Abstract
Objectives
The aim of our study was to evaluate the tumour volume doubling time (TVDT) of molecular breast cancer subtypes by serial ultrasound (US).
Methods
Sixty-six patients (mean age, 50 years; range, 29–78 years) with invasive breast cancer underwent initial and follow-up breast US examinations (at least three months apart) with no intervention. TVDT was determined using the tumours’ greatest dimensions in two orthogonal planes. The results were compared with clinical, imaging, and tumour variables and molecular subtypes (oestrogen receptor [ER]-positive, human epidermal growth factor receptor 2 [HER2]-positive, and triple negative) using a multiple linear regression analysis.
Results
TVDT exhibited a wide range (46–825 days; median, 141 days) with an overall mean of 193 ± 141 days and mean values of 241 ± 166 days for ER-positive tumours (n = 37), 162 ± 60 days for HER2-positive tumours (n = 12), and 103 ± 43 days for triple-negative tumours (n = 17) (P < 0.0001). In a multivariate regression analysis, compared to other features, only the different molecular breast cancer subtypes showed significant difference in TVDT (P < 0.0001).
Conclusions
TVDT differed significantly among the three molecular breast cancer subtypes, with the triple-negative tumours showing the fastest growth.
Key Points
• Knowledge of tumour volume doubling time provides clues for improving screening.
• TVDT assessed by serial US differed significantly between breast cancer subtypes.
• Triple-negative tumours had 2.4-fold shorter TVDT compared to ER-positive tumours.
• Tumours classified as BI-RADS 3 had shorter TVDT than BI-RADS 4.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The characterisation of tumour volume doubling time (TVDT) is important not only for determination of the optimal interval for screening and follow-up but also for developing new strategies for treatment [1]. A few reports have evaluated breast cancer TVDT on mammography [2, 3]. However, mammography is not a reliable imaging modality for breast cancer size measurement and detection, particularly in dense breast tissue [4]. Sectional imaging modalities, such as breast magnetic resonance imaging (MRI) and ultrasound (US), are more accurate in the measurement of tumour size and volume, and they could be used to evaluate TVDT of breast cancers [5–7]. With the increasing use of US for both screening and diagnostic purposes, breast cancers initially assessed as benign or suspicious undergo serial breast US examinations without any intervention, thus providing TVDT assessment [8].
Breast cancer is a heterogeneous disease with different clinicopathological features, recurrence patterns, and survival [9–11]. Three major molecular breast cancer subtypes can be distinguished by immunohistochemistry (IHC): oestrogen receptor (ER)-positive, human epidermal growth factor receptor 2 (HER2)-positive, and triple-negative [10]. This classification has been widely used for predicting the prognosis and response to treatment in breast cancer patients [12]. Dawson et al. investigated the distribution of these subtypes in breast cancers detected at screening and those detected without screening [13]. Tumours detected at screening were more likely to belong to the ER-positive subtype and were less likely to belong to the triple-negative subtype. Imaging findings of the three molecular breast cancer subtypes have recently been published and triple-negative tumours frequently have benign or indeterminate features on mammography and US [14–16]. To our knowledge however, no data have been published on TVDT according to molecular breast cancer subtype.
The primary objective of this study was to determine the TVDT of molecular breast cancer subtypes on a serial US; secondary objectives were to identify clinical, imaging, and tumour variables linked to TVDT, and to assess the reproducibility and accuracy of the measurement.
Materials and methods
Patients
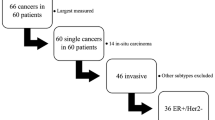
This retrospective study was approved by the institutional review board (Seoul National University Hospital) and informed consent was waived. A search of departmental preoperative US databases identified 2030 female patients with invasive breast cancer between January 2003 and May 2012, and a total of 305 patients underwent serial breast US with no intervention (i.e., needle biopsy or treatment). Cases were then excluded for the following reasons: no visible lesion on previous breast US (155 patients), an interval of less than three months between the first and last breast US examinations (52 patients), and no immunohistochemistry results (32 patients). These exclusions yielded a final cohort of 66 female patients (mean and median age, 50 and 52 years; range, 29–78 years) with 66 cases of invasive breast cancer. To minimize measurement bias related to varying US data acquisition, we restricted our analyses to patients who had undergone two US examinations that were at least three months apart. In three patients who had more than two breast US examinations, the initial and final follow-up breast US examinations were selected for TVDT analysis.
