Abstract
Antibodies to cyclic citrullinated peptides (anti-CCP) are highly specific for the diagnosis of rheumatoid arthritis (RA) with a marginal increased prediction of the disease. In this study, we aimed to investigate the relation of the presence of anti-CCP with clinical manifestations and disease activity in a cohort of RA patients. A total of 61 RA patients were included in this study. Data of disease-related parameters such as duration of disease, medications, degree of pain (visual analog scale, VAS), disease activity score 28 (DAS-28) and health assessment questionnaire (HAQ) were recorded. Laboratory workup included erythrocyte sedimentation rate (ESR), plasma C-reactive protein (CRP), rheumatoid factor (RF), anti-CCP, complete blood count and anemia parameters. Anti-CCP positivity was associated with higher scores of DAS-28, longer duration of morning stiffness, serum RF positivity and low levels of serum ferritin, while it was not associated with disease duration, VAS, HAQ, ESR, CRP and hemoglobin.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rheumatoid arthritis (RA) is the most common autoimmune disease affecting approximately 1% of the world’s population [1]. Chronic inflammation of the involved joints results in progressive loss of function, which, together with the extraarticular manifestations and adverse events of therapy, may increase morbidity and mortality of patients with RA [2, 3].
Rheumatoid factor (RF), which is an antibody directed against the Fc region of IgG, has been used as a diagnostic marker of RA. Rheumatoid factor has an acceptable sensitivity, but may be present in healthy elderly persons or in patients with other autoimmune and infectious diseases [4]. Other rheumatoid arthritis-associated autoantibodies include antiperinuclear factor and antikeratin antibodies [5, 6]. The epitopes of their antigens are arginyl residues citrullinated by peptidyl arginine deiminase [7–9]. Some enzyme-linked immunosorbent assays (ELISA) use linear citrulline-containing peptides that have similar sensitivity to and higher specificity than RF for diagnosing rheumatoid arthritis [10]. Assays that use cyclic citrullinated peptide (CCP) that were developed to detect anti-CCP antibody have higher sensitivity [11].
Although the presence of anti-CCP is accepted to be a reliable diagnostic and prognostic tool in RA [12, 13], its association with disease activity and severity remains unclear. In the present study, we have investigated the relation of the presence anti-CCP with clinical manifestations and disease activity in a cohort of patients with established diagnosis of RA.
Patients and methods
A total of 61 patients, attending the clinic of Ankara Physical Medicine and Rehabilitation Training and Research Hospital, fulfilled the American College of Rheumatology criteria for RA [14]. Data regarding disease variables were collected during clinical evaluation of the patient as well as based on information available in the case records. Patients with other connective tissue disease, acute or chronic infectious diseases or malignancy were not included in the study to avoid positive anti-CCP results associated with other conditions [15]. All patients gave their informed consent and the study was approved by the local ethics committee at the Ankara Physical Medicine and Rehabilitation Training and Research Hospital.
In addition to physical examination, data of disease-related parameters such as duration of disease, medications, degree of pain (visual analog scale, VAS), disease activity score 28 (DAS-28), health assessment questionnaire (HAQ) were recorded. Laboratory workup included erythrocyte sedimentation rate (ESR > 20 mm/h considered positive), plasma C-reactive protein (CRP > 0.5 mg/dl considered positive), RF (determined by the standard latex agglutination technique, Olympus America Inc.), anti-CCP (Microparticle Enzyme Immunoassay-MEIA, Abbott Laboratories Inc., USA), complete blood count and anemia parameters (serum iron, iron binding capacity, transferin, ferritin, vitamin B12).
Statistics
At study end, patients were divided into anti-CCP positive and anti- CCP negative and comparisons between the two groups were performed in all the above-mentioned characteristics. Constant variables were compared with the use of independent- samples t-test, while in comparisons regarding categorical variables the chi-square test was applied. Logistic regression was used to detect factors associated with anti-CCP positivity. The level of two-sided statistical significance was set at 0.05. All data were analyzed using SPSS, version 16.
Results
The socio-demographic and disease-associated characteristics of the patient with rheumatoid arthritis are summarized in Table 1. Of the 61 patients, 51 (83.6%) were women and 10 (16.4%) were men with a mean age of 53.27 years (±11.26) and the mean duration of disease was 108.5 months (±102.8). As much as 43 (70.5%) patients were positive for IgM RF, and 38 (62.3%) were positive for anti-CCP. Of the 18 RF-negative patients, 6 (33.3%) were anti-CCP positive.
A total of 54 patients were treated with disease-modifying anti-rheumatic drugs (DMARDS). The most common treatment was with methotrexate (68.8%), followed by corticosteroid (44.3%), sulfosalazine (31.1%), hydroxychloroquine (24.6%) and leflunomide (13.1%).
