Abstract
The purposes of this study were to determine the effect of structural change on the intestinal disposition of isoflavones and to elucidate the mechanisms responsible for transport of phase II isoflavone conjugates. Transport and metabolism of six isoflavones (i.e., genistein, daidzein, glycitein, formononetin, biochanin A, and prunetin) were studied in the human intestinal Caco-2 model and mature Caco-2 cell lysate. Glucuronides were the main metabolites in intact Caco-2 cells for all isoflavones except prunetin, which was mainly sulfated. In addition, the 7-hydroxy group was the main site for glucuronidation whereas the 4′-hydroxy group was only one of the possible sites for sulfation. Glucuronidated isoflavones (except biochanin A) were preferably excreted to the basolateral side, whereas sulfated metabolites (except genistein and glycitein) were mainly excreted into the apical side. Polarized excretion of most isoflavone conjugates was inhibited by the multidrug resistance-related protein (MRP) inhibitor leukotriene C4 (0.1 μM) and the organic anion transporter (OAT) inhibitor estrone sulfate (10 μM). When formation and excretion rates of isoflavones were determined simultaneously, the results showed that formation served as the rate-limiting step for all isoflavone conjugates (both glucuronides and sulfates) except for genistein glucuronide, which had comparable excretion and formation rates. In conclusion, the intestinal disposition of isoflavones was structurally dependent, polarized, and mediated by MRP and OAT. Formation generally served as the rate-limiting step in the cellular excretion of conjugated isoflavones in the Caco-2 cell culture model.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Isoflavonoids belong to a class of plant non-nutrients called phytoestrogens that have estrogen-like activities. Epidemiological studies have shown that consumption of an isoflavonoid-rich diet is associated with a decrease in the incidence of hormone-related cancers (e.g., breast and prostate cancer) ([1, 14, 24] and references therein). Isoflavonoids are widely available as dietary supplements in pharmacies and health food stores. The most commonly consumed isoflavones are derived from soy (mainly a mixture of genistein, daidzein, and glycitein) and red clover (mainly a mixture of formononetin, biochanin A, genistein, and daidzein). Other popular supplements that contain isoflavones also include black cohosh (containing formononetin) and a mixture of soy and black cohosh. Currently, soy isoflavones are undergoing clinical trials for prostate cancer prevention.
Despite the assertion that isoflavones are effective cancer prevention agents and have a variety of other health benefits, a significant gap exists between what has been shown in vitro and what can be achieved in vivo. This is because the in vivo concentration of intact isoflavones (nanomolar range) achieved with a normal soy diet is much lower than that needed in vitro to achieve their reported anticancer and chemopreventive effects, which generally have IC50 values in the micromolar range [1, 4, 14, 24]. The low bioavailability is usually not the result of poor absorption but of extensive metabolism as has been shown for genistein and apigenin [5, 15]. However, much work is needed to understand why isoflavones have poor bioavailability so we can develop isoflavones or their analogs into viable cancer chemopreventive agents.
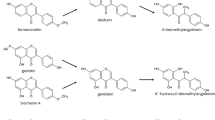
Our understanding of the absorption mechanism and metabolic pathways of isoflavones is limited, which makes it difficult to understand the reasons for their poor bioavailability. Previously, it has been also shown that intestinal metabolism and subsequent metabolite excretion is the main contributor of genistein’s first-pass metabolism [5, 15]. Furthermore, the main metabolites of genistein in Caco-2 cells are similar to those found in humans in vivo, namely genistein glucuronide and genistein sulfate [12, 13, 18]. Therefore, the goals of this study were to characterize the metabolic pathways of six isoflavones (i.e., genistein, daidzein, glycitein, formononetin, biochanin A, and prunetin; Fig. 1) and how conjugated metabolites of isoflavones are excreted in the Caco-2 cell culture model.
HPLC elution profiles of six isoflavones (shown with chemical structures) and their conjugated metabolites. Representative samples after 24-h incubation with the Caco-2 cell monolayers were used. Isoflavone glucuronides, sulfates, and their own original aglycones (or isoflavones) are labeled G, S, and A, respectively. Biochanin A (Bio) and prunetin (Pru) generated two glucuronidated metabolites labeled G1 and G2, respectively. The other four isoflavones, genistein (Gen), daidzein (Dai), glycitein (Gly), and formononetin (For) only generated one sulfate and one glucuronide. Testosterone was used as an internal standard (IS, 4 nmol), which had different retention times in two slightly different HPLC methods (see “ HPLC analysis of isoflavones and their conjugates ”)
Materials and methods
Materials
Cloned Caco-2 TC7 cells were a kind gift from Dr. Moniqué Rousset of INSERM U178 (Villejuit, France). Leukotriene C4 was purchased from BIOMOL (Plymouth Meeting, Pa.). Isoflavones genistein, daidzein, glycitein, formononetin, and prunetin were purchased from Indofine Chemical Company (Wayne, N.J.) or LC Laboratories (Woburn, Mass.). β-Glucuronidase with sulfatase (catalog #G1512) or without sulfatase (catalog #G7396), sulfatase without glucuronidase (catalog #S1629), uridine diphosphoglucuronic acid, 3′-phosphoadenosine 5′-phosphosulfate, alamethicin, d-saccharic-1,4-lactone monohydrate, biochanin A, magnesium chloride, estrone sulfate, and Hank’s balanced salt solution (HBSS, powder form) were purchased from Sigma Chemical Company (St Louis, Mo.). All other materials (typically analytical grade or better) were used as received.
