Abstract
Antibody-mediated in vivo T cell depletion is common prior to unrelated (URD) or mismatched allogeneic stem cell transplantation (alloSCT) and optional in HLA-identical sibling (FAM) alloSCT. While anti-thymocyte globulin (ATG) is the current standard, alemtuzumab is an alternative. The optimal dose of alemtuzumab has not been defined. This retrospective analysis compares low-dose alemtuzumab with ATG in URD alloSCT and with no antibody in FAM alloSCT. Twenty-eight patients treated with alemtuzumab (10 mg; HLA mismatch, 20 mg) were matched to 28 patients who have either received ATG (URD) or no antibody (noAB) according to disease, disease stage, age, transplant type and risk state. Both groups were compared for engraftment, outcome, disease-free (DFS) and overall survival (OS), graft-versus-host disease (GvHD), freedom from GvHD (ffGvHD) and transplant-related mortality (TRM). No significant differences were found between the groups for leukocyte engraftment, GvHD, ffGvHD, TRM, DFS and OS. There was a trend for reduction of cGvHD by alemtuzumab (p = 0.05). A transplant-type stratified subanalysis consolidated equivalency of alemtuzumab and ATG in URD-SCT and indicates possible superiority of low-dose alemtuzumab compared to noAB in FAM-SCT. Low-dose alemtuzumab, as part of conditioning regimen prior to alloSCT, is safe and comparable to standard ATG. Prospective trials, particularly comparing alemtuzumab vs. noAB in FAM alloSCT, should be conducted.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Antibodies against T lymphocytes have become an integral part of conditioning regimens for allogeneic stem cell transplantation (alloSCT) [1]. Application of T cell-depleting antibodies as part of the conditioning regimens leads to depletion of donor- and recipient-derived T cells. This facilitates engraftment of donor haemopoiesis and reduces the risk of severe graft-versus-host disease (GvHD) [2]. Today, T cell-depleting antibodies are commonly used in transplantation of stem cells from unrelated and HLA–antigen mismatched donors. Their application is optional in HLA-matched sibling transplantation [1, 3].
Polyclonal anti-thymocyte globulin (ATG) generated by immunisation of rabbits or, formerly, also of horses is most often applied for T cell depletion prior to unrelated/HLA-mismatched alloSCT. An evolving alternative to ATG is alemtuzumab (MabCampath®) [4, 5].
Alemtuzumab is the humanised version of the monoclonal antibody Campath-1H which recognises the epitope CD52 [6, 7]. This antigen is expressed on T cells and distinct B cell populations. Campath has been used successfully during the late 1980s in higher doses to suppress the graft-versus-host reaction after alloSCT, mainly in chronic myeloid leukaemia (CML) patients. However, this was associated with a considerable high relapse rate of the underlying disease [8, 9], a significant increase of infectious complications after transplantation, and potentially also associated with a higher rejection rate [9, 10]. This led to withdrawal of Campath-1H from the conditioning regimen and its replacement by ATG [2]. However, the experience with Campath helped to identify the importance of donor-derived T cells for the control of disease after transplantation and to discover the graft-versus-leukaemia effects [11].
Although it is now well accepted that the benefit–risk ratio disfavours high doses of the anti-CD52 T cell-depleting antibody, data on lower doses of alemtuzumab are sparse, and the optimal dose of alemtuzumab has not yet been established in different settings of alloSCT. Indeed, alemtuzumab is administered at doses between 20 and 100 mg either intravenously or subcutaneously prior to alloSCT [12, 13]. With these doses of alemtuzumab, Kröger et al. found a reduction in acute GvHD but a higher relapse rate [14], while Norlin et al. reported less acute but more chronic GvHD [15]. Only in the setting of reduced intensity conditioning regimens has a dose reduction for alemtuzumab been investigated by two groups [16, 17, 18].
Few data are available comparing low doses of alemtuzumab with current standard T cell depletion using ATG in unrelated alloSCT. We now report pilot data of our transplant unit, which indicate that low-dose alemtuzumab (10 and 20 mg) for T cell depletion prior to alloSCT may be advantageous compared to the standard ATG regimen, especially in regard to treatment-associated adverse effects and a reduction of chronic GvHD.
