Abstract
Purpose
Irreversible electroporation (IRE) is a newly developed nonthermal tissue-ablation technique in which high-voltage electrical pulses of microsecond duration are applied to induce irreversible permeabilisation of the cell membrane, presumably through nanoscale defects in the lipid bilayer, leading to apoptosis. The purpose of this study was to assess the feasibility and safety of ablating renal cell carcinoma (RCC) tissue by IRE.
Methods
Six patients scheduled for curative resection of RCC were included. IRE was performed during anaesthesia immediately before the resection with electrographic synchronisation. Central haemodynamics were recorded before and 5 min after electroporation. Five-channel electrocardiography (ECG) was used for detailed analysis of ST waveforms. Blood sampling and 12-lead ECG were performed before, during, and at scheduled intervals after the intervention.
Results
Analysis of ST waveforms and axis deviations showed no relevant changes during the entire study period. No changes in central haemodynamics were seen 5 min after IRE. Similarly, haematological, serum biochemical, and ECG variables showed no relevant differences during the investigation period. No changes in cardiac function after IRE therapy were found. One case of supraventricular extrasystole was encountered. Initial histopathologic examination showed no immediate adverse effects of IRE (observation of delayed effects will require a different study design).
Conclusion
IRE seems to offer a feasible and safe technique by which to treat patients with kidney tumours and could offer some potential advantages over current thermal ablative techniques.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Like radiofrequency ablation (RFA), irreversible electroporation (IRE) is performed by applying a voltage to needle-like electrodes inserted at the site of the tumour to be ablated. However, the physiologic principles underlying the two techniques are entirely different: whereas RFA depends on thermal destruction of carcinoma tissue, IRE, a novel technique, employs high-voltage electrical pulses on the microsecond timescale to induce irreversible opening of pores in the cell membrane, with consequent cell death [1]. IRE has a clear potential for application in surgical oncology, and special instrumentation has been designed for the clinical use of IRE [1–3].
For this first-in-man study, renal tumours were selected as suitable targets because IRE may offer a clinical advantage over local thermal ablation in this organ. They can be detected at a relatively early stage (<3 cm) and may thus be treated by procedures that conserve the renal parenchyma, such as partial resection in case of contraindication for surgical treatment. They are also excellently accessible for local ablative procedures. However, they are associated with several problems. Certain options, such as chemotherapy or radiotherapy, are not promising because in general renal tumours respond poorly to these treatments. Furthermore, the location of the tumour within the kidney may influence the success of thermal ablation because regional vascular flow can provide a heat sink, especially in cases in which renal tumours are centrally located [4, 5]. Renal cell carcinoma represents 80 to 90% of all renal tumours and is the most malignant of these, with poor (60% at 5 years) survival prognosis (see [6] for an up-to-date review).
The potential of IRE to overcome the limitations of RFA has been demonstrated in animal models [7–10]. It appears to offer a number of advantages as follows:
-
1.
Because it does not require heating of the tissue to be ablated, it is not sensitive to the cooling effects that result from normal circulation or hypervascularisation.
-
2.
Important structures are conserved [10].
-
3.
Shrinkage of the renal pelvis and calices or necrosis in the access area, a special risk in centrally located tumours, is avoided [4].
Surgical nephrectomy of renal carcinoma leads to 5-year survival rates from 97 to 87% (pT1a/pT1b) to only 20% (pT4) [11], with similar rates for partial resection. For tumours with a largest diameter measuring <4 cm, the 5- and 10-year survival rates are 92 and 80%, respectively [12]. The progression of small renal masses is not well defined: Tumours measuring <3 cm diameter were formerly classified as adenomas [13] and later as “renal carcinomas of low metastatic potential” [14]. The growth rate of such small neoplasms is 0 to 1.3 cm/year [15], and only few cases exhibit multicentricity (<4% [16]). They have an average metastatic rate of approximately 1% after 34 months of follow-up [17]. However, total and nephron-sparing nephrectomy are not ideal for many patients, such as those who cannot tolerate surgery because of serious comorbidities or those in whom the tumour is unfavourably located. Alternative procedures, above all involving thermoablative procedures, for parenchyma-sparing renal tumour excision are topics of much current research. Dependence of the degree of tumour destruction on tumour size, geometry, and vascularisation has been demonstrated [18–21].
