Abstract
Juvenile onset systemic sclerosis (JoSSc) is a rare disease, and there are no studies focusing in bone mineral density and biochemical bone parameters. Ten consecutive patients with JoSSc and 10 controls gender, age, menarche age, and physical activity matched were selected. Clinical data were obtained at the medical visit and chart review. Laboratorial analysis included autoantibodies, 25-hydroxyvitamin D (25OHD), intact parathyroid hormone, calcium, phosphorus, alkaline phosphatase and albumin sera levels. Bone mineral density was analyzed by dual-energy X-ray absorptiometry, and bone mineral apparent density (BMAD) was calculated. A lower BMAD in femoral neck (0.294 ± 0.060 vs. 0.395 ± 0.048 g/cm3, P = 0.001) and total femur (0.134 ± 0.021 vs. 0.171 ± 0.022 g/cm3, P = 0.002) was observed in JoSSc compared to controls. Likewise, a trend to lower BMAD in lumbar spine (0.117 ± 0.013 vs. 0.119 ± 0.012 g/cm3, P = 0.06) was also found in these patients. Serum levels of 25OHD were significantly lower in JoSSc compared to controls (18.1 ± 6.4 vs. 25.1 ± 6.6 ng/mL, P = 0.04), and all patients had vitamin D insufficiency (<20 ng/mL) compared to 40% of controls (P = 0.01). All other biochemical parameters were within normal range and alike in both groups. BMAD in femoral neck and total femur was correlated with 25OHD levels in JoSSc (r = 0.82, P = 0.004; r = 0.707, P = 0.02; respectively). We have identified a remarkable high prevalence of 25OHD insufficiency in JoSSc. Its correlation with hip BMAD suggests a causal effect and reinforces the need to incorporate this hormone evaluation in this disease management.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Systemic sclerosis (SSc) is a chronic disease characterized by increased synthesis and deposition of collagen in skin and connective tissue, vascular alteration and immunological disturbances. Although uncommon in children and adolescent, it is one of the most challenging rheumatic conditions in pediatric rheumatology. The overall outcome of juvenile onset systemic sclerosis seems to be better than adults [1], with a more favorable survival even after 20-year disease course.
We have recently demonstrated that adult SSc itself is a risk factor for osteoporosis [2]. In fact, previous studies had observed a relationship between low bone density and scleroderma [3–5]. This risk may be even greater in JoSSc due to the longer survival of most of these patients.
Vitamin D insufficiency is recognized as one of the important causal factor for low bone mass, since it has well-known control function of calcium and phosphorus metabolism, bone formation and mineralization. Of interest, vitamin D has also immunomodulatory properties with a potential benefit for autoimmune disease [6]. In fact, a high prevalence of vitamin D deficiency has been reported in rheumatologic outpatients [7–13], including autoimmune diseases such as systemic lupus erythematosus [8–12] and arthritis rheumatoid [9, 10, 13, 14]. Recently, high frequency of low levels of vitamin D and secondary hyperparathyroidism was described in Mediterranean SSc patients partially explained by traditional dress habits and low exposure to sun [15].
In contrast to adult SSc, no data are available in the literature regarding bone metabolism analysis in juvenile onset SSc (JoSSc), including vitamin D status. Therefore, the authors analyzed its levels and also evaluated its possible association with clinical and laboratorial parameters in JoSSc.
Materials and methods
Patients
Ten consecutive JoSSc who met LeRoy and Medsger [16] criteria and ten gender, physical activity, chronological and menarche age matched healthy children were selected for the study. All patients were followed in the Division of Rheumatology, School of Medicine, and University of São Paulo. Exclusion criteria were secondary causes of low bone mass such as clinical history of poor absorption, renal or hepatic insufficiency, hyperthyroidism or hypothyroidism, smoking and glucocorticoid use.
Clinical data including gender, ethnicity, age at onset and disease duration (JoSSc patients), CHAQ score [17], number of limited joints, Rodnan score, treatment (cyclophosphamide or glucocorticoids), risk factors for osteoporosis (familial history of osteoporosis and/or fractures, physical activity, sun exposure, personal fracture history, menstrual cycles, menarche age) were obtained by medical visit and chart review.
Physical activity was measured in patients and controls using 60-min screening measure for moderate to vigorous physical activity (MVPA) [18]. Accumulation of 60 min of MVPA corresponds with recent recommendations for youth physical activity that are important for healthy bone [19]. Sun exposure was quantified in number of minutes per week that the patient or healthy control was exposure.
