Abstract
Although the most involved organs are liver and lung, hydatid cysts occur in adrenal glands, rarely, and constitute only 0.5% of hydatid cysts. Herein, we demonstrate and discuss, for the first time in the literature to the best of our knowledge, the radiological features of adrenal hydatid disease and evaluate the long-term results (57 months of follow-up) of the percutaneous treatment of hydatid cyst in the adrenal gland in a patient.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hydatid disease, also known as echinococcosis granulosus, caused by Echinococcus granulosus larvae, remains a common health problem in endemic areas, such as most of the Mediterranean, the Middle East, Eastern Europe, Africa, Latin American countries, and China, where livestock is raised in association with dogs [1]. Hydatid cysts are usually found in liver (45–75%) and lungs (10–50%), but can occur almost anywhere in the body, including kidneys, heart, spleen, musculoskeletal system, and brain, which altogether represent about 10% of the total number of cases. Hydatid cysts of the adrenal gland are unusual, comprising 0.5% of all cases [2, 3].
Traditionally, the treatment of adrenal hydatid disease consists of open surgical removal, with support of antiparasitic agents prior to and after surgery, in an effort to prevent secondary dissemination. The current trend toward minimal invasive surgery has introduced laparoscopic techniques for uncomplicated adrenal hydatid disease [3]. Percutaneous treatment of hydatid cyst has gained wide acceptance in liver hydatidosis, with successful long-term results [4–6]. Percutaneous treatment is also an effective management option in atypically located hydatid cysts such as in the kidney, pulmonary, spleen, orbita, parotid gland, and musculoskeletal system [7–13].
Herein we demonstrate and discuss, for the first time in the literature to the best of our knowledge, the radiological features of adrenal hydatid disease and evaluate the long-term results of percutaneous treatment of hydatid cyst in the adrenal gland.
Case Report
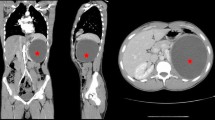
A 70-year-old man with a history of splenectomy by reason of hydatid cyst 6 years previously was admitted to our department for treatment of a cystic mass in the splenectomy region. The patient had been put on medical treatment (100 mg acetylsalicylic acid) because of coronary artery disease. His physical examination revealed a palpable mass in the left lumbar region. Laboratory investigation including whole blood count and biochemistry profile were within normal limits. Echinococcal hemagglutination and specific echinococcus IgE tests were positive. Abdominal ultrasonography (US) showed an 8 × 11 × 13 cm (~600 ml) cystic mass including structures resembling germinative membrane in the left suprarenal region. Abdominal multidetector computed tomography (MDCT) documented the presence of a hydatid cyst in the left adrenal gland. According to the WHO classification, a CE-3A hydatid cyst (type 2 according to the Gharbi classification) was considered in this patient (Fig. 1).
As prophylaxis to reduce the risk of secondary dissemination, albendazole was given orally, at a dose of 10 mg/kg/day, for 1 week before and 4 weeks after the percutaneous procedure. The procedure was performed with the patient under conscious sedation and local anesthesia. A single dose of cefazoline was given for antibiotic prophylaxis. Standard catheterization technique [14] with hypertonic saline and alcohol was preferred, owing to the large size of the cyst. Procedure-related complications did not occur. The cyst substance was in the form of clear-crystal fluid. No protoscolices was detected in microscopic examinations of the sample. Fluid drainage from the catheter was 40, 15, and 5 ml on days 1, 2, and 3 after the procedure, respectively, and the catheter was withdrawn on the third day after performing sclerosis with ethyl alcohol.
The size of the hydatid cyst had decreased to 7.3 × 4.5 × 3.1 cm (~53 ml) at 1-month follow-up and was measured as 6 × 4.5 × 3 cm (~42 ml) consistently. One-year follow-up MDCT and US examinations showed that the fluid component of the cyst had largely disappeared and the cyst had gained a solid appearance. Two-year follow-up US and MDCT examinations revealed an increase in cyst dimensions (9 × 5 × 5 cm; ~119 ml) and a 1.5-cm daughter cyst (Fig. 2). This hydatid cyst lesion, with findings again consistent with recurrence, was treated percutaneously using standard catheterization technique with hypertonic saline and alcohol. During follow-up, the cyst decreased in size (to 8.3 × 4.2 × 4.7 cm; ~87 ml); no feature of viability in the remnant was observed until the 57th month of follow-up (Fig. 3). No other hydatid cyst lesion was found during the follow-up.
