Abstract
Objectives
To prospectively evaluate image quality (IQ) and radiation dose of dual-source cardiac computed tomography (CCTA) using different imaging protocols.
Methods
CCTA was performed in 150 patients using the retrospective ECG-gated spiral technique (rECG) the prospective ECG-gated technique (pECG), or the prospective ECG-gated technique with systolic imaging and automated tube voltage selection (pECGsys). IQ was rated using a 16-segment coronary artery model. Techniques were compared for overall IQ, IQ of the large and the small coronary artery segments. Effective dose was used for comparison of radiation dose.
Results
Overall IQ and IQ of the large segments showed no differences between the groups. IQ analysis of the small segments showed lowered IQ in pECGsys compared to rECG (p = 0.02), but not to pECG (p = 0.6). Effective dose did not differ significantly between rECG and pECG (p = 0.13), but was significantly lower for pECGsys (p < 0.001 vs. rECG and pECG).
Conclusion
Radiation dose of dual-source CCTA in heart transplant recipients is significantly reduced by using prospective systolic scanning and automated tube voltage selection, while overall IQ and IQ of the large coronary segments are maintained. IQ appears to be lower compared to retrospective techniques with regard to small coronary segments.
Key points
• Cardiac computed tomography angiography is useful for cardiac allograft vasculopathy assessment.
• Despite elevated heart rate, dose reduction in cardiac computed tomography is possible.
• Prospective systolic gating and automated tube voltage selection enable 50 % dose reduction.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Despite major progress in immunosuppressive therapy, cardiac allograft vasculopathy (CAV) remains the main cause of morbidity and mortality in the late phase after cardiac transplant [1]. CAV is characterized by concentric myointimal thickening of intramural and epicardial arteries, which leads to vascular stenosis and occlusion, resulting in graft failure and graft loss [2]. As a result of the denervation of the transplanted heart, CAV is initially a clinically silent disease that presents in a later stage of disease with heart failure, arrhythmias and sudden cardiac death. Therefore, early detection of CAV is of paramount importance for patients, as changes in drug regimen may avoid disease progression [3, 4].
Screening of CAV is traditionally performed by invasive coronary angiography (ICA) repeated on a regular basis [5]. However, ICA does not allow evaluation of the vessel wall; therefore, the early stages of CAV may be missed if no additional intravascular ultrasound is performed [6].
During recent years, CCTA has emerged as a promising tool for the assessment of CAV [7, 8]. Contrary to ICA, CCTA is non-invasive and able to assess both luminal and vessel wall changes [9]. However, there are concerns about the radiation dose associated with CCTA in heart transplant recipients owing to the need for repetitive examination, which results in a high cumulative radiation dose [10]. With the introduction of the second generation of dual-source CT technology, enormous reductions of radiation dose have been realized [11]. However, dose reduction in CCTA for the evaluation of CAV is challenging because, as a result of the denervation of the graft, patients usually present with elevated heart rates that are not sensitive to beta-blockers. Therefore the aim of this study was to prospectively evaluate image quality (IQ) and radiation dose using three different CT imaging protocols for dual-source CCTA in heart transplant recipients.
Methods
The study was approved by the local institutional review board. Power calculation revealed a sample size of 50 per group in order to detect a mean difference in overall IQ of 1 per patient with a power of 90 %. All patients gave written, informed consent prior to study enrolment. Patients were randomized using a computer-generated list (www.randomizer.org).
Patients
A total of 150 consecutive patients (121 male/29 female) after cardiac transplant were included in this prospective randomized trial. Patients with known allergic reaction to iodinated contrast agents, hyperthyroidism and elevated serum creatinine (>1.5 mg/dl) were considered ineligible.
CT protocols
All examinations were performed using a 128-row, dual-source, multi-slice CT scanner (Somatom Definition Flash, Siemens Medical Systems, Germany), with 2 × 128 × 0.6 mm collimation and a 280 ms gantry rotation. Patients were randomized to one of the following three study protocols:
-
1.
