Abstract
Objective
To investigate the safety and efficacy of transcatheter arterial embolisation (TAE) in the management of lower gastrointestinal bleeding (LGIB) and to identify predictors of clinical outcomes.
Methods
Between December 2005 and April 2017, 274 patients underwent diagnostic angiography for signs and symptoms of LGIB; 134 patients with positive angiographic findings were retrospectively analysed. The technical success of TAE and clinical outcomes, including recurrent bleeding, major complications, and in-hospital mortality were evaluated. The associations of various clinical and technical factors with clinical outcomes were analysed. Predictors for clinical outcomes were evaluated using univariate and multivariate logistic regression analyses.
Results
A total of 134 patients (mean age, 59.7 years; range, 14–82 years) underwent TAE for LGIB. The bleeding foci were in the small bowel in 74 patients (55.2%), colon in 35 (26.1%), and rectum in 25 (18.7%). Technical success was achieved in 127 patients (94.8%). The clinical success rate was 63% (80/127). The rates of recurrent bleeding, major complications, and in-hospital mortality were 27.9% (31/111), 18.5% (23/124), and 23.6% (33/127), respectively. Superselective embolisation and the use of N-butyl cyanoacrylate (NBCA) were significant prognostic factors associated with reduced recurrent bleeding (OR, 0.258; p = 0.004 for superselective embolisation, OR, 0.313; p = 0.01 for NBCA) and fewer major complications (OR, 0.087; p ˂ 0.001 for superselective embolisation, OR, 0.272; p = 0.007 for NBCA).
Conclusions
TAE is an effective treatment modality for LGIB. Superselective embolisation is essential to reduce recurrent bleeding and avoid major complications. NBCA appears to be a preferred embolic agent.
Key Points
• Transcatheter arterial Embolisation (TAE) is a safe and effective treatment for lower gastrointestinal tract haemorrhage.
• Superselective embolisation is essential to improve outcomes.
• N-butyl cyanoacrylate (NBCA) appears to be a preferred embolic agent with better clinical outcomes.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Acute lower gastrointestinal bleeding (LGIB) refers to acute bleeding from the gastrointestinal tract distal to the ligament of Treitz. LGIB accounts for 20% of all cases of acute gastrointestinal bleeding (GIB), of which 15% require invasive treatment [1, 2]. Emergent endoscopy is the mainstay for diagnosis and subsequent haemostasis in cases of acute LGIB [3]. However, endoscopic treatment has limitations in patients with haemodynamic instability, poor bowel preparation, small bowel bleeding, or whose bleeding focus is not localised because of massive haemorrhage [4, 5]. In cases of endoscopic failure, therapeutic options include surgery and transcatheter treatment. Emergent surgery has mortality rates as high as 15–30% [6]. Moreover, when the bleeding focus is not localised, the surgery-related mortality rate increases up to 50% [4].
Transcatheter arterial embolisation (TAE) has been used for the treatment of LGIB for decades. In the past, vasopressin infusion or non-superselective embolisation using a large-profile catheter was performed, which led to an unacceptably high rate of recurrent bleeding and bowel infarction [4, 6]. Recently, the development of microcatheters with a small caliber and various embolic materials has enabled superselective embolisation; thus, TAE is now being used as a first-line treatment for LGIB [4, 7]. However, one of the remaining major complications is bowel infarction because the bowel has a relatively smaller vascular network than the upper gastrointestinal tract. Only a few studies with small sample sizes have investigated rates of recurrent bleeding, complications, and mortality after TAE for LGIB [8,9,10,11]; therefore, a study with a larger population is needed.
The purpose of this study was to evaluate the efficacy and safety of superselective TAE for LGIB in a large population in tertiary care over a 12-year period and to assess factors associated with clinical outcomes.
Materials and methods
Patient selection
Our Institutional Review Board approved this single-centre retrospective study, waiving the usual informed consent requirement. Figure 1 demonstrates the study flowchart. From December 2005 to April 2017, 919 patients were admitted to our hospital with signs and symptoms of gastrointestinal bleeding. Of these, we excluded patients with upper gastrointestinal bleeding (n=496) and variceal bleeding (n=149) based on clinical symptoms and imaging findings upon computed tomography (CT) and/or endoscopy. Diagnostic angiography was performed for LGIB (n=274). All patients with positive angiographic findings for LGIB (n=134) received TAE and were included in the analysis.