Cell culture
The culture conditions for growing Caco-2 TC7 cells have been described previously [8, 9, 15]. The seeding density (100,000 cells/cm2), growth medium (DMEM supplemented with 10% fetal bovine serum), and quality control criteria were all implemented in the present study as described previously [8, 9, 15]. Caco-2 TC7 cells were fed every other day, and the monolayers were ready for experiments from 19 to 22 days after seeding. The TC7 strain of Caco-2 cells was developed in the laboratories of Dr. Moniqué Rousset of INSERM U178 (Villejuit, France) and is generally similar to wild-type Caco-2 cells used in our previous studies [10]. More than 55 published studies have used this strain of Caco-2 cells according to a recent PubMed search.
Transport experiments in the Caco-2 cell culture model
Experiments in triplicate were performed in pH 7.4 HBSS [8, 9]. The protocol for performing cell culture experiments was from those described previously [11]. Briefly, the cell monolayers were washed three times with 37°C pH 7.4 HBSS. The transepithelial electrical resistance values were measured, and those with values less than 420 Ohm cm2 were discarded. The monolayers were incubated with the buffer for 1 h and the incubation medium was then aspirated. Afterwards, 2.5 ml of an isoflavone solution was loaded onto the apical side and 2.5 ml HBSS onto the basolateral side. Amounts of transepithelial transport were followed as a function of time, which was accomplished by four sequential samples every 30 min from the apical and basolateral sides. After each sampling, the donor or the receiver side was immediately replenished with the same volume (i.e., 500 μl) of fresh isoflavone solution or HBSS, respectively. As internal standard, 100 μl methanol/acetonitrile containing 100 μM testosterone was added to each sample. The mixture was then centrifuged at 16,000 g for 8 min, and the supernatant was analyzed by HPLC (see below).
Caco-2 cell lysate
Six mature Caco-2 cell monolayers were removed with a sharp blade from the culture inserts, immersed in 6 ml 50 mM potassium phosphate buffer (pH 7.4), and sonicated in an Aquasonic 150D sonicator (VWR Scientific, Bristol, Ct.) for 20 min at the maximum power (average 135 W) in an ice-cold water bath [11]. The resulting cell lysate was then harvested and used fresh in the experiments or frozen at −80°C until use.
Lysate protein concentration
The concentrations of lysate protein were determined using the Bio-Rad protein assay, using bovine serum albumin as the standard.
Measurement of UGT activities using cell lysate
The incubation procedures for measuring UDP-glucuronosyltransferase (UGT) activities using cell lysate were as follows: (1) mix 135 μl cell lysate (final concentration approximately 0.7 mg protein/ml), magnesium chloride (0.88 mM), saccharolactone (4.4 mM), alamethicin (0.022 mg/ml), different concentrations of substrates in a 50 mM potassium phosphate buffer (pH 7.4), and uridine diphosphoglucuronic acid (UDPGA, 3.3 mM, add last) to a final volume of 200 μl; (2) incubate the mixture at 37°C for 120 min; (3) stop the reaction by the addition of 50 μl 94% acetonitrile/6% glacial acetic acid containing 100 μM testosterone as an internal standard; (4) centrifuge the sample at 16,000 g for 8 min; and (5) assay the sample via HPLC as described below.
Measurement of sulfotransferase activities using cell lysate
The incubation conditions for measuring sulfotransferase activities were as follows: (1) mix 90 μl cell lysate (final concentration approximately 0.9 mg protein/ml), 3′-phosphoadenosine 5′-phosphosulfate (PAPS, 100 μM), and substrate of an appropriate concentration (total volume 100 μl); (2) incubate the mixture at 37°C for 120 min; (3) stop the reaction by the addition of 25 μl 94% acetonitrile/6% glacial acetic acid containing 100 μM testosterone as an internal standard; (4) centrifuge the sample at 16,000 g for 8 min; and (5) assay the sample via HPLC as described below.