Patients and methods
Patients and diagnoses
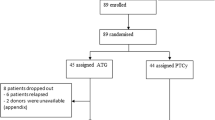
We treated 28 consecutive patients with alemtuzumab (MabCampath®, Genzyme, Cambridge, MA, USA) as part of the conditioning regimen from June 2009 until November 2010. The dose was 10 mg in HLA-matched transplantations (family and unrelated donors) and 20 mg in HLA-mismatched transplantations at day 1. The controls were 28 patients transplanted between July 1999 and May 2009. The switch to alemtuzumab was based on the promising data published by other groups [16]. These patients were matched to the patients from the investigational group according to age, diagnoses, disease state, high-risk state and transplant type. The median age was 52 years (range, 20–70) in the investigational group and 47 years (range, 20–67) in the control group (p = 0.15, chi2). The percentage of male gender was slightly higher in the control group compared to the alemtuzumab group (79 vs. 61 %, p = 0.39, Mann–Whitney U test (MWU)). Patients of the control group received either ATG-Fresenius® (30 mg/kg, Fresenius-Biotech, Munich, Germany) or Thymoglobin® (6 mg/kg, Genzyme, Cambridge, MA, USA) prior to unrelated or mismatched transplantation or no antibody (noAB) in case of HLA-identical sibling transplantation.
Diagnoses, conditioning and transplantation
In each group, 14 patients suffered from acute leukaemia, 1 patient from chronic myeloid leukaemia, 3 from multiple myeloma, and 5 and 3 from high-grade and indolent non-Hodgkin's lymphoma, respectively. Conditioning was myeloablative (MAC) in 22 patients with busulfan16(or i.v. equivalent)/cyclophosphamid120 ± VP1630 (n = 21) or total body irradiation (TBI)12Gy + cyclophosphamide120 (n = 1) [19]. Reduced intense conditioning (RIC; n = 34) was performed with TBI2Gy/fludarabin150 (n = 10), treosulfan36/fludarabin150 (n = 15) or melphalan100-140/fludarabin150 (n = 6; Table 1) [20, 21, 22]. In general, MAC was preferred in younger patients and in patients with aggressive disease, and RIC was chosen for elderly patients or patients with co-morbidity and for patients with less aggressive disease.
Seventeen patients were grafted from matched donors and 39 from unrelated donors. A serological HLA–antigen mismatch was present in nine cases and a molecular HLA-disparity in 22 transplantations. Ten male patients received a graft from a female donor, an ABO blood group disparity was evident in 28 transplantations and 11 CMV+ patients were grafted from a CMV− donor. Generally, an HLA mismatch was preferred about the CMV− to CMV+ graft constellation. All patients were grafted with granulocyte colony-stimulating factor (G-CSF)-mobilised peripheral stem cells (Table 1).
Supportive care, GvHD prophylaxis and haematological recovery
Third-generation quinolones were given as antibacterial prophylaxis. Systemic antifungal prophylaxis was initiated with fluconazole and switched at day +1 to either itraconazole or voriconazole. Also, metronidazole for anaerobic gut decontamination was initiated at day +1. Quinolones were discontinued after engraftment or at initiation of broad-spectrum antibiosis for treatment of infections. Metronidazole was discontinued at day +30, and antifungal prophylaxis was given until day +70, depending on absence of GvHD. All patients received acyclovir from day +1 at least until day +30 to prevent reactivation of herpes simplex virus. Pneumocystis jirovecii prophylaxis was conducted until day +180 either with TMP-SMZ or with aerosolised pentacarinate [23]. Anti-infective prophylaxis did not change over the observation time.
GvHD prophylaxis after transplantation was performed with cyclosporine A and short-course methotrexate. Cyclosporine A, in association with mycophenolate mofetil, was given after conditioning with TBI2Gy/fludarabine conditioning. In general, it was scheduled to discontinue GvHD prophylaxis between days +100 and +180.
Forty-one patients received G-CSF after transplantation to stimulate haemopoiesis. Erythrocytes were substituted when haemoglobin decreased below 8 mg/dl, and prophylactic transfusion of platelets was made in the case of platelets less than 10/nl. The haematological recovery was related to the day of stem cell infusion (day 0). Leukocyte engraftment was defined as the first of three consecutive days with an absolute leukocyte count exceeding 1.0/nl in the peripheral blood and platelet engraftment as the first day that the platelet count exceeded 20/nl, independent of platelet transfusions. The day of engraftment was ‘0’ when cells never declined below the threshold.