Diagnosis of lower gastrointestinal bleeding
Contrast-enhanced CT scan, endoscopic findings, and/or clinical symptoms were used to localise the LGIB. Colonoscopy was used as a primary diagnostic tool when bleeding from the colorectal region was clinically suspected. Endoscopic haemostasis was attempted when the bleeding focus was located, but if haemostasis failed, patients were referred for embolisation. Some patients underwent CT scan if endoscopy failed to localise bleeding foci; other patients had immediate angiography if their vital signs were unstable despite medical treatment.
Definitions
Patient characteristics, aetiologies of bleeding, haemodynamic status, transfusion and/or inotropic use, laboratory findings, and details of angiographic findings and embolisation procedure were obtained for analysis.
Vascular disease included arteriovenous malformation, angiodysplasia, vasculitis, and Dieulafoy’s lesion. Coagulopathy was defined by (i) prolonged prothrombin time (international normalised ratio ˃ 1.5); (ii) thrombocytopenia (platelet count ˂ 80,000/mL); or (iii) activated partial thromboplastin time (˃ 45 seconds) [12, 13]. Enterocolitis included diverticulitis, inflammatory bowel disease, infectious enterocolitis, and ischaemic enterocolitis. Haemodynamic instability was defined as hypotension (systolic pressure ˂ 100 mmHg) and/or tachycardia (heart rate ˃ 100 beats/min). Superselective embolisation was defined as fewer than three embolised vasa recta. Parent artery refers to the origin of the affected branch including the superior mesenteric artery (SMA), inferior mesenteric artery (IMA), and internal iliac artery (IIA).
Angiography and Embolisation Technique
The right common femoral artery was used for ultrasound-guided access under local anesthesia. A 5-F vascular access sheath (Terumo) was inserted into the right common femoral artery. A 0.035-inch hydrophilic guide wire (Terumo) and a 5-F angiographic catheter (RHR catheter; Cook Medical) were used to image the SMA and IMA. When rectal bleeding was suspected, a bilateral IIA angiographic image was obtained.
Once the active bleeding focus was located, a 2.0-F microcatheter (Progreat Alpha, Terumo) was advanced coaxially for superselective embolisation. For embolisation, various materials including N-butyl cyanoacrylate (NBCA) glue (Histoacryl; B. Braun), absorbable gelfoam sponge particles (Spongostan; Ferrosan), or microcoils (Tornado or Micronestor, Cook) were used according to the following conditions. NBCA was used as a primary embolic agent when a bleeding branch was superselected with a microcatheter. The tip of the microcatheter was advanced as close as possible to the bleeding focus and wedged into the target artery whenever possible. Afterwards, the NBCA mixture with lipiodol (Guerbet) was injected under fluoroscopy monitoring to embolise the selected vessel (Fig. 2). The ratio of the mixture was 1:1 to 1:3, depending on the distance between the bleeding focus and the tip of the microcatheter: we diluted the mixture more for longer distances to prevent premature polymerisation of the NBCA. If the bleeder could not be superselected, absorbable gelfoam sponge or microcoils were used as primary embolic agents (Fig. 3). After embolisation, completion angiography was performed to check the extent of embolisation and ensure complete occlusion of the bleeding arteries.