Glucuronidated and sulfated isoflavones were identified by treating samples with β-glucuronidase or sulfatase, which would result in the disappearance of the corresponding peaks in the relevant HPLC chromatograms.
HPLC analysis of isoflavones and their conjugates
The conditions for HPLC analysis of isoflavones were modified based on a previously reported method [5]. The conditions for analyzing isoflavones other than daidzein and glycitein and their conjugates were: system, HP/Agilent 1090 with a diode array detector and HP Chemstation; column, Aqua (Phenomenex, Guilory, Calif.), 5 μm, 150×0.45 cm; mobile phase A, 0.04% (v/v) phosphoric acid plus 0.06% (v/v) triethylamine (pH 2.8); mobile phase B, 100% acetonitrile; mixed isocratic gradient elution (0–3 min, 20% B; 2–22 min, 20–49% B; 22–23 min, 49% B), wavelength 254 nm; injection volume, 200 μl. The assay method for glycitein and daidzein was similar to that for the other isoflavones but with a small modification to the elution profile: mixed isocratic gradient elution (0–3 min, 10% B; 3–19 min, 10–34% B; 19–28 min, 34–52% B). There was a 5-min interval between the end of the run and the next injection to allow the column re-equilibrate. Representative profiles of six isoflavones and their conjugates are shown in Fig. 1.
Data analysis
Rates of transport (Vt) were obtained using the concentration of transported isoflavones or their metabolites as a function of time (Eq. 1). Permeability (P) across a cellular membrane was calculated using the rate of transport divided by the surface area (A) of the monolayer and the initial concentration of these isoflavone compounds at the loading side (Ci; Eq. 2). Lastly, the clearance (CLmt) of a metabolite from the cell monolayer was defined as the rate of metabolite excretion (Vmt) divided by the loading concentration (Eq. 3). Vmt was determined in the same way as Vt.
Statistical analysis
One-way ANOVA or Student’s t-test was used to analyze the data. The level of significance was set at 5% or P<0.05.
Results
Apical to basolateral transport of intact isoflavones in Caco-2 cell monolayers
Isoflavones genistein, daidzein, glycitein, biochanin A, formononetin (10 μM dosing solution on the apical side), and prunetin (5 μM dosing solution due to solubility limit) were rapidly transported and metabolized during their transport from the apical to the basolateral side. The amounts of isoflavones transported increased linearly as a function of time. The apparent transport rates of intact isoflavones were quite different and ranged from 4.77 to 20.82 pmol/min/cm2 of cell membrane (P<0.05; Fig. 2). The transport rate of glycitein was the fastest, followed closely by genistein, then daidzein, biochanin A, and formononetin (Fig. 2). The transport rate of prunetin was determined at a concentration of 5 μM, and therefore was not used in comparison because the rate is dependent on concentration. The permeabilities of these isoflavones (Fig. 2) followed the same trend as the transport rate, except the permeability of prunetin which was higher than that of biochanin A or formononetin. We used 10 μM (except for prunetin) for the rest of the study since this concentration is achievable in vivo, and initial results showed sufficient metabolites and aglycone on the donor and receiver side to allow accurate measurement and the determination of inhibitor effects.
Initial transport rate and permeability of isoflavones in the Caco-2 cell culture model. The first hour transport rate (bars, left axis) and permeability (points, right axis) of genistein (Gen), daidzein (Dai), glycitein (Gly), biochanin A (Bio), formononetin (For), and prunetin (Pru) to the basolateral side in the cell culture model were determined. The isoflavone concentration in the donor solution was 10 μM except that of prunetin, which was 5 μM due to solubility limitation. Transport experimental details and assay conditions are described in “Materials and methods”. Each point or bar represents the average of three determinations, and the error bars are the standard deviations of the means. *Significant difference in the transport rate, one-way ANOVA; #significant difference in the permeability value, one-way ANOVA
Excretion of isoflavone metabolites in intact Caco-2 cell monolayers
We also followed the metabolism of all six isoflavones after loading 10 μM of each isoflavone (5 μM for prunetin due to solubility limitation) to the apical side. Metabolites were excreted to both sides of the cell monolayers, and the amounts of metabolites excreted increased linearly as a function of time (Fig. 3).