Retrospective gated spiral mode (rECG): The tube voltage was set to 120 kVp for both tubes. Tube current was set to 320 reference mAs. Adaptive tube current modulation was used, which translates into an optimal pulsing window chosen by the scanner depending on the heart rate.
-
2.
Prospective triggered sequential scan (pECG): The tube voltage was set to 120 kVp for both tubes. Tube current was set to 320 reference mAs. The main padding window was set at 30–70 % of the RR ECG cycle.
-
3.
Prospective triggered sequential scan with a narrow systolic window (pECGsys): The main pulsing window was set to 35–45 % of the RR cycle. Automated tube voltage settings, calculated automatically and based on the acquired topogram (CAREkv), were switched on. Reference mAs values were chosen by the scanner, depending on the kVp settings.
For the completion of the prospective triggered sequential scans 3–4 slabs (each 64 × 0.6 mm) were required.
Contrast agent injection protocols
Contrast agent injections were performed using a programmable power injector (CT Expres, Swiss Medical Care, Switzerland). A non-ionic iodinated contrast medium was used at a concentration of 400 mg iodine/mL (Iomeron 400, Bracco, Austria). The flow was set to 5 mL/s and the amount of the contrast medium depended on the body weight of the patients (70–110 mL). A bolus tracking technique was applied, with the reference image at the level of the ascending aorta (threshold 150 HU; delay 5 s).
Image reconstruction
Images were reconstructed at 0.6 mm slice thicknesses on a Syngo® workstation (Siemens Medical System, Germany) using filtered back-projection and a medium soft kernel (B26F). An increment of 10 % RR was used for image reconstruction. Curved planar reformats for all three main epicardial vessels, as well as thin maximum intensity projections (MIP), were additionally reconstructed. MIP images were reconstructed in the systolic and diastolic phase in rECG and pECG, whereas only the systolic phase was provided in pECGsys.
Image quality rating
For IQ analysis, the coronary artery tree was divided into 16 segments. Fifteen segments were classified according to the American Heart Association (AHA) model, and the intermediate branch artery was assigned to segment 16 [12]. For further analysis, coronary artery segments were divided into large (AHA segments 1–3, 5–7, 11, 13) and small segments (4, 8–10, 12, 14–16). Small vessels with a diameter below 1.5 mm were not excluded from the analysis. Two experienced readers performed the IQ reading separately, using a four-point scale:
-
Grade 1 (excellent): excellent IQ enabling sufficient differentiation of even small structures; very good vessel wall visualization.
-
Grade 2 (good quality): good IQ providing all details necessary for an adequate diagnosis; good vessel wall visualization.
-
Grade 3 (poor-diagnostic): only patency or occlusion of the vascular segment can be assessed; details of vessel wall pathologies cannot be distinguished.
-
Grade 4 (non-diagnostic): vascular segments cannot be assessed; details of vessel wall pathologies are not provided.
Occluded vessel segments and segments that were not visible owing to anatomical variance were graded zero.
For IQ group comparison, the means of the per segment ratings from both readers were calculated. A higher mean per segment score translates into a reduced IQ.
Radiation dose calculation
For the estimation of the radiation dose, the dose length product (DLP) was calculated by the CT scanner by multiplication of the computed tomography dose index (CTDI) by the imaging range. Effective dose (ED) was subsequently calculated by using a standard conversion factor of 0.014 mSv mGy−1 cm−1 [13].
Statistical analysis
Statistical analyses were performed using the Statistical Package for the Social Sciences, version 20.0 (SPSS, Chicago, Illinois). Descriptive statistics were used for nominal data. An intraclass correlation coefficient (ICC) was calculated to test for agreement between the two raters. For each subject, three additional variables were defined to serve as a measure of IQ: means were calculated for large, small and for all segments on the basis of the mean ratings from both raters. Segment ratings that were rated zero (i.e. occluded or not visible) were excluded to avoid any bias in the group comparison. An analysis of variance was conducted to test for differences between the three different subgroups. The alpha level for all tests was set at p = 0.05, and correction for multiple comparisons was performed using least significant difference (LSD).
Results
Patient population
Mean age, mean BMI and mean heart rate showed an equal distribution, without significant differences among the study groups (Table 1).
