Abstract
The arbitrary threshold of 5 × 109/L chronic lymphocytic leukemia (CLL)-like lymphocytes differentiates monoclonal B lymphocytosis (MBL) from CLL. There are no prospective studies that search for the optimal cut-off of monoclonal lymphocytes able to predict outcome and simultaneously analyze the prognostic value of classic, immunophenotypic, and cytogenetic variables in patients with asymptomatic clonal CLL lymphocytosis (ACL), which includes MBL plus Rai 0 CLL patients. From 2003 to 2010, 231 ACL patients were enrolled in this study. Patients with 11q deletion and atypical lymphocyte morphology at diagnosis had shorter progression-free survival (PFS) (p = 0.007 and p = 0.015, respectively) and treatment-free survival (TFS) (p = 0.009 and p = 0.017, respectively). Elevated beta-2 microglobulin (B2M) also correlated with worse TFS (p = 0.002). The optimal threshold of monoclonal lymphocytes independently correlated with survival was 11 × 109/L (p = 0.000 for PFS and p = 0.016 for TFS). As conclusion, monoclonal lymphocytosis higher than 11 × 109/L better identifies two subgroups of patients with different outcomes than the standard cut-off value of 5 × 109/L. Atypical lymphocyte morphology, 11q deletion and elevated B2M had a negative impact on the survival in ACL patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Main diagnostic criteria for B-cell chronic lymphocytic leukemia (CLL) according to 2008 NCI Guidelines is the presence of at least 5 × 109/L monoclonal B lymphocytes in the peripheral blood with the characteristic CLL immunophenotype by flow cytometry during a period longer than 3 months. Asymptomatic patients who present smaller monoclonal lymphocyte population with a CLL phenotype who do not have lymphadenopathies, organomegalies, or cytopenias shall be classified as monoclonal B lymphocytosis (MBL) [1]. However, the threshold of 5 × 109/L monoclonal lymphocytes is arbitrary and it has not been properly validated in prospective studies.
Two different types of MBL can be identified. Individuals with normal blood counts in whom monoclonal lymphocytes are detected during population screening for epidemiological purposes (low count MBL) show a very small probability, if any, of progression to CLL [2]. On the other hand, those patients who are referred for hematological evaluation because of lymphocytosis (clinical MBL) will develop CLL and require treatment at a rate of 1.1 % per year [3]. According to previous reports, clinical MBL is very similar to Rai 0 CLL regarding microRNA and immunogenetic profiles [4–6]. These data suggest that clinical MBL and Rai 0 CLL should be seen as a unique group of patients in different phases of the same disease who presents asymptomatic clonal CLL-like lymphocytosis (ACL).
Some studies have analyzed the prognostic value of chromosomal alterations by fluorescent in situ hybridization (FISH) while others have searched the most appropriate monoclonal lymphocyte count able to predict outcome in ACL [3, 6–11]. None of them, though, have addressed both issues and also analyzed the prognostic role of classic variables in a prospective and multivariate way in an ACL cohort.
We describe a series of 307 consecutive patients presenting with CLL clonal lymphocytes in one single reference center for hematological malignancies and belonging to the same geographic area. A prospective follow-up was focused in 231 ACL patients without any other sign of disease. The main objectives were to find a significant monoclonal lymphocyte count able to predict outcome as well as identify other prognostic variables for PFS and TFS in this group of patients. In this study, all laboratory and biological parameters tested are routinely used in clinical practice in CLL and were available in virtually all cases at diagnosis.
Patients and methods
Patients
Three hundred and seven consecutive untreated patients diagnosed with MBL, small lymphocytic lymphoma (SLL), or CLL in one single center from January 2003 to December 2010 were prospectively included in this study. Clinical and laboratory data, Rai staging, immunophenotyping, and FISH cytogenetics were reported at diagnosis. Analysis of prognostic factors and survival was performed in the cohort of 231 patients with ACL, which included MBL plus Rai 0 CLL patients.
The study was approved by our Institution’s Ethic Committee Board, and all patients provided informed consent. All procedures followed were in accordance with the Helsinki Declaration of 1975, as revised in 2008.
Classification into MBL, SLL, or CLL was made according to 2008 NCI Guidelines and based in absolute number of monoclonal B lymphocytes by flow cytometry [1]. Those cases diagnosed before 2008 had their classification retrospectively reviewed.
Patients were prospectively followed up for progression of disease, need for treatment, or death. Treatment was initiated following standard guidelines criteria [1, 12].
