Abstract
Background
The presence of gastric ectopic mucosa in Meckel’s diverticulum is associated with a higher risk of development of complications. The aim of the present study was to investigate which demographic/clinical parameters predict the presence of gastric heterotopia in Meckel’s diverticulum.
Methods
This was a retrospective cohort study conducted in a single institution (University Hospital Ostrava, Czech republic). All children who underwent laparoscopic/open resection of Meckel’s diverticulum within a 20-year study period were included in the study.
Results
In total, 88 pediatric patients underwent analysis. The mean age of the children was 4.6 ± 4.73 years; the male–female ratio was approximately 2:1. There were 50 (56.8%) patients with asymptomatic Meckel’s diverticulum in our study group. Laparoscopic resection was performed in 24 (27.3%) patients; segmental bowel resection through laparotomy was performed in 13 (14.8%) patients. Gastric heterotopia was found in 39 (44.3%) patients; resection margins of all patients were clear of gastric heterotopia. No correlation was found between the presence of gastric heterotopia and the following parameters: age, gender, maternal age, prematurity, low birth weight, perinatal asphyxia, distance from Bauhin’s valve and length of Meckel’s diverticulum. The width of the diverticulum base was significantly higher in patients with gastric heterotopia (2.1 ± 0.57 vs. 1.2 ± 0.41 cm; p < 0.001).
Conclusions
According to the study outcomes, the width of the diverticulum base seems to be a significant predictive factor associated with the presence of gastric heterotopia in Meckel’s diverticulum. The laparoscopic/open resection of asymptomatic MD with a wide base should therefore be recommended.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Meckel’s diverticulum (MD) is the remnant of the omphalomesenteric (vitelline) duct which is present in 0.3–2.0% of population [1,2,3]. It generally remains asymptomatic, but 4–16% of individuals with MD will become symptomatic, presenting most commonly with bleeding, obstruction, diverticulitis or perforation [4, 5]. These complications are seen more often in neonates, infants and young children compared to adults [2, 6].
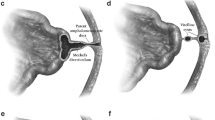
MD containing gastric heterotopia is present in approximately half of the overall MD cases and in up to 80% of symptomatic cases [3, 7]. Gastric heterotopia is a predisposition to local hyperacidity, mucosal ulceration of adjacent ileal mucosa and gastrointestinal bleeding. That is why diverticula that harbor ectopic mucosa carry a significantly higher risk of development of complications [1, 8]. Direct visual examination or palpation of the diverticulum is unreliable in the detection of gastric ectopic mucosa [8].
Although it is widely accepted that symptomatic MD should be resected, the management of MD incidentally detected during an abdominal surgery is still controversial [4, 8,9,10,11,12]. The main argument against routine resection is a low risk of asymptomatic MD becoming symptomatic. A recent systematic review demonstrated that leaving an incidental MD in situ reduces the risk of postoperative complications without increasing late complications [11].
In our opinion, incidentally detected MD with gastric heterotopia should be resected. However, the question remains which asymptomatic MD comprises gastric heterotopia? During an operation, it is usually impossible to determine by inspection or palpation whether an incidentally found MD is at an increased risk of developing future complications or not [4]. Above all, if gastric heterotopia in MD is suspected, segmental bowel resection may be preferred to simple diverticulectomy [5, 11, 12].
To the best of our knowledge, there is no study in the available literature focused on the investigation of risk factors associated with the presence of gastric heterotopia in MD. The aim of the present study was to determine which parameters predict the presence of gastric heterotopia in MD (and should be removed if discovered as an incidental finding).
Materials and methods
Design and setting
This was a retrospective cohort study conducted in University Hospital Ostrava, Czech Republic. The study was approved by the Ethics Committee of the University Hospital Ostrava. All children who underwent laparoscopic/open resection of MD within a 20-year study period (January 1, 1996–December 31, 2016) were assessed for the study eligibility. The exclusion criteria were age ≥ 18 years and incomplete data in operative protocols or histopathology reports.
