Abstract
The decolorization of dye and textile effluent by Trametes hirsuta was studied in both induced and non-induced media. A removal of 70–100% of the color was achieved through adsorption and the action of laccases. Laccase activity was increased significantly with the addition of grapefruit peel (4000 U/mL) and effluent with grapefruit peel (16,000 U/mL) in comparison with the basal medium (50 U/mL). Analysis of the expression of laccase isoenzymes lac-B and lac-T revealed clear differences in the expression of these genes. The low levels of expression of lac-B in all media suggest a basal or constitutive gene expression, whereas lac-T was over-expressed in the media with effluent, and showed an up/down regulation depending on culture conditions and time. The results obtained suggest that the lac-T gene of T. hirsuta is involved in the decolorization of dyes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The textile industry generates large volumes of complex effluents which contain high concentrations of dyes, salts, metals and phenols, and demonstrate high biological oxygen demand (BOD) and high chemical oxygen demand, COD [1, 2]. They contain mixes of dyes with different chemical structures, such as azo, triphenylmethane, anthraquinone and indigoid. The dyes used in the textile industry are characterized by their resistance to light and heat, and furthermore, many dyes may be mutagenic or carcinogenic. Owing to their low fixation rate, 10–50% of dyes are released into waste water, and when these waters are discharged into the environment they damage the ecosystem [3].
White rot fungi (WRF) are the most efficient organisms for degrading lignin polymer by means of the extracellular oxidative enzyme complex which they produce. Microbial and/or enzymatic treatments are developing technologies which have advantages over physical and chemical methods, which are expensive and often generate large quantities of unwanted toxic by-products. Laccases (benzenediol:oxidoreductase, EC 1.10.3.2) are enzymes secreted by WRF which are characterized by being very versatile, since they are able to oxidize a wide range of phenolic compounds and substrates with aromatic rings in a very unspecific manner, and they use molecular oxygen as final electron acceptor, generating water as an end product [4,5,6]. Laccases enzymes are codified by families of genes and the isoenzymes produced may differ in their affinity for substrates, stability, and in their mechanisms for regulation and localization. The group of isoenzymes expressed depends on the source of carbon and nitrogen, culture conditions and medium, as well as the addition of inductors to the medium [7,8,9]. The diversity of laccase genes in WRF suggests that the function of these enzymes may be variable, for example ligninolysis, lignin synthesis, morphogenesis, or defense against stress, and that their action may be individual, cooperative or synergetic [8]. In order to determine the function of different isoforms, it is important to study the regulation of laccase gene expression, and to identify the isoenzymes involved in any particular process. Owing to their high non-specificity, laccases can act on monophenols, diphenols, polyphenols and aromatic compounds, and they have a wide range of biotechnological applications such as detoxifying xenobiotic compounds, decolorizing dyes, functioning as biosensors, clarifying wines and juices, and delignifying paper pulp [10, 11]. In addition, catalysts based on laccases have proved to be efficient, sustainable and environmentally friendly.
Because of the foregoing, these enzymes have enormous potential in the treatment of complex effluents such as those from the textile industry, where the pollutants can be destroyed or transformed into less toxic compounds [12]. Studies of decolorization have been carried out on individual synthetic dyes using Basidiomycete fungi of the genera Pleurotus [13], Trametes [14] and Ganoderma [15], among others. It has also been established that the addition of synthetic and natural mediators to the laccases can increase the range of action of these enzymes upon dyes with different chemical structures or with a high redox potential that laccases alone cannot oxidize [16]. For example, the addition of hydroxybenzotriazole to laccases of Trametes trogii permitted maximum decolorization of six dyes [17].
However, there have been few studies reporting decolorization of real effluents by white rot fungus. Most studies have been carried out on synthetic effluents containing mixtures of dyes or diluted effluent. Trametes versicolor succeeded in efficiently removing the color from a synthetic effluent with three dyes (97%) and a textile effluent (92%) even though this latter was diluted 7-fold [18]. Pycnoporus sp. SYBC decolorized up to 70% of a diluted textile effluent supplemented with a lignocellulosic waste. Based on the foregoing, it is clear that the application of these microbian systems to waste waters is still a huge challenge, and therefore it is important to expand our knowledge of the regulation of laccase gene expression to identify the time and level of expression of the isoenzymes that participate in dye removal.