This first-in-man pilot study with IRE was therefore designed to investigate the feasibility and safety of IRE treatment. This study addressed in particular the possible risks associated with the need for general anaesthesia (to induce muscular relaxation) and with the need to synchronise IRE pulses with the refractory period of the cardiac rhythm (to avoid rhythm disorders).
Patients and Methods
Approval for this good clinical practice (GCP)-compliant study was granted by the Ethics Committee of the Medical Faculty of the University of Magdeburg. The study design and conduct complied with the precepts of good clinical practice. Patients were only treated by IRE if they had given their written informed consent to do so.
Six patients, already scheduled for surgical resection of renal tumours of size measuring <4 cm and without any signs of metastasis, were treated with IRE during their surgery, and the entire procedure was monitored by ultrasonography. For all patients we used a NanoKnife bipolar probe (length 15 cm) and a NanoKnife IRE electroporator (AngioDynamics, Latham, NY). The electrode was positioned under ultrasound guidance. On the basis of in vitro porcine data with a bipolar probe, this electrode typically ablates a prolate ellipsoid with axes of approximately 30 and 15 mm. IRE was followed by a 15-min rest period; after this, blood was sampled, and the resection was conducted according to the normal procedure.
For the safety assessment, laboratory values, electrocardiography (ECG), and blood gas analyses were performed. Blood was sampled before, during, 6 h after, and 1, 3, and 5 days after the intervention. Twelve-lead ECGs were taken before, 6 h after, 1, 3, and 5 days, and 12 weeks after the intervention. The laboratory values recorded and analysed included erythrocyte count, haemoglobin, haematocrit, mean corpuscular volume, mean corpuscular haemoglobin (amount and concentration), leucocyte count, electrolytes (Na+, K+, Cl−, Ca2+), urea, creatinine, creatine kinase MM (isoenzymic form for skeletal muscle), creatine kinase MB (isoenzymic form for myocardium), troponin T, alanine and aspartate aminotransferases, lactate and lactate dehydrogenase and C-reactive protein, prothrombin time, partial thromboplastin time, thrombocyte count, and D-dimers.
Standard histopathologic analyses of the electroporated and resected tissue were performed with haematoxylin-and-eosin staining. (It should be noted that these can only be of preliminary value when the tissue is removed immediately after IRE; see “Discussion” section.)
In a further analysis, the changes in the variables assessed were compared with those found for six patients selected from the institution’s database and matched with the study patients with respect to tumour size, operative technique, age, and renal function.
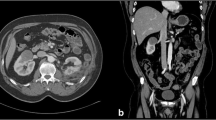
Cardiac and laboratory variables were subjected to statistical testing at an exploratory level; in view of the very small sample size of this first-in-man study, no formal testing was appropriate. Cardiac, respiratory, and acid–base values (before and 15 min after IRE) were compared by Wilcoxon’s test, and coagulation values (before IRE and 5 min, 6 h, and 1, 3, and 5 days after IRE) were compared by the Kruskal–Wallace test. A view of the operative procedure is shown in Fig. 1, and a typical sonographic monitor image is shown in Fig. 2.
A typical sonographic image obtained during monitoring of the positioning of the needle electrode (see Fig. 1) for IRE. The “hockey-stick” curve shows the envelope of the kidney; the dark central mass is the tumour; and the slightly off-horizontal line passing through it is the electrode
Results
The patients’ basic demographic and disease characteristics are listed in Table 1. The IRE procedure was conducted in each case according to plan. The lesions were fully covered by the IRE (dimensions listed in Table 1); depending on the shape and size of the tumour growth, and one to three positionings of the needle electrodes were required. All resections were conducted by the same surgeon with approximately 10 years’ experience, and the needles were positioned by a nephrological surgeon with approximately 15 years’ experience.
The most important (as presumably most sensitive) cardiac values were those measured during the surgery. These are listed in Table 2. None of the individual or mean differences were clinically significant, and all statistical comparisons (Wilcoxon’s test) yielded p > 0.05.
There was a single case of intraoperative supraventricular extrasystole. In the postoperative monitoring phase (≤5 days) and at follow-up examination (after 12 weeks), no ECG-related changes were detected. No changes in cardiac function were found after IRE.
