Abstract
An efficient system for the establishment and multiplication of highly prolific embryogenic cell cultures of grapevine (Vitis sp.) was developed. Using anther-derived pro-embryogenic masses as starting material, cell suspensions of different grapevine cultivars (Tempranillo, Cabernet-Sauvignon) and rootstocks (Kober 125 AA, Kober 5 BB, 110 Richter) were initiated in liquid medium containing NOA (1.0 mg l-1) and BAP (0.25 mg l-1) as growth regulators. Conditioned medium was recovered and utilised for establishing new, highly totipotent cell cultures. The suspensions obtained, showed embryogenic competence resulting in somatic embryo induction and subsequent plant regeneration. In this study, a simplified establishment procedure for grapevine embryogenic cell suspension allowing the fast multiplication of embryogenic material is described. Evidence for the promoting effect of the protein fraction derived from conditioned medium, on cell proliferation was found. In bioassays, addition of ß-d-GlcY affect cell proliferation suggesting that arabinogalactan proteins are required for growth processes in grapevine cell cultures.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Embryogenic suspension cultures have been found to be a useful system for plant regeneration (Reustle et al. 1995; Taylor et al. 1996; Gupta and Conger 1999). Furthermore, somatic embryo development and synchronisation under liquid conditions were reported as crucial steps in taking full advantage of somatic embryogenesis for high production potential and reduction of unit cost through automation (Tonon et al. 2001). In grapevine, the world’s most widely grown fruit crop, somatic embryogenesis represents the best system for plant regeneration and gene transfer technology (Perl et al. 1996) as well as for cryo-preservation (Dussert et al. 1992; Wang et al. 2004). Somatic embryogenesis has been reported for several Vitis species (Rajasekaran and Mullins 1979; Mauro et al. 1986; Stamp and Meredith 1988; Martinelli and Gribaudo 2001). Embryogenic suspension establishment was described by Coutos-Thévenot et al. (1992a) and Jayasankar et al. (1999) and was shown to be the most suitable material for grapevine genetic engineering (Hebert et al. 1993; Kikkert et al. 1996; Vidal et al. 2003). However, embryogenic suspension establishment is often considered as a time-consuming procedure requiring high amounts of embryogenic material to get the suspension started. In addition, growth inhibition and browning of the suspension as well as loss of embryogenic competence of the cells was frequently observed.
van Hengel et al. (1998) postulated secretion of metabolites by cultured cells, contributing to the conditioning of the medium. Beside growth regulators, other classes of molecules have been recently identified as embryo-stimulating factors, especially, those secreted into the culture medium (Massonneau et al. 2005). It was suggested that some extra-cellular proteins such as arabinogalactan proteins (AGP), receptor protein kinases (RPK) and endochitinases were involved in embryogenesis (De Jong et al. 1992; van Engelen and De Vries 1992; Becraft 1998; van Hengel et al. 2002). AGP represent a protein family in which the core protein is linked to arabinogalactan residues. These compounds play an important role in plant development and morphogenesis (Majewska-Sawka and Nothnagel 2000), and were found in the culture media of various plant tissue culture systems (Immerzeel et al. 2004, 2006). For rose cell cultures, AGPs were reported to induce cell proliferation (Kimberly and Nothnagel 1997; Serpe and Nothnagel 1994).
The present study reports an efficient and reproducible procedure for the establishment of embryogenic cell suspension of grapevine using conditioned medium. The procedure results in highly proliferating embryogenic cell suspensions with competence for successful plant regeneration, offering a useful target material for grapevine transformation and the production of transgenic plants. The protocol presented in this paper suggests the presence of extra-cellular proteins, stimulating cell division and leading to a significant increase of callus fresh weight. Experiments were performed to identify some of these extra-cellular substances involved in cell proliferation.
Materials and methods
Induction of somatic embryogenesis
Five grapevine varieties were used in this study, the rootstocks 110 Richter (Vitis berlandieri × Vitis rupestris), Kober 5 BB (Vitis berlandieri × Vitis riparia), Kober 125 AA (Vitis berlandieri × Vitis riparia) and two cultivars of Vitis vinifera (cv. Tempranillo and cv. Cabernet-Sauvignon). Embryogenic callus was induced from anther and ovary cultures on solid NN medium (Nitsch and Nitsch 1969) containing 2,4-D (1.0 mg l−1) and BAP (0.25 mg l−1) according to Mauro et al. (1986). After induction, maintenance and propagation of the embryogenic callus was performed on the same medium as for induction. The embryogenic material was subcultured onto fresh medium in 4-week intervals.