Time course of conjugate excretion in the Caco-2 cell culture model. Amounts of the glucuronide and sulfate of genistein, daidzein, glycitein, biochanin A, formononetin, and prunetin efflux to both sides as a function of incubation time. The incubation time points were 30, 60, 90 and 120 min for genistein, daidzein and glycitein, and 60, 120, 180 and 240 min for biochanin A, formononetin and prunetin. The prunetin concentration in the donor solution was 5 μM, and the concentration of the other isoflavones was 10 μM
The excretion of both conjugates was polarized and the rates of conjugate excretion were determined (Fig. 4). The efflux rates of glucuronides to the basolateral side were higher (30–280%, P<0.05) than those to the apical side, except for the efflux rates of biochanin A glucuronide which were similar to both sides (P>0.05). On the other hand, the sulfate efflux rates favored the apical side for biochanin A, formononetin, and prunetin (15–163% higher, P<0.05), but not for daidzein (44% more to the basolateral side, P<0.05). The effluxes were not polarized for genistein sulfate (P>0.05; Fig. 4a) at this concentration.
Rates of conjugate excretion in the Caco-2 cell culture model. The polarized excretion rate (a) and the total excretion rate (b) of isoflavone conjugates were determined at a parent compound concentration of 10 μM, except for prunetin whose concentration was 5 μM. The transport experimental details and the metabolite assay are described in “Materials and methods”. Each bar represents the average of three determinations, and the error bars are the standard deviations of the means. *Significant difference in the polarized excretion rate (a) or types of metabolites formed (b); #significant differences in total conjugate excretion rates of the six isoflavones according to a one-way ANOVA (b)
In addition to polarized excretion, the total amounts of conjugates excreted from the Caco-2 cells were also significantly different (P<0.05) among the five isoflavones (Fig. 4b). The total excretion rates of glucuronide were the highest for glycitein (66.56 pmol/min per monolayer or 15.85 pmol/min/cm2 for a monolayer of 4.2 cm2) followed by biochanin A, formononetin, daidzein, and genistein (11.75 pmol/min per monolayer or 2.80 pmol/min/cm2), whereas the total excretion rates of sulfates in decreasing order were biochanin A (15.95 pmol/min per monolayer, 3.8 pmol/min/cm2), glycitein, genistein, daidzein, biochanin A, and formononetin (2.85 pmol/min per monolayer or 0.68 pmol/min/cm2). On the other hand, the rank order of the total clearance of glucuronide (which is the rate of glucuronide excretion divided by the loading concentration of parent compound) was slightly different: glycitein > biochanin A > formononetin > prunetin > daidzein > genistein. When comparing the total clearance of sulfates, prunetin showed the most rapid clearance because its loading concentration was half that of the other isoflavones.
Comparing the excretion rates of both conjugates (Fig. 4a), the excretion rates of formononetin glucuronide were much higher than those of its sulfate (19.5-fold, P<0.05), and the excretion rates of glycitein, daidzein and biochanin A, and genistein glucuronides were also higher than that of their sulfates (4.5, 3.3, 2.0, and 1.2-fold, respectively, P<0.05). In contrast, the excretion rate of prunetin sulfate was much higher than that of its glucuronide (2.1-fold, P<0.05).
The total conjugate excretion rates (Fig. 4b) in decreasing order at 10 μM were: glycitein, biochanin A, formononetin, daidzein and genistein, with values of 19.4, 16.2, 13.9, 5.7, and 5.2 pmol/min/cm2, respectively. The total clearance values of conjugates, on the other hand, were in the rank order: glycitein > biochanin A > formononetin > prunetin > daidzein > genistein.
Effects of MRP and OAT inhibitors on excretion of isoflavone metabolites in intact Caco-2 cells
The multidrug resistance-related protein (MRP) inhibitor leukotriene C4 (LTC4, 0.1 μM) and the organic anion transporter (OAT) inhibitor estrone sulfate (10 μM) were used to determine the mechanisms by which conjugated genistein and its analogs are transported out of Caco-2 cells (Fig. 5). LTC4 or estrone sulfate was present in the transport medium bathing both sides of Caco-2 cell monolayers.
Effects of MRP and OAT inhibitors on the excretion of conjugated genistein and its analogs in the Caco-2 cell culture model. Genistein or its analogs at 10 μM in dosing solutions (prunetin at 5 μM) were on the apical side of cell monolayers, whereas MRP inhibitor LTC4 (0.1 μM) or OAT inhibitor estrone sulfate (10 μM) were on both sides of the monolayers. Excretion rates of glucuronidated (a, b) and sulfated (c, d) isoflavones to the apical (a, c) and basolateral (b, d) sides were determined in the absence (control) or presence of MRP inhibitor (LTC4) or OAT inhibitor estrone sulfate (ES). Transport experimental details and the metabolite assay are described in “Materials and methods”. Each bar represents the average of three determinations, and the error bars are the standard deviations of the means. *Significant difference in excretion rate following inhibition, Student’s t-test
MRP inhibitor LTC4 (0.1 μM) significantly (P<0.05) decreased the apical and basolateral efflux rates of genistein, daidzein, glycitein, biochanin A, and formononetin glucuronides, but did not affect the excretion of prunetin glucuronide (P>0.05; Fig. 5a, b). LTC4 also decreased the efflux rates (P<0.05) of genistein, daidzein, glycitein, and biochanin A sulfates from both sides, but did not change the efflux rate of prunetin sulfate (P>0.05). For formononetin sulfate, excretion from the apical side was significantly decreased (P<0.05), but the excretion from the basolateral side was increased by 36% (P<0.05; Fig. 5c, d).