Group comparison of image quality
In all patients, 339 (14.1 %) of the coronary artery segments were rated zero owing to anatomic variance or occlusion and were excluded from the further analysis (rECG: n = 110 [13.8 %]; pECG: n = 111 [13.9 %]; pECGsys: n = 118 [14.8 %]).
For the calculation of the ICC, all 150 patient ratings were included. ICC revealed excellent agreement between the two raters (0.905, CI [0.868; 0.931]).
Descriptive statistics from IQ reading are provided in Table 2. A non-diagnostic image (grade 4) quality rating was assigned, overall, to 0.3 % (n = 7) of the segments (rECG: 0, n = 0; pECG: 0.7 %, n = 5; pECGsys: 0.3 %, n = 2).
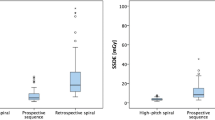
Overall mean IQ per segment rating was 1.45 ± 0.36, IQ rating of the large epicardial segments reached a mean of 1.33 ± 0.35, and both showed no significant differences between the study groups. Analysis of the small segments delivered an overall mean per segment score of 1.62 ± 0.43. Post hoc tests of the small segments resulted in a significant difference between the rECG and the pECGsys group (1.50 ± 0.40 vs. 1.70 ± 0.43, p = 0.02). Mean per segment score of the pECG group was 1.65 ± 0.48, with no statistical significance compared to the other groups (Fig. 1; Table 3).
Radiation dose
Group comparison of the radiation dose values showed significant differences between pECG vs. pECGsys (p < 0.001) and rECG vs. pECGsys (p < 0.001) (Fig. 2; Table 4). In pECGsys, one patient was examined at 80 kV, 22 patients were examined at 100 kV, 25 patients were examined at 120 kV and one patient was examined at 140 kV.
Discussion
The results of our study suggest that the radiation dose of CCTA in heart transplant recipients can be significantly reduced by using an ECG-gated sequential scan technique, with scanning in the systolic phase only, and automated tube voltage selection. Using prospective imaging and placing a narrow padding window into the systolic phase only, we maintained the overall diagnostic IQ in cardiac allograft recipients presenting with elevated heart rates. However, IQ in our study was found to be slightly lower compared to retrospective gating, in particular with regard to the small coronary artery segments; however, the IQ remains at an acceptable level.
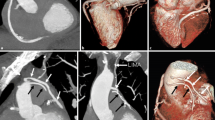
In this study of 150 patients, we could further show that, despite higher mean heart rates, overall IQ and the assessability of coronary segments from a dual-source CCTA exam were excellent, with an excellent inter-rater correlation (Fig. 3).
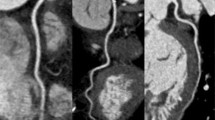
CCTA in patients with cardiac transplant using three different scan protocols. a–c Curved planar reformation of the left anterior descending artery in patients with no evidence of allograft vasculopathy. d, f Curved planar reformation of the right coronary artery in patients with allograft vasculopathy. Similar image quality is evident in the pECGsys group with considerably reduced radiation dose values in c, f. rECG retrospective gating, pECG prospective gating, pECGsys systolic prospective gating + automated tube voltage selection, mSv millisievert
Different imaging techniques may be used for CCTA [11, 14]. The retrospective gating technique with dose-modulation algorithms, used in rECG, is defined by low-pitch scanning with full tube current throughout a predefined window of the cardiac cycle, while the rest of the cardiac cycle is scanned with reduced tube current. In our study, rECG showed the lowest mean per segment ratings, representing the best IQ. However, there were no significant differences with regard to overall IQ and IQ of the main epicardial coronary artery segments compared to the other groups. At the same time, the highest radiation dose values were present in the rECG group.
The prospective ECG-gated sequence technique used in pECG is defined by applying the full tube current only at predefined phases of the cardiac cycle. The magnitude of dose-saving potential depends on how narrow the padding window is set. Commonly, for elevated heart rates, a wider padding window has been suggested to ensure the availability of multiple phases for image reconstruction. Therefore, no significant dose savings when compared to rECG were evident in our study. With regard to IQ, no differences were found when pECG was compared to the other study groups. In conclusion, prospective gating with a wide padding window does not offer any benefits to cardiac allograft recipients when compared to retrospective gating technique.
