Abstract
Backgrounds
Some disorders, such as otitis media and Eustachian tube dysfunction, may cause the temporal bone to become sclerotic. A sclerotic temporal bone has the tendency to shrink. The aim of this study was to evaluate the morphologic changes that result from sclerosis of the temporal bone.
Methods
We measured 9 variables on 2 axial images, and 8 variables on 2 coronal images in healthy ears and diseased ears in 37 patients with unilateral chronic otitis media. We also measured the volume of mastoid pneumatization.
Results
The distance from sigmoid sinus to Henle’s spine was correlated to the degree of volume reduction, and it accounted for about 17.7% of the total variation in volume reduction. There was no difference in the sigmoid sinus type in comparisons between sclerotic and pneumatic mastoids.
Conclusions
The sclerosis of the temporal bone was observed to reduce the volume of the mastoid pneumatization. However, a large portion of the volume reduction may result from the sclerotic change in the air cell system, rather than from shrinkage of the mastoid bone. Therefore, the location of surgically-important structures, in the middle and inner ear, is only rarely changed in sclerotic temporal bone.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Because the degree of mastoid pneumatization is closely related to the status of the Eustachian tube or middle ear/mastoid mucosa, most cases of chronic otitis media and cholesteatoma have a poorly-pneumatized mastoid bone [2, 5]. Therefore, sclerotic change in the mastoid bone may result in a change of several important surgical landmarks especially for a mastoidectomy procedure.
Objective data concerning the influence of sclerotic change on adjacent structures in the temporal bone are scarce. Although several reports [7, 9, 11, 13] have investigated the influence of mastoid pneumatization on the size of the temporal bone, these studies focused only on the location of the sigmoid sinus, especially its anterior displacement. Therefore, prior reports have not explained changes in volume of the mastoid bone in detail, and have ignored other variations that can result from sclerosis of the mastoid bone. In addition, it is important to evaluate the location of the landmarks and provide measurements based on known reference points that are not influenced by the sclerotic change in the mastoid bone.
In this study, we reconstructed 3-dimensional images of the temporal bone based on the temporal bone high-resolution CT scans and evaluated the morphological differences between the normal pneumatic and sclerotic mastoid bones in the patients with unilateral sclerotic temporal bone.
Materials and methods
This retrospective, institutional review board-approved study reviewed the temporal bone computed tomography (CT) images that were obtained at a university-based hospital. Patients, who had perforation of their tympanic membrane on physical exam, as well as the sclerotic change of the mastoid bone on CT, were included in the study. Exclusion criteria were a history of mastoid/middle ear surgery, CT showing a destructive lesion in the temporal bone, and cholesteatoma case that was diagnosed clinically or confirmed pathologically.
A total of 37 patients consisted of 18 males and 19 females were enrolled in this study. Their mean age was 44.6 years (SD 13.4 years). Eighteen patients had the sclerotic mastoid on right side and 19 patients on the left side.
The temporal bone high-resolution CT scans were obtained using a Somatom Plus 4 (Siemens Medical Systems, Erlangen, Germany). Continuous non-overlapping sections were obtained with a 1.0 mm slice thickness, 120 kV and 100 mAs. After the imaging data were stored in a DICOM (Digital Imaging and Communication in Medicine) file, they were imported to a personal computer that ran Vworks™ 4.0 software (Cybermed Inc, Seoul, Korea).
To investigate the changes in location of the anatomical landmarks within the mastoid bone properly, we defined some reference lines that were located outside of the mastoid, or were not influenced by the sclerotic changes in the mastoid bone. Our reference lines were as follows: (1) an extension line from the internal auditory canal axis on an axial image at level of the lateral semicircular canal, (2) a line between the center of the cochlear basal turn and posterior attachment of auricle on an axial image at the level of cochlear basal turn, (3) a horizontal line at the level of the facial nerve on a coronal image at the level of the incudostapedial joint, and (4) a horizontal line at the level of posterior semicircular canal on a coronal image at the level of the posterior semicircular canal.
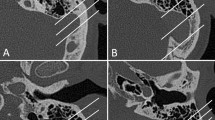
On the basis of these reference lines, we measured 17 length and 1 volume parameters. The selected length parameters were as follows: (1) 4 length parameters on an axial image at level of lateral semicircular canal (Fig. 1a), (2) 5 length parameters on an axial image at the level of cochlear basal turn (Fig. 1b), (3) 4 length parameters on a coronal image at the level of the incudostapedial joint (Fig. 1c), and (4) 4 length parameters on a coronal image at the level of the posterior semicircular canal (Fig. 1d, Table 1 and 2).