In an effort to determine which demographic/clinical parameters are predicting the presence of gastric heterotopia in MD, the study patients were divided into two study subgroups (patients with gastric heterotopia vs. patients without heterotopia). The differences in the following parameters between the study subgroups were tested: child’s age and sex, maternal age, prematurity (gestational age under 37 weeks), low birth weight (< 2500 grams), perinatal asphyxia (5th minute Apgar score < 7), distance between MD and Bauhin’s valve, diverticulum length and width of diverticulum base.
Surgical technique
Surgery was performed under general anesthesia in acute or elective settings; the choice of surgical approach (laparoscopic/open) depended on the operating surgeon’s preferences. After proper abdominal cavity exploration, the parameters regarding MD were recorded (length, width, distance from Bauhin’s valve, palpable mass within MD and the presence of inflammation/complications regarding MD). Incidentally detected MD was resected in all patients. Several types of MD resection were performed—wedge resection of MD, segmental bowel resection or resection using an endostapler.
Data collection and analysis
The demographic data, clinical features and histopathology data of all the study patients were extracted from medical records. The acquired data underwent analysis by means of descriptive statistics. The differences between the subgroups (patients with gastric heterotopia vs. patients without heterotopia) were tested using Chi-square test and Fisher’s exact test for categorical variables. For continuous variables, test of normality was performed and Wilcoxon test or two-sample t test was used to test the differences. A level of significance of α = 0.05 (p values < 0.05) were considered statistically significant.
Results
During the study period, 102 pediatric patients underwent surgical treatment of Meckel’s diverticulum in University Hospital Ostrava. Of these, 14 patients were excluded due to exclusion criteria (10 patients with incomplete data in operative protocols; 4 patients with incomplete data in histolopathology reports). In total, 88 pediatric patients were enrolled in the study and underwent analysis.
Surgical resection of symptomatic MD (bleeding, diverticulitis, perforation, obstruction, intussusception) was performed in 38 (43.2%) patients; resection of an incidentally found MD (during laparotomy/laparoscopy for another surgical problem) was performed in 50 (56.8%) patients.
The mean age of children with symptomatic MD was 4.1 ± 4.62 (mean ± SD) years; the mean age of children with asymptomatic MD was 5.0 ± 4.83 years. The frequency of MD decreased with age in our study group (Fig. 1); more than half of the patients were less than 3 years old. There were 59 (67.1%) boys and 29 (32.9%) girls in our study group. The male–female ratio was approximately 2:1 for symptomatic MD cases (68.4 vs. 31.6%) and also for asymptomatic MD cases (66.0 vs. 34.0%).
The majority of symptomatic MD were resected via laparotomy (81.6% of symptomatic patients); diagnostic laparoscopy followed by MD resection through laparotomy was performed in 3 (7.9%) and laparoscopic resection in 4 (10.5%) symptomatic cases. There were 5 stapled resections, 10 segmental bowel resections and 23 diverticulectomies in patients with symptomatic MD. In a subgroup of patients with incidentally found MD, laparoscopic diverticulectomy was performed in 20 (40%) patients. There were 21 stapled MD resections, 3 segmental bowel resections and 26 diverticulectomies.
Histopathology examination revealed the presence of gastric heterotopia in MD in 39 (44.3%) study patients; gastric heterotopia was present in 29 (76.3%) patients with symptomatic MD. Resection margins of all study patients were clear of gastric heterotopia. A palpable mass within asymptomatic MD was detected in 18 patients (36.0%) during surgery; gastric heterotopia was found in all these patients.
A detailed data regarding demographic/clinical parameters of the study patients and the results of the statistically tested differences between both study subgroups are clearly presented in Table 1. There were no statistically significant differences between the study subgroups regarding age, gender, maternal age, prematurity, low birth weight, perinatal asphyxia, distance from Bauhin’s valve and length of MD.
