techniques on study heterogeneity because of a lack of information and difficulties in subgrouping.
In conclusion, our study found that MA-MRI with gadoxetate disodium provided a higher rate of adequate AP imaging than SA-MRI, but the differences for lesion detection rate were not statistically significant. On the basis of the published literature, we consider MA-MRI to be more clinically useful than SA-MRI, although at this stage it might be inappropriate to fully adopt multiple AP acquisitions in clinical practice because of substantial study heterogeneity and publication bias. Therefore, further study is necessary to establish more solid evidence supporting the use of MA-MRI.
Abbreviations
- AP :
-
Arterial-phase
- APHE :
-
Arterial-phase hyperenhancement
- HCC :
-
Hepatocellular carcinoma
- MA-MRI :
-
Multiple arterial-phase magnetic resonance imaging
- PRISMA :
-
Preferred Reporting Items for Systematic Reviews and Meta-Analyses
- QUADAS-2 :
-
Quality Assessment of Diagnostic Accuracy Studies-2
- SA-MRI :
-
Single arterial-phase magnetic resonance imaging
References
Fowler KJ, Brown JJ, Narra VR (2011) Magnetic resonance imaging of focal liver lesions: approach to imaging diagnosis. Hepatology 54:2227–2237
Huh J, Kim SY, Yeh BM et al (2015) Troubleshooting arterial-phase MR images of gadoxetate disodium-enhanced liver. Korean J Radiol 16:1207–1215
Chernyak V, Fowler KJ, Kamaya A et al (2018) Liver Imaging Reporting and Data System (LI-RADS) Version 2018: imaging of hepatocellular carcinoma in at-risk patients. Radiology 289:816–830
Lencioni R, Llovet JM (2010) Modified RECIST (mRECIST) assessment for hepatocellular carcinoma. Semin Liver Dis 30:52–60
European Association for the Study of the Liver (2018) EASL Clinical Practice Guidelines: management of hepatocellular carcinoma. J Hepatol 69:182–236
Tirkes T, Mehta P, Aisen AM, Lall C, Akisik F (2015) Comparison of dynamic phase enhancement of hepatocellular carcinoma using gadoxetate disodium vs gadobenate dimeglumine. J Comput Assist Tomogr 39:479–482
Chandarana H, Block TK, Ream J et al (2015) Estimating liver perfusion from free-breathing continuously acquired dynamic Gd-EOB-DTPA enhanced acquisition with compressed sensing reconstruction. Invest Radiol 50:88
Pietryga JA, Burke LM, Marin D, Jaffe TA, Bashir MR (2014) Respiratory motion artifact affecting hepatic arterial phase imaging with gadoxetate disodium: examination recovery with a multiple arterial phase acquisition. Radiology 271:426–434
Yoon JH, Nickel MD, Peeters JM, Lee JM (2019) Rapid imaging: recent advances in abdominal MRI for reducing acquisition time and its clinical applications. Korean J Radiol 20:1597–1615
Gruber L, Rainer V, Plaikner M, Kremser C, Jaschke W, Henninger B (2018) CAIPIRINHA-Dixon-TWIST (CDT)-VIBE MR imaging of the liver at 3.0T with gadoxetate disodium: a solution for transient arterial-phase respiratory motion-related artifacts? Eur Radiol 28:2013–2021
Hong SB, Lee NK, Kim S et al (2019) Modified CAIPIRINHA-VIBE without view-sharing on gadoxetic acid-enhanced multi-arterial phase MR imaging for diagnosing hepatocellular carcinoma: comparison with the CAIPIRINHA-Dixon-TWIST-VIBE. Eur Radiol 29:3574–3583
Hope TA, Saranathan M, Petkovska I, Hargreaves BA, Herfkens RJ, Vasanawala SS (2013) Improvement of gadoxetate arterial phase capture with a high spatio-temporal resolution multiphase three-dimensional SPGR-Dixon sequence. J Magn Reson Imaging 38:938–945
Ichikawa S, Motosugi U, Sato K, Shimizu T, Wakayama T, Onishi H (2020) Transient respiratory-motion artifact and scan timing during the arterial phase of gadoxetate disodium-enhanced MR imaging: the benefit of shortened acquisition and multiple arterial phase acquisition. Magn Reson Med Sci 20:280–289
Kazmierczak PM, Theisen D, Thierfelder KM et al (2015) Improved detection of hypervascular liver lesions with CAIPIRINHA-Dixon-TWIST-volume-interpolated breath-hold examination. Invest Radiol 50:153–160