Measurements of mastoid size-related parameters that were measured in this study. a At the level of the lateral semicircular canal, b at the level of the cochlear basal turn on axial views of right temporal bone. c At the level of incudostapedial joint, d at the level of the posterior semicircular canal on coronal views of right temporal bone. a a; MCF-IAC, b; SS-IAC, c; MH-PSCC, d; MCx-PSCC. b a; HS-CoAu, b; SS-CoAu, c; SS-HS, d; SS-MCx, e; EAC-FN(a). c a; MCx-FN, b; Tg-EAC, c; Tg-FN, d; EAC-FN(c). d a; Tg-MTip, b; PSCC-Tg, c; PSCC-MTip, d; PSCC-MCx
When reconstructed as a surface-rendering algorithm, a lower threshold of −1,024 Hounsfield units (HU) and an arbitrary upper threshold of −200 HU were used to represent the air cells only. The volume of mastoid pneumatization was automatically calculated by Vworks™ 4.0 software (Cybermed Inc, Seoul, Korea). All mastoid air cells were included into the volume calculation, but the air volume in the external auditory canal, middle ear cavity and epitympanum was excluded.
All statistical analyses were performed using the SPSS software programs (SPSS Inc., Chicago, IL), and a P value < 0.05 was considered significant.
Results
Gender difference of measurements in normal pneumatic and sclerotic mastoid
There was no significant difference in the pneumatic mastoid volume between male and female (male 9880.1 mm2, female 8062.9 mm2, Mann–Whitney U test; P = 0.107). Also, there was no significant difference in sclerotic mastoid volume in the male and female (male 3784.9 mm2, female 3148.3 mm2, Mann–Whitney U test; P = 0.300). MH-PSCC, MCx-FN, Tg-MTip, and PSCC-Tg showed a significant gender difference in the pneumatic mastoids (Mann-Whitney U test; P = 0.034, P = 0.001, P = 0.026 and P = 0.019, respectively) as well as in the sclerotic mastoids (Mann–Whitney U test; P = 0.007, P = 0.001, P = 0.029 and P = 0.016, respectively). SS-HS showed a significant gender difference only in the pneumatic mastoid (Mann–Whitney U test, P = 0.014) and SS-MCx showed a significant gender difference only in the sclerotic mastoid (Mann–Whitney U test, P = 0.007; Table 3).
Comparison of measurements in normal pneumatic and sclerotic mastoid
The parameters that showed a significant difference between pneumatic and sclerotic mastoids were six length parameters and a mastoid pneumatization volume (paired T test, Table 4).
Multiple regression analysis showed that the distance from the sigmoid sinus to Henle’s spine (SS-HS) was significantly correlated with a reduction in mastoid pneumatization volume (R = 0.421, P = 0.011). In addition, it showed that the SS-HS parameter accounted for 17.7% of the total variation in the mastoid pneumatization volume (R 2 = 0.177).
Correlation of changes in measurements with sclerotic change in the mastoid
To evaluate the variation of measurements by sclerotic change in the mastoid and measure their association, Pearson correlation analysis was used. The significant correlations in parameters studied were classified into four categories (Table 5). Among these parameters, SS-HS, SS-CoAu, and EAC-FN(c) showed a significant correlation with reduction in mastoid pneumatization volume by sclerotic change. Among 22 significant correlations, the correlation between SS-CoAu and SS-HS had the largest coefficient (r = 0.762, P = 0.000).
Analysis of measurements according to the type of sigmoid sinus
The type of sigmoid sinus was classified as: saucer, half-moon and protrusive according to Ichijo’s classification [6]. In the pneumatic mastoid, the saucer type included 15 ears, the half-moon type 11 ears and the protrusive type 11 ears. For the sclerotic mastoid, the saucer type included 10 ears, the half-moon type 10 ears and the protrusive type 17 ears. There was no significant association of the sigmoid sinus types with the pneumatic or sclerotic mastoid (chi-squared test; P = 0.311, Fig. 2).
SS-IAC, SS-HS, SS-MCx and MCx-FN showed a significant difference according to sigmoid sinus types (ANOVA; P = 0.004, P = 0.005, P = 0.013, and P = 0.022, respectively). SS-IAC, SS-HS and MCx-FN were significantly shorter in the protrusive type than in the saucer type, and SS-MCx were significantly shorter in the half-moon type than in the saucer type.