The prospective ECG-gated sequence technique with scanning in the late systolic phase only has recently been reported as feasible in patients with elevated heart rates and atrial fibrillation, while reducing radiation dose at the same time [15, 16]. Using this method, scanning is only performed in a narrow systolic phase from 35 to 45 % of the cardiac cycle. Bastarrika et al. proved that image reconstructions obtained from the systolic phase in CCTA of heart transplant recipients showed excellent IQ, concluding that this approach seems to be suitable for the evaluation of CAV [17]. In our study, the application of a systolic pulsing window showed no significant differences with regard to overall IQ or for IQ of the large epicardial arteries. In contrast to previous studies, we evaluated IQ of the large and the small coronary artery segments separately, as CAV may also involve small segments at an early stage [17]. Analysis of the small segments revealed a significant difference between rECG and pECGsys, with lower IQ in the pECGsys group. Although we obtained a statistically significant difference, it remains questionable whether this result is also clinically relevant and implies that scanning in the systolic phase only should not be used for CAV assessment. A mean per segment score of 1.7 represents good diagnostic IQ, still suitable for the evaluation of CAV.
In addition to tailoring the CT protocol to the heart rate, further strategies to achieve a dose reduction include automated, attenuation-based tube voltage selection [18]. While using this technique, kVp values of 80, 100, 120 and 140 are chosen by the scanner system, based on the pre-scan topogram [19]. This offers the possibility of significant radiation dose reduction, without the need for manual intervention [20]. In our study, the combined application of automated tube voltage selection and prospective systolic imaging resulted in a significant dose reduction of 54 %, compared to retrospective gating, and 50 %, as compared to prospective gating, using a wide padding window.
Among the diverse imaging modalities used for the follow-up of heart transplant recipients, ICA is considered the gold standard [5]. However, its invasive nature, and the inability to assess the vessel wall, has led to further development of alternative non-invasive techniques [21]. During the last decade, CCTA has emerged as a robust imaging modality for the evaluation of CAV, with sensitivity values ranging from 71 to 100 % and high negative predictive values of 93–100 % when compared to ICA [9]. A recent meta-analysis by Wever-Pinzon et al., including mainly studies using single-source and first-generation dual-source CT systems, strongly suggests CCTA for the screening of CAV, in order to avoid invasive procedures [7]. However, most of the papers included in this meta-analysis did not use any specific CT protocols tailored to imaging CAV at a lowered radiation dose, and some did not even report on radiation dose at all. On the other hand, radiation dose has received more attention during recent years, as there are major concerns about the increase in cumulative radiation dose from medical imaging [10, 22]. Cardiac transplant patients have a 3.5 times greater exposure to radiation and receive a mean cumulative radiation dose of 84 mSv during a 10-year follow-up [23]. Therefore, dose reduction in these patients is of even higher relevance. Using 64-slice scanners, high average dose values from 8 to 18 mSv have been reported in CCTA of heart transplant recipients, and, despite the technical progress, patients with arrhythmia and elevated heart rate still represent a so-called difficult-to-image population in CCTA [24].
Within the last decade, tremendous progress has been made in terms of radiation dose reduction in CCTA. Radiation dose exposure in CCTA has dropped from 15–20 mSv down to 1 mSv [25]. However, most of the studies reporting on low dose in CCTA included non-obese patients with low and regular heart rates, but hardly any patients with heart rate values above 70 bpm [26]. Although not yet evaluated in comparison to the gold standard ICA, CCTA, using recent scanner techniques, may overcome these limitations and may become a reliable non-invasive test for CAV even at lowered dose, as shown by our study results.