Acute and chronic GvHD were graded and treated according to international standard [24, 25]. Freedom from GvHD was calculated using log-rank test and Kaplan–Meier analysis. Events were defined as the onset of either acute or chronic GvHD or death. Overall and relapse-free survival was calculated using the log-rank test and the Kaplan–Meier analysis. For calculation of event-free survival, either relapse or death was defined as events.
Statistics
Data were collected using the computer software MS Office (Microsoft, Munich, Germany) and analysed with GraphPad Prism (GraphPad Software, San Diego, CA, USA). The statistical tests used were the chi2 test, MWU test, t test, log-rank test and Kaplan–Meier survival analysis. A p value <0.05 was considered as statistically significant.
Results
Engraftment and discharge/transfer from the transplant unit
The percentage of patients who have received G-CSF for stimulation of haemopoiesis after alloSCT was significantly higher in the control group than in the investigational group (100 vs. 46 %, p < 10−5, chi2; Table 1). The median dose of CD34+ cells transplanted was 7.1 (range, 2.7–11.3) in the alemtuzumab group and 5.29 (0.9–19.1) in the control group (p = 0.15). Patients engrafted with leukocytes at day +15 (median, range, 11–28 [alemtuzumab]; median, 9–31 [control], p = 0.80, MWU). Twenty platelets per nanolitre were significantly earlier reached after alemtuzumab (median day +11.5; range, 0–26) compared to the control group (median day +20.5; range, 0–54), (p < 10−4, MWU). Patients from the control group had a non-significantly longer stay on the transplant unit calculated from day 0 until discharge or transfer than patients after alemtuzumab-containing conditioning (46.5 days [median; range, 24–76] vs. 36.5 days [median; range, 22–82], p = 0.06, MWU; Table 2).
Acute and chronic graft-versus-host disease
The incidence of acute GvHD (aGvHD) was 36 % (n = 10) after alemtuzumab compared to 50 % (n = 14) in the control group (p = 0.28, chi2). A view on the severity of aGvHD also gave no evidence for differences between both groups (Table 2). Six patients had mild aGvHD in each group, and four and eight patients had moderate to severe aGvHD in the investigational and in the control group, respectively (p = 0.16, chi2). Chronic GvHD (cGvHD) occurred in 12 patients. Nine of these had prior aGvHD, and three patients developed de novo cGvHD. No patient had a so-called ‘overlap syndrome’. There was a borderline significance (p = 0.05, chi2) for a lower incidence of cGvHD after alemtuzumab conditioning compared to the control group (11 % [n = 3] vs. 32 % [n = 9]; Table 2).
To consider the different follow-up time in both groups, the ‘freedom from GvHD (ffGvHD)’ period was calculated using the log-rank test. The median interval was 12.6 months (range, 0.5–31.2) after alemtuzumab compared to 3.0 months (range, 0.3–106.5) in the control group (Table 2, Fig. 1). To consider the different biology of matched sibling and unrelated transplantation and the fact that familiar transplantation was performed with alemtuzumab in the investigational group and without antibody in the control group, a subanalysis for transplant type was conducted. The Kaplan–Meier plot here shows parallel curves for patients after unrelated transplantation and a trend for a superior ffGvHD for the alemtuzumab group after familiar transplantation (p = 0.06; Fig. 2, Table 3). This trend was significant when data from sibling transplantation were analysed alone (p = 0.01, log-rank test). The ffGvHD after sibling transplantation was 18.2 months (median; range, 0.8–30.6) after alemtuzumab conditioning compared to 1.4 months (median; range, 0.7–21.4) without antibody in conditioning. The ffGvHD after unrelated transplantation was comparable for the investigational and the control group with 5.2 months (median; range, 0.5–31.2) after alemtuzumab and 4.3 months (median; range, 0.3–106.5) after (p = 0.06; Fig. 2, Table 3).