Acid–base and respiratory values are listed in Table 3. Again, none of the individual or group differences were clinically significant, and all comparisons yielded p > 0.05.
Relative changes in the most important clinical laboratory variables, as measured at the various time points, are shown in Figs. 3, 4 and 5. Changes are expressed as percentages of the mean of the respective normal range. Coagulation values showed no changes: Specifically, the absence of changes in creatine kinase MB and troponin T indicated no risk of cardiac infarction, although the stability of the lactate and lactate dehydrogenase values indicated, as expected, no ischaemia or cell death (these would only be observed later). Myoglobin and creatine kinase MM increased because of the surgery.
Creatine kinase MB, troponin T, lactate, and lactate dehydrogenase values. Details as in Figure 3
Myoglobin and creatine kinase MM. Details as in Figure 3
Urea and creatinine are markers of renal function. Increases in creatinine values are to be expected because a part or the whole of a kidney is removed in the surgery, with resulting impairment of the detoxification function (Fig. 3).
The destruction of muscle cells (including heart muscle) results in increased creatine kinase MM levels; in the case of heart muscle, CK-MB and troponin levels are also increased. Therefore, these were monitored to allow differentiation between the effects of the surgical intervention and those of the high-voltage electroporation. Results are shown in Figs. 4 and 5.
The kidneys play an important part in the regulation of arterial blood pressure. The relative changes in systolic and diastolic arterial pressure are shown in Fig. 6. As expected, a decrease was seen for the perioperative period, and values were restored by 24 h after surgery.
Statistically relevant changes were observed in the coagulation variables as follows: partial thromboplastin time (p = 0.015), thrombin time (p = 0.025), thromboplastin time (p = 0.010), and D-dimers (p < 0.001). All of these changes are expected in connection with standard surgical intervention, including administration of heparin. None of the six patients showed any signs of an infection, as assessed by C-reactive protein or leucocyte count.
The other laboratory values (as listed in the “Patients and methods” section; results not shown) did not show any conspicuous changes or effects of IRE. No complications associated with loss of blood (e.g., decreased red cell counts) were encountered.
In the matched-pair comparison, no statistically significant differences were found except for duration of surgery, which was longer for the study subjects, as expected in view of the additional procedure that was conducted.
In the histopathologic examination, cells showed a mismatch between plasma and nuclear volume, i.e., the cells had begun to swell but were not dead (no dead cells were found in the specimens).
In summary, the safety assessments gave no suggestion of any deleterious short-term effect of intraoperative IRE. Most importantly, the application of electrical pulses did not interfere with the patients’ cardiac parameters as measured by ECG.
Discussion
Because no clinically or statistically significant changes were found in the laboratory analysis (including the respiratory values) or in the ECG assessment, IRE can be regarded as safe enough to allow the design of a further study. The first task of such a study will be to conduct IRE several days before resection to investigate the success of this technique in destroying tumour cells. Because the results presented previously suggest that IRE may be a useful complement to, or may even one day supplant, thermal ablation, we therefore consider in turn the relevant technical aspects and associated safety factors.
In thermal ablation, the cooling effect in hypervascularised lesions and the enlargement of renal tumours has been successfully avoided by selective preinterventional embolisation [22, 23]. However, the nature of the surrounding perirenal fatty tissue also influences the ablation zone: Both normal fatty tissue and fibrotic tissue (the latter resulting from chronic renal disorder) have an insulating effect and can decrease heat dissipation, thus increasing the temperature attained in the tumour and amplifying the thermal effect [20]. It is possible that the rates of local tumour recurrence found in a meta-analysis of 71 studies of RFA in the kidney region were due to the problem of thermal ablation and temperature monitoring. Other serious complications that can occur with RFA include uretral stricture and loss of a renal unit [24]. From a technical perspective, IRE—as a nonthermal procedure—may possess considerable potential in the treatment of inoperable renal tumours, as appears to be confirmed by animal studies in which cell destruction right up to the large “cooling” vessels has been observed [25] as has preservation of the integrity of the ureter, the nerve trunk, and the renal blood vessels [10, 26].