Discussion
Poor mastoid pneumatization is found to be correlated with a number of conditions in which the aeration of the middle ear and mastoid is compromised. In addition, the extent of mastoid pneumatization influences the shape and size of the temporal bone. It is well known that there is a considerable variation in the size of mastoid pneumatization among individuals, and the development of mastoid pneumatization changes with age [8]. Although the individual factors that influence mastoid pneumatization have been debated for years, two theories have been widely accepted. According to the genetic theory, the extent of mastoid pneumatization is determined genetically, and its variation may become an anatomical and etiological factor for chronic otitis media. By contrast, the environmental theory emphasizes that the extent of mastoid pneumatization is determined by environmental factors. According to this concept, the pathologic influences on the middle ear and mastoid air cell system during childhood affect the pneumatization process, resulting in a sclerotic mastoid [5, 9, 10, 12, 13].
The invasion of the epithelium of the pharyngeal pouch forms the mucosal lining of the middle ear. After the precartilage in the mastoid changes into bone and connective tissue, the epithelium invades from the attic the antrum. During the same period, growth of the temporal bone is accompanied by bone resorption, creating spaces filled with connective tissue. The largest mastoid air cell, the antrum appears at 22 weeks gestation and reaches adult size by 35 weeks gestation. The full incorporation of the mastoid air cells occurs only with postnatal development; mastoid pneumatization reaches a maximum by the second decade of life in men and during the first decade in women [3, 4, 8].
When sclerotic change develops within the mastoid bone, mastoid pneumatization is reduced and may result in variation of some of the structures within the temporal bone. This possible variation of structures may lead to a change in the important surgical landmarks; it is very important for a surgeon to be able to predict possible variation prior to surgery of a sclerotic mastoid. This is why a preoperative temporal bone CT is crucial to prevent unwanted surgical complications, and was the motive for this study.
Several reports [7, 9, 11, 13] have investigated the influence of mastoid pneumatization on the position of several structures in the temporal bone. However, these focused only on one or two structures, such as sigmoid sinus; there have been no studies that have ever attempted an overall evaluation of a sclerotic mastoid bone.
The parameters that showed a significant difference in the pneumatic and sclerotic mastoids in this study, were MCx-PSCC, SS-CoAu, SS-HS, SS-MCx, Tg-MTip and PSCC-Tg. These results are consistent with those of other studies [7, 11, 13] that demonstrated that the “under-developed” mastoid process could be a consequence of hampered pneumatization. The significant reduction of MCx-PSCC, SS-HS and SS-MCx in the sclerotic mastoid can be explained by medial displacement of the outer mastoid cortex or lateral displacement of the sigmoid sinus and the posterior cranial fossa. To evaluate which of these was more influenced by the sclerotic change, the reference structure was set to the cochlea that was rarely affected.
This study also showed that SS-HS and SS-CoAu were significantly reduced in the sclerotic mastoid, but HS-CoAu was not. This result suggests that change of SS-CoAu could form the majority of change of SS-HS; that is, the lateral displacement of the sigmoid sinus and the posterior cranial fossa. In addition, considering that there was no significant association of sigmoid sinus types in the pneumatic or sclerotic mastoid in this study, the most likely explanation is the lateral displacement of the posterior cranial fossa, rather than that of sigmoid sinus.
The correlation analysis showed the following: first, the displacement of the sigmoid sinus was directed toward the anterior as well as the lateral aspect in most sclerotic mastoids. However, as noted above, the displaced structure was the posterior cranial fossa rather than the sigmoid sinus; second, displacement of the posterior cranial fossa was related to the inferior displacement of the middle cranial fossa; third, the tegmen tympani was displaced inferiorly together with tegmen mastoideum in the sclerotic mastoid whereas the middle cranial fossa was displaced inferiorly; finally, in the sclerotic mastoid, the length decrement in the mid-inferior portion of the mastoid accounted mainly for the whole length decrement of the mastoid process.
This study demonstrated that the sigmoid sinus was displaced forward in the mastoid having the protrusive-typed sigmoid sinus, and outward in the mastoid having the half-moon-typed sigmoid sinus. Therefore, when surgeons drill out the perilabyrinthine air cells in the mastoid of the protrusive-typed sigmoid sinus, they must consider that the posterior semicircular canal may be closer to sigmoid sinus than expected. In addition, when surgeons perform a cortical mastoidectomy in the mastoid of the half-moon-typed sigmoid sinus, the sigmoid sinus may be closer to the cortex than expected.