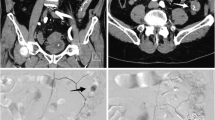
A 71-year old woman taking warfarin after coronary artery stent insertion presented with haematochezia. (a) Contrast-enhanced CT scan showed the active bleeding focus at the pelvic small bowel (arrow). (b) Superior mesenteric artery angiography demonstrated extravasation of contrast medium into the lumen of the ileum (arrow). (c) The feeding artery was superselected with microcatheter (arrowhead) and successfully embolised with a mixture of NBCA and lipiodol at a 1:2 ratio. (d) Angiogram obtained after NBCA TAE showed NBCA and lipiodol casts (open arrow) in one vasa rectum and two branches. The patient stabilised after TAE without recurrent bleeding or complications
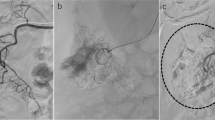
A 65-year old female presented with haematochezia of unknown aetiology. (a) Contrast-enhanced CT scan showed an active bleeding focus at the pelvic small bowel (arrow). (b) Angiography demonstrated extravasation of contrast medium at the ileal branch of the superior mesenteric artery (arrow). (c) A microcatheter was advanced to the target artery, and gelfoam particle embolisation was performed. Angiogram obtained after TAE shows > 3 vasa recta were embolised (arrowhead). (d) Patient had abdominal pain 2 days after TAE, and small bowel infarction was revealed on contrast-enhanced CT scan (open arrow). Patient underwent segmental resection of small bowel and transmural infarction was confirmed on pathologic examination
Technical and clinical outcomes
Technical success was defined as the complete cessation of blood flow from the bleeding artery. Technical failure was defined as follows: (i) spontaneous cessation of bleeding as a result of arterial spasm during guidewire or catheter negotiation; (ii) inability to perform embolisation because of concerns about non-target embolisation (failure of superselection); (iii) inability to reach the target vessel due to stenosis or occlusion at the proximal portion of the vessel being treated. Clinical success was defined as the absence of the following within 30 days of TAE: recurrent bleeding, in-hospital mortality, or a major complication as a result of surgical resection. Recurrent bleeding was defined as the presence of bleeding signs that required immediate treatment within 30 days of embolisation. Complications were classified as either major or minor according to the guidelines of the Society of Interventional Radiology Standards of Practice Committee [14]. In-hospital mortality was defined as death from any cause during the admission period associated with the TAE procedure.
Clinical signs and symptoms, including haemodynamic instability, melena/haematochezia, and laboratory findings, were investigated during the admission period for evaluation of recurrent bleeding. Additional treatments for patients with recurrent bleeding included repeated TAE, endoscopic haemostasis, surgery, or conservative management. In patients who developed abdominal pain, distension, and/or tenderness after embolisation, a contrast-enhanced CT scan or colonoscopy was performed to assess post-procedural complications, such as bowel ischaemia or infarction.
Statistical analysis
All statistical analyses were performed using SPSS version 23.0 for Windows (SPSS Inc.). Predictors for recurrent bleeding, major complications, and in-hospital mortality were evaluated using univariate and multivariate logistic regression analyses. Variables with a p-value lower than 0.1 in the univariate analysis were selected for multivariate analysis. A two-sided p-value ˂ 0.05 was considered statistically significant. The odds ratios (ORs) and 95% confidence intervals (CIs) were calculated.
Results
Patient characteristics
During the period from December 2005 to April 2017, 134 patients (mean age, 59.7; range, 14–82) underwent TAE for LGIB. Table 1 shows the patients’ baseline characteristics. Before TAE, 69 patients (51.5%) had positive findings for active bleeding on contrast-enhanced CT scans, and 26 (19.4%) were diagnosed using endoscopy. The remaining 39 patients (29.1%) underwent immediate angiography. Bleeding foci were located in the small bowel in 74 patients (55.2%), colon in 35 (26.1%), and rectum in 25 (18.7%). Enterocolitis was the most common cause of colonic (45.7%) and rectal bleeding (48%). However, the most common cause of small bowel bleeding was unknown (31.1%). The incidence of coagulopathy was relatively higher in small bowel bleeding (21.6%) than in colorectal bleeding (11.7%), but the difference was not statistically significant (p = 0.129).
Technical and clinical outcomes
Procedure details are summarised in Table 2. Technical success was achieved in 127 patients (94.8%). Of the seven technical failures (5.2%), superselection was not feasible in three patients, who subsequently underwent surgery. In two patients, embolisation was not attempted because the bleeding foci disappeared as a result of vasospasm during the selection of the feeding artery. Angiography of the remaining two patients with rectal bleeding revealed bilateral IIA occlusion with fine collateral supply from an external iliac artery, resulting in technical failure; these patients were treated with repeated endoscopic haemostasis (n=1) or surgery (n=1).