Similar to the effects of LTC4, the OAT inhibitor estrone sulfate also inhibited the excretion of genistein, daidzein, glycitein, biochanin A, and formononetin glucuronides from both sides of the cell monolayers (P<0.05). However, it did not decrease the apical excretion of prunetin glucuronide (P>0.05; Fig. 5a, b). Estrone sulfate also inhibited the efflux of genistein, daidzein, glycitein, biochanin A, and prunetin sulfates from both sides of cells (P<0.05), but did not decrease the excretion of formononetin sulfate from the apical side (P>0.05; Fig. 5c, d).
Formation rates of isoflavone conjugates in Caco-2 cell lysate and comparison to their excretion rate in intact cell monolayers
Glucuronidation and sulfation of six isoflavones (each at 10 μM, except prunetin at 5 μM) were determined using Caco-2 cell lysate (Fig. 6) with a reaction time of 120 min and a protein concentration of about 0.7 mg/ml based on preliminary studies that demonstrated linearity for this length of reaction and enough metabolite production at this protein concentration and time (not shown). Similar to cellular excretion of isoflavone conjugates, formation rates of glycitein glucuronide and sulfate (15.2 and 4.6 pmol/min per monolayer, respectively) were the highest among these six isoflavones. In addition, formation rates of genistein, glycitein, biochanin A, and formononetin glucuronides were also much higher (3.0 to 3.9-fold) than the formation rates of their sulfates (P<0.05). In contrast, the sulfation rate of daidzein was higher (20%, P<0.05) than its glucuronidation rate but the difference (20%) was much smaller than for prunetin (120% higher sulfation rate, P<0.05).
Effects of structural change on the formation and excretion of isoflavone conjugates. Genistein and its analogs (10 μM) were incubated in the glucuronidation or sulfation reaction mixture solution with cell lysate (about 0.7 mg/ml) for 120 min. Formation rates (pmol/min per mg of protein) of glucuronidated and sulfated isoflavones were determined and plotted along with the corresponding excretion rates. Each bar represents the average of three determinations, and the error bars are the standard deviations of the means. *Significant difference between excretion rate and formation rate, Student’s t-test
To facilitate the comparison of formation and excretion, both the formation rate (picomoles per minute per milligram) and excretion rate were converted to the unit of picomoles per minute per monolayer. When excretion rates were plotted against formation rates, formation rates were the rate-limiting step in cellular excretion for all the isoflavone conjugates except for genistein glucuronide, whose formation and excretion rates were similar.
Discussion
Many isoflavones undergo extensive conjugated metabolism in rats and humans [4, 5, 15, 18]. The conjugated isoflavones are present at a much higher concentrations than their aglycone forms in human blood [4, 18]. In a rat perfusion model, genistein conjugates but not intact genistein can be detected in blood [5]. Our previous studies have also indicated that the intestine is the main site of genistein conjugation and that intestinal conjugation enables enteric recycling of genistein [5]. In the present study, we compared intestinal disposition of six isoflavones. This study focused on the effects of structure on the excretion of conjugated metabolites, mechanisms of conjugated excretion, and the determination of the rate-limiting step in the cellular excretion of isoflavone conjugates.
Both the cell lysate model and the intact Caco-2 cell culture model were used for the present study because together they can provide more useful information than either model alone. The cell lysate model is best used to determine the formation rates that reflect the metabolic rate of a compound. The intact cell culture model is best used to determine the cellular excretion of metabolites. Excretion is a more complex process than metabolite formation, but is more reflective of what may occur in vivo. This is because multiple metabolic pathways are present in the cells and excretion may limit the formation of a particular metabolite [11]. In other words, conjugated metabolites inside the cytosolic domain must be efficiently pumped out of the cells to achieve rapid excretion. Accumulation of metabolites inside cells due to lack of excretion could cause “product inhibition” of the enzyme reaction or lead to reconversion back to the aglycones via the action of hydrolases (e.g., glucuronidase and sulfatase). Therefore, the intact cell model can be used to measure the coupling effects of conjugation enzyme and efflux transporters.