All patients were asymptomatic at the initial breast US examination. Of these patients, two neglected the biopsy recommendation for suspicious breast lesions on US, and the others were scheduled at six (n = 8) or 12 (n = 56) months follow-up for probably benign or benign lesions on US. The reason for prompt biopsy at the time of diagnosis was either the presence of a palpable mass (n = 21) or an increase in tumour size on follow-up US (n = 45). Mammograms were available in 56 women at the time of the initial US examination, and all 66 women had mammograms at the time of diagnosis.
US examinations and imaging review
All US images were obtained using an HDI 5000 system (Advanced Technology Laboratories, Bothell, Wash) with a 12-5 MHz linear transducer or an EUB-8500 system (Hitachi Medical, Tokyo, Japan) with a 14-6 MHz linear transducer. Twelve breast radiologists with 2–25 years of experience in breast US performed the breast US examination. The imaging protocol included transverse and longitudinal real-time imaging of the lesions. All images were sent and saved in a picture archiving and communications system (PACS). The 5,000-pixel monochrome liquid crystal display monitor (ME1i2-BC, Totoku, Electric Co., Tokyo, Japan) and PACS workstation were used to review the images.
For this study, initial breast US were retrospectively reviewed in consensus by two breast radiologists without clinical or pathologic information. Imaging findings were analysed according to BI-RADS US lexicon (shape, orientation, margin, echo pattern, lesion boundary, posterior acoustic features, and calcifications), and a final assessment category was provided to indicate the probability of malignancy [17]. A solid mass with ovoid shape, circumscribed margin, and parallel orientation was assessed as BI-RADS category 3, and masses with any suspicious findings were assessed as BI-RADS category 4 or 5. We performed this retrospective review because the BI-RADS US lexicon was not used until 2005 in our institution.
Calculation of TVDT
An investigator who was not involved in the reader study retrieved the representative transverse and longitudinal images of lesions at initial and follow-up breast US. A single transverse image and a single longitudinal image for each lesion were provided for the readers.
The longest dimension and maximal perpendicular dimension of each lesion were measured by four radiologists, who did not perform the US examinations and who were blinded to the image acquisition technique by using electronic callipers on the transverse images. Additionally, the longest dimension of each lesion on the longitudinal scans was measured. The volume of each tumour was estimated using the formula for oblate spheroids [5]: V = 4/3π • a/2 • b/2 • c/2, where a, b, and c denote the means of the four observers’ measurements.
By using the volumes from the initial and follow-up examinations, TVDT was calculated in days by using the equation: TVDT = t • log 2/(log V2– log V1), where t represents the interval in days between the two examinations and V1 and V2 represent lesion volume on the initial and follow-up studies, respectively [2, 18]. Tumours were assumed to have exponential growth, as this is usually the best approximation for the range of tumour sizes in our study [2].
Histological evaluation
Breast conserving surgery (n = 45) or mastectomy (n = 21) was performed for all breast cancers. The histological type, invasive tumour size, histological grade, and lymph node status were determined from the surgically excised specimens. The expression of ER, progesterone receptor (PR), HER2, and the Ki-67 index were evaluated in the surgically excised specimens by standard avidin–biotin complex IHC staining methods.
Based on the expression of ER, PR, and HER2 status, breast cancers were classified into three molecular subtypes: ER-positive, HER2-positive, and triple-negative. The ER subtype was defined by a finding of at least 1 % positive tumour cells on either or both of the ER and PR assays [19]. The HER2 status could be either negative or positive. The HER2 subtype was defined to include ER-negative cancers showing HER2 over-expression and/or HER2 gene amplification [20]. The triple-negative subtype was defined as cancers with ER-negative/PR-negative/HER2-negative results. For the Ki-67 index, a cutoff value of 14 % was used as a cellular marker for proliferation [21].
Data analysis
The documented information on clinical, imaging, and tumour variables included patient age at diagnosis, menopausal status, previous history of breast cancer, symptoms at diagnosis, mammographic density, BI-RADS category and tumour size (defined as maximal diameter) on initial US, histologic type, invasive tumour size, histologic grade, lymph node metastases, ER, PR, HER2, Ki-67, and molecular subtypes. Univariate analysis was performed to compare TVDT and variables of breast cancer. Analysis of variance (ANOVA) or Kruskal-Wallis analysis was used to compare continuous variables, and Fisher’s exact test or the χ2 test was used for categorical variables. Multiple linear regression analysis was used to perform multivariate analyses to determine which variables were most influential on TVDT. A t-test or Wilcoxon’s rank sum test was used to assess the differences of TVDT according to BI-RADS US features. The analyses were performed using SAS 9.2 software (SAS Institute, Cary, NC). A P-value less than 0.05 was considered to indicate a significant difference.