Body mass index (BMI) was calculated by measuring the weight and height of each patient and control during the interview.
All patients and controls or their legal guardians gave their written informed consent, and the study was approved by the Ethics Committee of the University Hospital.
Laboratory evaluation
The serum concentrations of calcium, phosphorus and alkaline phosphatase were determined according to standardized methods.
Blood samples for 25OHD and iPTH were collected for patients and controls during summer, under fasting, between 8 and 10 a.m., stored at −70°C and analyzed at the same time. The serum concentration of 25OHD was used to measure the vitamin D reserves, using a radioimmunoassay technique (DiaSorin, Stillwater, MN, USA). The intra and inter-assay variation coefficients in the laboratory were 16.6 and 22.6%, respectively [20]. The definition of vitamin D insufficiency was based on the suggested cutoff for healthy schoolchildren, 25OHD <20 ng/mL [21]. Intact parathyroid hormone (iPTH) serum concentrations were measured by immunoradiometric assay (ELSA-PTH, CIS bio international, France), with reference variations ranging from 11 to 62 pg/mL.
Autoantibodies including anti-nuclear antibodies (indirect immunofluorescence with Hep-2 cell), anticentromere (indirect immunofluorescence with Hep-2 cell) and Scl70 (immunoblotting) were tested for both groups.
Dual-energy X-ray absorptiometry (DXA)
Bone mineral densities were determined by dual-energy X-ray absorptiometry using a Hologic Discovery densitometer. Bone mineral density (BMD; g/cm2) and bone area (BA; cm2) were calculated in lumbar spine (L1-L4), femoral neck and total femur. In order to minimize the confounding effect of skeletal size on DXA measures, bone mineral apparent density (BMAD; g/cm3) was calculated using the volumetric analysis. The calculation of volumetric density was obtained by dividing the bone mineral density in lumbar spine, femoral neck and total femur by the square root of their respective lumbar spinal, femoral neck and total femur area [BMAD = BMD/√BA (where BA = bone area of the region to be analyzed)] [22–24]. Coefficients of variation for bone mineral density in our laboratory were 0.7% for lumbar spine (L1-L4), 1.5% for femoral neck, 1.3% for total femur [20]. Z-score less than −2 SD in lumbar spine, femoral neck and total femur were defined as low bone mass for chronological age [22].
Statistical analysis
The results are reported as mean standard deviation and percentage. The two groups were compared by the Student t-test. The percentage of each variable was compared between groups by Fisher’s exact test. Pearson correlation was used to analyze the BMAD in lumbar spine, femoral neck, total femur with following variables: disease duration, Rodnan score, presence of autoantibodies (Scl70 and anticentromere), BMI, menarche age, physical activity and 25OHD serum level. Values of P < 0.05 were considered to be significant.
Results
As expected by the matching design of the study, chronological age (P = 0.81), gender (P = 1.00), sun exposure (0.11) and menarche age (P = 0.62) were comparable in patients and controls. Other relevant factors associated with bone mass were also alike in both groups: race (P = 1.00), BMI (P = 0.07), regular menstrual cycle (P = 1.00) and familial history of osteoporosis/fracture (P = 1.00), except weight (46.4 ± 8.5 vs. 56.2 ± 3.5 kg, P = 0.01) (Table 1). Physical activity, measure by MVPA (moderate and vigorous physical activity) was similar in patients and controls (0.56 ± 0.9 vs. 0.78 ± 1.23, P = 0.68) (Table 1).
Disease was classified according to LeRoy as diffuse SSc in seven and limited in three patients [16]. The mean Rodnan score was 21.9 ± 15.5. The mean disease duration for all patients was 7.2 ± 3.5 years, the mean of CHAQ scores were 0.88 ± 0.93, and the number of limited joints were 17.5 ± 18.25.
Two patients had anti-topoisomerase (Scl70) and one anticentromere antibody, and controls were uniformly negative. The frequency of current and previous use of was 30% d-penicillamine, 30% methotrexate and 40% other immunosuppressors (azathioprine or cyclophosphamide).
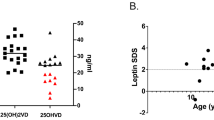
Biochemical bone metabolism evaluation revealed that vitamin D levels were lower in JoSSc patient when compared to control group (18.1 ± 6.4 vs. 25.1 ± 6.6 ng/mL; P = 0.04) in spite of calcium (P = 0.69), phosphorus (P = 0.12), alkaline phosphatase (0.11) and iPTH (0.33) sera levels within normal range and alike in both groups (Table 2).