Another issue is the recovery of normal tissue in the treated region. Early work in liver showed rapid resolution of IRE lesions to a minimal scar in approximately 2 weeks [2]; this was attributed to preservation of the microvasculature throughout the IRE lesion. Compared with this, lesions deriving from RFA and cryotherapy must resolve from the edges inward [10]. Therefore, a lower complication rate may be expected with IRE than with thermoablation or cryoablation.
Another major limitation of thermal ablation technologies has been the nonselective nature of the destructive process. IRE destroys the cellular components of a tissue but does not affect the underlying collagen network, thereby allowing the basic tissue structure to be preserved. Consequently, tissue with regenerative capacities (such as the ureter) may replace its mucosal cells with time. Although RFA and cryoablation cause complications to normal structures within the ablation zone, IRE lesions have been observed to leave intact bile ducts within liver lesions, and similar results in the prostatic urethra and the periprostatic nerves have been obtained (reviewed by Onik et al. [10]). Similar considerations should apply to the possible sparing of healthy nephron tissue in renal IRE. Thus, again, IRE appears to offer a treatment with a lower complication rate than thermoablation or cryoablation.
A further anticipated difference between IRE and RFA in clinical application is the overall duration of the electrical treatment. IRE is coupled to the cardiac rate only and typically requires 90 to 100 pulses (corresponding to the same number of heartbeats) for treatment. In contrast, thermal procedures can require up to 30 min.
Finally, studies in vitro suggest that different tumour cells may respond with greater or lesser sensitivity to treatment by IRE and that ablation may be steered by optimising such parameters as the voltage gradient and the duration, number, frequency, and polarity of the pulses applied [27]. Optimising these could provide further possibilities for minimising the side effects of treatment.
Potential limitations of the IRE procedure, and of the selection of appropriate patients, could be the need for the use of muscle relaxant, electrocardiographic synchronisation, and placement of several electrodes in cases of larger tumours. Our results give a first indication that the procedure is generally safe and does not constitute an additional risk for patients when applied in an intraoperative setting.
The present pilot study was intentionally restricted to a small patient population and to an overall procedure that deviated as little as possible from the standard resection. The next step in development of the method will be a further study to establish the long-term safety of the procedure. Furthermore, to assess the efficacy of this technique, it will be necessary to conduct the IRE well in advance of resection to discover the histological effects of IRE, which necessarily require some time to develop. In the present study, the 15 min between IRE and resection was only sufficient to show any immediate histological effects, but the longer-term effects will be decisive for possible application of this method. In addition, it will be necessary to include larger patient cohorts into the design strategy. If the long-term safety of IRE is confirmed, then it is anticipated that the way will be open for the first clinical studies of the method’s efficacy.
Conclusion
This safety-only study indicates that IRE is potentially a feasible and safe technique to treat patients with tumour of the kidney.
References
Bertacchini C, Margotti PM, Bergamini E, Lodi A, Ronchetti M, Cadossi R (2007) Design of an irreversible electroporation system for clinical use. Technol Cancer Res Treat 6(4):313–320
Rubinsky B, Onik G, Mikus P (2007) Irreversible electroporation: a new ablation modality—clinical implications. Technol Cancer Res Treat 6(1):37–48
Rubinsky B (2007) Irreversible electroporation in medicine. Technol Cancer Res Treat 6(4):255–260
Hines-Peralta A, Goldberg SN (2004) Review of radiofrequency ablation for renal cell carcinoma. Clin Cancer Res 10(18 Pt 2):6328S–6334S