Clinical outcomes are summarised in Table 3. Clinical success was achieved in 80 out of 127 patients (63%). Recurrent bleeding occurred in 31 patients (27.9%) within 30 days among 111 patients (16 of the 127 patients were excluded because their follow-up periods were less than 30 days). These patients were treated with repeated TAE (n=6), endoscopic haemostasis (n=10), surgery (n=7), or conservative medical treatment (n=8). Major complications after TAE occurred in 23 of 124 patients (18.5%): three of the 127 patients were excluded because of early deaths within 30 days not associated with complications. The severity of major complications was classified based on contrast-enhanced CT findings [15]. Ten patients had transient bowel ischaemia managed with conservative treatment, and these patients were included in the clinical success category. By contrast, the remaining 13 patients with transmural bowel infarction underwent surgery (n=10) or died (n=3) within 30 days. The overall in-hospital mortality rate was 26% (33 of 127 patients). Thirteen of 33 (39.4%) deaths were related to recurrent bleeding, and three patients (9.1%) died of complications after TAE. The remaining 17 patients (51.5%) died due to underlying conditions, including malignancy (n=6), cardiovascular disease (n=6), disseminated intravascular coagulopathy (n=3), or pneumonia (n=2), despite successful TAE.
Predictors of outcomes
Tables 4 and 5 show associations of selected clinical and technical parameters with clinical outcomes. Univariate analysis showed that superselective embolisation was related to decreased recurrent bleeding (OR, 0.258; 95% CI, 0.103-0.642; p = 0.004), major complications (OR, 0.087; 95% CI, 0.029-0.258; p ˂ 0.001), and in-hospital mortality (OR, 0.319; 95% CI, 0.140-0.724; p = 0.006). The use of NBCA was associated with lower rates of recurrent bleeding (OR, 0.313; 95% CI, 0.129-0.760; p = 0.01) and major complications (OR, 0.272; 95% CI, 0.106-0.695; p = 0.007). In terms of the parent artery, bleeding foci originating from the SMA were associated with a lower recurrent bleeding rate (OR, 0.369; 95% CI, 0.145-0.939; p = 0.036).
In-hospital mortality was associated with recurrent bleeding (OR, 3.229; 95% CI, 1.331-7.833 ; p = 0.01), GI surgery complication (OR, 2.670; 95% CI, 1.006-7.087; p = 0.045), malignancy (OR, 10.222; 95% CI, 1.950-53.590; p = 0.006), haemodynamic instability (OR, 10.971; 95% CI, 1.421-84.684; p = 0.022), blood transfusion (OR, 1.402; 95% CI, 1.249-1.575; p = 0.033), inotropic use (OR, 3.138; 95% CI, 1.381-7.132; p = 0.006), or a haemoglobin level ˂ 7 g/dL before TAE (OR, 3.163; 95% CI, 1.329-7.526; p = 0.009). There was no significant association between mean ages and major complication rate and in-hospital mortality rate. Also, there was no significant difference in clinical outcomes between patients with coagulopathy and those with normal coagulation status. Results from multivariate analysis showed that superselective embolisation had negative associations with recurrent bleeding (OR, 0.295; 95% CI, 0.111-0.779; p = 0.014) and major complications (OR, 0.094; 95% CI, 0.027-0.320; p ˂ .001). The presence of recurrent bleeding (OR, 3.330; 95% CI, 1.161-9.551; p = 0.025), GI surgery complication (OR, 3.273; 95% CI, 1.078-9.940; p = 0.036), malignancy (OR, 14.165; 95% CI, 1.962-102.289; p = 0.009), and haemodynamic instability (OR, 12.846; 95% CI, 1.478-111.663; p = 0.021) were independently related to in-hospital mortality.
Discussion
The present study of 134 patients is one of the largest studies describing the technical and clinical success rates for TAE for LGIB as well as the predictors associated with clinical outcomes. The rate of technical success was 94.8% (127/134), and the rates of recurrent bleeding, major complications, and in-hospital mortality were 27.9% (31/111), 18.5% (23/124), and 23.6% (33/127), respectively.
Recently, the development of microcatheters and guidewires has allowed superselection of vessels during the TAE procedure. Previous studies reported high technical success rates of TAE for LGIB (89.5–100%) [3, 7, 8, 11, 16,17,18,19,20,21,22,23,24]. In our study, the technical success rate was 94.8%. One of the major causes of technical failure was vasospasm during catheterisation and subsequent disappearance of the bleeding focus of a target vessel. Vasospasm normally resolves in a few minutes with catheter tip adjustment, and then the bleeding focus can be visualised [5]. In the present study, vasospasm occurred in two patients and subsided within a few minutes. However, bleeding stopped spontaneously, thus TAE was not performed, which was considered a technical failure.