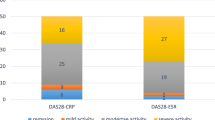
DAS-28 scores (4.82 ± 1.48 and 3.84 ± 1.50; P = 0.01), rate of patients with morning stiffness (50 and 21.73%; P = 0.05) and serum RF positivity (84.2 and 47.8%; P = 0.006) were significantly higher in anti-CCP-positive patients compared to anti-CCP-negative patients. Anti-CCP-positive patients had longer duration of disease than the negative patients (125.71 ± 104.4 and 77.38 ± 94.30; P = 0.08), but the difference was not statistically significant. Anti-CCP-positive patients had significantly lower serum ferritin levels than anti-CCP-negative patients (36.74 ± 39.15 and 80.91 ± 71.28; P = 0.02). Serum hemoglobin and thrombocyte levels were similar for anti-CCP-positive and negative patients (Table 2).
In multivariate logistic regression analysis, DAS-28 (OR: 2.0, 95% CI: 1.0–3.98, P = 0.049) and RF positivity (OR: 9.892, 95% CI: 2.1–47.5, P = 0.004) were associated with anti-CCP positivity (Table 3).
Discussion
The present study of a cohort of 61 patients with RA demonstrated the association of anti-CCP positivity with DAS-28, morning stiffness, serum RF positivity and low serum ferritin levels. Anti-CCP positivity was not associated with HAQ, ESR and CRP. DAS-28 was the only clinical independent determinant of anti-CCP positivity.
Inclusion of anti-CCP into standard diagnostic criteria of RA was proposed by some authors [16–19] to increase the diagnostic specificity, but others demonstrated a marginal increase in prediction of the disease [20, 21]. In this study, only 33.3% of RF-negative RA patients were anti-CCP positive. This result is similar with the results of studies where anti-CCP positivity in RF-negative patients were found to be between 20 and 43% [11, 22].
Joint erosions and deformities are the major adverse outcomes. Several studies demonstrated the association of anti-CCP positivity and joint destruction in patients with established RA [18, 21, 23].
Follow-up parameters of disease activity in RA patients are duration of morning stiffness, degree of joint pain, HAQ, DAS-28, CRP, ESR, hemoglobin and serum RF positivity [24–32]. Limited number of studies demonstrated an association between ESR, HAQ and anti-CCP positivity [33, 34]. In this study, HAQ, VAS, CRP, ESR, hemoglobin, anemia rate and platelet levels were not associated with anti-CCP positivity.
DAS-28 is a composite index that measures disease activity in patients with RA [32]. There is considerable disagreement in the relation between anti-CCP positivity and DAS-28 scores in RA patients [17, 33–36]. In this study, DAS-28, serum RF positivity and morning stiffness were found to be associated with anti-CCP positivity. In logistic regression analysis of multiple parameters, higher DAS-28 scores and RF positivity were associated with 2 and 9.89 times greater risk of being anti-CCP positive, respectively.
The reason for anemia in rheumatoid arthritis is multifactorial. The majority of the anemia cases encountered in RA patients were reticuloendothelial iron-replete anemia of chronic disease. Anemia in active RA has been associated with impaired erythropoiesis due to elevated inflammatory cytokines, defective erythropoietin production or response in bone marrow and blockade of iron reticuloendothelial cells leading to erythroblast iron deprivation. Ferritin is an acute phase reactant, which has moderate association with the degree of inflammation and it is also used as a surrogate marker of iron stores in anemia patients [37]. In this study, 21.3% of the patients had anemia, 75.4% of the patients had active disease according to DAS-28 scores, and 73.7% of the patients had elevated serum CRP levels or ESR. Only two (3.3%) patients had elevated serum ferritin levels in this study group; which was similar to previous result [38]. In our study, low levels of ferritin was found to be associated with anti-CCP positivity, which may indicate the negative effects of active RA on iron metabolism. We did not find any association between serum hemoglobin, thrombocyte levels or anemia rate and anti-CCP positivity. A limitation of our study was the insufficient number of patients in whom the association of anti-CCP positivity with anemia markers was investigated. Further studies are needed to investigate the possible association of ferritin and anti-CCP positivity.
Conclusion
Anti-CCP is a candidate marker for routine diagnosis and prognostic evaluation of RA patients. Our study demonstrates an association between clinical activity of the disease and anti-CCP positivity. Further studies are needed to incorporate anti-CCP tests into the routine management of rheumatoid arthritis patients.