We acknowledge some study limitations. A comparison to the gold standard ICA is lacking, as not all patients underwent consecutive ICA within a reasonable time frame [5]. Second, patients examined a long time after transplantation might have reacted to beta-blockers because of cardiac reinnervation, which might have had a benefit on radiation dose and IQ. When looking at the CT technique, two dose-saving algorithms were combined in pECGsys and, from our data, it is not clearly distinguishable how much each of these factors contributed to the overall dose reduction. However, this approach represents the future direction of modern CT, in order to achieve a maximum dose reduction [11, 27]. Furthermore, the positive trend of a superiority of rECG over pECGsys regarding overall IQ would have probably been reached if more patients had been included. Nevertheless, although this significance was not met in the present study, this would not necessarily affect the clinical recommendations.
In conclusion, automated tube voltage selections, combined with prospective systolic scanning, enable a significant dose reduction of approximately 50 %, compared to retrospective scanning, with comparable overall diagnostic IQ and lowered, but acceptable, IQ in the small coronary artery segments. In the context of recently published papers, this underlines the role of CCTA in the evaluation of CAV [7, 8]. With further reduction of radiation dose values using the next generation of CT scanner systems, patient-tailored scan protocols and iterative reconstruction algorithms, CCTA could take the next step and become a useful, non-invasive screening method for CAV, replacing ICA [28].
Abbreviations
- CAV:
-
Cardiac allograft vasculopathy
- CCTA:
-
Cardiac computed tomography angiography
- CTDI:
-
Computed tomography dose index
- DLP:
-
Dose length product
- ED:
-
Effective dose
- HU:
-
Hounsfield units
- ICA:
-
Invasive coronary angiography
- IQ:
-
Image quality
- MIP:
-
Maximum intensity projection
- mSv:
-
Millisievert
References
Christie JD, Edwards LB, Kucheryavaya AY et al (2012) The Registry of the International Society for Heart and Lung Transplantation: 29th adult lung and heart-lung transplant report—2012. J Heart Lung Transplant 31:1073–1086
Rahmani M, Cruz RP, Granville DJ, McManus BM (2006) Allograft vasculopathy versus atherosclerosis. Circ Res 99:801–815
Mancini D, Pinney S, Burkhoff D et al (2003) Use of rapamycin slows progression of cardiac transplantation vasculopathy. Circulation 108:48–53
Aliabadi A, Cochrane AB, Zuckermann AO (2012) Current strategies and future trends in immunosuppression after heart transplantation. Curr Opin Organ Transplant 17:540–545
Mehra MR, Crespo-Leiro MG, Dipchand A et al (2010) International Society for Heart and Lung Transplantation working formulation of a standardized nomenclature for cardiac allograft vasculopathy—2010. J Hear Lung Transplant 29:717–727
St Goar FG, Pinto FJ, Alderman EL et al (1992) Intracoronary ultrasound in cardiac transplant recipients. In vivo evidence of “angiographically silent” intimal thickening. Circulation 85:979–987
Wever-Pinzon O, Romero J, Kelesidis I et al (2014) Coronary computed tomography angiography for the detection of cardiac allograft vasculopathy: a meta-analysis of prospective trials. J Am Coll Cardiol 63:1992–2004
Mittal TK, Panicker MG, Mitchell AG, Banner NR (2013) Cardiac allograft vasculopathy after heart transplantation: electrocardiographically gated cardiac CT angiography for assessment. Radiology 268:374–381
Khan R, Jang I-K (2012) Evaluation of coronary allograft vasculopathy using multi-detector row computed tomography: a systematic review. Eur J Cardiothorac Surg 41:415–422
Einstein AJ, Henzlova MJ, Rajagopalan S (2007) Estimating risk of cancer associated with radiation exposure from 64-slice computed tomography coronary angiography. JAMA 298:317–323
Yin W-H, Lu B, Hou Z-H et al (2013) Detection of coronary artery stenosis with sub-millisievert radiation dose by prospectively ECG-triggered high-pitch spiral CT angiography and iterative reconstruction. Eur Radiol 23:2927–2933
Cerqueira MD (2002) Standardized myocardial segmentation and nomenclature for tomographic imaging of the heart: a statement for healthcare professionals from the Cardiac Imaging Committee of the Council on Clinical Cardiology of the American Heart Association. Circulation 105:539–542