Methods
The following classic variables were analyzed for prognosis: atypical lymphocyte morphology, hypogammaglobulinemia, monoclonal protein, elevated lactate dehydrogenase (LDH), and elevated beta-2 microglobulin (B2M). Lymphocyte morphology was evaluated in all cases in the same laboratory and by the same experts according to the French-American-British (FAB) classification of leukemias [13]. Atypical CLL included two morphology subtypes: the CLL/prolymphocytic leukemia, with more than 10 % circulating prolymphocytes and the mixed subtype, with more than 15 % elements with lymphoplasmacytic aspect and cleaved cells.
Flow cytometry, FISH, and conventional cytogenetic techniques were performed as previously described [14]. CD38 was considered positive when at least 30 % of pathologic lymphocytes expressed the corresponding antibody, while ZAP70 was considered positive above 20 %. FISH results were divided into the five hierarchical categories classically described by Dohnner: normal FISH, presence of isolated 13q deletion, presence of 12 trisomy alone or with 13q deletion, presence of 11q deletion with or without other abnormality but 17p deletion, and presence of 17p deletion with or without other abnormality [15]. The presence of more than 10 % nuclei with the referred abnormality was considered a positive result.
Statistical analysis
Crosstabs were used to compare the frequencies of variables between groups. Comparison of categorical variables was performed using X 2 test.
Progression-free survival (PFS) was defined as the time between diagnosis and the first sign of disease progression as described previously [14]. Treatment-free survival (TFS) was defined as the time from diagnosis until the first treatment required. Survival curves were plotted using Kaplan-Meier’s method, and log-rank test was used to identify differences in survival between subgroups. Multivariate analysis for survival was performed with a Cox regression model.
Receiver operating characteristic (ROC) curves for censored survival data were used to test the various thresholds of monoclonal lymphocyte count for PFS and TFS [16].
All analyses were performed at a 95 % confidence interval and differences were considered statistically significant when p value was less than 0.05. Statistical analysis was made using SPSS software, version 17.0 (SPSS Inc., Chicago, IL, USA).
Results
Clinical, laboratory, and biological data of the 307 patients at diagnosis did not differ from any CLL series previously reported (data not shown). Diagnosis and Rai staging data are shown in Table 1.
Two hundred thirty-one patients had ACL, which included 81 patients classified as MBL plus 150 patients classified as Rai 0 CLL. One hundred forty-five (62.8 %) patients were male and median age at diagnosis was 68 years (limits 27–93 years). Table 2 shows prognostic factors at diagnosis in patients with ACL and differences between Rai 0 CLL and MBL subgroups. Deletions of 13q and 11q were significantly more frequent in Rai 0 CLL than in MBL patients while a normal FISH was found in a higher proportion of MBL patients.
With a median follow-up time of 61 months, overall survival (OS) for 231 ACL patients was 86.5 %. Seventeen (7.4 %) patients died and only 3 (1.3 %) due to CLL related causes. Forty-six (19.9 %) patients presented disease progression and 36 (15.6 %) patients required treatment during the follow-up period.
Variables included in the analysis for prognostic factors were: hypogammaglobulinemia, monoclonal protein, atypical lymphocyte morphology, elevated LDH, elevated B2M, positive CD38, and positive ZAP70 by flow cytometry, abnormal conventional karyotype, and FISH categorical results. Elevated LDH, elevated B2M, atypical lymphocyte morphology, CD38 expression, abnormal karyotype, and 11q deletion by FISH had all a negative impact on PFS and TFS in univariate analysis. Atypical lymphocyte morphology and 11q deletion were correlated with worse PFS and TFS in the multivariate analysis. Elevated B2M was also significant for TFS (Table 3).
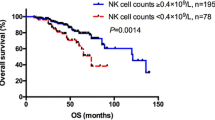
To search for the optimal monoclonal lymphocyte count able to predict survival in the ACL patients, we measured the risk of each increase of 1 × 109/L monoclonal lymphocytes regarding to PFS and TFS, starting from 1 × 109/L until 20 × 109/L cells. According to this analysis, 11 × 109/L was the monoclonal lymphocyte threshold able to identify two groups with different PFS and TFS in our series (Figs. 1 and 2). This did not happen either when the chosen value was the standard 5 × 109/L or with any other limit beyond 11 × 109/L cells.
When included as a new variable in the multivariate analysis, the cut-off of 11 × 109/L monoclonal lymphocytes had an independent prognostic impact in both PFS (OR 3.9, CI 1.8–8.3, p = 0.000) and TFS (OR 2.9, CI 1.2–7.3, p = 0.016) together with the other variables described before.