Trametes hirsuta Bm-2 is an easily cultivatable fungus, it produces extracellular laccases and has proved to be capable of removing color from dyes [19,20,21]. Recently, two laccase genes from T. hirsuta Bm-2, lac-B and lac-T, have been isolated and described [22]. The objective of this study was to determine the potential of T. hirsuta Bm-2 for decolorization of índigo carmíne and a textile effluent, and also to analyze the levels of expression of the lac-B and lac-T genes during fungal treatment. The transcriptional profiles of these genes were examined over the different treatments, as were laccase activity and decolorization of the effluent.
Materials and Methods
Raw Materials
Grapefruit peelings were used as lignocellulosic waste. The particle size was approximately 7.5 mm. They were immersed for 1 h in 83.17 mM KOH, rinsed with distilled water and dried at 65 °C. They were subsequently ground in a commercial Waring laboratory blender model 51BL30 for 60 s.
The textile effluent was collected from Maquiladora Lee de México, S.A. de C.V., located in the municipality of Acanceh, Yucatan. Sampling was carried out in the effluent storage tanks after having gone through the screening process, but before moving on to biological treatment. The pH was measured and the effluent stored at 4 °C.
Fungal Strain, Media, and Culture Conditions
Trametes hirsuta strain Bm-2 (GQ280373.1) was isolated from decaying wood in Yucatan, Mexico [19], and used throughout this study. This strain was grown in six culture media: (1) Kirk’s liquid basal medium (BM) pH 6, which consists of 10 g glucose, 5 g ammonium tartrate, 0.2 g MgSO4·7H2O, 2 g KH2PO4, 0.1 g CaCl2·2H2O, 1 mg thiamine, and 1 mL trace compound solution, without Tween 20 or veratryl alcohol [23]; (2) BM supplemented with carmine indigo (0.1%), (3) Rodríguez-Couto Medium (RC) [24], g/L: glucose, 2; malt extract, 15; (NH4)2SO4, 0.9; KH2PO4, 2; MgSO4·7H2O, 0.5; CaCl2·2H2O, 0.1; thiamine–HCl, 1 mg/L, supplemented with 3% (w/v) of grapefruit peel (GP), (4) RC medium supplemented with 3% (w/v) of grapefruit peel (GP) and carmine indigo (0.1%), (5) Effluent with the addition of RC and GP, (6) effluent with the addition of 250 mL BM. Erlenmeyer flasks containing 100 mL of each medium were inoculated with 1 mL of cell suspension, produced in accordance with [25]. The flasks were incubated at 35 °C and shaken at 150 rpm. As controls, uninoculated media were used. Samples were taken every 24 h. They were centrifuged at 5000 rpm for 15 min.
Decolorization of Indigo Carmine and Textile Effluent
The decolorization of the indigo carmine was measured spectrophotometrically at the maximum absorbance of the dye (600 nm) after different time intervals. In the effluent an absorbance scan was performed in a wavelength range of 200 to 1100 nm, in which maximum absorbance was obtained at 778 nm. The decolorization was expressed in terms of the percentage calculated according to the following equation:
where A0 is initial absorbance at λmax of the dye and effluent before inoculating with the fungus and At is the absorbance after each time interval [26]. All experiments were performed in triplicate.
Laccase Assays
Laccase activity in cell-free filtrates was measured at 40 °C using 2,2′-azino-bis (3-ethylbenzthiazoline-6-sulphonic acid) (ABTS). The assay mixture contained 1 M sodium acetate buffer (pH 4.5) and 0.5 mM ABTS in a total volume of 1 mL. The oxidation of ABTS was measured by the increase in absorbance at 420 nm as described by Johannes and Majcherczyk [27]. One enzyme unit (U) is defined as the amount of enzyme required to oxidize 1 μmol of ABTS per min under the assay conditions. The amount of the enzyme production was expressed as U/mL.