Deak PD, Smal Y, Kalender WA (2010) Multisection CT protocols: sex- and age-specific conversion factors used to determine effective dose from dose-length product. Radiology 257:158–166
Hou Y, Yue Y, Guo W et al (2012) Prospectively versus retrospectively ECG-gated 256-slice coronary CT angiography: image quality and radiation dose over expanded heart rates. Int J Cardiovasc Imaging 28:153–162
Srichai MB, Barreto M, Lim RP et al (2013) Prospective-triggered sequential dual-source end-systolic coronary CT angiography for patients with atrial fibrillation: a feasibility study. J Cardiovasc Comput Tomogr 7:102–109
Kim HY, Lee JW, Hong YJ et al (2012) Dual-source coronary CT angiography in patients with high heart rates using a prospectively ECG-triggered axial mode at end-systole. Int J Cardiovasc Imaging 28(Suppl 2):101–107
Bastarrika G, De Cecco CN, Arraiza M et al (2008) Dual-source CT coronary imaging in heart transplant recipients: image quality and optimal reconstruction interval. Eur Radiol 18:1791–1799
Layritz C, Muschiol G, Flohr T et al (2013) Automated attenuation-based selection of tube voltage and tube current for coronary CT angiography: reduction of radiation exposure versus a BMI-based strategy with an expert investigator. J Cardiovasc Comput Tomogr 7:303–310
Yu L, Li H, Fletcher JG, McCollough CH (2009) Automatic selection of tube potential for radiation dose reduction in CT: a general strategy. Med Phys 37:234–243
Gonzalez-Guindalini FD, Ferreira Botelho MP, Töre HG et al (2013) MDCT of chest, abdomen, and pelvis using attenuation-based automated tube voltage selection in combination with iterative reconstruction: an intrapatient study of radiation dose and image quality. AJR Am J Roentgenol 201:1075–1082
Miller CA, Chowdhary S, Ray SG et al (2011) Role of noninvasive imaging in the diagnosis of cardiac allograft vasculopathy. Circ Cardiovasc Imaging 4:583–593
Mettler FA Jr, Bhargavan M, Faulkner K et al (2009) Radiologic and nuclear medicine studies in the United States and worldwide: frequency, radiation dose, and comparison with other radiation sources–1950–2007. Radiology 253:520–531
Noor M, Shekhdar J, Banner NR (2011) Radiation exposure after heart transplantation: trends and significance. J Heart Lung Transplant 30:309–314
Westwood ME, Raatz HDI, Misso K et al (2013) Systematic review of the accuracy of dual-source cardiac CT for detection of arterial stenosis in difficult to image patient groups. Radiology 267:387–395
Neefjes LA, Dharampal AS, Rossi A et al (2011) Image quality and radiation exposure using different low-dose scan protocols in dual-source CT coronary angiography: randomized study. Radiology 261:779–786
Sun Z, Ng K-H (2012) Prospective versus retrospective ECG-gated multislice CT coronary angiography: a systematic review of radiation dose and diagnostic accuracy. Eur J Radiol 81:e94–100
Fuchs TA, Stehli J, Bull S et al (2014) Coronary computed tomography angiography with model-based iterative reconstruction using a radiation exposure similar to chest X-ray examination. Eur Heart J 35:1131–1136
Kobashigawa J (2014) Coronary computed tomography angiography: is it time to replace the conventional coronary angiogram in heart transplant patients? J Am Coll Cardiol 63:2005–2006
Acknowledgments
The scientific guarantor of this publication is Florian Wolf. The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article. The authors state that this work has not received any funding. Veronika Schöpf kindly provided statistical advice for this manuscript. Institutional review board approval was obtained. Written informed consent was obtained from all subjects (patients) in this study. None of the study subjects or cohorts have been previously reported. Methodology: prospective, randomised controlled trial, performed at one institution.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Beitzke, D., Berger-Kulemann, V., Schöpf, V. et al. Dual-source cardiac computed tomography angiography (CCTA) in the follow-up of cardiac transplant: comparison of image quality and radiation dose using three different imaging protocols. Eur Radiol 25, 2310–2317 (2015). https://doi.org/10.1007/s00330-015-3650-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-015-3650-2