Discussion
Some retrospective studies analyzed the prognostic value of monoclonal lymphocyte count in ACL patients but did not simultaneously test the role of CLL cytogenetic prognostic factors [8–10]. Other few authors explored the prognostic role of cytogenetic, but not of classic variables in ACL patients [6, 11, 17]. In our study, we search for the optimal monoclonal lymphocyte cut-off able to predict survival in ACL patients and prospectively analyze the correlation of classic, immunophenotypic, and cytogenetic variables with outcome.
In our ACL series, no differences were found between MBL and Rai 0 CLL regarding the frequency of classic variables at diagnosis. However, among MBL patients, normal FISH was more frequent (p = 0.007) than in Rai 0 CLL, while cases with Rai 0 CLL showed a higher frequency of either 13q deletion (p = 0.027) or 11q deletion (p = 0.018) than MBL cases. In a study published before the 2008 NCI Guidelines [1], Tsimberidou et al. [18] retrospectively analyzed the biological characteristics of more than 2000 patients with CLL and separated them according to the limit of 5 × 109/L absolute lymphocytes (not monoclonal). Despite being a heterogeneous group of patients, they found karyotype and FISH abnormalities to happen in both groups but in a higher frequency in patients with more than 5 × 109/L lymphocytes. Kern et al. [11] also reported a higher frequency of chromosomal aberrations by FISH in patients with Rai 0 CLL comparing with MBL while Rossi et al. [17] and Morabito et al. [6] described a lower prevalence of 11q and 17p deletions in MBL cases. On the other hand, clinical MBL seem to be very similar to Rai 0 CLL regarding to microRNA signature and immunogenetic profile [5, 6]. Altogether, these findings suggest that clinical MBL and Rai 0 CLL could simply correspond to different phases of the same disease and that, the larger the clone, the more frequent the acquisition of cytogenetic abnormalities.
We have then analyzed the impact of different variables in the survival of ACL as a unique group, based on the findings described above. Deletion of 11q was the only cytogenetic parameter associated with worse PFS and TFS. In CLL patients, it has a clear correlation with advanced disease, extensive lymph node involvement, and a shorter time to treatment (TTT) [15, 19, 20]. In patients with early-stage CLL expansions, as in our cohort, this cytogenetic aberration has no well-established role. Rossi et al. [17] found 17p deletion and 12 trisomy, but not 11q deletion, to be related to worse PFS and TFS in a prospective series of MBL patients. On the other hand, Kern et al. [11] reported a shorter TTT in patients with 11q deletion in both MBL and Rai 0 CLL cohorts. In a previous series described by our group with a shorter follow-up and a smaller number of cases, we found 17p deletion, but not 11q deletion, to show prognostic value in multivariate analysis regarding PFS in Rai 0–1 CLL patients [14]. It is possible that 17p deletion does not have a prognostic relevance in our present study because it is found with a very low frequency in these first steps of monoclonal lymphocytosis as it occurs later in lymphocyte clonal evolution.
Regarding classic prognostic factors, atypical morphology was a strong predictor of both PFS and TFS. In CLL, it has been classically correlated with a worse outcome. In 1997, Oscier et al. [21] reported its association with disease progression, and in 2006, Schwarz et al. [22] described its correlation with a shorter OS. Since then, no studies have been published addressing this subject. We think it is an interesting finding that a cheap and easily available tool as microscopic examination of lymphocyte morphology can have a strong prognostic value in ACL patients. Also interestingly, B2M was the other classic variable with significance in multivariate analysis regarding TFS. The negative impact of this prognostic marker in CLL was first described in 1996 by Hallek et al. [23]. Lately, other authors also reported its correlation with survival in patients diagnosed with CLL in all stages of the disease [17, 19, 24]. More recently, Letestu et al. [25] have proposed a prognostic score based on routine parameters with significance for PFS in a multivariate analysis of 339 Binet A stage CLL patients and have found elevated B2M to be one of the strongest variables.
We also have searched for the most appropriate monoclonal lymphocyte threshold able to identify different prognostic subgroups within ACL group and found that patients with more than 11 × 109/L at diagnosis had significantly worse PFS and TFS. Other authors have described similar values of monoclonal lymphocytes to be correlated with outcome in retrospective cohorts. Molica et al. [10] found the limit of 10 × 109/L absolute B lymphocytes to be associated with TTT in a multivariate analysis, and Scarfò et al. [7] described the same cut-off regarding PFS and TFS while Shanafelt et al. [8] reported 11 × 109/L to be the one that predicted TFS an OS in their series. Only one prospective study correlated the 5 × 109/L absolute B lymphocytes cut-off with the likelihood of progression when three or more unfavorable prognostic variables were present in the same patient [6]. Although most of these studies are based in the absolute (and not monoclonal) B lymphocyte count, their data suggest that maybe ACL patients should be stratified into two prognostic subgroups based in a higher threshold of monoclonal lymphocytes.