RNA Extraction, cDNA Synthesis
After 7 days, mycelia were harvested from the liquid culture by vacuum filtration, and frozen in liquid nitrogen. The total RNA was extracted in accordance with the TRIzol™ procedure (Invitrogen, Carlsbad, CA, USA). RNA quantification was measured in a UV–Vis spectrophotometer (NanoDrop 2000, Thermo Scientific™, Waltham, MA, USA). RNA integrity was visualized in 0.8% (w/v) agarose gel electrophoresis prepared in 1 × Tris–borate–EDTA buffer (0.9 M Tris–borate pH 8; 20 mM EDTA) and stained with ethidium bromide. The images were taken with a Gel Doc™ EZ imager (BioRad). First-strand cDNA synthesis was performed using 200 ng of total RNA as a template and 500 μg/mL of primer dT12-18, with the enzyme SuperScript II RT from Life Technologies (New York, NY, USA) according to the manufacturer’s instructions.
qPCR Amplification
RT-qPCRs were performed in a StepOnePlus (Applied Biosystems), using SYBR green dye to detect product amplification. A set of specific primers (Table 1) was designed for the amplification of the transcript from the two laccase genes (lac-B and lac-T) identified in the genome [22].
All experiments were performed in triplicate. Actin was the housekeeping gene used as an endogenous control. The following primers were used to amplify the housekeeping gene actin: Act-512F 5′-ATGTGCAA GGCCGGTTTCGC-3′ and Act-783R 5′-TACGAGTCCTTCTGGCCCAT-3′ [28].
The amplifications were performed in a final volume of 20 µL. Each reaction mixture contained 10 µL RadiantTM Green 2X qPCR Mix Hi-Rox (Alkali Scientific), 10 mM forward and reverse primer, and a 2 µL 1:10 dilution of the RT product. Amplifications were performed with an initial 5 min step at 95 °C followed by 40 denaturation cycles at 95 °C for 30 s and primer annealing and extension at 60 °C for 40 s. The melting curves ranged from 60 to 95 °C and temperature was increased in increments of 0.3 °C. StepOne software was used to confirm the occurrence of specific amplification peaks.
Data pre-processing were performed using Microsoft Excel 2007 and included efficiencies and reference gene normalization. The fold expression was calculated by the \(2^{{ - \Delta \Delta C_{{\text{t}}} }}\) method as described by [29] (Eq. 1).
In the above equation Etarget is the real-time PCR efficiency of target gene transcript; Eref is the real-time PCR efficiency of a reference gene transcript; ΔCptarget is the CP deviation of control–sample of the target gene transcript. All other multiple comparisons were performed using the statistical analysis software.
Statistical Analysis
The data are the average of the results of three replicates and are presented as the mean ± SD. One-way ANOVA was used to determine the difference between treatments at a significant level of 0.01 or 0.05.
Results and Discussion
Decolorization of Dyes and Textile Industry Effluent
In this study, expression of laccase genes from T. hirsuta Bm-2 was analyzed during treatment of indigo carmine dye and an effluent in different culture conditions: (1) basal medium, (2) dye in basal medium, (3) grapefruit peel, (4) dye with grapefruit peel, (5) effluent with basal medium, (6) effluent with grapefruit peel. In these media, the percentage of decolorization, laccase activity and expression of lac-B and lac-T was determined at different culture times.
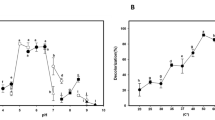
Figure 1a shows that dye decolorization in the BM was 48% after 24 h, and reached a maximum decolorization after 72 h (100%). Laccases were not induced by the addition of dye, since the maximum activity (50 U/mL) was similar to that attained in the BM after 96 h of culture. A colorless extract was obtained (Fig. 1b) and the mycelium was blue. The results show that the low laccase activity and high efficiency at decolorization were linked to the adsorption of dye to the mycelium. Various studies have shown that the biomass of fungi is capable of adsorbing dyes, metals and other dangerous pollutants due to the presence of functional groups [30]. However, the transfer of dye to a substrate is not a desirable process, since the biomass is difficult to store for long periods of time [31].