Procedures for cell suspension establishment and maintenance
Embryogenic callus and proembryogenic masses (PEM) derived from anther-/ovary-culture were used for the initiation of embryogenic suspensions. As grapevine suspension medium, a modified NN69 liquid medium (Nitsch and Nitsch 1969) containing 0.5 gl−1 MES, 20 gl−1 maltose and 4.6 gl−1 glycerol instead of sucrose (Coutos-Thévenot et al. 1992a) supplemented with growth regulators (1.0 mg l−1 NOA, 0.25 mg l−1 BAP) was used. The pH was adjusted to 5.8 with KOH before autoclaving (20 min at 120°C). For initiation of suspension, 0.5–2 g fresh weight of fine embryogenic callus (2–3 weeks post subculture) was transferred into a 500 ml Erlenmeyer flask containing 100 ml of medium. Suspensions were incubated in the dark on an orbital rotary shaker (100 rpm) at 24°C, and maintained by weekly exchange of 50% of the old medium with fresh medium.
For cell suspensions establishment, three different approaches were developed and compared:
-
1.
Fresh medium procedure: suspensions were initiated and maintained as described above.
-
2.
Increased volume procedure: suspensions were initiated in 50 ml fresh medium (FM) followed by weekly subculture, removing an increasing volume (10, 20, 30, 40 ml) of old medium and addition of an increasing volume (20, 30, 40, 50 ml) of FM until 100 ml final volume had been reached. Then, 50% of the medium was exchanged by FM in 7-day intervals.
-
3.
Conditioned medium procedure: suspensions were initiated in a mixture of 50 ml conditioned medium (CM) and 50 ml FM, and subcultured in 1-week intervals by exchanging 50% of the medium by FM. The CM was collected from suspensions already established for about 2 months. Therefore, cells were allowed to settle down and 50% of the supernatant was collected and filter-sterilised (0.45 μm) using reusable bottle top filter unit.
Suspensions were initiated with 0.5, 1.0 and 2.0 g of grapevine embryogenic callus and cultivated for 6 weeks. Fresh weight development of embryogenic callus in suspension was measured weekly by removing the entire liquid medium. For each treatment, three suspensions were initiated and fresh weight of these replicates was determined.
Embryogenic cell suspensions of zucchini (Cucurbita pepo) and non-embryogenic suspension of parsley (Petroselinum crispum) cultivated in MS medium (5 mg l−1 2,4-D, 3 mg l−1 Kinetin) and Gamborg B5 medium (1 mg l−1 2,4-D) respectively were transferred into grapevine suspension medium (modified NN69) and cultivated for about 2 weeks before collecting CM as described before.
Concentration of extra-cellular proteins
Seven-day old CM (500 ml) from each grapevine variety was collected and filter-sterilised (0.45 μm). Proteins were concentrated 200-fold by 3.5 kDa cut-off membrane (Membra-Cel™ Dialysis Membranes, Serva). Resin (StragaClean™ Resin, Stratagene) was added to the concentrated protein fraction and shaken overnight at 4°C before centrifugation at 13,000 g to concentrate the resin-protein binding complex. The resin–protein complex was resuspended in glycine solution (pH 2) to separate proteins from the complex. Samples of concentrated CM (40 μl) from each variety were loaded on a nitrocellulose membrane (Hybond-P, Amersham Biosciences) using a Dot-blot device (Easy-Titer® Elifa System, Pierce, USA). Immuno-detection was performed as described by Burnette (1981). AGPs were detected with MAC 207 monoclonal antibody (Carbosource Services, USA) against α-Glc α1−>3 α-Gal α1−>2 Rha carbohydrate chains. This antibody was recognized using a secondary mouse anti-rat antibody conjugated with peroxidase and revealed by chemiluminescent detection (lumilight western blotting kit, Roche Diagnostics).
Bioassays with extra cellular protein fraction
-
1.
Concentrated extra-cellular protein fraction (CEPF) of each CM were added to initiate cell suspensions using 0.5 g callus fresh weight in 250 flasks containing 50 ml liquid FM.
-
2.
Arabinogalactan (AG) from larch wood (Sigma-Aldrich, A-9788) was added into the FM at the initiation step for cell suspension and at each weekly subculture.
-
3.
The β-D-GlcY reagent [1,3,5-tris (4-β-D-glycopyranosyloxyphenylazo)-2,4,6-trihydroxybenzene] is a synthetic phenylglycoside which specifically binds to AGP. β-D-GlcY was added to grapevine embryogenic suspensions to determine the effect on cell proliferation. For this, 25 ml of CM were treated with β-D-GlcY (25 μM) for 2 h at room temperature. Half a gram of Tempranillo callus fresh weight was used to start suspension with 25 ml FM and 25 ml β-D-GlcY treated CM.