The overall recurrent bleeding rate in our study was 27.9%, which is comparable to those reported in previous studies (8.8-26%) [3, 7, 8, 11, 16,17,18,19,20,21,22,23,24]. In the present study, superselective embolisation was independently correlated with recurrent bleeding (OR, 0.295; p = 0.014). One possible explanation is that non-superselective embolisation may lead to proximal vessel embolisation, if particulate embolic materials (gelfoam sponge or PVA) or microcoils are too large or if NBCA is premature polymerised. In such instances, recurrent bleeding from collateral vessels can occur. Therefore, superselective embolisation is important not only to reduce complication rates but also to reduce recurrent bleeding.
In the present study, although the use of NBCA was associated with lower rates of recurrent bleeding, there was no independent relationship (OR, 0.739; p = 0.614). Nevertheless, NBCA has several advantages as an embolic agent for LGIB. First, NBCA can be delivered through a smaller microcatheter than that used for particulate agents or microcoils, which makes it easier to perform superselective embolisation of a small-branch bleeding focus [25]. Second, it can be delivered distally of the tip of the microcatheter and occlude a bleeding focus that is inaccessible with the microcatheter because of small diameter, tortuosity, stenosis, or spasm of the target artery [24, 25]. Third, although gelfoam sponge or microcoils are known as safe embolic materials, they have a higher recanalisation rate than NBCA [28]. The recurrent bleeding rate of TAE with NBCA for LGIB was reported to range from 4.7 to 21.4% [16, 24, 26, 27]. In the present study, the recurrent bleeding rate of the NBCA group was 17.5%.
Coagulopathy is associated with recurrent bleeding after TAE for acute gastrointestinal bleeding [24, 29,30,31,32,33]. When gelfoam sponge, polyvinyl alcohol (PVA) particles, or microcoils were used in patients with coagulopathy, the successful bleeding control rates were between 43% and 46% [33, 34]. Yonemitsu et al reported that the recurrent bleeding rate was significantly higher in the gelfoam sponge group (23.1%) than in the NBCA group (0%) [31]. This is because gelfoam sponge, PVA particles, and microcoils rely on patients’ coagulation status for occlusion of the vessel. By contrast, NBCA polymerises immediately upon contacting blood and occludes the vessel, regardless of the patient’s coagulation status [16, 24]. In our study, the recurrent bleeding rate in patients with coagulopathy was 33.3% (6 of 18 patients), and there was no statistical significance when compared with patients with normal coagulation status (OR, 1.614; p = 0.390). Possible explanation is that NBCA is effective for treatment of LGIB in patients with coagulopathy. In terms of embolic agents, of the six patients with recurrent bleeding, NBCA was used in one patient, and other embolic agents, including gelfoam (n=4) and microcoils (n=1), were used in five patients. By contrast, of 12 patients without recurrent bleeding, NBCA was used in 11 patients, and gelfoam sponge was used in one patient. As a result, the recurrent bleeding rate was significantly lower in the NBCA group than in the other embolic agent groups (OR, 55.000; p = 0.001) in the presence of coagulopathy.
In the present study, LGIB originating from the SMA branches was associated with significantly better outcomes than LGIB originating from the IMA or IIA branches (OR, 0.033; p = 0.036). This finding can be explained by greater collateral circulation in the IMA or IIA region compared with the SMA region. For example, the rectum is fed by rich collateral blood flow from the IMA and IIA or middle sacral artery, resulting in clinical failures.
The overall complication rate in the present study was 18.3%, which is slightly higher than that reported in previous studies (3.3-15.0%) [16, 24, 26, 27, 35]. The complication rate may be overestimated since major complications were classified as patients who required treatment with hospitalisation after TAE regardless of whether they were treated with conservatively management or not. Indeed, 10 patients were included in the major complication category, despite complete recovery after conservative management for transient bowel ischaemia and absence of recurrent bleeding.
Superselective embolisation for GIB is one of the important factors related to complications. In particular, LGIB requires superselective embolisation because collateral circulation is less plentiful than in the upper gastrointestinal tract; thus, ischaemic complications are more likely to occur [26]. In previous studies, superselective embolisation involving three or fewer feeding arteries for LGIB was reported to be relatively tolerable with silent or self-limiting complications [9,10,11]. This is in line with our findings that the major complication rate was significantly lower in the superselective embolisation group (OR, 0.087; p < 0.001).