Interobserver agreement in three-dimensional measurements, and between the greatest dimension on follow-up US and at pathologic examination, was assessed by using intraclass correlation coefficient (ICC) values. ICCs were defined as follows: an ICC of 0–0.20 indicated no agreement; an ICC of 0.21–0.40, poor agreement; an ICC of 0.41–0.60, moderate agreement; an ICC of 0.61–0.80, good agreement; and an ICC greater than 0.80, excellent agreement.
Results
Clinical, imaging, and tumour characteristics
There were 37 ER-positive (including one ER-positive and HER2-positive cancer), 12 HER2-positive, and 17 triple-negative tumours. Clinical, imaging, and tumour characteristics according to molecular breast cancer subtypes are summarised in Table 1. Women with triple-negative subtypes more often had symptoms at diagnosis, a larger tumour size and volume on follow-up US, larger invasive tumour size, and higher tumour grade compared to women with ER-positive subtypes. The three molecular breast cancer subtypes did not differ in terms of age at diagnosis, menopausal status, previous history of breast cancer, mammographic density, BI-RADS category, tumour size and volume on initial US, interval between initial and follow-up US examinations, histologic type, and lymph node metastases.
Tumour volume doubling time
All tumours increased in diameter and volume between initial and follow-up US examinations. A large variation in TVDT was observed from 46 days to 825 days (mean, 193 ± 141 days; median, 141 days). There were significant differences in TVDT among molecular breast cancer subtypes (P < 0.0001) (Fig. 1) with mean values of 241 ± 166 days (range, 75-825 days; median, 194 days) for ER-positive tumours (Fig. 2), 162 ± 60 days (range, 97-312 days; median, 154 days) for HER2-positive tumours (Fig. 3), and 103 ± 43 days (range, 46-205 days; median, 93 days) for triple-negative tumours (Fig. 4). In ER-positive tumours, TVDT increased with increasing tumour size whereas TVDT remained constant in HER2-positive and triple-negative tumours (Fig. 5).
Transverse US images in a 69-year-old woman with ER-positive, grade 1 invasive ductal carcinoma. a US image shows an 8x4-mm hypoechoic, oval-shaped mass (arrow). The mass was initially classified as probably benign and follow-up was chosen as a management. b US image obtained after 12 months shows a 10x6-mm hypoechoic mass (arrow). Initial and follow-up tumour volumes were 835 mm3 and 2010 mm3, respectively, and the tumour volume doubling time was 276 days
Transverse US images in a 75-year-old woman with HER2-positive, grade 3 invasive ductal carcinoma. a US image shows a 6x4-mm hypoechoic, irregularly-shaped mass (arrow). The mass was initially classified as suspicious; however, the patient neglected a core biopsy. b US image obtained after 11 months (now palpable) shows a 9x8-mm hypoechoic mass. Initial and follow-up tumour volumes were 807 mm3 and 2398 mm3, respectively, and the tumour volume doubling time was 224 days
Transverse US images in a 59-year-old woman with triple-negative, grade 3 invasive ductal carcinoma. a US image shows an 11x7-mm hypoechoic, oval-shaped mass (arrow) with posterior enhancement. The mass was initially classified as probably benign and follow-up was chosen as a management. b US image obtained after seven months (now palpable) shows a 20x19-mm hypoechoic mass (arrow). Initial and follow-up tumour volumes were 3,730 mm3 and 23,864 mm3, respectively, and the tumour volume doubling time was 81 days
According to the retrospective review, 58 tumours were classified as BI-RADS category 3 and eight tumours were classified as category 4, with TVDT values of 176 ± 111 days and 302 ± 264 days, respectively (P = 0.001) (Table 2). However, there were no significant US features related to shorter TVDT (Table 3).
In univariate analysis, significant differences in TVDT were found with respect to symptoms at diagnosis (P = 0.005), BI-RADS category (P = 0.001) on initial US, ER status (P = 0.001), PR status (P = 0.002), Ki-67 index (P = 0.004), and molecular subtypes (P < 0.0001). However, age at diagnosis, menopausal status, previous history of breast cancer, mammographic density, histologic grade, HER2 status, and lymph node metastases did not affect TVDT (Table 2). By multivariate regression analysis, only the molecular breast cancer subtype was significantly associated with TVDT (P < 0.0001).