The BMAD was significantly decreased in JoSSc compared to controls in femoral neck (0.294 ± 0.060 vs. 0.351 ± 0.048 g/cm3, P = 0.001) and total femur (0.134 ± 0.021 vs. 0.171 ± 0.022 g/cm3, P = 0.002). A trend to lower BMAD in lumbar spine (0.117 ± 0.013 vs. 0.119 ± 0.012 g/cm3, P = 0.06) was also found in these patients (Table 3).
Low bone density for chronological age (Z score < −2.0 SD) was observed in lumbar spine in two (20%) patients, femoral neck in four (40%) and total femur in two (20%) as showed in Table 3. None of the controls had bone density bellow the expected range for age in the three sites analyzed.
Pearson correlation analysis revealed that BMAD in femoral neck and total femur was correlated to 25OHD (r = 0.82, P = 0.004; r = 0.707, P = 0.02). In contrast, no correlation was observed between BMAD in femoral neck and total femur with weight (r = 0.06, P = 0.87; r = 0.42, P = 0.23, respectively).
Discussion
We have identified that JoSSc patients have a lower BMAD and lower 25OHD levels compared to controls. The status of this hormone seems to be an important contributing factor for cortical bone mass in this disease.
In the present study, the careful matching for age and gender is an essential factor to interpret the results. It is well known that age has a major impact not only in bone mass loss but also in acquisition. In fact, bone accrual in adolescence is a major determinant of peak bone mass and risk of osteoporotic fractures later in life [23]. The pathogenesis of bone fragility is also distinct in male and female gender since boys have higher bone mineral content than girls [24]. The homogenous distribution of ethnicity between groups is also relevant since there is a known disparity in BMD and osteoporosis fractures between white and black race.
In addition, there are strong evidences that estrogen deficiency is a risk factor for later development of osteoporosis and hip fracture [25, 26]. The comparable frequency of menstrual irregularities and age of menarche is therefore important parameters to evaluate bone health [27].
With regard to physical activity, it has been demonstrated a relevant beneficial effect with a significant increase in bone mineral content during the adolescent years [28] which is maintained into young adulthood [29]. Thus, we have matched this factor in order to minimize the influence of this variable.
Bone mineral apparent density, a volumetric three-dimensional approximation of bone density was calculated to diminish the effect of skeletal size on DXA measurement. This method appears to offer advantages to areal BMD technique due to intrinsic biases caused by bone thickness differences [20, 30, 31].
To our knowledge, this is the first study to describe a universal vitamin D deficiency in JoSSc patients. We have used serum concentration of 25-OH vitamin D, since it is the best marker of total body reserve of this hormone, and the cutoff of <20 ng/mL established for adults to define deficiency was also validated for healthy children [21].
Small bowel involvement manifesting with malabsorption may develop in adult and pediatric SSc patients during the disease course [1]. This clinical complication does not seem to have contributed to the finding of low vitamin D in our patients, since none of them referred diarrhea or weight loss and all of them had normal albumin levels.
Low sun exposure may explain in part our observation in both groups. In spite of living in a tropical area with reasonable sunlight availability, vitamin D deficiency is not uncommon in our country [32], particularly in those with a poorer physical health. In this regard, it has been demonstrated that SSc adversely affects quality of life and functional status in systemic sclerosis compared to other rheumatic diseases [33].
Another cause of vitamin D deficiency in patients and controls is the fact that in our country, food is not enriched with this hormone, vitamin supplementation is uncommon, and a diet high in fish oil is not part of the traditional Brazilian food. The seasonal effect was excluded since all sera during summer time.
We have identified in JoSSc patients a lower bone mineral apparent density compared to controls, particularly in cortical bone and a tendency in trabecular bone. This finding is similar to previous studies in adult SSc; however, none have evaluated concomitantly the level of vitamin D [2–5, 15]. Other biochemical parameters within the normal range have excluded additional causal factors for this bone metabolic disturbance such as renal and hepatic insufficiency, osteomalacia and malabsorption.
