Abstract.
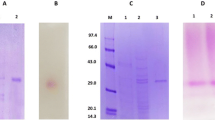
The invertase of Lactobacillus reuteri CRL 1100 is a glycoprotein composed by a single subunit with a molecular weight of 58 kDa. The enzyme was stable below 45°C over a wide pH range (4.5–7.0) with maximum activity at pH 6.0 and 37°C. The invertase activity was significantly inhibited by bivalent metal ions (Ca++, Cu++, Cd++, and Hg++), β-mercaptoethanol, and dithiothreitol and partially improved by ethylenediaminetetraacetic acid. The enzyme was purified 32 times over the crude extract by gel filtration and ion-exchange chromatography with a recovery of 17%. The K m and Vmax values for sucrose were 6.66 mM and 0.028 μmol/min, respectively. An invertase is purified and characterized for the first time in Lactobacillus, and it proved to be a β-fructofuranosidase.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Received: 13 August 1999 / Accepted: 15 September 1999
Rights and permissions
About this article
Cite this article
Cuezzo de Ginés, S., Maldonado, M. & Font de Valdez, G. Purification and Characterization of Invertase from Lactobacillus reuteri CRL 1100. Curr Microbiol 40, 181–184 (2000). https://doi.org/10.1007/s002849910036
Issue Date:
DOI: https://doi.org/10.1007/s002849910036