The addition of grapefruit peel to the medium allowed a high level of color removal and laccase activity (Fig. 2a, b). Laccase activity after 72 h was 1480 U/mL and continued to increase until reaching 4500 U/mL after 96 h of culture, which suggests an inductive effect on laccase synthesis due to the presence of lignocellulosic waste. The high color removal after 48 h was attributed to the adsorption of dye to the fungal biomass. However, after 96 h the blue color of the mycelium was eliminated, which was attributed to the action of the extracellular laccases which were produced.
Decolorization of indigo carmine and laccase activity during culture of T. hirsuta supplemented with grapefruit peel. a Laccase activity and decolorization, and b color before and after treatment. Results were represented as means ± standard deviation of three parallel measurements (n = 3) (Color figure online)
Figure 2b shows the decolorization of the dye after treatment. The change in coloring of the blue dye to green was caused by the presence of polymeric substances or carotenoids in the lignocellulosic waste, as has been previously reported [25]. Various studies have managed to increase laccase activity when lignocellulosic waste was added to the culture [32,33,34]. For example: Bakkiyaraj et al. [35] performed cultures of T. hirsuta 141 using different lignocellulosic wastes. The maximum laccase activity was obtained in wheat bran acquired from the wheat milling industry as a source of carbon (9300 U/L), 8512 U/L with lemon peel, 6543 U/L with bagasse and 6284 U/L with orange peel. The addition of lignin monomers such as ferulic acid, guaiacol and coumaric acid to fungal cultures increases laccase production [35,36,37].
Owing to the extreme complexity of effluents, which contain cocktails of phenols, metals, antibiotics and various aromatic compounds, studies in these systems with white rot fungus and laccase enzymes has been little reported. In this project, raw undiluted effluent was used, supplemented with salts from the basal medium (Fig. 3). There was little increase during 72 h of culture, with no laccase activity or decolonization. Later there was a growth in decolorization, which reached 95%, an in laccase activity (1100 U/mL). The correlation between decolorization and laccase activity is to be expected, since in short time spans adsorption to the mycelium takes place, which reduces the concentration of compounds such as phenols and metals that negatively affect enzyme synthesis, and enzyme production is facilitated [38, 39]. Laccase induction in several basidiomycetes grown in effluent has previously been reported in waste waters [40, 41]. On the other hand, it has been shown that phenols present at low concentrations in the culture can also act as redox mediators, encouraging enzyme action and the biotransformation of dyes [42]. Recently members of our work team have reported the obtaining of ultrafiltered (UF) phenolic extracts from T. hirsuta Bm-2 cultivated in wheat bran. The UF-laccase system increased laccase activity in ABTS by 12.4 times, and decolorization of indigo carmine by 12 times, in comparison with laccase alone [21].
Decolorization of indigo carmine and laccase activity during culture of T. hirsuta in a textile effluent with basal medium. a Laccase activity and decolorization, and b color before and after treatment. Results were represented as means ± standard deviation of three parallel measurements (n = 3) (Color figure online)
The addition of grapefruit to the textile effluent produced similar behavior to the medium with textile effluent supplemented with salts, showing a high level of efficiency in decolorization (90%), although in these conditions a notable increase in laccase activity was observed, rising to 16,000 U/mL after 144 h (Fig. 4). The high inductive effect may be caused by phenolic and/or metal components present in the effluent and the grapefruit. For example, copper present in effluents has a great influence on white rot fungus laccase synthesis [43]. Also, the synergetic effect of copper and phenols such as ferulic acid present in lignin has been reported to increase laccase levels up to 40 times in cultures of Pleurotus ostreatus, compared to a control medium [8]. Some reports of fungus used in decolorization of textile effluents include T. versicolor [44], Trametes troggi [45], Aspergillus ochraceus [46] and Schyzohyllum commune [47], the action of which has been linked to adsorption processes and laccase activity. Kamida et al. [48] studied decolorization of textile effluent with sugar cane baggasse by two strains of P. ostreatus and found that both strains decolorized the effluent completely after 14 days. Wesenberg et al. [49] reported decolorization of waste water from the textile industry with Clitocybula dusenii fungus, achieving a maximum decolorization of 87% after 20 days, using effluent diluted four times.