Our studies demonstrated that the transport rates of intact isoflavones (the parent compounds, aglycone) in the Caco-2 cell model were all rapid but there were substantial differences in the apparent rate of transport (Fig. 2). Many isoflavones are also extensively metabolized during transport and there was no apparent relationship between total conjugate excretion rates and apparent rates of transport or permeabilities. However, if we defined the total uptake rates of isoflavones as rates of intact isoflavone transport (Fig. 2) plus total rates of conjugate excretion, since conjugating enzymes are present inside the cells (Fig. 4b), the highest uptake was achieved with glycitein, followed by biochanin A, genistein, daidzein, and formononetin. Prunetin was not used for comparison because it was used at a lower concentration. Its total rate of uptake would have been slightly higher than that of formononetin if we assume that a doubling of concentration will result in a doubling of transport rates and excretion rates. This result demonstrated that changes in isoflavone structure would affect total uptake and cellular excretion of metabolites.
The polarization of the excretion rates of conjugates was also highly dependent on the isoflavone structure. All isoflavone glucuronides except biochanin A glucuronide were mainly excreted to the basolateral side, whereas biochanin A glucuronide were excreted equally to both sides (Fig. 4). In addition, the sulfates of three of the six isoflavones (biochanin A, formononetin, and prunetin) were mainly excreted apically, whereas daidzein sulfate was mainly to the basolateral side. The remaining two isoflavone sulfates were not preferentially distributed at this concentration (Fig. 4).
In order to determine the rate-limiting step of isoflavone conjugate excretion at 10 μM for all isoflavones except prunetin, we determined the formation and excretion rates and normalized these rates to a “per monolayer” basis (Fig. 6). The glucuronide formation rate was much lower than the excretion rate for all isoflavones except genistein, whose formation rate was similar to its excretion rate. Assuming the lower rate was the rate-limiting step, glucuronide formation was the rate-limiting step for all isoflavones except genistein. For isoflavone sulfates, the sulfate formation rate always represented the rate-limiting step. Taken together, these results indicate that conjugate formation is the rate-limiting step in the cellular excretion of isoflavone conjugates. Coupling of the conjugating enzymes and efflux transporters determines the rate and direction of conjugate excretion, based on the assumption that these hydrophilic conjugates are evenly distributed within the cellular water space.
To further study the effect of structural change on conjugation, we sought to determine how a change in the number and position of hydroxy groups would affect their conjugation pathways. Previous studies has shown that isoflavone glucuronidation mostly occurs at the 7-hydroxyl position whereas sulfation mostly occurs at the 4′-hydroxy group [12, 18]. In the present study, blockage of the 4′-hydroxyl position diminished but did not prevent sulfation in the Caco-2 cell lysate when the only metabolic pathway was sulfation (because of the presence of co-enzyme PAPS). For example, biochanin A and formononetin were both sulfated. However, the rate of sulfation was typically lower than the rate of glucuronidation for an isoflavone with 7-hydroxyl and 4′-methoxy groups (e.g., formononetin). These results suggest that sulfation could occur at a hydroxy group other than 4′-hydroxyl. In contrast to sulfation, glucuronidation appeared to mainly occur at the 7-hydroxy group since the only compound with a blocked 7-hydroxy group (i.e., prunetin) was very poorly glucuronidated (Fig. 6).
In the intact cell culture model, in which an isoflavone could either be glucuronidated or sulfated, the preference for glucuronidation (at the 7-hydroxy group) over sulfation (at the 4′-hydroxy group) was more obvious. For example, excretion rates of the glucuronides of biochanin A and formononetin (both lacking the 4′-hydroxy group) were much higher than that of their sulfates (Fig. 6). In contrast, the excretion rate of prunetin sulfate was more than double that of prunetin glucuronide. For isoflavones which have both hydroxy groups (7 and 4′ positions) available (genistein and glycitein), the main excretion products were also glucuronides. Taken together, these results indicate a preference for glucuronidation over sulfation for the six isoflavones at this concentration. In addition, the 7-hydroxy group remains a major site of isoflavone glucuronidation, whereas the 4′-hydroxy group is only one of the sites for isoflavone sulfation since compounds without a 4′-hydroxy group were sulfated at approximately the same rates.