Interobserver agreement
There was excellent ICC between the dimensions measured by four readers: the longest dimension on transverse scan (ICC = 0.95, 95 % CI: 0.92 – 0.97), maximal perpendicular dimension on transverse scan (ICC = 0.91, 95 % CI: 0.83 – 0.98), and longest dimension on longitudinal scan (ICC = 0.87, 95 % CI: 0.79 – 0.90) (Table 4). TVDT measured by four readers also showed excellent ICC (ICC = 0.91, 95 % CI: 0.80 – 0.96).
The greatest dimension measured on US and at pathologic examination showed good ICC (ICC = 0.77, 95 % CI: 0.64 – 0.86). Differences in measurements of the greatest dimension by US and pathologic examination were found in six invasive ductal carcinomas with ductal carcinoma in situ. After exclusion of these six cases, an excellent ICC for the greatest dimension measured by US and pathologic examination was observed (ICC = 0.91, 95 % CI: 0.83 – 0.95).
Discussion
Although, there were several clinical, imaging, and tumour variables that exhibited association with TVDT by univariate analysis, only the molecular breast cancer subtype was significantly associated with TVDT according to multiple linear regression analysis. We measured TVDT using the greatest dimensions of the tumour in two orthogonal planes at serial breast US, and the measurement showed high reproducibility among four observers, as well as a good correlation with pathology. Breast US with its sectional imaging capability is more accurate in the measurement of tumour size and volume compared to mammography, and could be used as a nonionizing method to evaluate TVDT of breast cancers. The TVDT values (mean, 193 days; median, 141 days; range, 46–825 days) of the breast cancers in our study were within the range of previously measured or estimated TVDTs using mammography or MRI [2–6]. Most mammography studies, however, were conducted before the molecular breast cancer subtype was published [22]. This study is, to our knowledge, the first to investigate TVDT according to molecular breast cancer subtypes.
Our results showed significantly different TVDT values among the three molecular breast cancer subtypes. Triple-negative tumours, the most aggressive form of breast cancer, had 2.4-fold and 1.6-fold shorter TVDT (103 ± 43 days) compared to ER-positive (241 ± 166 days) and HER2-positive (162 ± 60 days) tumours, respectively. Because we are now able to identify the type of breast cancer for which women are at risk, this information will provide important clues for designing demonstration projects to associate risk-based screening and tumour molecular subtype [10, 11, 23, 24]. Our findings on TVDT also support that molecular breast cancer subtype should be considered when planning ongoing management and surveillance for breast cancer patients [25]. In previous studies, the peak recurrence for ER-positive tumours was after 36 months post-operation, while the recurrence of HER2-positive and triple-negative tumours peaked at 12-24 months [26, 27].
Clinical and experimental observations show that cancer growth follows an S-shaped or Gompertzian curve between linear and exponential kinetics [28, 29]. The Gompertzian model predicts that TVDT does not remain constant over time but is related to tumour size, such that TVDT increases with increasing tumour size. Our results showed that there was, however, a significant difference in tumour growth patterns according to the molecular breast cancer subtype. In ER-positive tumours, TVDT increased with increasing tumour size whereas TVDT remained constant in HER2-positive and triple-negative tumours. This finding, to the best of our knowledge, has not been previously reported and will be helpful to develop mathematical modelling of tumour growth and metastases according to molecular breast cancer subtypes. In contrast to previous studies [2, 6], no significant difference in TVDT was found between young and old women in our study.
Notably, breast cancer classified as BI-RADS category 3, probably benign, on initial US has a significantly shorter TVDT than category 4, suspicious (176 ± 111 days and 302 ± 264 days, P = 0.001). This finding is concordant with previous reports that triple-negative and high-grade tumours have a tendency to exhibit benign US features such as round or oval shape, circumscribed margins, and posterior enhancement due to necrosis and lack of a host desmoplastic reaction [14, 30]. In our study, however, no US features within the BI-RADS lexicon were significantly related to shorter TVDT. Additional studies such as colour Doppler and elastography will be helpful to avoid misclassifying triple-negative and high-grade tumours as probably benign on US [31, 32]. Triple-negative tumours more often presented as interval cancer compared to other subtypes in our study (59 % in triple negative vs. 22 % in ER positive and 25 % in HER2 positive, P = 0.021).