Decolorization of indigo carmine and laccase activity during culture of T. hirsuta in a textile effluent supplemented with grapefruit peel. a Laccase activity and decolorization, and b color before and after treatment. Results were represented as means ± standard deviation of three parallel measurements (n = 3) (Color figure online)
Effect of Dyes and Textile Industry Effluent on the Expression of Laccase Genes
The results of expression in qPCR showed that lac-T is the gene more predominantly expressed in all the conditions evaluated, and differential patterns of expression were observed (Fig. 5). It could be noted in the medium with dye and the basal medium that the expression of lac-T was double (2 UR) that of lac-B, however, both genes showed constitutive levels (Fig. 5a).
These results coincide with the low extracellular laccase activity detected in the culture medium. When the dye was supplemented with grapefruit peel the level of expression of both genes was low (Fig. 5b) and laccase activity increased 90-fold compared to the basal medium with the dye. This indicates that there was no correlation between the transcriptional levels of both genes and the increase observed in the extracellular laccase extracts.
The transcriptional response in the media with the effluent shows over-expression of lac-T in the medium supplemented with salts, 30 to 50 times in comparison with the basal expression detected during culture (Fig. 5c). It is important to point out that expression was not determined at shorter time spans because of the scarce mycelium production. The high level of induction detected in the medium with effluent supplemented with salts was correlated with the increase in decolorization activity that occurred. On the contrary, the expression profile of lac-T when the grapefruit was added to the effluent showed an up/down regulation over the time span of the culture. After 72 h there was a high level of lac-T induction (50 UR) but laccase activity was low. On the other hand at 96 h the transcript level of lac-T dropped to basal levels and activity in the extracts was the highest of all the cultures to be evaluated (16,000 U/mL). Considering the multiplicity of laccases in WRF, it is possible that other laccases were expressed in these conditions. The results coincide with the findings of other authors, who indicate that the expression of lignolithic enzymes may or may not be correlated with extracellular activity and decolorization of dyes. Palmieri et al. [50], observed a large quantity of LACC2 transcripts with low laccase activity, and this finding was linked to the possible lack of post-translational modifications (proteolysis) to activate the enzyme. Cuamatzi-Flores et al. [51] found that the highest levels of expression of peroxidase dye decolorant Pleos-dyP1 occurs when maximum activity and decolorization are present. Goudopoulou et al. (2010) mentioned that the differential expression of laccase genes from lignolithic fungus varies as a function of nutritional conditions and the physiological state of the fungus. Moreover, the accumulation of toxic compounds and/or the degradation of products in a specific time could be another reason for the transcriptional induction or suppression of laccase genes. The transcriptional response may involve complex regulation mechanisms linked to the presence in the promoter of putative elements such as xenobiotic response elements (XRE), heat shock elements (HSE), or metal response elements (MRE) among others [52]. On the other hand, various studies with purified laccases have shown that laccases are responsible for the decolorization of waste water from olive processing [53]. Also, partially purified laccases of T. hirsuta Bm-2 have proved to be capable of carrying out complete decolorization of indigo carmine and partial decolorization of textile effluent [54].
There has recently been a report of the analysis in silico of the non-codifying 5′-UTR region of lacB and lacT genes from T. hisruta Bm2 (Pereira-Patrón et al. 2019). The variation in length and number of regulatory motifs in the 5′-UTR region constitutes an important factor for accessing regulatory proteins that modify gene expression by increasing or decreasing transcription and/or translation of proteins (Barret et al. 2013). In this study, we found differences of expression in both genes, where lacT was over-expressed during treatment of effluent, while the expression of lacB was practically unchanged. Kozak (1989) pointed out that long 5′-UTR structured from proteins involved in cell development can be regulated more finely. LacT has a longer 5′-UTR and double the regulatory elements of lacB, and it is therefore possible that these differences influence the variations in transcriptional response detected during the treatment of dyes and effluent.