Subanalysis for transplant type and antibody. Freedom from GvHD (months, top, p = 0.06), event-free survival (years, middle, p = 0.47) and overall survival (years, bottom, p = 0.34). Fam Cam related donor with alemtuzumab (Campath), Fam no AB related donor without antibody, URD ATG unrelated donor with ATG, URD Cam unrelated donor with alemtuzumab (Campath), p = 0.06 (log-rank test)
Reactivation of latent EBV and CMV infection
Patients were monitored for CMV reactivation at least weekly by PCR and were considered as positive when ≥1,000 genomes per millilitre were detected in two consecutive samples. The episodes of CMV reactivation until day +100 were compared between the alemtuzumab and the control group by the independent t test without detection of significant differences. After alemtuzumab, a median of 0.53 episode per patient (±0.22, 95 % CI) was seen compared to 0.32 (±0.18, 95 % CI) in the control group (p = 0.14).
Routine monitoring for EBV reactivation was not performed. Only one case of EBV reactivation was observed in the entire group. This patient died subsequently from EBV-lymphoproliferative disease (LPD; Table 4).
Disease-free and overall survival
The cumulative incidence of relapse calculated by log-rank test was 23 % after a median follow-up of 1.3 years (median; range, 0.1–2.6 years) compared to 39 % in the control group after 1.6 years (median; range, 0.1–11.4 years; p = 0.95, log-rank test, graph not shown). The median event-free survival was 1.3 years (range, 0.1–2.6) in the alemtuzumab group and 1.7 years (0.1–11.4) in the control group. Log-rank test (p = 0.67) and Kaplan–Meier plot showed no differences between both groups (Table 2, Fig. 1). The corresponding median overall survival was also comparable in both groups (alemtuzumab, 1.3 years [range, 0.1–2.6], vs control, 2.6 [range, 0.1–11.4], p = 0.55; log-rank test; Table 2, Fig. 1). The overall follow-up was identical to the overall survival.
The different biology of sibling and unrelated transplantation and the fact that familiar transplantation was conducted in the control group without in vivo T cell depletion suggested here also a subanalysis stratified for transplant type. The median event-free survival and overall survival after matched sibling transplantation were 1.5 years (median; range, 0.1–2.6) after alemtuzumab conditioning and 1.8 years (median; range, 0.1–11.4) in the control group (p = 0.47 and 0.34, respectively, log-rank test; Table 3). After unrelated transplantation, the event-free survival was 0.7 years (median; range, 0.1–2.6) in the alemtuzumab group compared to 1.5 years (median; range, 0.1–8.9) in the control group (p = 0.47, log-rank test; Table 3). The overall survival periods were 1.1 years (median; range, 0.2–2.6 [alemtuzumab]) and 2.6 years (median; range, 0.1–8.9 [control group]; p = 0.34, log-rank test; Table 3). The corresponding Kaplan–Meier plots are shown in Fig. 2, middle and bottom.
Mortality
Fifteen patients (54 %) after alemtuzumab conditioning and 11 (39 %) patients from the control group are well and alive. The data in Table 2 suggest a trend for a non-significantly higher numerical relapse rate and mortality rate after conventional conditioning policy compared to the alemtuzumab-containing regimen. However, Kaplan–Meier plots show here that this observation is mainly an effect of the different observation periods in both groups. Eight patients from the alemtuzumab group (29 %) and 12 patients (43 %) from the control group have died from relapsing or progressive disease. The absolute non-relapse mortality (NRM) was n = 5 (18 %) in both groups. Here, infections were the leading events in four and two cases, respectively, followed by GvHD (n = 2), toxicity (n = 1) and post-transplant lymphoproliferative disease (EBV-LPD; n = 1; Table 4). The cumulative non-relapse mortality calculated by log-rank test was comparable in both groups with 22 % after 1.3 years (median; range, 0.1–2.6 years) in the alemtuzumab group and 16 % in the control group after 2.6 years (median; range, 0.1–11.4 years; p = 0.80, log-rank test, graph not shown).
Discussion
This matched pair analysis provides evidence that low-dose alemtuzumab (10/20 mg) prior to alloSCT is a feasible strategy for in vivo T cell depletion before haematopoietic stem cell transplantation (HSCT). Compared to standard conditioning, using ATG in patients undergoing unrelated (URD)-HSCT low-dose alemtuzumab resulted in similar outcomes and showed a potential benefit in patients undergoing matched sibling alloSCT by reducing chronic GvHD and improving freedom from GvHD.
This makes low-dose alemtuzumab an interesting alternative to ATG during conditioning for alloSCT, as alemtuzumab treatment may improve the quality of life of the patient. Alemtuzumab was associated with reduced acute toxicity compared to ATG, but more importantly with a trend of lowering the incidence and severity of cGvHD compared to both standard ATG and noAB policy [2].