Regeneration of plants from embryogenic suspensions
Six weeks after suspension establishment, induction of embryo development was started from the different grapevine varieties. Approximately, 500 μl of packed cell volume (PCV) determined after sedimentation of the cells in a 10 ml sterile pipette, was inoculated in 250 ml flasks containing 50 ml of growth regulator free medium. After 2 weeks of cultivation with a daily transfer of the material in growth regulator free FM, PEM progressively produced embryos at globular, heart and torpedo stage. Following 3–6 weeks development under daily subculture conditions, 25 elongated somatic embryos were transferred to growth regulator free NN69 medium (Nitsch and Nitsch 1969) supplemented with activated charcoal and solidified with 0.3% gelrite. Embryos were adapted stepwise to standard culture room conditions (16 h photoperiod, 60 μmol m−2 s−1 photon fluence rate) for conversion and plantlets where transferred to greenhouse conditions. For each variety, 25 somatic embryos were transferred per plate and 20 replicates were tested.
Results
Establishment of grapevine embryogenic cell suspensions
Under standard conditions (FM procedure), when embryogenic callus of grapevine was transferred from solid culture into fresh liquid medium, suspensions were established within 2–3 months. Frequently, cell suspensions turned brown with a dramatic decrease of growth when initiated with 2 g of callus (Fig. 1a). In contrast, the use of less than 1 g of callus for suspension establishment could not initiate cell division and proliferation (data not shown). However, with the increased volume procedure for suspension initiation, PEM started to grow gradually indicating improved growth conditions (Fig. 1b). Suspensions initiated with CM procedure showed vigorous proliferation of fine embryogenic cell aggregates and significant increase of fresh weight was observed (Fig. 1c).
Efficiency of suspension establishment procedures. Experiments were carried out with embryogenic material of Vitis vinifera cv. Tempranillo. Starting inoculum: 0.5 g filled circle, 1.0 g filled square and 2.0 g filled triangle embryogenic tissue. a Fresh medium procedure with 100 ml of fresh liquid medium. b Cell suspensions initiated by using increased volume procedure. c Cell suspensions initiated with 50% of conditioned medium (CM). Data represent average of three independent replications, bars indicate SD values
Following the CM procedure, cell suspensions from Tempranillo, Cabernet Sauvignon, Kober 125 AA, Kober 5 BB and 110 Richter could be established within 3–5 weeks (Fig. 2a) using CM of Vitis vinifera cv. Tempranillo. A reciprocal experiment was performed using CM of these varieties for the initiation of new embryogenic cell suspensions of Tempranillo (Fig. 2b). Most of the suspensions showed a good multiplication rate and growth remained in an exponential phase. Furthermore, CM derived from embryogenic suspension of zucchini (Cucurbita pepo) and non-embryogenic parsley suspensions (Petroselinum crispum) could also induce embryogenic suspension establishment and cell proliferation of grapevine (Fig. 2c). This indicates the presence of common compounds in conditioned medium related to cell division processes.
Effect of conditioned medium (CM) for establishment of grapevine embryogenic cell suspension. a Initiation of embryogenic cell suspensions of different grapevine varieties using CM of Vitis vinifera cv. Tempranillo. b Initiation of embryogenic cell suspension of Vitis vinifera cv. Tempranillo using CM collected from suspensions of different grapevine varieties: Kober 125 AA filled square, Kober 5 BB cross mark, Cabernet Sauvignon filled triangle and 110 Richter open circle. c Establishment of Vitis vinifera cv. Tempranillo embryogenic cell suspension with CM from parsley filled diamond and zucchini filled square suspensions, CM from Tempranillo as positive control filled triangle, FM as negative control filled circle. Data represents average of three independent replications, bars indicate SD values
To elucidate if peptides or extra-cellular proteins are responsible for this effect, autoclaving of CM was performed for protein denaturation. Results showed that the promoting effect of CM on biological activity of cell suspensions was lost when autoclaved, resulting in growth performance as obtained with the FM procedure (data not shown).
Effect of extra-cellular proteins on grapevine suspensions
Supplementing concentrated extra-cellular proteins to fresh medium resulted in a promoting effect on suspension initiation and cell proliferation (Table 1). Fractionation of concentrated CM proteins by anion exchange chromatography (Mono-Q) yielded more than 100 separate fractions (data not shown), making it difficult to test the activity of each fraction in bioassays.