Conclusion
The results obtained in this study highlight the efficiency of T. hirsuta-Bm2 in the decolorization of indigo carmine and an undiluted textile effluent. It is also important to point out that waste such as effluent and grapefruit peel are an important source for the production of high laccase levels. The complexity of the regulation of the laccase system was evident, especially with the use of waste; however, the results showed the possible contribution of lac-T in the process of dye decolorization. This finding is particularly important for future studies of genetic improvement. However, further studies are required to elucidate the mechanisms that regulate transcription expression of laccase genes in T. hirsuta Bm-2.
References
Ghaly A, Ananthashankar R, Alhattab M et al (2014) Chemical engineering and process technology production, characterization and treatment of textile effluents: a critical review. J Chem Eng Process Technol. https://doi.org/10.4172/2157-7048.1000182
Mani S, Chowdhary P, Bharagava RN (2019) Textile wastewater dyes: toxicity profile and treatment approaches. In: Emerging and eco-friendly approaches for waste management. Springer, Singapore, pp 219–244
Kumar PS, Saravanan A (2017) Sustainable wastewater treatments in textile sector. Sustainable fibres and textiles. Elsevier, Amsterdam, pp 323–346
Osma JF, Toca-Herrera JL, Rodríguez-Couto S (2011) Cost analysis in laccase production. J Environ Manag 92:2907–2912
Giardina P, Faraco V, Pezzella C et al (2010) Laccases: a never-ending story. Cell Mol Life Sci 67:369–385
Dwivedi UN, Singh P, Pandey VP, Kumar A (2011) Structure–function relationship among bacterial, fungal and plant laccases. J Mol Catal B 68:117–128
Garrido-Bazán V, Téllez-Téllez M, Herrera-Estrella A et al (2016) Effect of textile dyes on activity and differential regulation of laccase genes from Pleurotus ostreatus grown in submerged fermentation. AMB Express 6:93
Pezzella C, Lettera V, Piscitelli A et al (2013) Transcriptional analysis of Pleurotus ostreatus laccase genes. Appl Microbiol Biotechnol 97:705–717
Wang W, Liu F, Jiang Y et al (2015) The multigene family of fungal laccases and their expression in the white rot basidiomycete Flammulina velutipes. Gene 563:142–149
Viswanath B, Rajesh B, Janardhan A et al (2014) Fungal laccases and their applications in bioremediation. Enzyme Res 2014:163242
Senthivelan T, Kanagaraj J, Panda RC (2016) Recent trends in fungal laccase for various industrial applications: an eco-friendly approach—a review. Biotechnol Bioprocess Eng 21:19–38
Unuofin JO, Okoh AI, Nwodo UU (2019) Aptitude of oxidative enzymes for treatment of wastewater pollutants: a laccase perspective. Molecules 24:2064
Kunjadia PD, Sanghvi GV, Kunjadia AP, et al (2016) Role of ligninolytic enzymes of white rot fungi (Pleurotus spp.) grown with azo dyes. SpringerPlus 5:1487
Si J, Cui B-K, Dai Y-C (2013) Decolorization of chemically different dyes by white-rot fungi in submerged cultures. Ann Microbiol 63:1099–1108
Qin P, Wu Y, Adil B et al (2019) Optimization of laccase from Ganoderma lucidum decolorizing Remazol brilliant blue R and Glac1 as main laccase-contributing gene. Molecules 24:3914
Grassi E, Scodeller P, Filiel N et al (2011) Potential of Trametes trogii culture fluids and its purified laccase for the decolorization of different types of recalcitrant dyes without the addition of redox mediators. Int Biodeterior Biodegrad 65:635–643
Forootanfar H, Rezaei S, Zeinvand-Lorestani H et al (2016) Studies on the laccase-mediated decolorization, kinetic, and microtoxicity of some synthetic azo dyes. J Environ Health Sci Eng 14:7
Amaral PFF, Fernandes DLA, Tavares APM et al (2004) Decolorization of dyes from textile wastewater by Trametes versicolor. Environ Technol 25:1313–1320