The major complication rate of TAE with NBCA was 11.3%, which was significantly lower than in the other embolic agent groups (OR, 0.272; p = 0.007). Possible explanations comprise the following: (i) the viscosity and speed of polymerisation of NBCA prevented the agent from traveling too distally into the capillary bed, and the circulation to the post-embolic tissue can be preserved by collateral channels in the intramural microcirculation [26]. (ii) NBCA prevented non-target embolisation because it was easier to control under fluoroscopy monitoring than gelfoam sponge or microcoils. (iii) As mentioned above, TAE with NBCA was performed superselectively in a majority of cases (67 of 80 patients).
Overall in-hospital mortality rate after TAE was 24.2% in our study and was similar to that reported in previous studies (9–27%) [3, 5, 18, 24, 32]. Many factors are known to affect mortality after TAE. One of the most important factors is recurrent bleeding [3, 5, 36, 37]. Dempsey et al reported that the mortality rate decreased by 50% when there was no evidence of recurrent bleeding [37]. In our study, the mortality rate was 41.9% in patients with recurrent bleeding (13 of 31 patients), which was significantly higher than the mortality rate in patients without recurrent bleeding (OR, 3.229; p = 0.01). In addition, superselective embolisation was a significant prognostic factor of not only in-hospital mortality (OR, 0.319; p = 0.006) but also recurrent bleeding.
Haemodynamic instability, low haemoglobin levels, blood transfusions, and inotropic use are associated with recurrent bleeding, which results in increased in-hospital mortality [5, 36, 38, 39]. Defreyne et al reported that survival was inversely correlated with pre-embolisation clinical parameters, such as shock therapy, blood transfusion, and decrease in haemoglobin concentration [5]. Our results also indicated that the presence of haemodynamic instability was a significant prognostic factor for in-hospital mortality (OR, 10.971; p = 0.022). Other prognostic factors were recent surgery (OR, 2.670; p = 0.045) and underlying malignancy (OR, 10.222; p = 0.006), which support other studies [3, 5, 24, 36].
The current study has a few limitations. First, this study has a retrospective design. Second, the embolic agent was not randomly chosen. Further study is warranted to compare outcomes based on randomised selection of embolic materials. Third, colonoscopy or CT scan was not performed in all patients after TAE, and some ischaemic complications without clinical symptoms may have been missed. However, complications without clinical symptoms are classified as a minor complication and are considered to have no effect on clinical outcomes of the patients.
In conclusion, TAE is a safe and effective treatment modality for LGIB. In addition, NBCA potentially could be a preferred embolic material. Moreover, whenever possible, superselective embolisation should be used to avoid major complications and reduce recurrent bleeding.
Abbreviations
- CT:
-
Computed tomography
- GIB:
-
Gastrointestinal bleeding
- IIA:
-
Internal iliac artery
- IMA:
-
Inferior mesenteric artery
- LGIB:
-
Lower gastrointestinal bleeding
- NBCA:
-
N-butyl cyanoacrylate
- PVA:
-
Polyvinyl alcohol
- SMA:
-
Superior mesenteric artery
- TAE:
-
Transcatheter arterial embolisation
References
Longstreth GF (1997) Epidemiology and outcome of patients hospitalized with acute lower gastrointestinal hemorrhage: a population-based study. Am J Gastroenterol 92:419–424
Abbas SM, Bissett IP, Holden A, Woodfield JC, Parry BR, Duncan D (2005) Clinical variables associated with positive angiographic localization of lower gastrointestinal bleeding. ANZ J Surg 75:953–957
Maleux G, Roeflaer F, Heye S et al (2009) Long-term outcome of transcatheter embolotherapy for acute lower gastrointestinal hemorrhage. Am J Gastroenterol 104:2042–2046