To investigate the presence of AGP in our embryogenic cell suspension, ß-d-GlcY, known to specifically bind and inhibit AGP, was applied to CM before suspension initiation. As shown in Fig. 3a, addition of 25 μM ß-d-GlcY results in a prominent inhibition of the cell proliferation. In contrast, supplementing 1 and 2 mg of arabinogalactan from larch to FM demonstrated a stimulating effect on embryogenic cell proliferation (Fig. 3b). Weekly supplementing of arabinogalactan into suspensions did not show any difference of growth compared to arabinogalactan supply at suspension initiation only (data not shown). This points out to a secretion of AGP into the medium by growing cell suspension and to a saturation of the promoting effect of AG/AGP to cell proliferation. We confirmed the presence of AGP in CM by immuno-detection technique. Figure 4 shows the detection of AGP for all grapevine varieties tested. A strong signal was observed in zucchini CM.
Influence of ß-d-GlcY (a) and larch wood arabinogalactan (b) on growth of grapevine cell suspension starting with 0.5 g of embryogenic cells of Vitis vinifera cv. Tempranillo. a Addition of 25 μM of ß-d-GlcY penylglycoside filled triangle to CM at the beginning of culture. Filled square CM, open square FM, b initiation of embryogenic cell suspensions of Vitis vinifera cv. Tempranillo with 1 or 2 mg of arabinogalactan. Starting inoculum: 0,5 gram of callus fresh weight. Filled square CM, open square FM, filled circle treatment with 1 mg arabinogalactan, filled triangle treatment with 2 mg arabinogalactan. Data represent average of three independent replications, bars indicate SD values
Embryo maturation and plant regeneration
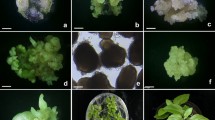
Established grapevine embryogenic cell suspensions were transferred to growth regulator free liquid medium to induce embryo formation and conversion into plantlets (Fig. 5). PEM showed the ability to produce globular embryo-like structures after removal of growth regulators. Heart and early torpedo stages were observed in suspensions after about 3 weeks of a daily fresh medium transfer procedure. A high capacity of synchronised somatic embryo development was observed 5–6 weeks after induction of regeneration. Elongated structures were counted and transferred onto solid medium (Fig. 5). The system produced thousands of somatic embryos per 50 ml of medium. Somatic embryo induction and conversion efficiency strongly differed between the different varieties (Table 2). Elongated embryos transferred onto fresh medium for regeneration, started to form green, shoot-like structures. Depending on the genotype, 11.8 to 36.8% of these embryos could finally be converted into intact plants. Generally, plantlets were obtained from all the varieties tested within about 3 months. However, plant regeneration from Kober 5 BB suspensions was achieved within 6 weeks and the time required for the complete plant development process starting from a proliferating embryogenic suspension was reduced to 8 weeks (Fig. 5).
Plant regeneration process from embryogenic cell suspensions of Kober 5BB. a Kober 5BB embryogenic cell suspension, b Embryo induction after cultivation in growth regulator free liquid medium for 2 weeks, c Mature and elongated embryos cultivated on solid medium for 10 days, d Somatic embryos picked separately and transferred to light (16 h light/8 h dark) for conversion, e Plantlets obtained from germinated embryos after about 1 month, f Regenerated grapevine plantlets transferred to greenhouse conditions
Discussion
The establishment and maintenance of friable embryogenic cell lines in suspension is difficult particularly for woody species. Previous reports on grapevine cell suspensions describe procedures to produce germinating embryos from few varieties (Coutos-Thévenot et al. 1992a; Jayasankar et al. 1999, 2001). A number of studies have been attempted to improve productivity of the system by antioxidant supply or by culturing at low cell density and following frequent subcultures (Coutos-Thévenot et al. 1992a; Perl et al. 1996; Maes et al. 1997). However, the way to initiate liquid medium-based culture is still the main critical point.
Simultaneous embryo development was observed in Fraxinus (Tonon et al. 2001) and soybean (Kim et al. 2004), whereas a mixture of asynchronised embryo stages has been reported in grapevine and switchgrass (Jayasankar et al. 1999; Gupta and Conger 1999). Our data demonstrate an improved procedure for the establishment of highly totipotent embryogenic suspensions of different grapevine varieties using conditioned culture medium. The system allows the production of homogeneous suspensions with a good potential for plant regeneration ability. In contrast to other reports on grapevine and potato (Jayasankar et al. 1999; Vargas et al. 2005) we obtained a high number of embryos suggesting our CM-procedure as being very efficient. Suspension-derived somatic embryos germinated precociously without undergoing a typical dormancy phase as previously reported by morphological and anatomical studies (Jayasankar et al. 1999, 2003).