Yoon SK, Choi JC, Cho JH, Oh JY, Nam KJ et al (2009) Radiofrequency ablation of renal VX2 tumors with and without renal artery occlusion in a rabbit model: feasibility, therapeutic efficacy, and safety. Cardiovasc Intervent Radiol 32(6):1241–1246
Ljungberg B, Hanbury DC, Kuczyk MA, Merseburger AS, Mulders PF, Patard JJ et al (2007) Renal cell carcinoma guideline. Eur Urol 51(6):1502–1510
Al Sakere B, André F, Bernat C, Connault E, Opolon P, Davalos RV, et al (2007) Tumor ablation with irreversible electroporation. PLoS One 2(11):e1135
Garcia PA, Rossmeisl JH, Robertson J, Ellis TL, Davalos RV (2009) Pilot study of irreversible electroporation for intracranial surgery. Conf Proc IEEE Eng Med Biol Soc 1:6513–6516
Granot Y, Ivorra A, Maor E, Rubinsky B (2009) In vivo imaging of irreversible electroporation by means of electrical impedance tomography. Phys Med Biol 54(16):4927–4943
Onik G, Mikus P, Rubinsky B (2007) Irreversible electroporation: implications for prostate ablation. Technol Cancer Res Treat 6(4):295–300
Frank I, Blute ML, Leibovich BC, Cheville JC, Lohse CM, Zincke H (2005) Independent validation of the 2002 American Joint Committee on cancer primary tumor classification for renal cell carcinoma using a large, single institution cohort. J Urol 173(6):1889–1892
Hafez KS, Fergany AF, Novick AC (1999) Nephron-sparing surgery for localized renal cell carcinoma: impact of tumor size on patient survival, tumor recurrence and TNM staging. J Urol 162(6):1930–1933
Bell ET (1950) Renal diseases. Lippincott, Philadelphia, PA, pp 428–439
Peterson RO (1986) Urologic pathology. Lippincott, Philadelphia, PA, pp 85–110
Bosniak MA (1995) Observation of small incidentally detected renal masses. Semin Urol Oncol 13(4):267–272
Nissenkorn, Bernhelm J (1995) Multicentricity in renal cell carcinoma. J Urol 153(3 Pt 1):620–622
Chawla SN, Crispen PL, Hanlon AL, Greenberg RE, Chen DY, Uzzo RG (2006) The natural history of observed enhancing renal masses: meta-analysis and review of the world literature. J Urol 175(2):425–431
Mayo-Smith WW, Dupuy DE, Parikh PM, Pezzullo JA, Cronan JJ (2003) Imaging-guided percutaneous radiofrequency ablation of solid renal masses: techniques and outcomes of 38 treatment sessions in 32 consecutive patients. AJR Am J Roentgenol 180(6):1503–1508
Matlaga BR, Zagoria RJ, Woodruff RD, Torti FM, Hall MC (2002) Phase II trial of radio frequency ablation of renal cancer: evaluation of the kill zone. J Urol 168(6):2401–2405
Gervais DA, McGovern FJ, Arellano RS (2003) Renal cell carcinoma: clinical experience and technical success with radio-frequency ablation of 42 tumors. Radiology 226(2):417–424
Pavlovich C, Walther M, Choyke P, Pautler SE, Chang R, Linehan WM et al (2002) Percutaneous radiofrequency ablation of small renal tumors: initial results. J Urol 167(1):10–15
Tacke J, Mahnken A, Bucker A, Rohde D, Günther RW (2001) Nephron-sparing percutaneous ablation of a 5 cm renal cell carcinoma by superselective embolization and percutaneous RF-ablation. Rofo 173(11):980–983
Gebauer B, Werk M, Lopez-Hanninen E, Felix R, Althaus P (2007) Radiofrequency ablation in combination with embolization in metachronous recurrent renal cancer in solitary kidney after contralateral tumor nephrectomy. Cardiovasc Intervent Radiol 30(4):644–649
Dib RE, Touma NJ, Kapoor A (2009) Review of the efficacy and safety of radiofrequency ablation for the treatment of small renal masses. Can Urol Assoc J 3(2):143–149
Lee EW, Loh CT, Kee ST (2007) Imaging-guided percutaneous irreversible electroporation: ultrasound and immunohistological correlation. Technol Cancer Res Treat 6(4):287–294
Maor E, Ivorra A, Leor J, Rubinsky B (2007) The effect of irreversible electroporation on blood vessels. Technol Cancer Res Treat 6(4):307–312
Miller L, Leor J, Rubinsky B (2005) Cancer cells ablation with irreversible electroporation. Technol Cancer Res Treat 4(6):699–705
Conflict of interest
The authors declare that they have no conflict of interest. This study was performed independently of the manufacturer of the devices used.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pech, M., Janitzky, A., Wendler, J.J. et al. Irreversible Electroporation of Renal Cell Carcinoma: A First-in-Man Phase I Clinical Study. Cardiovasc Intervent Radiol 34, 132–138 (2011). https://doi.org/10.1007/s00270-010-9964-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-010-9964-1