Discussion
Cystic adrenal lesions are uncommon, with a prevalence of about 0.06%. Although they may occur in all age groups, they are mostly seen at between 50 and 60 years. They are primarily unilateral, with a prevalence of 92% [15]. Adrenal gland cysts, which may pose diagnostic and management problems, have been classified as pseudocysts (39–78%), endothelial cysts (20–45%), epithelial cysts (2–9%), and parasitic cysts (0–7%). Adrenal neoplasms including adrenal cortical adenomas, adrenal cortical carcinomas, and pheochromocytomas may present with benign-appearing cysts [16, 17].
Adrenal hydatid cysts are generally asymptomatic masses, usually discovered incidentally during radiological examinations performed for other reasons. Rarely, adrenal hydatid cysts may get complicated and become symptomatic. The most common symptoms are flank discomfort and gastrointestinal complaints (bloating, fullness, nausea, vomiting, constipation, and anorexia), which are related to the mass effect of the cysts [15, 18, 19]. The risks for and major complications of hydatid cysts located in adrenal glands are rupture, with possible anaphylactic shock and hemorrhage, similar to those at other locations. Arterial hypertension due to adrenal hydatid cyst, which has been called the Goldblatt phenomenon, and fistulization of adrenal gland hydatid cyst into the proximal jejunal loop have also been rarely reported as site-specific complications [20–22].
US is the primary imaging tool in the diagnosis and classifications of liver hydatid cysts. US is also a primary modality in the assessment of viability of hydatid cysts during interventional procedures and posttreatment follow-up [23]. MDCT and magnetic resonance imaging (MRI) can detect cystic lesions including hydatid cysts of the adrenal gland with a high sensitivity. When large cysts occur, for which the diagnosis is usually confused, multiplanar images on MDCT and MRI are helpful for localizing the origin of the cyst, as in our case. Additionally, MDCT can be a useful alternative tool for guiding percutaneous treatment of hydatid cyst of the adrenal gland [24].
Some of the serologic tests, such as the Casony intradermal test, the Weinberg test, complement fixation and indirect hemagglutination tests are not reliable. There are many new sensitive and specific serological tests available, such as complement fixation, enzyme-linked immunosorbent assay, ARC 5 precipitation, and specific hydatid IgE tests. However, positive results on these tests do not confirm the existence of active disease, whereas negative results do not exclude it [5, 25].
Previously, it has been postulated that hydatid cysts should not be punctured or drained, and surgery was considered the first-line treatment. Accordingly, all hydatid cysts of the adrenal gland reported in the literature so far had been treated with partial and total excision, with and without preservation of the adrenal gland [25]. Simple cystectomy for treatment of a hydatid cyst of the adrenal gland has also rarely been performed [26]. Traditionally open surgery is effective but it has several disadvantages such as surgical scarring, operative pain, risk of anesthesia, risk of contamination, and long hospital stay. Since surgeons usually use the transabdominal approach in adrenal hydatid surgery, risk of contamination of the peritoneal cavity with spilled cyst contents containing viable protoscolices is also considerable [25]. Transabdominal laparoscopic and endoscopic retroperitoneal adrenalectomies, which have recently been performed, offer less surgical scarring, less operative pain, and a shorter postoperative hospital stay compared to traditional open surgery [3]. Percutaneous treatment has the advantages of minimal risk of contamination, preservation of adrenal glands, lack of operative scar, minimal operative pain, and only 1 day of postoperative hospitalization.
In conclusion, approximately 20 years of experience with percutaneous treatment of hydatid cysts in different body locations, except the adrenal glands, shows excellent long-term results. We think that similar principles for percutaneous treatment of hydatid cysts may be applied to adrenal hydatid cysts with successful results as an effective alternative to surgery.