In the present study, the use of conditioned medium strongly enhanced cell growth after transfer into liquid culture. Our results suggest a beneficial effect of conditioned medium at an intra-specific (Vitis vinifera) and inter-specific level (Vitis spp.) which so far has not been reported for grapevine. In both cases, conditioned medium components have a potential effect to increase grapevine PEM fresh weight. However, stimulation was completely lost by media autoclaving suggesting that secreted substances could have protein nature and thus are heat-denaturated. CM collected from liquid culture of other species (zucchini and parsley) could also perform a stimulating effect on grapevine suspensions. These results support the hypothesis that cell cultures secrete proteins into the medium which are likely genotype-independent and responsible for the promoting effect on the initiation of new cell suspensions.
Several reports indicated that plant glycoproteins secreted into the medium accompany the formation of embryogenic clusters. So far, three different extra-cellular proteins (EP1, EP2, EP3) have already been identified in carrot cell cultures that however do not show any effect on cell division (De Jong et al. 1992). But it’s also known, that secreted proteins from cell suspension such as arabinogalactan proteins (AGP) play a major role in cell proliferation and embryogenesis (Becraft et al. 1998; De Jong et al. 1992, 1993; Majewska-Sawka and Nothnagel 2000; Showalter 2001; van Hengel et al. 1998, 2001).
For that reason, we focused our investigation on the presence of AGP in the CM and its effect on grapevine embryogenic suspension. However, AGPs are secreted into the culture media at very low concentrations that do not exceed nanomolar ranges (van Hengel et al. 2001) making purification and direct analysis difficult. Addition of larch wood arabinogalactan (2 mg l-1) for initiation of grapevine embryogenic suspensions strongly stimulates cell proliferation and production of PEM. Continuous weekly addition of arabinogalactan did not increase the multiplication of the PEM. We suggest that after the initiation process, secretion of AGP from proliferating embryogenic cells is sufficient to continuously stimulate growth of suspension. Also Kaparakis and Alderson (2003) showed the increase of pepper callus mass and viability by AGP addition. This indicates that AGPs are involved in induction of cell division.
Due to their high content of polysaccharide chains (Mau et al. 1995), AGPs do not migrate in SDS-PAGE and thus cannot be detected by Western blot analysis. However, dot-blot immuno-detection by a specific antibody (MAC207) demonstrated presence of AGP in grapevine and zucchini CM. For zucchini Penell et al. (1989) and Showalter (2001) already reported detection of AGP by dot-blot immuno assays with this antibody. A clear correlation between the amount of AGP in conditioned medium and cell proliferation could not be identified. A high AGP content was detected for zucchini CM; however, the promoting effect of zucchini conditioned medium on grapevine cell suspension development was lower compared to conditioned medium originating from grapevine cell suspensions.
The results support the hypothesis that cell cultures secrete proteins into the medium which are likely genotype-independent and responsible for the promoting effect on the initiation and proliferation of cell suspensions. The amount of secreted AGP seems to depend on the structure of PEM leading to the suggestion that fine material allows more surface contact with liquid medium and provide better cell wall protein release.
Application of ß-d-GlcY resulted in a strong inhibition of cell proliferation, indicating the importance of AGP for suspension initiation. ß-d-GlcY binds selectively to arabinogalactan side chain by a mechanism that is not fully understood (Jermyn 1978; Guan and Nothnagel 2004) resulting in precipitation of an AGP-ß-d-GlcY complex. The low proliferation of grapevine cell suspensions in the presence of ß-d-GlcY is hypothesised to be due to the perturbation of AGP. Altogether it confirms that extra-cellular AGP in the conditioned media are somehow involved in the induction of grapevine cell proliferation. These are the first results detecting AGP in grapevine embryogenic cell suspension and demonstrating their involvement in the cell proliferation process.
In rose (Serpe and Nothnagel 1994), carrot (Willats et al. 1999), sugar beet (Butowt et al. 1999) and tobacco (Qin and Zhao 2006) cell cultures, application of ß-d-GlcY resulted in the arrest of growth and cell division. Similarly, experiments showed that inactivation of AGP alters the progress of cell division and cell wall expansion in undifferentiated cell cultures (Kimberly and Nothnagel 1997; Majewska-Sawka and Nothnagel 2000). Under moderate treatment with ß-d-GlcY, the inhibition of proliferation is reversible after washing the cells and growth recovery could be partially achieved (Kimberly and Nothnagel 1997; Majewska-Sawka and Nothnagel 2000). In Arabidopsis cells, higher concentrations of ß-d-GlcY induced programmed cell death within 3 days (Majewska-Sawka and Nothnagel 2000).