As conclusion, MBL and Rai 0 CLL correspond to a single group of patients in different phases of the same disease. In clinical practice, the identification of patients with ACL who will develop progressive CLL and require treatment is difficult. Monoclonal lymphocytosis higher than 11 × 109/L better identifies two subgroups with different outcomes than the standard cut-off value of 5 × 109/L. Moreover, atypical lymphocyte morphology and 11q deletion by FISH at diagnosis correlated to progression of disease and need for treatment. Elevated B2M also had a negative impact in TFS.
References
Hallek M, Cheson BD, Catovsky D, Caligaris-Cappio F, Dighiero G, Döhner H, Hillmen P, Keating MJ, Montserrat E, Rai KR, Kipps TJ (2008) Guidelines for the diagnosis and treatment of chronic lymphocytic leukemia: a report from the International Workshop on Chronic Lymphocytic Leukemia updating the National Cancer Institute-Working Group 1996 guidelines. Blood 111:5446–5456
Fazi C, Scarfò L, Pecciarini L, Dagklis A, Janus A, Talarico A, Scielzo C, Sala C, Toniolo D, Caligaris-Cappio F, Ghia P (2011) General population low-count CLL-like MBL persists over time without clinical progression, although carrying the same cytogenetic abnormalities of CLL. Blood 118:6618–6625
Rawstron AC, Bennet FL, O’Connor SJM, Kwok M, Fenton JA, Plummer M, de Tute R, Owen RG, Richards SJ, Jack AS, Hillmen P (2008) Monoclonal B-cell lymphocytosis and chronic lymphocytic leukemia. N Engl J Med 359:575–583
Dagklis A, Fazi C, Sala C, Cantarelli V, Scielzo C, Massacane R, Toniolo D, Caligaris-Cappio F, Stamatopoulos K, Ghia P (2009) The immunoglobulin gene repertoire of low-count chronic lymphocytic leukemia (CLL)-like monoclonal B lymphocytosisis different from CLL: diagnostic implications for clinical monitoring. Blood 114:26–32
Vardi A, Dagklis A, Scarfò L et al (2013) Immunogenetics shows that not all MBL are equal: the larger the clone, the more similar to CLL. Blood 121:4521–4528
Morabito F, Mosca L, Cutrona G et al (2013) Clinical monoclonal B lymphocytosis versus Rai 0 chronic lymphocytic leukemia: a comparison of cellular, cytogenetic, molecular and clinical features. Clin Cancer Res 19:5890–5900
Scarfò L, Zibellini S, Tedeschi A, Maura F, Neri A, Bertazzoni P, Sarina B, Nalli G, Motta M, Rossini F, Cortelezzi A, Montillo M, Orlandi E, Ghia P (2012) Impact of B-cell count and imaging screening in cMBL: any need to revise the current guidelines? Leukemia 26:1703–1707
Shanafelt TD, Kay NE, Jenkins G, Call TG, Zent CS, Jelinek DF, Morice WG, Boysen J, Zakko L, Schwager S, Slager SL, Hanson CA (2009) B-cell count and survival: differentiating chronic lymphocytic leukemia from monoclonal B-cell lymphocytosis based on clinical outcome. Blood 113:4188–4196
Shanafelt TD, Kay NE, Rabe KG, Call TG, Zent CS, Maddocks K, Jenkins G, Jelinek DF, Morice WG, Boysen J, Schwager S, Bowen D, Slager SL, Hanson CA (2009) Brief report: natural history of individuals with clinically recognized monoclonal B-cell lymphocytosis compared with patients with Rai 0 chronic lymphocytic leukemia. J Clin Oncol 27:3959–3963
Molica S, Mauro FR, Giannarelli D, Lauria F, Cortelezzi A, Brugiatelli M, Liso V, Cuneo A, Foà R (2011) Differentiating chronic lymphocytic leukemia from monoclonal B-lymphocytosis according to clinical outcome: on behalf of GIMEMA chronic lymphoproliferative diseases working group. Hematologica 96:277–283
Kern W, Bacher U, Haferlach C, Dicker F, Alpermann T, Schnittger S, Haferlach T (2012) Monoclonal B-cell lymphocytosis is closely related to chronic lymphocytic leukaemia and may be better classified as early-stage CLL. Br J Haematol 157:86–96
Cheson BD, Bennett JM, Grever M, Kay N, Keating MJ, O’Brien S, Rai KR (1996) National Cancer Institute-Sponsored Working Group guidelines for chronic lymphocytic leukemia: revised guidelines for diagnosis and treatment. Blood 87:4990–4997