References
Czermak BV, Akhan O, Hiemetzberger R et al (2008) Echinococcosis of the liver. Abdom Imaging 33:133–143
Bastounis E, Pikoulis E, Leppaniemi A, Cyrochristos D (1996) Hydatid disease: a rare cause of adrenal cyst. Am Surg 62:383–385
Dionigi G, Carrafiello G, Recaldini C et al (2007) Laparoscopic resection of a primary hydatid cyst of the adrenal gland: a case report. J Med Case Reports 1:61
Ustünsöz B, Akhan O, Kamiloğlu MA, Somuncu I, Uğurel MS, Cetiner S (1999) Percutaneous treatment of hydatid cysts of the liver: long-term results. AJR Am J Roentgenol 172:91–96
Akhan O, Ozmen MN (1999) Percutaneous treatment of liver hydatid cysts. Eur J Radiol 32:76–85 (Review)
Akhan O, Ozmen MN, Dinçer A, Sayek I, Göçmen A (1996) Liver hydatid disease: long-term results of percutaneous treatment. Radiology 198:259–264
Akhan O, Ustünsöz B, Somuncu I et al (1998) Percutaneous renal hydatid cyst treatment: long-term results. Abdom Imaging 23:209–213
Akhan O, Ozmen MN, Dinçer A, Göçmen A, Kalyoncu F (1994) Percutaneous treatment of pulmonary hydatid cysts. Cardiovasc Interv Radiol 17:271–275
Ormeci N, Soykan I, Palabiyikoğlu M et al (2002) A new therapeutic approach for treatment of hydatid cysts of the spleen. Dig Dis Sci 47:2037–2044
Akhan O, Koroglu M (2007) Hydatid disease of the spleen. Semin Ultrasound CT MR 28:28–34
Akhan O, Ensari S, Ozmen M (2002) Percutaneous treatment of a parotid gland hydatid cyst: a possible alternative to surgery. Eur Radiol 12:597–599
Akhan O, Bilgiç S, Akata D, Kiratli H, Ozmen MN (1998) Percutaneous treatment of an orbital hydatid cyst: a new therapeutic approach. Am J Ophthalmol 125:877–879
Akhan O, Gumus B, Akinci D, Karcaaltincaba M, Ozmen M (2007) Diagnosis and percutaneous treatment of soft-tissue hydatid cyst. Cardiovasc Interv Radiol 30:419–425
Akhan O, Dincer A, Gököz A et al (1993) Percutaneous treatment of abdominal hydatid cysts with hypertonic saline and alcohol. An experimental study in sheep. Invest Radiol 28:121–127
Yeniyol CO, Minareci S, Ayder AR (2000) Primary cyst hydatid of adrenal: a case report. Int Urol Nephrol 32:227–229 (Review)
Danza FM, De Marinis L, Mancini A et al (1993) Adrenal gland cysts. Our experience. Minerva Chir 48:1325–1330
Erickson LA, Lloyd RV, Hartman R, Thompson G (2004) Cystic adrenal neoplasms. Cancer 101:1537–1544
Ozbey I, Aksoy Y, Polat O, Atmaca AF, Demirel A (2002) Clinical management of hydatid disease of the urinary tract. J Int Med Res 30:346–352
Gurbuz R, Guven S, Kilinc M, Abasiyanik F, Gokce G, Piskin MM (2005) Primary hydatid cyst in adrenal gland: a case report. Int Urol Nephrol 37:21–23
Escudero MD, Sabater L, Calvete J et al (2002) Arterial hypertension due to primary adrenal hydatid cyst. Surgery 132:894–895
Ruiz-Rabelo JF, Gomez-Alvarez M, Sanchez-Rodriguez J, Rufian Pena S (2008) Complications of extrahepatic echinococcosis: fistulization of an adrenal hydatid cyst into the intestine. World J Gastroenterol 14:1467–1469
Ozarmagan S, Erbil Y, Barbaros U, Salmaslioglu A, Bozbora A (2006) Primary hydatid disease in the adrenal gland: a case report. Braz J Infect Dis 10:362–363
WHO informal working group (2003) International classification of ultrasound images in cystic echinococcosis for application in clinical and field epidemiological settings. Acta Tropica 85:253–261
Guo YK, Yang ZG, Li Y et al (2007) Uncommon adrenal masses: CT and MRI features with histopathologic correlation. Eur J Radiol 62:359–370 (Review)
Akcay MN, Akcay G, Balik AA, Böyük A (2004) Hydatid cyst of the adrenal gland: review of nine patients. World J Surg 28:97–99
el Idrissi Dafali A, Dahami Z, Zerouali NO (2002) Hydatid cyst of the adrenal gland. Ann Urol 36:99–103
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Akhan, O., Canyigit, M., Kaya, D. et al. Long-Term Follow-Up of the Percutaneous Treatment of Hydatid Cyst in the Adrenal Gland: A Case Report and Review of the Literature. Cardiovasc Intervent Radiol 34 (Suppl 2), 256–259 (2011). https://doi.org/10.1007/s00270-009-9672-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-009-9672-x