Glycoproteins splitted by cell wall enzymes might generate molecules like lipochito-oligosaccharides (LCO) homologous with Rhizobium-Nod factors (De Jong et al. 1993; Dyachock et al. 2002). These small molecules could stimulate proliferation of PEM as described in several reports (Oropeza et al. 2001; van Hengel et al. 2002; Matthys-Rochon 2005). van Hengel et al. (2002) suggested that AGPs can be activated by endochitinase and may give rise to several molecules implicated in signalling pathway and controlling proliferation of plant cells. AGPs are substrate of enzymes to produce some components with hormone-like activity or act as a messenger for signal transduction which may contribute in development regulation and cell division (for review, see Showalter 2001).
To initiate embryo maturation and germination, beside the use of hormone free media, a frequent medium change resulted in the highest rate of regenerating embryos. As already reported by Coutos-Thevenot et al. (1992b), accumulation of secreted proteins in the suspension medium completely blocks embryo differentiation. Obviously, with the daily transfer procedure into fresh medium, secreted proteins were diluted to a non-inhibiting concentration, resulting in improved embryo elongation and plant regeneration efficiency. From our experiments it is not clear, if AGP inducing intensive proliferation of embryogenic grapevine suspensions, become inhibitors of embryo differentiation during regeneration steps. However, it seems that cell proliferation and differentiation appear as two distinct morphogenetic pathways which require different environmental conditions. Previous studies proposed that AGPs stimulate somatic embryogenesis (Chapman et al. 2000; Majewska-Sawka and Nothnagel 2000; van Hengel et al. 2002).
Grapevine embryogenic cell suspensions are a useful tool for regeneration but remained difficult to establish. Here we describe a standard procedure, allowing the establishment of embryogenic suspension of grapevine. The improvement of our protocol is related to the fast establishment of productive suspensions yielding mature somatic embryos with competence for plant regeneration. The results of the current study suggest an important role of conditioned culture medium with a significant effect of extra-cellular proteins to promote cell division and effective multiplication with plant regeneration ability. Evidence of the involvement of AGP in the control of cell proliferation is given. However, the molecular mechanism involved remains unknown. Identification of AGP and other secreted components accumulated in conditioned media are currently in process, and may give a better understanding of cell proliferation and differentiation of grapevine embryogenic cell cultures.
Abbreviations
- AGPs:
-
Arabinogalactan-proteins
- BAP:
-
6-benzylaminopurine
- ß-D-Glc-Y:
-
ß-d-Glucosyl Yariv phenylglycoside
- CEPF:
-
Concentrated extra-cellular protein fraction
- 2,4-D:
-
2,4-Dichlorophenoxyacetic acid
- EP:
-
Extra-cellular protein
- NOA:
-
Naphthoxyacetic acid
- MES:
-
2-(N-Morpholino) ethanesulfonic acid
- FM:
-
Fresh medium
- CM:
-
Conditioned medium
- PEM:
-
Pro-embryogenic masses
- PCV:
-
Packed cell volume
References
Becraft PW (1998) Receptor kinases in plant development. Trends Plant Sci 3:384–388
Burnette WN (1981) “Western blotting”: electrophoretic transfer of proteins from sodium dodecyl sulfate-polyacrylamide gels to unmodified nitrocellulose and radiographic detection with antibody and radioiodinated protein A. Anal Biochem 112:195–203
Butowt R, Niklas A, Rodriguez-Garcia MI, Majewska-Sawka A (1999) Involvement of JIM13- and JIM8-responsive carbohydrate epitopes in early stages of cell formation. J Plant Res 112:107–116
Chapman A, Blervacq AS, Vasseur J, Hilbert JL (2000) Arabinogalactan-proteins in Cichorium somatic embryogenesis: effect of beta-glucosyl Yariv reagent and epitope localisation during embryo development. Planta 211:305–314
Coutos-Thévenot P, Goebel-Tourand I, Mauro MC, Jouanneau JP, Boulay M, Deloire A, Guern J (1992a Somatic embryogenesis from grapevine cells. I-Improvement of embryo development by changes in culture conditions. Plant Cell Tiss Org 29:125–133