Tapia-Tussell R, Pérez-Brito D, Rojas-Herrera R et al (2011) New laccase-producing fungi isolates with biotechnological potential in dye decolorization. Afr J Biotechnol 10(50):10134–10142
Tapia-Tussell R, Pérez-Brito D, Torres-Calzada C et al (2015) Laccase gene expression and vinasse biodegradation by Trametes hirsuta strain Bm-2. Molecules. https://doi.org/10.3390/molecules200815147
Ancona-Escalante W, Tapia-Tussell R, Pool-Yam L, et al (2018) Laccase-mediator system produced by Trametes hirsuta Bm-2 on lignocellulosic substrate improves dye decolorization. 3 Biotech. https://doi.org/10.1007/s13205-018-1323-y
Pereira-Patrón A, Solis-Pereira S, Lizama-Uc G, et al (2019) Molecular characterization of laccase genes from the basidiomycete Trametes hirsuta Bm-2 and analysis of the 5′ untranslated region (5′ UTR). 3 Biotech 9:160
Kirk TK, Croan S, Tien M et al (1986) Production of multiple ligninases by Phanerochaete chrysosporium: effect of selected growth conditions and use of a mutant strain. Enzyme Microb Technol 8:27–32
Rodríguez-Couto S, Rodríguez A, Paterson RRM et al (2006) High laccase activity in a 6 l airlift bioreactor by free cells of Trametes hirsuta. Lett Appl Microbiol 42:612–616
Zapata-Castillo P, Villalonga-Santana M, Tamayo-Cortés J et al (2012) Purification and characterization of laccase from Trametes hirsuta Bm-2 and its contribution to dye and effluent decolorization. Afr J Biotechnol 11:3603–3611
Sirianuntapiboon S, Sihanonth P, Somchai P et al (1995) An adsorption mechanism for melanoidin decolorization by Rhizoctonia sp. Biosci Biotechnol Biochem 59:1185–1189
Johannes C, Majcherczyk A (2000) Natural mediators in the oxidation of polycyclic aromatic hydrocarbons by laccase mediator systems. Appl Environ Microbiol 66:524–528
Carbone I, Kohn LM (1999) A method for designing primer sets for speciation studies in filamentous ascomycetes. Mycologia 91:553–556
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29:e45
Kaushik P, Malik A (2009) Fungal dye decolourization: recent advances and future potential. Environ Int 35:127–141
Kahraman S, Kuru F, Dogan D, Yesilada O (2012) Removal of indigo carmine from an aqueous solution by fungus Pleurotus ostreatus. Arch Environ Prot 38:51–57
Gonzalez JC, Medina SC, Rodriguez A et al (2013) Production of Trametes pubescens laccase under submerged and semi-solid culture conditions on agro-industrial wastes. PLoS ONE 8:e73721
Imran M, Asad MJ, Hadri SH, Mehmood S (2012) Production and industrial applications of laccase enzyme. J Cell Mol Biol 10(1):1–11
Larsson S, Cassland P, Jönsson LJ (2001) Development of a Saccharomyces cerevisiae strain with enhanced resistance to phenolic fermentation inhibitors in lignocellulose hydrolysates by heterologous expression of laccase. Appl Environ Microbiol 67:1163–1170
Bakkiyaraj S, Aravindan R, Arrivukkarasan S, Viruthagiri T (2013) Enhanced laccase production by Trametes hirusta using wheat bran under submerged fermentation. Int J ChemTech Res 5:1224–1238
Cambria MT, Ragusa S, Calabrese V, Cambria A (2011) Enhanced laccase production in white-rot fungus Rigidoporus lignosus by the addition of selected phenolic and aromatic compounds. Appl Biochem Biotechnol 163:415–422
Elisashvili V, Kachlishvili E, Khardziani T, Agathos SN (2010) Effect of aromatic compounds on the production of laccase and manganese peroxidase by white-rot basidiomycetes. J Ind Microbiol Biotechnol 37:1091–1096
de la Rubia T, Ruiz E, Pérez J, Martínez J (2002) Properties of a laccase produced by Phanerochaete flavido-alba induced by vanillin. Arch Microbiol 179:70
Goudopoulou A, Krimitzas A, Typas MA (2010) Differential gene expression of ligninolytic enzymes in Pleurotus ostreatus grown on olive oil mill wastewater. Appl Microbiol Biotechnol 88:541–551