We next determined the mechanisms responsible for the conjugate efflux employing the MRP inhibitor LTC4 and the OAT inhibitor estrone sulfate. LTC4 inhibited the apical and basolateral efflux of five isoflavone glucuronides but not that of prunetin glucuronide (Fig. 5a, b). Estrone sulfate was similarly effective in inhibiting the efflux of isoflavone glucuronides from both the apical and the basolateral domains, except it did not change the apical efflux of prunetin glucuronide (Fig. 5a, b). These results suggest that excretion of isoflavone glucuronides is mediated by MRP and OAT. Based on earlier observations that Caco-2 cells mostly express MRP2 and MRP3 with low expression of MRP1 and MRP5 [7, 22], and that MRP3 and MRP2 are located at the basolateral and apical membranes of the Caco-2 cells [2, 3, 22]. MRP3 and MRP2 are probably responsible for the efflux of isoflavone glucuronides since they have been shown to be involved in the excretion of flavone conjugates [11, 16, 21, 23]. On the other hand, OAT4 is probably also involved in the cellular excretion of isoflavone glucuronides since this organic efflux transporter is capable of exporting cellular organic anions [17].
The effects of these two efflux transporter inhibitors on sulfate excretion were mostly similar to their effects on glucuronide excretion. However, LTC4 was not effective in inhibiting the efflux of formononetin and prunetin sulfates, whereas estrone sulfate was not effective in inhibiting the basolateral efflux of formononetin sulfate. Estrone sulfate was, somewhat surprisingly, highly effective against the apical and basolateral excretion of prunetin conjugates, inhibiting its excretion by more than 80% from both sides.
Taken together, the results of our study clearly demonstrate that human intestinal Caco-2 cells can form isoflavone conjugates, and the conjugated metabolites of isoflavones are excreted into both the apical and basolateral sides. Our results also indicate that the formation of conjugates and excretion of conjugated metabolites are coupled, which is an enabling process for enteric recycling [15]. Together with enterohepatic recycling proposed previously by other investigators, these recycling mechanisms help explain the pharmacokinetic behavior of isoflavonoids. In other words, as a consequence of this recycling, the apparent plasma half-lives of isoflavonoids (e.g., genistein) after oral administration are greatly increased (typically more than 5 h [4, 6, 19, 20]) when compared to i.v. administration (typically less than 1 h [6]). In essence, coupling-enabled recycling processes (e.g., enteric and enterohepatic recycling) are critical for the biological effects of isoflavonoids, because they allow these molecules to stay in vivo for a much longer period of time. The prolonged presence in the systemic circulation enables these compounds to display prolonged antioxidant effects in vivo, and provides the opportunity for their conversion into more active aglycones at the target tissues by the action of conjugate hydrolases (i.e., sulfatase and glucuronidase).
In summary, our results suggest that changes in the structure of isoflavones affects the type of conjugates formed, and rate of conjugate excretion. In human intestinal Caco-2 cells, glucuronidation primarily occurs at the 7-hydroxyl position and is the main metabolic pathway. On the other hand, sulfation appears to occur at positions in addition to the 4′-hydroxyl position as described in the literature and is the secondary metabolic pathway for isoflavones other than prunetin. Conjugated metabolites are excreted via the action of MRPs and OATs, since their excretion was inhibited by prototypical inhibitors of these efflux transporters.
Abbreviations
- HBSS:
-
Hank’s balanced salt solution
- LTC4 :
-
Leukotriene C4
- MRP:
-
Multidrug resistance related protein
- OAT:
-
Organic anion transporter
- PAPS:
-
3′-Phosphoadenosine 5′-phosphosulfate
- SULT:
-
Sulfotransferases
- UDPGA:
-
Uridine diphosphoglucuronic acid
- UGT:
-
Uridine diphosphate-5′-glucuronosyltransferase or UDP-glucuronosyltransferase
References
Birt DF, Hendrich S, Wang W (2001) Dietary agents in cancer prevention: flavonoids and isoflavonoids. Pharmacol Ther 90:157–177
Bock KW, Eckle T, Ouzzine M, Fournel-Gigleux S (2000) Coordinate induction by antioxidants of UDP-glucuronosyltransferase UGT1A6 and the apical conjugate export pump MRP2 (multidrug resistance protein 2) in Caco-2 cells. Biochem Pharmacol 59:467–470