Coutos-Thévenot P, Maes O, Jouenne T, Mauro MC, Jouanneau JP, Boulay M, Deloire A, Guern J (1992b Extracellular protein patterns of grapevine cell suspensions in embryogenic and non-embryogenic situations. Plant Sci 86:137–145
De Jong AJ, Cordewener J, Lo Schiavo F, Terzi M, Vanderkerckhove J, Van Kammen A, De Vries SC (1992) A carrot somatic embryo mutant is rescued by chitinase. Plant Cell 4:425–433
De Jong AJ, Heidstra R, Spaink HP, Vartog MV, Meijer EA, Hendriks T, Schiavo FL, Terzi M, Bisseling T, Van Kammen A, De Vries SC (1993) Rhizobium lipooligosaccharides rescue a carrot somatic embryo mutant. Plant Cell 5:615–620
Dussert S, Mauro MC, Engelmann F (1992) Cryopreservation of grape embryogenic cell suspensions. II-Influence of post-thaw culture conditions and application to differents strains. Cryo Lett 13:15–22
Dyachock JV, Wiweger M, Kenne L, Von Arnold S (2002) Endogenous Nod-factor-like signal molecules promote early somatic embryo development in Norway spruce. Plant Physiol 128:523–533
Guan Y, Nothnagel EA. (2004) Binding of Arabinogalactan proteins by Yariv phenylglycoside triggers wound-like responses in Arabidopsis cell cultures. Plant Physiol 135(3):1346–1366
Gupta DS, Conger BV (1999) Somatic embryogenesis and plant regeneration from suspension cultures of Switchgrass. Crop Sci 39:243–247
Hebert D, Kikkert JR, Smith FD, Reisch BI (1993) Optimization of biolistic transformation of embryogenic grape cell suspensions. Plant Cell Rep 12:585–589
Immerzeel P, Schols HA, Voragen AGJ, De Vries S (2004) Different arabinogalactan proteins are present in carrot (Daucus carota) cell culture medium and in seeds. Physiol Plant 122:181–190
Immerzeel P, Eppink MM, De Vries S, Schols HA, Voragen AGJ (2006) Carrot arabinogalactan proteins are interlinked with pectins. Physiol Plant 128:18–27
Jayasankar S, Gray DJ, Litz RE (1999) High-efficiency somatic embryogenesis and plant regeneration from suspension cultures of grapevine. Plant Cell Rep 18:533–537
Jayasankar S, Van Aman M, Li Z, Gray DJ (2001) Direct seeding of grapevine somatic embryos and regeneration of plants. In Vitro Cell Dev Plant 37:476–479
Jayasankar S, Bonbada BR, Li Z, Gray DJ (2003) Comparative anatomy and morphology of Vitis vinifera (Vitaceae) somatic embryos from solid- and liquid-culture-derived proembryogenic masses. Am J Bot 90:973–979
Jermyn MA (1978) Comparative specificity of concanavalin A and the ß-lectins. Aust J Plant Physiol 5:687–696
Kaparakis G, Alderson G (2003) Enhancement of in vitro cell proliferation of pepper (Capsicum annuum L.) by pluronic F-68, haemoglobin and arabinogalactan proteins. J Hortic Sci Biotech 78:647–649
Kikkert JR, Hubert D, Wallace PG, Streim MJ, Reisch BI (1996) Transgenic plantlets of “Chancellor” grapevine Vitis sp. from biolistic transformation of embryogenic cell suspensions. Plant Cell Rep 15:311–316
Kim YJ, Park T, Kim HS, Such SK, Kim HS, Yun SJ (2004) Effective multiplication of somatic embryos using suspension culture and regeneration in soybean. J Plant Biotech 6:91–96
Kimberly JL, Nothnagel EA (1997) Cell surface arabinogalactan-proteins and their relation to cell proliferation and viability. Protoplasma 196:87–98
Maes O, Coutos-Thevenot P, Jouenne T, Boulay M, Guern J (1997) Influence of extracellular proteins, proteases and protease inhibitors on grapevine somatic embryogenesis. Plant Cell Tiss Org 50:97–105
Majewska-Sawka A, Nothnagel E (2000) The multiple roles of arabinogalactan proteins in plant development. Plant Physiol 122:3–9
Martinelli L, Gribaudo I (2001) Somatic embryogenesis in grapevine. In: Roubelakis-Angelakis KA (ed) Molecular biology and biotechnology of the grapevine. Kluwer, Dordrecht, pp 327–351
Massonneau A, Coronado MJ, Audran A, Bagniewska A, Mol R, Testillano PS, Goralski G, Dumas C, Rieueno MC, Matthys-Rochon E (2005) Multicellular structures developing during maize microspore culture express endosperm and embryo-specific genes and show different embryogenic potentialities. Eur J Cell Biol 84:663–675
Matthys-Rochon E (2005) Secreted molecules and their role in embryo formation in plants: a mini review. Acta Biol Cracov Bot 47:23–29
Mau SL, Chen CG, Pu ZY, Moritz RL, Simpson RJ, Bacic A, Clarke AE (1995) Molecular cloning of cDNAs encoding the protein backbones of arabinogalactan-proteins from the filtrate of suspension-cultured cells of Pyrus communis and Nicotiana alata. Plant J 8:269–281