González T, Terrón MC, Yagüe S et al (2008) Melanoidin-containing wastewaters induce selective laccase gene expression in the white-rot fungus Trametes sp. I-62. Res Microbiol 159:103–109
González T, Terrón MC, Yagüe S et al (2000) Pyrolysis/gas chromatography/mass spectrometry monitoring of fungal-biotreated distillery wastewater using Trametes sp. I-62 (CECT 20197). Rapid Commun Mass Spectrom 14:1417–1424
Li X, La G, Cheng Q et al (2014) Profile of natural redox mediators production of laccase-producing fungus Pleurotus ostreatus. Bull Environ Contam Toxicol 93:478–482
Elisashvili V, Kachlishvili E (2009) Physiological regulation of laccase and manganese peroxidase production by white-rot Basidiomycetes. J Biotechnol 144:37–42
Blánquez P, Guieysse B (2008) Continuous biodegradation of 17β-estradiol and 17α-ethynylestradiol by Trametes versicolor. J Hazard Mater 150:459–462
Khlifi R, Belbahri L, Woodward S et al (2010) Decolourization and detoxification of textile industry wastewater by the laccase-mediator system. J Hazard Mater 175:802–808
Lade HS, Waghmode TR, Kadam AA, Govindwar SP (2012) Enhanced biodegradation and detoxification of disperse azo dye Rubine GFL and textile industry effluent by defined fungal-bacterial consortium. Int Biodeterior Biodegrad 72:94–107
Asgher M, Yasmeen Q, Iqbal HMN (2014) Development of novel enzymatic bioremediation process for textile industry effluents through response surface methodology. Ecol Eng 63:1–11
Kamida HM, Durrant LR, Monteiro RTR, de Armas ED (2005) Biodegradação de efluente têxtil por Pleurotus sajor-caju. Quim Nova
Wesenberg D, Buchon F, Agathos SN (2002) Degradation of dye-containing textile effluent by the agaric white-rot fungus Clitocybula dusenii. Biotechnol Lett 24:989–993
Palmieri G, Cennamo G, Faraco V et al (2003) Atypical laccase isoenzymes from copper supplemented Pleurotus ostreatus cultures. Enzyme Microb Technol 33:220–230
Cuamatzi-Flores J, Esquivel-Naranjo E, Nava-Galicia S et al (2019) Differential regulation of Pleurotus ostreatus dye peroxidases gene expression in response to dyes and potential application of recombinant Pleos-DyP1 in decolorization. PLoS ONE 14:e0209711
Xiao YZ, Hong YZ, Li JF et al (2006) Cloning of novel laccase isozyme genes from Trametes sp. AH28-2 and analyses of their differential expression. Appl Microbiol Biotechnol 71:493–501
D’Souza TM, Boominathan K, Reddy CA (1996) Isolation of laccase gene-specific sequences from white rot and brown rot fungi by PCR. Appl Environ Microbiol 62:3739–3744
Zapata-Castillo P, Villalonga-Santana L, Islas-Flores I et al (2015) Synergistic action of laccases from Trametes hirsuta Bm2 improves decolourization of indigo carmine. Lett Appl Microbiol 61:252–258
Kozak M (1989) Circumstances and mechanisms of inhibition of translation by secondary structure in eukaryotic mRNAs. Mol Cell Biol 9:5134–5142
Acknowledgements
The names of funding organizations were Consejo Nacional de Ciencia y Tecnología (CONACYT)-FOMIX Award Number 108415 and Tecnológico Nacional de México Award Number 7409.19-P.
Author information
Authors and Affiliations
Contributions
All the authors contributed to this work. T-T and S-P conceived, designed and wrote the paper; P-P performed the experiments and analyzed the data; A-G, P-B, participated in the data analysis and writing of the paper; L-UC in the review and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tapia-Tussell, R., Pereira‑Patrón, A., Alzate-Gaviria, L. et al. Decolorization of Textile Effluent by Trametes hirsuta Bm-2 and lac-T as Possible Main Laccase-Contributing Gene. Curr Microbiol 77, 3953–3961 (2020). https://doi.org/10.1007/s00284-020-02188-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00284-020-02188-9