Bennett JM, Catovsky D, Daniel MT, Flandrin G, Galton DA, Gralnick HR, Sultan C (1989) Proposals for the classification of chronic (mature) B and T lymphoid leukaemias. French-American-British (FAB) Cooperative Group. J Clin Pathol 42:567–584
Oliveira AC, De La Banda E, Domingo-Domènech E, Encuentra M, Mercadal S, Domingo A, Alonso E, Espinet B, Grau J, De Sevilla AF, Gonzalez-Barca E (2011) Prospective study of clinical and biological prognostic factors at diagnosis in patients with early stage B-cell chronic lymphocytic leukemia. Leuk Lymph 52:429–435
Dohner H, Stilgenbauer S, Benner A, Leupolt E, Kröber A, Bullinger L, Döhner K, Bentz M, Lichter P (2000) Genomic aberrations and survival in chronic lymphocytic leukaemia. N Engl J Med 343:1910–1916
Heagerty PJ, Lumley T, Pepe MS (2000) Time-dependent ROC curves for censored survival data and a diagnostic marker. Biometrics 56:337–344
Rossi D, Sozzi E, Puma A et al (2009) The prognosis of clinical monoclonal B cell lymphocytosis differs from prognosis of Rai 0 chronic lymphocytic leukaemia and is recapitulated by biological risk factors. Br J Haematol 146:64–75
Tsimberidou AM, Wen S, O’Brien S et al (2007) Assessment of chronic lymphocytic leukemia and small lymphocytic lymphoma by absolute lymphocyte counts in 2126 patients: 20 years of experience at the University of Texas MD Anderson Cancer Center. J Clin Oncol 25:4648–4656
Wierda WG, O’Brien S, Wang X et al (2011) Multivariable model for time to first treatment in patients with chronic lymphocytic leukemia. J Clin Oncol 29:4088–4095
Dohner H, Stilgenbauer S, James MR, Benner A, Weilguni T, Bentz M, Fischer K, Hunstein W, Lichter P (1997) 11q deletions identify a new subset of B-cell chronic lymphocytic leukemia characterized by extensive nodal involvement and inferior prognosis. Blood 89:2516–2522
Oscier DG, Matutes E, Copplestone A, Pickering RM, Chapman R, Gillingham R, Catovsky D, Hamblin TJ (1997) Atypical lymphocyte morphology: an adverse prognostic factor for disease progression in stage A CLL independent of trisomy 12. Br J Haematol 98:934–939
Schwarz J, Mikulenkova D, Cermakova K et al (2006) Prognostic relevance of the FAB morphological criteria in chronic lymphocytic leukemia: correlations with IgVh gene mutational status and other prognostic markers. Neoplasma 53:219–225
Hallek M, Wanders L, Ostwald M, Busch R, Senekowitsch R, Stern S, Schick HD, Kuhn-Hallek I, Emmerich B (1996) Serum beta(2)-microglobulin and serum thymidine kinase are independent predictors of progression-free survival in chronic lymphocytic leukemia and immunocytoma. Leuk Lymphoma 22:439–447
Delgado J, Pratt G, Phillips N, Briones J, Fegan C, Nomdedeu J, Pepper C, Aventin A, Ayats R, Brunet S, Martino R, Valcarcel D, Milligan D, Sierra J (2009) Beta2-microglobulin is a better predictor of treatment-free survival in patients with chronic lymphocytic leukaemia if adjusted according to glomerular filtration rate. Br J Haematol 145:801–805
Letestu R, Lévy V, Eclache V, Baran-Marszak F, Vaur D, Naguib D, Schischmanoff O, Katsahian S, Nguyen-Khac F, Davi F, Merle-Béral H, Troussard X, Ajchenbaum-Cymbalista F (2010) Prognosis of Binet stage A chronic lymphocytic leukemia patients: the strength of routine parameters. Blood 116:4588–4590
Acknowledgments
This study was supported by “Instituto de Salud Carlos III” (RTICC RD12/0036/0029).
Conflict of interest
The authors have no conflicts of interest to declare.
Funding
No funding was received for this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Oliveira, A.C., Fernández de Sevilla, A., Domingo, A. et al. Prospective study of prognostic factors in asymptomatic patients with B-cell chronic lymphocytic leukemia-like lymphocytosis: the cut-off of 11 × 109/L monoclonal lymphocytes better identifies subgroups with different outcomes. Ann Hematol 94, 627–632 (2015). https://doi.org/10.1007/s00277-014-2263-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-014-2263-1