References
Lee DM, Weinblatt ME (2001) Rheumatoid arthritis. Lancet 358:903–911
Hochberg MC (1993) Predicting the prognosis of patients with rheumatoid arthritis: is there a crystal ball? J Rheumatol 20:1265–1267
Turesson C, O’Fallon MW, Crowson CS, Gabriel SE, Matteson EL (2002) Occurrence of extra-articular disease manifestation is associated with excess mortality in a community-based cohort of patients with rheumatoid arthritis. J Rheumatol 29:62–67
Dörner T, Egerer K, Feist E, Burmester GR (2004) Rheumatoid factor revisited. Curr Opin Rheumatol 16:246–253
Nienhuis RL, Mandema E (1964) A new serum factor in patients with rheumatoid arthritis: the antiperinuclear factor. Ann Rheum Dis 23:302–305
Young BJ, Mallya RK, Leslie RD, Clark CJ, Hamblin TJ (1979) Anti-keratinantibodies in rheumatoid arthritis. Br Med J 2:97–99
Vossenaar ER, Despre′s N, Lapointe E, van der Heijden A, Lora M, Senshu T et al (2004) Rheumatoid arthritis specific anti-Sa antibodies target citrullinated vimentin. Arthritis Res Ther 6:R142–R150
Vossenaar ER, Radstake TR, van der Heijden A, van Mansum MA, Dieteren C, de Rooij DJ et al (2004) Expression and activity of citrullinating peptidylarginine deiminase enzymes in monocytes and macrophages. Ann Rheum Dis 63:373–381
Vossenaar ER, Zendman AJ, van Venrooij WJ, Pruijn GJ (2003) PAD, a growing family of citrullinating enzymes: genes, features and involvement in disease. Bioessays 25:1106–1118
Schellekens GA, de Jong BA, van den Hoogen FH, van de Putte LB, van Venrooij WJ (1998) Citrulline is an essential constituent of antigenic determinants recognized by rheumatoid arthritis-specific autoantibodies. J Clin Invest 101:273–281
Schellekens GA, Visser H, de Jong BA, van den Hoogen FH, Hazes JM, Breedveld FC et al (2000) The diagnostic properties of rheumatoid arthritis antibodies recognizing a cyclic citrullinated peptide. Arthritis Rheum 43:155–163
Zendman AJW, van Venrooij WJ, Pruijn GJM (2006) Use and significance of anti-CCP autoantibodies in rheumatoid arhritis. Rheumatology 45:20–25
Ates A, Karaaslan Y, Aksaray S (2006) Predictive value of autoantibodies to cyclic citrullinated peptide in patients with early arthritis. Clin Rheumatol 25:743–745
Arnett FC, Edworthy SM, Bloch DA, McShane DJ, Fries JF, Cooper NS, Healey LA, Kaplan SR, Liang MH, Luthra HS et al (1998) The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum 31:315–324
Bizzaro N, Mazzanti G, Tonutti E, Villalta D, Tozzoli R (2001) Diagnostic accuracy of the anti-citrulline antibody assay for rheumatoid arthritis. Clin Chem 47(6):1089–1093
Nishimura K, Sugiyama D, Kogata Y, Tsuji G, Nakazawa T, Kawano S, Saigo K, Morinobu A, Koshiba M, Kuntz KM, Kamae I, Kumagai S (2007) Meta-analysis: diagnostic accuracy of anti-cyclic citrullinated peptide antibody and rheumatoid factor for rheumatoid arthritis. Ann Intern Med 146(11):797–808
Alexiou I, Germenis A, Ziogas A, Theodoridou K, Sakkas LI (2007) Diagnostic value of anti-cyclic citrullinated peptide antibodies in Greek patients with rheumatoid arthritis. BMC Musculoskelet Disord 8:37
Nieto-Colonia AM, Santos WS, Keusseyan SP, Caldana W, Fernandes AR, Andrade LE (2008) Antibodies to citrullinated peptides are not associated with the rate of joint destruction in patients with a well-established diagnosis of rheumatoid arthritis. Braz J Med Biol Res 41(3):188–192
Vittecoq O, Incaurgarat B, Jouen-Beades F, Legoedec J, Letourneur O, Rolland D, Gervasi G, Ménard JF, Gayet A, Fardellone P, Daragon A, Jolivet M, le Loët X, Tron F (2004) Autoantibodies recognizing citrullinated rat filaggrin in an ELISA using citrullinated and non-citrullinated recombinant proteins as antigens are highly diagnostic for rheumatoid arthritis. Clin Exp Immunol 135(1):173–180
Berglin E, Padyukov L, Sundin U, Hallmans G, Stenlund H, Van Venrooij WJ, Klareskog L, Dahlqvist SR (2004) A combination of autoantibodies to cyclic citrullinated peptide (CCP) and HLA-DRB1 locus antigens is strongly associated with future onset of rheumatoid arthritis. Arthritis Res Ther 6(4):R303–R308