Vitamin D levels are directly related to bone mineral density in both gender and in diverse races with a maximum density accomplished with levels of 40 ng/mL or more [34]. The finding of a direct correlation of bone mineral apparent density in hip with this hormone in JoSSc patients emphasizes its possible role in this metabolic disturbance. In contrast, weight, a known relevant factor for BMD [35], did not seem to contribute to this finding in this subgroup of patients with a lack of correlation in hip BMD. Likewise, the absence of correlation of 25OHD with lumbar site is probably due to the small sample analyzed, or to a preferential involvement of hip in JoSSc. In fact, we have demonstrated previously a low bone mineral apparent density in juvenile dermatomyositis in hip and not in lumbar spine [20]. Likewise, rheumatoid arthritis followed for up 20 years also had a predominant decrease in BMD of femoral neck [36].
In summary, this is the first study to identify a remarkable high prevalence of vitamin D insufficiency in JoSSc. The 25OHD level correlation with hip BMAD suggests a causal effect and reinforces the need to incorporate this hormone evaluation in the optimal management of this disease.
References
Foeldvari I (2008) Update on pediatric systemic sclerosis: similarities and differences from adult disease. Curr Opin Rheumatol 20:608–612
Souza RB, Borges CT, Takayama L, Aldrighi JM, Pereira RM (2006) Systemic sclerosis and bone loss: the role of the disease and body composition. Scand J Rheumatol 35:384–387
Carbone L, Tylavsky F, Wan J, McKown K, Cheng S (1999) Bone mineral density in scleroderma. Rheumatology (Oxford) 37:1–2
Di Munno O, Mazantini M, Massel P, Ferdeghini M, Petaro N, Latorraca A, Ferri C (1995) Reduced bone mass and normal calcium metabolism in systemic sclerosis with and without calcinosis. Clin Rheumatol 14:407–412
La Montagna G, Vatti M, Valenti G, Tirri G (1991) Osteopenia in systemic sclerosis evidence of a participating role of earlier menopause. Clin Rheumatol 10:18–22
Szodoray P, Nakken B, Gaal J, Jonsson R, Szegedi A, Zold E, Bodolay E (2008) The complex role of vitamin D in autoimmune diseases. Scand J Immunol 68:261–269
Mouyis M, Ostor AJK, Crisp AJ, Ginawi A, Halsall DJ, Shenker N, Poole KE (2008) Hypovitaminosis D among rheumatologic outpatients in clinical practice. Rheumatology 47:1348–1351
Kamen D, Aranow C (2008) Vitamin D in systemic lupus erythematosus. Curr Opin Rheumatol 20:532–537
Costenbader KH, Feskanich D, Holmes M, Karlson EW, Benito-Garcia E (2008) Vitamin D intake and risks of systemic lupus erythematosus and rheumatoid arthritis in women. Ann Rheum Dis 67:530–535
Adorini L, Penna G (2008) Control of autoimmune diseases by the vitamin D endocrine system. Nat Clin Pract Rheumatol 4:404–412
Cutolo M, Otsa K (2008) Review: vitamin D, immunity and lupus. Lupus 17:6–10
Kamen DL, Cooper GS, Bouali H, Shaftman SR, Hollis BW, Gilkeson GS (2006) Vitamin D deficiency in systemic lupus erythematosus. Autoimmun Rev 5:114–117
Merlino LA, Curtis J, Mikuls TR, Cerhan JR, Criswell LA, Saag KG, Iowa Women’s Health Study (2004) Vitamin D intake is inversely associated with rheumatoid arthritis: results from the Iowa Women’s Health Study. Arthritis Rheum 50:72–77
Cutolo M, Otsa K, Uprus M, Paolino S, Seriolo B (2007) Vitamin D in rheumatoid arthritis. Autoimmun Rev 7:59–64
Braun-Moscovici Y, Furst DE, Markovits D, Rozin A, Clements PJ, Nahir AM, Balbir-Gurman A (2008) Vitamin D, parathyroid hormone, and acroosteolysis in systemic sclerosis. J Rheumatol 35:2201–2205
LeRoy EC, Medsger TA (2001) Criteria for the classification of early systemic sclerosis. J Rheumatol 28:1573–1575
Len C, Goldenberg J, Ferraz MB, Hilário MO, Oliveira LM, Sacchetti S (1994) Crosscultural reliability of the Childhood Health Assessment Questionnaire. J Rheumatol 21:2349–2352
Prochaska JJ, Sallis JF, Long B (2001) A physical activity screening measure for use with adolescents in primary care. Arch Pediatr Adolesc Med 155:554–559