This was driven by matched sibling alloSCT. In sibling transplantation, the use of alemtuzumab compared to ‘noAB’ improved ffGvHD (p = 0.01 [only sibling SCT], p = 0.06 [all patients], log-rank test), without influence on event-free and overall survival.
In contrast to the experience with high-dose alemtuzumab, we found no evidence that alemtuzumab limits the graft-versus-leukaemia/myeloma effects as shown by the Kaplan–Meier plots (Fig. 1) [8, 9]. Also, the NRM did not differ between both groups. Particularly, we found no increase in mortality by opportunistic infections. However, the generally low NRM in this high-risk collective allows no further conclusions.
Our study has the strength that we followed the same protocol for supportive measures and therapy during the entire period of analysis, i.e. the control group and the alemtuzumab group were treated very similarly. Nevertheless, the study has all the limitations of a retrospective, cohort study with non-contemporaneous cohorts. While this prevents any definite conclusions, our observations may prompt a sufficiently powered prospectively randomised trial.
In conclusion, the use of low-dose alemtuzumab within conditioning prior to allogeneic SCT seems to result in comparable outcomes in comparison to ATG in URD-SCT and might be associated with a benefit in regard to chronic GvHD in matched sibling SCT. This should be further assessed in a prospective trial.
References
Bacigalupo A (2005) Antilymphocyte/thymocyte globulin for graft versus host disease prophylaxis: efficacy and side effects. Bone Marrow Transplant 35(3):225–231
Finke J, Bethge WA, Schmoor C, Ottinger HD, Stelljes M, Zander AR et al (2009) Standard graft-versus-host disease prophylaxis with or without anti-T-cell globulin in haematopoietic cell transplantation from matched unrelated donors: a randomised, open-label, multicentre phase 3 trial. Lancet Oncol 10(9):855–864
Ayuk F, Diyachenko G, Zabelina T, Panse J, Wolschke C, Eiermann T et al (2008) Anti-thymocyte globulin overcomes the negative impact of HLA mismatching in transplantation from unrelated donors. Exp Hematol 36(8):1047–1054
Hale G (2002) Alemtuzumab in stem cell transplantation. Med Oncol 19(Suppl):S33–S47
Shaw BE, Apperley JF, Russell NH, Craddock C, Liakopoulou E, Potter MN et al (2011) Unrelated donor peripheral blood stem cell transplants incorporating pre-transplant in-vivo alemtuzumab are not associated with any increased risk of significant acute or chronic graft-versus-host disease. Br J Haematol 153(2):244–252
Frampton JE, Wagstaff AJ (2003) Alemtuzumab. Drugs 63(12):1229–1243
Hale G, Cobbold S, Waldmann H (1988) T cell depletion with CAMPATH-1 in allogeneic bone marrow transplantation. Transplantation 45(4):753–759
Apperley JF, Jones L, Hale G, Waldmann H, Hows J, Rombos Y et al (1986) Bone marrow transplantation for patients with chronic myeloid leukaemia: T-cell depletion with Campath-1 reduces the incidence of graft-versus-host disease but may increase the risk of leukaemic relapse. Bone Marrow Transplant 1(1):53–66
Bunjes D (2000) T cell depletion of allogeneic stem cell grafts with anti-CD 52 monoclonal antibodies: the Ulm experience from 1983–1999. Transfus Sci 23(2):151–162
Chakrabarti S, Avivi I, MacKinnon S, Ward K, Kottaridis PD, Osman H et al (2002) Respiratory virus infections in transplant recipients after reduced-intensity conditioning with Campath-1H: high incidence but low mortality. Br J Haematol 119(4):1125–1132
Kolb HJ, Schattenberg A, Goldman JM, Hertenstein B, Jacobsen N, Arcese W et al (1995) Graft-versus-leukemia effect of donor lymphocyte transfusions in marrow grafted patients. Blood 86(5):2041–2050
Marsh JC, Gupta V, Lim Z, Ho AY, Ireland RM, Hayden J et al (2011) Alemtuzumab with fludarabine and cyclophosphamide reduces chronic graft-versus-host disease after allogeneic stem cell transplantation for acquired aplastic anemia. Blood 118(8):2351–2357