Mauro MC, Nef C, Fallot J (1986) Stimulation of somatic embryogenesis and plant regeneration from anther culture of Vitis vinifera cv. Cabernet-Sauvignon. Plant Cell Rep 5:377–380
Nitsch JP, Nitsch C (1969) Haploid plants from pollen grains. Science 163:85–87
Oropeza M, Marcano AK, Garcia ED (2001) Proteins related with embryogenic potential in callus and cell suspensions of sugarcane (Saccharum sp.). In Vitro Cell Dev Plant 37:211–216
Pennell RI, Knox JP, Scofield GN, Selvendran RR, Roberts K (1989) A family of abundant plasma membrane-associated glycoproteins related to arabinogalactan proteins is unique to flowering plants. J Cell Biol 108:1967–1977
Perl A, Lotan O, Abu-Abied M, Holland D (1996) Establishment of an Agrobacterium-mediated transformation system for grape (Vitis vinifera L.): the role of antioxidants during grape-Agrobacterium interactions. Nature Biotechnol 14:624–628
Qin Y, Zhao J (2006) Localization of arabinogalactan proteins in eggs cells, zygotes and two-celled proembryos and effects of ß-d-glucosyl Yariv reagent on egg cell fertilization and zygote division in Nicotiana tabacum. J Exp Bot 57:2061–2074
Rajasekaran K, Mullins MG (1979) Embryos and plantlets from cultured anthers of hybrid grapevines. J Exp Bot 30:399–407
Reustle G, Harst M, Alleweldt G (1995) Plant regeneration of grapevine (Vitis sp) protoplasts isolated from embryogenic tissue. Plant Cell Rep 15:238–241
Serpe MD, Nothnagel EA (1994) Effects of Yariv phenylglycosides on Rosa cell suspensions: evidence for the involvement of arabinogalactan-proteins in cell proliferation. Planta 193:542–550
Showalter A (2001) Arabinogalactan-proteins: structure, expression and function. Cell Mol Life Sci 1399–1417
Stamp JA, Meredith CP (1988) Somatic embryogenesis from leaves and anthers of grapevine. Scientia Hort 35:235–250
Taylor NJ, Edwards M, Kiernan RJ, Davey CD, Blakesley D, Henshaw GG (1996) Development of friable embryogenic callus and embryogenic suspension culture systems in cassava (Manihot esculenta Crantz). Nature Biotechnol 14:726–730
Tonon G, Berardi G, Rossi C, Bagnaresi U (2001) Synchronized somatic embryo development in embryogenic suspensions of Fraxinus angustifolia. In Vitro Cell Dev Plant 37:462–465
van Engelen FA, De Vries SC (1992) Extracellular proteins in plant embryogenesis. Trends Genet 8:66–70
van Hengel AJ, Gruzzo F, Van Kammen A, De Vries SC (1998) Expression pattern of the carrot EP3 endochitinase genes in suspension cultures and in developing seeds. Plant Physiol 117:43–53
van Hengel AJ, Tadesse Z, Immerzeel P, Schols H, Van Kammen A, De Vries SC (2001) N-acetylglucosamine and glucosamine-containing arabinogalactan proteins control somatic embryogenesis. Plant Physiol 125:1880–1890
van Hengel AJ, Van Kammen A, De Vries SC (2002) A relationship between seed development, arabinogalactan-proteins and the AGP mediated promotion of somatic embryogenesis. Physiol Plant 114:637–641
Vargas TE, De Garcia E, Oroperza M (2005) Somatic embryogenesis in Solanum tuberosum from cell suspension cultures: histological analysis and extracellular protein patterns. J Plant Physiol 162:449–456
Vidal JR, Kikkert JR, Wallace PG, Reisch BI (2003) High-efficiency biolistic transformation and regeneration of “Chardonnay” (Vitis vinifera L.) containing npt-II and antimicrobial peptide genes. Plant Cell Rep 22:252–260
Wang Q, Mawassi M, Sahar N, Li Ping, Colova-Tsolova V., Gafny R, Sela I, Tanne E, Perl A (2004) Cryopreservation of grapevine (Vitis spp.) embryogenic cell suspensions by encapsulation-vitrification. Plant Cell Tiss Org 77:267–275
Willats GT, Steele-King CG, Marcus SE, Knox JP (1999) Side chains of pectic polysaccharides are regulated in relation to cell proliferation and cell differentiation. Plant J 20:619–628
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by R. Reski.
Rights and permissions
About this article
Cite this article
Ben Amar, A., Cobanov, P., Boonrod, K. et al. Efficient procedure for grapevine embryogenic suspension establishment and plant regeneration: role of conditioned medium for cell proliferation. Plant Cell Rep 26, 1439–1447 (2007). https://doi.org/10.1007/s00299-007-0341-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-007-0341-8