This study has several limitations. First, this is a retrospective study with a small sample size, and only one case of ER-positive and HER2-positive cancer was included. Selection bias was inevitable because only cancers visible on both initial and follow-up breast US were included. Many of the fast-growing tumours could be excluded and our results on TVDT may differ from those obtained from population-based screening [33]. Second, we did not evaluate the interobserver variability for data acquisition. Variability within data acquisition and tumour volume measurement could occur as a result of minor changes in position or degree of compression. An automated 3D US scanner has been developed and could be used to monitor changes in tumour volume during follow-up [34]. Third, we did not provide treatment response or prognosis according to TVDT. Previous studies have shown that patients with shorter TVDT tend to have a poorer prognosis, particularly when multiple nodal metastases are combined [35].
In conclusion, the TVDT of breast cancer differed significantly among the three molecular subtypes, with the triple-negative tumours showing the fastest growth. This information will provide important clues for improving screening and surveillance of breast cancer patients.
Abbreviations
- TVDT :
-
Tumour volume doubling time
- ER :
-
oestrogen receptor
- PR :
-
progesterone receptor
- HER2 :
-
human epidermal growth factor receptor 2
- IHC :
-
immunohistochemistry
- BI-RADS :
-
Breast Imaging Reporting and Data System
References
Friberg S, Mattson S (1997) On the growth rates of human malignant tumors: implications for medical decision making. J Surg Oncol 65:284–297
Peer PG, van Dijck JA, Hendriks JH, Holland R, Verbeek AL (1993) Age-dependent growth rate of primary breast cancer. Cancer 71:3547–3551
von Fournier D, Weber E, Hoeffken W, Bauer M, Kubli F, Barth V (1980) Growth rate of 147 mammary carcinomas. Cancer 45:2198–2207
Robertson JF, Caseldine J, Winfield S (1987) Breast mammography and tumour volume. Br J Cancer 56:902
Millet I, Bouic-Pages E, Hoa D, Azria D, Taourel P (2011) Growth of breast cancer recurrences assessed by consecutive MRI. BMC Cancer 11:155
Madeleine MA, Tilanus-Linthorst MM, Obdeijn IM et al (2007) BRCA1 mutation and young age predict fast breast cancer growth in the Dutch, United Kingdom, and Canadian magnetic resonance imaging screening trials. Clin Cancer Res 13:7357–7362
Bosch AM, Kessels AG, Beets GL et al (2003) Preoperative estimation of the pathological breast tumour size by physical examination, mammography and ultrasound: a prospective study on 105 invasive tumours. Eur J Radiol 48:285–292
Moon HJ, Kim EK, Kwak JY, Yoon JH, Kim MJ (2011) Interval growth of probably benign breast lesions on follow-up ultrasound: how can these be managed? Eur Radiol 21:908–918
Sorlie T, Perou CM, Tibshirani R et al (2001) Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci U S A 98:10869–10874
Carey LA, Perou CM, Livasy CA et al (2006) Race, breast cancer subtypes, and survival in the Carolina Breast Cancer Study. JAMA 295:2492–2502
Lin NU, Vanderplas A, Hughes ME et al (2012) Clinicopathologic features, patterns of recurrence, and survival among women with triple-negative breast cancer in the National Comprehensive Cancer Network. Cancer 118:5463–5472
de Ronde JJ, Hannemann J, Halfwerk H et al (2010) Concordance of clinical and molecular breast cancer subtyping in the context of preoperative chemotherapy response. Breast Cancer Res Treat 119:119–126
Dawson SJ, Duffy SW, Blows FM et al (2009) Molecular characteristics of screen-detected vs symptomatic breast cancers and their impact on survival. Br J Cancer 101:1338–1344
Dogan BE, Gonzalez-Angulo AM, Gilcrease M, Dryden MJ, Yang WT (2010) Multimodality imaging of triple receptor-negative tumors with mammography, ultrasound, and MRI. AJR Am J Roentgenol 194:1160–1166
Ko ES, Lee BH, Kim HA, Noh WC, Kim MS, Lee SA (2010) Triple-negative breast cancer: correlation between imaging and pathological findings. Eur Radiol 20:1111–1117