Juliusson G, Theorin N, Karlsson K, Frodin U, Malm C (2006) Subcutaneous alemtuzumab vs ATG in adjusted conditioning for allogeneic transplantation: influence of Campath dose on lymphoid recovery, mixed chimerism and survival. Bone Marrow Transplant 37(5):503–510
Kroger N, Shaw B, Iacobelli S, Zabelina T, Peggs K, Shimoni A et al (2005) Comparison between antithymocyte globulin and alemtuzumab and the possible impact of KIR-ligand mismatch after dose-reduced conditioning and unrelated stem cell transplantation in patients with multiple myeloma. Br J Haematol 129(5):631–643
Norlin AC, Remberger M (2011) A comparison of Campath and thymoglobulin as part of the conditioning before allogeneic hematopoietic stem cell transplantation. Eur J Haematol 86(1):57–66
Bertz H, Spyridonidis A, Wasch R, Grullich C, Egger M, Finke J (2009) A novel GVHD-prophylaxis with low-dose alemtuzumab in allogeneic sibling or unrelated donor hematopoetic cell transplantation: the feasibility of deescalation. Biol Blood Marrow Transplant 15(12):1563–1570
Chakrabarti S, MacKinnon S, Chopra R, Kottaridis PD, Peggs K, O’Gorman P et al (2002) High incidence of cytomegalovirus infection after nonmyeloablative stem cell transplantation: potential role of Campath-1H in delaying immune reconstitution. Blood 99(12):4357–4363
Chakraverty R, Orti G, Roughton M, Shen J, Fielding A, Kottaridis P et al (2010) Impact of in vivo alemtuzumab dose before reduced intensity conditioning and HLA-identical sibling stem cell transplantation: pharmacokinetics, GVHD, and immune reconstitution. Blood 116(16):3080–3088
Zander AR, Berger C, Kroger N, Stockschlader M, Kruger W, Horstmann M et al (1997) High dose chemotherapy with busulfan, cyclophosphamide, and etoposide as conditioning regimen for allogeneic bone marrow transplantation for patients with acute myeloid leukemia in first complete remission. Clin Cancer Res 3(12 Pt 2):2671–2675
Maris MB, Sandmaier BM, Storer BE, Chauncey T, Stuart MJ, Maziarz RT et al (2004) Allogeneic hematopoietic cell transplantation after fludarabine and 2 Gy total body irradiation for relapsed and refractory mantle cell lymphoma. Blood 104(12):3535–3542
Casper J, Knauf W, Kiefer T, Wolff D, Steiner B, Hammer U et al (2004) Treosulfan and fludarabine: a new toxicity-reduced conditioning regimen for allogeneic hematopoietic stem cell transplantation. Blood 103(2):725–731
Kroger N, Perez-Simon JA, Myint H, Klingemann H, Shimoni A, Nagler A et al (2004) Relapse to prior autograft and chronic graft-versus-host disease are the strongest prognostic factors for outcome of melphalan/fludarabine-based dose-reduced allogeneic stem cell transplantation in patients with multiple myeloma. Biol Blood Marrow Transplant 10(10):698–708
Kruger WH, Bohlius J, Cornely OA, Einsele H, Hebart H, Massenkeil G et al (2005) Antimicrobial prophylaxis in allogeneic bone marrow transplantation. Guidelines of the Infectious Diseases Working Party (AGIHO) of the German Society of Haematology and Oncology. Ann Oncol 16(8):1381–1390
Glucksberg H, Storb R, Fefer A, Buckner CD, Neiman PE, Clift RA et al (1974) Clinical manifestations of graft-versus-host disease in human recipients of marrow from HL-A-matched sibling donors. Transplantation 18(4):295–304
Vogelsang GB (1993) Acute and chronic graft-versus-host disease. Curr Opin Oncol 5(2):276–281
Acknowledgments
We wish to thank the nurses from the BMT unit for the excellent patient care.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Thomas Neumann and Meike Schulze contributed equally to this work.
Rights and permissions
About this article
Cite this article
Busemann, C., Neumann, T., Schulze, M. et al. Low-dose alemtuzumab vs. standard policy for prevention of graft-versus-host disease in unrelated and related allogeneic stem cell transplantation—a matched pair analysis. Ann Hematol 92, 945–952 (2013). https://doi.org/10.1007/s00277-013-1714-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-013-1714-4