Council for Physical Education for Children (1998) Physical activity for children: a statement of guidelines. National Association for Sport and Physical Education, Reston, VA
Santiago RA, Silva CA, Caparbo VF, Sallum AM, Pereira RM (2008) Bone mineral apparent density in juvenile dermatomyositis: the role of lean body mass and glucocorticoid use. Scand J Rheumatol 37:40–47
El-Hajj Fuleihan G, Nabulsi M, Choucair M, Salamoun M, Hajj Shahine C, Kizirian A, Tannous R (2001) Hypovitaminosis D in healthy schoolchildren. Pediatrics 107:E53
Khan AA, Bachrach L, Brown JP, Hanley DA, Josse RG, Kendler DL, Leib ES, Lentle BC, Leslie WD, Lewiecki EM, Miller PD, Nicholson RL, O’Brien C, Olszynski WP, Theriault MY, Watts NB (2004) Standards and guidelines for performing central dual-energy X-ray absorptiometry in premenopausal women, men, and children. J Clin Densitom 7:51–64
Cooper C, Harvey N, Javaid K, Hanson M, Dennison E (2008) Growth and bone development. Nestle Nutr Workshop Ser Pediatr Program 61:53–68
Cooper C, Westlake S, Harvey N, Javaid K, Dennison E, Hanson M (2006) Review: developmental origins of osteoporotic fracture. Osteoporos Int 17:337–347
Seeman E (2002) Pathogenesis of bone fragility in women and men. Lancet 25(359):1841–1850
Kamel HK (2006) Postmenopausal osteoporosis: etiology, current diagnostic strategies, and nonprescription interventions. J Manag Care Pharm 12(6 Suppl A):S4–S9
Popat VB, Prodanov T, Calis KA, Nelson LM (2008) The menstrual cycle: a biological marker of general health in adolescents. Ann N Y Acad Sci 1135:43–51
Bailey DA, McKay HA, Mirwald RL, Crocker PR, Faulkner RA (1999) A six-year longitudinal study of the relationship of physical activity to bone mineral accrual in growing children: the university of Saskatchewan bone mineral accrual study. J Bone Miner Res 14:1672–1679
Baxter-Jones AD, Kontulainen SA, Faulkner RA, Bailey DA (2008) A longitudinal study of the relationship of physical activity to bone mineral accrual from adolescence to young adulthood. Bone 43:1101–1107
Carter DR, Bouxsein ML, Marcus R (1992) New approaches for interpreting projected bone densitometry data. J Bone Miner Res 7:137–145
Eriksson S, Mellström D, Strandvik B (2009) Volumetric bone mineral density is an important tool when interpreting bone mineralization in healthy children. Acta Paediatr 98:374–379
Saraiva GL, Cendoroglo MS, Ramos LR, Araújo LM, Vieira JG, Kunii I, Hayashi LF, Corrêa MP, Lazaretti-Castro M (2005) Influence of ultraviolet radiation on the production of 25 hydroxyvitamin D in the elderly population in the city of São Paulo (23 degrees 34′S), Brazil. Osteoporos Int 16:1649–1654
Johnson SR, Glaman DD, Schentag CT, Lee P (2006) Quality of life and functional status in systemic sclerosis compared to other rheumatic diseases. J Rheumatol 33:1117–1122
Bischoff-Ferrari HA, Giovannucci E, Willett WC, Dietrich T, Dawson-Hughes B (2006) Estimation of optimal serum concentrations of 25-hydroxyvitamin D for multiple health outcomes. Am J Clin Nutr 84:18–28 (Erratum in: Am J Clin Nutr 84:1253)
Timpson NJ, Sayers A, Davey-Smith G, Tobias JH (2009) How does body fat influence bone mass in childhood? A Mendelian randomization approach. J Bone Miner Res 24:522–533
Hämäläinen H, Kautiainen H, Kaarela K, Kotaniemi A (2005) The development of bone mineral density and the occurrence of osteoporosis from 15 to 20 years of disease onset in patients with rheumatoid arthritis. Clin Exp Rheumatol 23:193–198
Acknowledgments
This work was supported by grants of Conselho Nacional de Desenvolvimento Científico e Tecnológico [300559/2009-7 to R.M.R.P., 305468/2006-5 to E.B.] and Federico Foundation to E.B.
Conflict of interest statement
All authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shinjo, S.K., Bonfá, E., de Falco Caparbo, V. et al. Low bone mass in juvenile onset sclerosis systemic: the possible role for 25-hydroxyvitamin D insufficiency. Rheumatol Int 31, 1075–1080 (2011). https://doi.org/10.1007/s00296-010-1421-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-010-1421-6