Boisserie-Lacroix M, Macgrogan G, Debled M et al (2013) Triple-negative breast cancers: associations between imaging and pathological findings for triple-negative tumors compared with hormone receptor-positive/human epidermal growth factor receptor-2-negative breast cancers. Oncologist 18:802–811
Mendelson EB, Baum JK, Berg WA, Merritt CRB, Rubin E (2003) Breast Imaging Reporting and Data System, BI-RADS: Ultrasound, 1st edn. American College of Radiology, Reston
Tilanus-Linthorst MM, Kriege M, Boetes C et al (2005) Hereditary breast cancer growth rates and its impact on screening policy. Eur J Cancer 41:1610–1617
Hammond ME, Hayes DF, Wolff AC, Mangu PB, Temin S (2010) American society of clinical oncology/college of american pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. J Oncol Pract 6:195–197
Wolff AC, Hammond ME, Schwartz JN et al (2007) American Society of Clinical Oncology/College of American Pathologists guideline recommendations for human epidermal growth factor receptor 2 testing in breast cancer. J Clin Oncol 25:118–145
Cheang MC, Chia SK, Voduc D et al (2009) Ki67 index, HER2 status, and prognosis of patients with luminal B breast cancer. J Natl Cancer Inst 101:736–750
Perou CM, Sorlie T, Eisen MB et al (2000) Molecular portraits of human breast tumours. Nature 406:747–752
Turkoz FP, Solak M, Petekkaya I et al (2013) Association between common risk factors and molecular subtypes in breast cancer patients. Breast 22:344–350
Eklund M, Esserman LJ (2013) Screening: biology dictates the fate of young women with breast cancer. Nat Rev Clin Oncol 10:673–675
de Melo Gagliato D, Gonzalez-Angulo AM, Lei X et al (2014) Clinical impact of delaying initiation of adjuvant chemotherapy in patients with breast cancer. J Clin Oncol(Jan 27)
Park S, Koo JS, Kim MS et al (2012) Characteristics and outcomes according to molecular subtypes of breast cancer as classified by a panel of four biomarkers using immunohistochemistry. Breast 21:50–57
Lee JA, Kim KI, Bae JW et al (2010) Triple negative breast cancer in Korea-distinct biology with different impact of prognostic factors on survival. Breast Cancer Res Treat 123:177–178
Norton L (1988) A Gompertzian model of human breast cancer growth. Cancer Res 48(24 Pt 1):7067–7071
Demicheli R, Terenziani M, Valagussa P, Moliterni A, Zambetti M, Bonadonna G (1994) Local recurrences following mastectomy: support for the concept of tumor dormancy. J Natl Cancer Inst 86:45–48
Stavros AT (2003) Breast ultrasound. Lippincott Williams & Wilkins, Philadelphia, PA, pp 490–491
Berg WA, Cosgrove DO, Doré CJ et al (2012) Shear-wave elastography improves the specificity of breast US: the BE1 multinational study of 939 masses. Radiology 262:435–449
Cho N, Jang M, Lyou CY, Park JS, Choi HY, Moon WK (2012) Distinguishing benign from malignant masses at breast US: combined US elastography and color doppler US–influence on radiologist accuracy. Radiology 262:80–90
Weedon-Fekjaer H, Lindqvist BH, Vatten LJ, Aalen OO, Tretli S (2008) Breast cancer tumor growth estimated through mammography screening data. Breast Cancer Res 10:R41
Kelly KM, Dean J, Lee SJ, Comulada WS (2010) Breast cancer detection: radiologists’ performance using mammography with and without automated whole-breast ultrasound. Eur Radiol 20:2557–2564
Kuroishi T, Tominaga S, Morimoto T et al (1990) Tumor growth rate and prognosis of breast cancer mainly detected by mass screening. Jpn J Cancer Res 81:454–462
Acknowledgments
The scientific guarantor of this publication is Woo Kyung Moon. The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article. This study has received funding by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MEST) (No. 2012R1A2A1A01010846). No complex statistical methods were necessary for this paper. Institutional Review Board approval was obtained. Written informed consent was waived by the Institutional Review Board. Methodology: retrospective, observational, performed at one institution.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ryu, E.B., Chang, J.M., Seo, M. et al. Tumour volume doubling time of molecular breast cancer subtypes assessed by serial breast ultrasound. Eur Radiol 24, 2227–2235 (2014). https://doi.org/10.1007/s00330-014-3256-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-014-3256-0