Barnert J, Messmann H (2009) Diagnosis and management of lower gastrointestinal bleeding. Nat Rev Gastroenterol Hepatol 6:637–646
Defreyne L, Vanlangenhove P, De Vos M et al (2001) Embolisation as a first approach with endoscopically unmanageable acute nonvariceal gastrointestinal hemorrhage. Radiology 218:739–748
Kuo WT, Lee DE, Saad WE, Patel N, Sahler LG, Waldman DL (2003) Superselective microcoil embolisation for the treatment of lower gastrointestinal hemorrhage. J Vasc Interv Radiol 14:1503–1509
Urbano J, Manuel Cabrera J, Franco A, Alonso-Burgos A (2014) Selective arterial embolisation with ethylene-vinyl alcohol copolymer for control of massive lower gastrointestinal bleeding: feasibility and initial experience. J Vasc Interv Radiol 25:839–846
Kickuth R, Rattunde H, Gschossmann J, Inderbitzin D, Ludwig K, Triller J (2008) Acute lower gastrointestinal hemorrhage: minimally invasive management with microcatheter embolisation. J Vasc Interv Radiol 19:1289–1296.e1282
Jae HJ, Chung JW, Kim HC et al (2008) Experimental study on acute ischaemic small bowel changes induced by superselective embolisation of superior mesenteric artery branches with N-butyl cyanoacrylate. J Vasc Interv Radiol 19:755–763
Ikoma A, Kawai N, Sato M et al (2010) Ischaemic effects of transcatheter arterial embolisation with N-butyl cyanoacrylate-lipiodol on the colon in a Swine model. Cardiovasc Intervent Radiol 33:1009–1015
Kodani M, Yata S, Ohuchi Y, Ihaya T, Kaminou T, Ogawa T (2016) Safety and Risk of Superselective Transcatheter Arterial Embolisation for Acute Lower Gastrointestinal Hemorrhage with N-Butyl Cyanoacrylate: Angiographic and Colonoscopic Evaluation. J Vasc Interv Radiol 27:824–830
O'Connor SD, Taylor AJ, Williams EC, Winter TC (2009) Coagulation concepts update. AJR Am J Roentgenol 193:1656–1664
Patel IJ, Davidson JC, Nikolic B et al (2012) Consensus guidelines for periprocedural management of coagulation status and hemostasis risk in percutaneous image-guided interventions. J Vasc Interv Radiol 23:727–736
Angle JF, Siddiqi NH, Wallace MJ et al (2010) Quality improvement guidelines for percutaneous transcatheter embolisation: Society of Interventional Radiology Standards of Practice Committee. J Vasc Interv Radiol 21:1479–1486
Wiesner W, Khurana B, Ji H, Ros PR (2003) CT of acute bowel ischaemia. Radiology 226:635–650
Huang CC, Lee CW, Hsiao JK et al (2011) N-butyl cyanoacrylate embolisation as the primary treatment of acute hemodynamically unstable lower gastrointestinal hemorrhage. J Vasc Interv Radiol 22:1594–1599
Koh DC, Luchtefeld MA, Kim DG et al (2009) Efficacy of transarterial embolisation as definitive treatment in lower gastrointestinal bleeding. Colorectal Dis 11:53–59
Tan KK, Wong D, Sim R (2008) Superselective embolisation for lower gastrointestinal hemorrhage: an institutional review over 7 years. World J Surg 32:2707–2715
Kwak HS, Han YM, Lee ST (2009) The clinical outcomes of transcatheter microcoil embolisation in patients with active lower gastrointestinal bleeding in the small bowel. Korean J Radiol 10:391–397
Gillespie CJ, Sutherland AD, Mossop PJ, Woods RJ, Keck JO, Heriot AG (2010) Mesenteric embolisation for lower gastrointestinal bleeding. Dis Colon Rectum 53:1258–1264
Lipof T, Sardella WV, Bartus CM, Johnson KH, Vignati PV, Cohen JL (2008) The efficacy and durability of super-selective embolisation in the treatment of lower gastrointestinal bleeding. Dis Colon Rectum 51:301–305
Sheth R, Someshwar V, Warawdekar G (2006) Treatment of acute lower gastrointestinal hemorrhage by superselective transcatheter embolisation. Indian J Gastroenterol 25:290–294
d’Othee BJ, Surapaneni P, Rabkin D, Nasser I, Clouse M (2006) Microcoil embolisation for acute lower gastrointestinal bleeding. Cardiovasc Intervent Radiol 29:49–58