The parameters that showed a significant correlation with reduction of mastoid pneumatization volume by sclerotic change were SS-HS, SS-CoAu, and EAC-FN(c). We found that SS-HS, the parameter with the highest correlation, accounted for only 17.7% of total variation in mastoid pneumatization volume. Thus, sclerosis of mastoid pneumatization is not directly nor proportionally related to the displacement of structures in the temporal bone.
Aoki et al. [1, 2] reported that the sclerosis developed from the hindrance to subperiosteal bone formation in the outer bone metabolic layer as well as lacunar resorption of spongiotic bone in the inner bone metabolic layer in the mastoid bone. If any pathologic lesion of the middle ear and Eustachian tube influence the development of the mastoid bone during early period of life, the hindrance to subperiosteal bone formation as well as to lacunar resorption of spongiotic bone might produce decreased mastoid pneumatization together with the size of the mastoid bone. However, our study showed that changes in 17 length parameters failed to account fully for the range of reduction in the mastoid volume, and this result presents that the lack of lacunar resorption of spongiotic bone in the inner bone metabolic layer may be the main mechanism of the sclerosis. This may explain why structures in the mastoid are rarely displaced in the sclerotic mastoid. This is also supported by the fact that most sclerotic mastoids, which we met clinically, develop from pathologic lesions of the middle ear and Eustachian tube during the period of life older than the period of prior study [1, 2].
Conclusion
These results demonstrate that the sclerotic change reduces the volume of the mastoid pneumatization. Also, our findings support that its volume reduction may attribute to the sclerotic change in the air cell system, rather than the shrinkage of the mastoid bone.
References
Aoki K, Esaki S, Morikawa K, Kikuti Y, Honda Y (1988) Process of the development of pneumatization in human fetal temporal bone: compared with the results of pneumatization by the experimental study of the pigs. J Otolaryngol Jpn 91:1220–1227
Aoki K, Esaki S, Honda Y, Tos M (1990) Effect of middle ear infection on pneumatization and growth of the mastoid process. An experimental study in pigs. Acta Otolaryngol 110:399–409
Austin DF (1977) On the function of the mastoid. Otolaryngol Clin North Am 10:541–547
Bayramoglu I, Ardic FN, Kara CO, Ozuer MZ, Katircioglu O, Topuz B (1997) Importance of mastoid pneumatization on secretory otitis media. Int J Pediatr Otorhinolaryngol 40:61–66
Diamant M (1940) Otitis and pneumatization of mastoid bone. Acta Otolaryngol 41:10
Ichijo H, Hosokawa M, Shinkawa H (1993) Differences in size and shape between the right and left sigmoid sinuses. Eur Arch Otorhinolaryngol 250:297–299
Ichijo H, Hosokawa M, Shinkawa H (1996) The relationship between mastoid pneumatization and the position of the sigmoid sinus. Eur Arch Otorhinolaryngol 253:421–424
Lee DH, Jun BC, Kim DG, Jung MK, Yeo SW (2005) Volume variation of mastoid pneumatization in different age groups: a study by three-dimensional reconstruction based on computed tomography images. Surg Radiol Anat 27:37–42
Shatz A, Sade J (1990) Correlation between mastoid pneumatization and position of the lateral sinus. Ann Otol Rhinol Laryngol 99:142–145
Silbiger H (1950) Uber das ausmass der mastoid pneumatisation beim menschen. Acta Anat 11:215–223
Sirikci A, Bayazit YA, Kervancioglu S, Ozer E, Kanlikama M, Bayram M (2004) Assessment of mastoid air cell size versus sigmoid sinus variables with a tomography-assisted digital image processing program and morphometry. Surg Radiol Anat 26:145–148
Tos M, Stangerup SE (1985) The causes of asymmetry of the mastoid air cell system. Acta Otolaryngol 99:564–570
Turgut S, Tos M (1992) Correlation between temporal bone pneumatization, location of lateral sinus and length of the mastoid process. J Laryngol Otol 106:485–489
Author information
Authors and Affiliations
Corresponding author
Additional information
Presented as a poster titled as “analysis of morphologic changes in the sclerotic temporal bone by spiral high-resolution computed tomography” at XVIII IFOS World Congress (June 25–30, 2006, Rome, Italy).
Rights and permissions
About this article
Cite this article
Lee, DH., Jung, MK., Yoo, YH. et al. Analysis of unilateral sclerotic temporal bone: how does the sclerosis change the mastoid pneumatization morphologically in the temporal bone?. Surg Radiol Anat 30, 221–227 (2008). https://doi.org/10.1007/s00276-008-0310-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00276-008-0310-0