Bock-Hennig BS, Kohle C, Nill K, Bock KW (2002) Influence of t-butylhydroquinone and beta-naphthoflavone on formation and transport of 4-methylumbelliferone glucuronide in Caco-2/TC-7 cell monolayers. Biochem Pharmacol 63:123–128
Busby MG, Jeffcoat AR, Bloedon LT, Koch MA, Black T, Dix KJ, Heizer WD, Thomas BF, Hill JM, Crowell JA, Zeisel SH (2002) Clinical characteristics and pharmacokinetics of purified soy isoflavones: single-dose administration to healthy men. Am J Clin Nutr 75:126–136
Chen J, Lin H, Hu M (2003) Metabolism of flavonoids via enteric recycling: role of intestinal disposition. J Pharmacol Exp Ther 304:1228–1235
Coldham NG, Zhang AQ, Key P, Sauer MJ (2003) Absolute bioavailability of [14C] genistein in the rat; plasma pharmacokinetics of parent compound, genistein glucuronide and total radioactivity. Eur J Drug Metab Pharmacokinet 27:249–258
Hirohashi T, Suzuki H, Chu XY, Tamai I, Tsuji A, Sugiyama Y (2000) Function and expression of multidrug resistance-associated protein family in human colon adenocarcinoma cells (Caco-2). J Pharmacol Exp Ther 292:265–270
Hu M, Chen J, Tran D, Zhu Y, Leonardo G (1994) The Caco-2 cell monolayers as an intestinal metabolism model: metabolism of dipeptide Phe-Pro. J Drug Target 2:79–89
Hu M, Chen J, Zhu Y, Dantzig AH, Stratford RE Jr, Kuhfeld MT (1994) Mechanism and kinetics of transcellular transport of a new β-lactam antibiotic loracarbef across a human intestinal epithelial model system (Caco-2). Pharm Res 11:1405–1413
Hu M, Li Y, Penman BW, Huang SM, Thummel K, Crespi CL (1999) Morphological and metabolic characterization of Caco-2 cells expressing high levels of cDNA-derived cytochrome P4503A4. Pharm Res 16:1352–1359
Hu M, Chen J, Lin H (2003) Metabolism of flavonoids via enteric recycling: mechanistic studies of disposition of apigenin in the Caco-2 cell culture model. J Pharmacol Exp Ther 307:314–321
King RA, Bursill DB (1998) Plasma and urinary kinetics of the isoflavones daidzein and genistein after a single soy meal in humans. Am J Clin Nutr 67:867–872
King RA, Broadbent JL, Head RJ (1996) Absorption and excretion of the soy isoflavone genistein in rats. J Nutr 126:176–182
Kurzer M, Xu X (1997) Dietary phytoestrogens. Annu Rev Nutr 17:353–381
Liu Y, Hu M (2002) Absorption and metabolism of flavonoids in the Caco-2 cell culture model and a perfused rat intestinal model. Drug Metab Dispos 30:370–377
O’Leary KA, Day AJ, Needs PW, Mellon FA, O’Brien NM, Williamson G (2003) Metabolism of quercetin-7- and quercetin-3-glucuronides by an in vitro hepatic model: the role of human beta-glucuronidase, sulfotransferase, catechol-O-methyltransferase and multi-resistant protein 2 (MRP2) in flavonoid metabolism. Biochem Pharmacol 65:479–491
Sekine T, Cha SH, Endou H (2000) The multispecific organic anion transporter (OAT) family. Pflugers Arch 440:337–350
Setchell KD, Brown NM, Desai P, Zimmer-Nechemias L, Wolfe BE, Brashear WT, Kirschner AS, Cassidy A, Heubi JE (2001) Bioavailability of pure isoflavones in healthy humans and analysis of commercial soy isoflavone supplements. J Nutr 131:1362S–1375S
Setchell KD, Faughnan MS, Avades T, Zimmer-Nechemias L, Brown NM, Wolfe BE, Brashear WT, Desai P, Oldfield MF, Botting NP, Cassidy A (2003) Comparing the pharmacokinetics of daidzein and genistein with the use of 13C-labeled tracers in premenopausal women. Am J Clin Nutr 77:411–419
Shelnutt SR, Cimino CO, Wiggins PA, Ronis MJ, Badger TM (2002) Pharmacokinetics of the glucuronide and sulfate conjugates of genistein and daidzein in men and women after consumption of a soy beverage. Am J Clin Nutr 76:588–594
Vaidyanathan JB, Walle T (2001) Transport and metabolism of the tea flavonoid (−)-epicatechin by the human intestinal cell line Caco-2. Pharm Res 18:1420–1425
Walgren RA, Lin JT, Kinne RK, Walle T (2000) Cellular uptake of dietary flavonoid quercetin 4′-β-glucoside by sodium-dependent glucose transporter SGLT1. J Pharmacol Exp Ther 294:837–843
Walle UK, Galijatovic A, Walle T (1999) Transport of the flavonoid chrysin and its conjugated metabolites by the human intestinal cell line Caco-2. Biochem Pharmacol 58:431–438
Yang CS, Landau JM, Huang MT, Newmark HL (2001) Inhibition of carcinogenesis by dietary polyphenolic compounds. Annu Rev Nutr 21:381–406
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, J., Lin, H. & Hu, M. Absorption and metabolism of genistein and its five isoflavone analogs in the human intestinal Caco-2 model. Cancer Chemother Pharmacol 55, 159–169 (2005). https://doi.org/10.1007/s00280-004-0842-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-004-0842-x