Hur S, Jae HJ, Lee M, Kim HC, Chung JW (2014) Safety and efficacy of transcatheter arterial embolisation for lower gastrointestinal bleeding: a single-centre experience with 112 patients. J Vasc Interv Radiol 25:10–19
Kim PH, Tsauo J, Shin JH, Yun SC (2017) Transcatheter Arterial Embolisation of Gastrointestinal Bleeding with N-Butyl Cyanoacrylate: A Systematic Review and Meta-Analysis of Safety and Efficacy. J Vasc Interv Radiol 28:522–531.e525
Frodsham A, Berkmen T, Ananian C, Fung A (2009) Initial experience using N-butyl cyanoacrylate for embolisation of lower gastrointestinal hemorrhage. J Vasc Interv Radiol 20:1312–1319
Yata S, Ihaya T, Kaminou T et al (2013) Transcatheter arterial embolisation of acute arterial bleeding in the upper and lower gastrointestinal tract with N-butyl-2-cyanoacrylate. J Vasc Interv Radiol 24:422–431
Vaidya S, Tozer KR, Chen J (2008) An overview of embolic agents. Semin Interv Radiol 25:204–215
Loffroy R, Guiu B, D'Athis P et al (2009) Arterial embolotherapy for endoscopically unmanageable acute gastroduodenal hemorrhage: predictors of early rebleeding. Clin Gastroenterol Hepatol 7:515–523
Aina R, Oliva VL, Therasse E et al (2001) Arterial embolotherapy for upper gastrointestinal hemorrhage: outcome assessment. J Vasc Interv Radiol 12:195–200
Yonemitsu T, Kawai N, Sato M et al (2009) Evaluation of transcatheter arterial embolisation with gelatin sponge particles, microcoils, and n-butyl cyanoacrylate for acute arterial bleeding in a coagulopathic condition. J Vasc Interv Radiol 20:1176–1187
Hongsakul K, Pakdeejit S, Tanutit P (2014) Outcome and predictive factors of successful transarterial embolisation for the treatment of acute gastrointestinal hemorrhage. Acta Radiol 55:186–194
Schenker MP, Duszak R Jr, Soulen MC et al (2001) Upper gastrointestinal hemorrhage and transcatheter embolotherapy: clinical and technical factors impacting success and survival. J Vasc Interv Radiol 12:1263–1271
Encarnacion CE, Kadir S, Beam CA, Payne CS (1992) Gastrointestinal bleeding: treatment with gastrointestinal arterial embolisation. Radiology 183:505–508
Koo HJ, Shin JH, Kim HJ et al (2015) Clinical outcome of transcatheter arterial embolisation with N-butyl-2-cyanoacrylate for control of acute gastrointestinal tract bleeding. AJR Am J Roentgenol 204:662–668
Loffroy R, Rao P, Ota S, De Lin M, Kwak BK, Geschwind JF (2010) Embolisation of acute nonvariceal upper gastrointestinal hemorrhage resistant to endoscopic treatment: results and predictors of recurrent bleeding. Cardiovasc Intervent Radiol 33:1088–1100
Dempsey DT, Burke DR, Reilly RS, McLean GK, Rosato EF (1990) Angiography in poor-risk patients with massive nonvariceal upper gastrointestinal bleeding. Am J Surg 159:282–286
Larson G, Schmidt T, Gott J, Bond S, O'Connor CA, Richardson JD (1986) Upper gastrointestinal bleeding: predictors of outcome. Surgery 100:765–773
Choudari CP, Rajgopal C, Elton RA, Palmer KR (1994) Failures of endoscopic therapy for bleeding peptic ulcer: an analysis of risk factors. Am J Gastroenterol 89:1968–1972
Funding
The author state that this work has not received any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Man-Deuk Kim, Severance hospital, Yonsei University.
Conflict of interest
The authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article.
Statistics and biometry
No complex statistical methods were necessary for this paper.
Informed consent
Written informed consent was waived by the Institutional Review Board.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• retrospective
• observational
• performed at one institution
Rights and permissions
About this article
Cite this article
Kwon, J.H., Kim, MD., Han, K. et al. Transcatheter arterial embolisation for acute lower gastrointestinal haemorrhage: a single-centre study. Eur Radiol 29, 57–67 (2019). https://doi.org/10.1007/s00330-018-5587-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-018-5587-8