Min JH, Kim YK, Kang TW et al (2018) Artifacts during the arterial phase of gadoxetate disodium-enhanced MRI: Multiple arterial phases using view-sharing from two different vendors versus single arterial phase imaging. Eur Radiol 28:3335–3346
Park YS, Lee CH, Kim JW, Lee YS, Paek M, Kim KA (2017) Application of high-speed T1 sequences for high-quality hepatic arterial phase magnetic resonance imaging: intraindividual comparison of single and multiple arterial phases. Invest Radiol 52:605–611
Wei Y, Deng L, Yuan Y et al (2019) Gadoxetate acid disodium-enhanced MRI: multiple arterial phases using differential sub-sampling with cartesian ordering (DISCO) may achieve more optimal late arterial phases than the single arterial phase imaging. Magn Reson Imaging 61:116–123
Yoon JH, Lee JM, Yu MH, Kim EJ, Han JK (2016) Triple arterial phase MR imaging with gadoxetic acid using a combination of contrast enhanced time robust angiography, keyhole, and viewsharing techniques and two-dimensional parallel imaging in comparison with conventional single arterial phase. Korean J Radiol 17:522–532
Yoon JH, Yu MH, Chang W et al (2017) Clinical feasibility of free-breathing dynamic T1-weighted imaging with gadoxetic acid-enhanced liver magnetic resonance imaging using a combination of variable density sampling and compressed sensing. Invest Radiol 52:596–604
Yoon JK, Kim MJ, Lee S (2019) Compressed sensing and parallel imaging for double hepatic arterial phase acquisition in gadoxetate-enhanced dynamic liver magnetic resonance imaging. Invest Radiol 54:374–382
Sofue K, Marin D, Jaffe TA, Nelson RC, Bashir MR (2016) Can combining triple-arterial phase acquisition with fluoroscopic triggering provide both optimal early and late hepatic arterial phase images during gadoxetic acid-enhanced MRI? J Magn Reson Imaging 43:1073–1081
Liberati A, Altman DG, Tetzlaff J et al (2009) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. Ann Intern Med 151:W65–W94
Kim KW, Lee J, Choi SH, Huh J, Park SH (2015) Systematic review and meta-analysis of studies evaluating diagnostic test accuracy: a practical review for clinical researchers-part i. General guidance and tips. Korean J Radiol 16:1175–1187
Higgins JP, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327:557–560
Egger M, Davey Smith G, Schneider M, Minder C (1997) Bias in meta-analysis detected by a simple, graphical test. BMJ 315:629–634
Leeflang MM, Deeks JJ, Gatsonis C, Bossuyt PM (2008) Systematic reviews of diagnostic test accuracy. Ann Intern Med 149:889–897
Kim KW, Lee J, Choi SH, Huh J, Park SH (2015) Systematic review and meta-analysis of studies evaluating diagnostic test accuracy: a practical review for clinical researchers-part I. General guidance and tips. Korean J Radiol 16:1175–1187
Borenstein M, Hedges LV, Higgins JP, Rothstein HR (2021) Introduction to meta-analysis. John Wiley & Sons
Funding
This work was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HI17C-1862-020018) and the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (grant number: NRF-2019R1G1A1099743).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
Institutional Review Board approval was not required because of the study nature of meta-analysis.
Informed consent
Informed consent was not required because of the study nature of meta-analysis.
Conflict of interest
Sang Hyun Choi receives research funding from Bayer Healthcare outside the submitted work. The other authors have no conflicts of interest to declare.
Guarantor
The scientific guarantor of this publication is Sang Hyun Choi.
Statistics and biometry
Sang Hyun Choi has significant statistical expertise
Methodology
• Systematic Review
• Meta-analysis
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 161 kb)
Rights and permissions
About this article
Cite this article
Hong, S., Choi, S.H., Hong, S.B. et al. Clinical usefulness of multiple arterial-phase images in gadoxetate disodium-enhanced magnetic resonance imaging: a systematic review and meta-analysis. Eur Radiol 32, 5413–5423 (2022). https://doi.org/10.1007/s00330-022-08620-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-022-08620-x