Bukhari M, Thomson W, Naseem H, Bunn D, Silman A, Symmons D, Barton A (2007) The performance of anti-cyclic citrullinated peptide antibodies in predicting the severity of radiologic damage in inflammatory polyarthritis: results from the Norfolk Arthritis Register. Arthritis Rheum 56(9):2929–2935
Kroot EJ, de Jong BA, van Leeuwen MA, Swinkels H, van den Hoogen FH, van’t Hof M, van de Putte LB, van Rijswijk MH, van Venrooij WJ, van Riel PL (2000) The prognostic value of anti-cyclic citrullinated peptide antibody in patients with recent-onset rheumatoid arthritis. Arthritis Rheum 43(8):1831–1835
Agrawal S, Misra R, Aggarwal A (2007) Autoantibodies in rheumatoid arthritis: association with severity of disease in established RA. Clin Rheumatol 26(2):201–204
American College of Rheumatology Ad Hoc Committee on Clinical Guidelines (2002) Guidelines for the management of rheumatoid arthritis: 2002 update. Arthritis Rheum 46:328
Fries JF, Spitz PW, Young DY (1982) The dimensions of health outcomes: the health assessment questionnaire, disability, pain scales. J Rheumatol 9:789
Fries JF, Spitz PW, Kraines RG et al (1980) Measurement of patient outcome in arthritis. Arthritis Rheum 23:137
Lane SK, Gravel JW Jr (2002) Clinical utility of common serum rheumatologic tests. Am Fam Physician 65:1073
Donald F, Ward MM (1998) Evaluative laboratory testing practices of United States rheumatologists. Arthritis Rheum 41:725
Listing J, Rau R, Muller B, Berlin Collaborating Rheumatological Study Group et al (2000) HLA-DRB1 genes, rheumatoid factor, elevated C-reactive protein: independent risk factors of radiographic progression in early rheumatoid arthritis. J Rheumatol 27:2100
Wolfe F, Kleinheksel SM, Cathey MA et al (1988) The clinical value of the Stanford Health Assessment Questionnaire Functional Disability Index in patients with rheumatoid arthritis. J Rheumatol 15:1480
Bruce B, Fries JF (2003) The Stanford Health Assessment Questionnaire: a review of its history, issues, progress, documentation. J Rheumatol 30:167
Prevoo MLL, van’t Hof MA, Kuper HH, van de Putte LBA et al (1995) Modified disease activity scores that include twenty-eight-joint counts: development and validation in a prospective longitudinal study of patients with rheumatoid arthritis. Arthritis Rheum 3:44
Papadopoulos NG, Tsiaousis GZ, Pavlitou-Tsiontsi A, Giannakou A, Galanopoulou VK (2008) Does the presence of anti-CCP autoantibodies and their serum levels influence the severity and activity in rheumatoid arthritis patients? Clin Rev Allergy Immunol 34(1):11–15
Inanc N, Dalkilic E, Kamali S, Kasapoglu-Günal E, Elbir Y, Direskeneli H, Inanc M (2007) Anti-CCP antibodies in rheumatoid arthritis and psoriatic arthritis. Clin Rheumatol 26(1):17–23
Serdaroğlu M, Cakirbay H, Değer O, Cengiz S, Kul S (2008) The association of anti-CCP antibodies with disease activity in rheumatoid arthritis. Rheumatol Int 28(10):965–970
Glasnović M, Bosnjak I, Vcev A, Soldo I, Glasnović-Horvatić E, Soldo-Butković S, Pavela J, Mićunović N (2007) Anti-citrullinated antibodies, radiological joint damages and their correlations with disease activity score (DAS28). Coll Antropol 31(1):345–348
Fitzsimons EJ, Houston T, Munro R, Sturrock RD, Speekenbrink AB, Brock JH (2002) Erythroblast iron metabolism and serum soluble transferrin receptor values in the anemia of rheumatoid arthritis. Arthritis Rheum 47(2):166–171
Orbach H, Zandman-Goddard G, Amital H, Barak V, Szekanecz Z, Szucs G, Danko K, Nagy E, Csepany T, Carvalho JF, Doria A, Shoenfeld Y (2007) Novel biomarkers in autoimmune diseases: prolactin, ferritin, vitamin D, and TPA levels in autoimmune diseases. Ann N Y Acad Sci 1109:385–400
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Önder, B., Kurtaran, A., Kimyon, S. et al. Association of anti-CCP positivity with serum ferritin and DAS-28. Rheumatol Int 30, 223–227 (2009). https://doi.org/10.1007/s00296-009-0941-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-009-0941-4