The width of the diverticulum base was significantly higher in patients with gastric heterotopia (2.1 ± 0.57) in comparison with patients without gastric heterotopia (1.2 ± 0.41); the significance was very strong (p < 0.001). Out of 39 patients with gastric heterotopia in our study group, heterotopia was found in 35 patients with diverticulum width ≥ 1.5 cm and in only 4 patients with diverticulum width < 1.5 cm. Our data analysis revealed no correlation between Meckel’s diverticulum width and child’s age.
Receiver operating characteristic (ROC) curve showing the relationship between sensitivity and specificity for gastric heterotopia presence and width of diverticulum base is presented in Fig. 2. Threshold of diverticulum width at 1.5 cm was found to have high specificity (87.76%) and sensitivity (79.49%); area under ROC curve = 0.8362. Thresholds of diverticulum width at 1 and 2 cm had worse detective values (area under ROC curve 0.6656 and 0.5000 respectively).
Discussion
In 1809, Johan Friedrich Meckel was the first to describe the most common congenital malformation of the gastrointestinal tract due to the persistence of the vitello-intestinal duct [13]. Patient with MD may develop symptoms at any point during their lifetime from infancy through to adulthood [10, 14].
Surgery presents a gold standard in the treatment of patients with symptomatic MD. However, the optimal management of MD incidentally detected during an abdominal surgery is controversial [4, 8,9,10,11]. Some authors recommend the resection of all asymptomatic diverticula—the rationale of this approach is to remove a potential cause of symptoms later on in life [8, 15, 16]. In other authors’ view, incidentally detected MD should be left in situ. Soltero et al. in his study of 202 patients revealed that the risk of postoperative complications after prophylactic MD resection was much higher than the risk of complications related to the MD itself. Authors calculated that it would be necessary to perform prophylactic MD resection in more than 800 patients to prevent one death caused by MD [17].
In a recent systematic review, very similar conclusions were drawn by Zani et al. The resection of asymptomatic MD had a significantly higher complication rate than leaving it in situ (5.3 vs. 1.3%; p < 0.001). Moreover, patients after prophylactic MD resection have a higher risk of developing intestinal obstruction or other complications [11]. Authors concluded that leaving an incidental MD in situ reduces the risk of postoperative complications without increasing late complications.
With regard to aforementioned controversies concerning the therapy of asymptomatic MD, it appears sensible to leave the final decision to the operating surgeon (so-called selective approach). Park et al. recommend resection of incidentally detected MD in case of: (1) patients younger than 50 years of age; (2) male patients; (3) MD length greater than 2 cm; (4) detection of an abnormal feature inside the diverticulum. The combination of these four factors was associated with a 70% risk of complications. Authors recommended resection of all incidentally found MD when at least one of these factors was present [9]. Similar risk factors were identified by Mackey et al. [18] in his study: age < 40 years, male gender, MD length > 2 cm, and macroscopic evidence of heterotopic mucosa.
The presence of gastric heterotopia is associated with a significantly higher risk of MD becoming symptomatic [8, 19]. Gastric mucosa releases acidic secretions that yields ulcers in the adjacent intestinal tissue. The most common clinical presentation of symptomatic MD in children is painless rectal bleeding; other symptoms such as abdominal pain, intestinal obstruction or acute inflammation may occur as well [1]. A recent systematic review analyzed data from more than 240 previously published papers; authors concluded that if gastric heterotopia is present, there is a relative risk of 3.64 (95% CI: 3.08–4.28) of the MD becoming symptomatic [11].
A strong correlation between the presence of gastric heterotopia and MD becoming symptomatic leads to the question which incidentally found MD contains gastric heterotopia. Unfortunately, direct visual examination or palpation of the MD is unreliable in the detection of gastric heterotopic mucosa. That is why other clinical factors predicting the presence of heterotopia are needed. Our study revealed a strong association between the diverticulum width and the presence of gastric heterotopia. Published data investigating Meckel’s diverticulum width as a risk factor (for asymptomatic MD becoming symptomatic) are severely insufficient. In the vast majority of available studies, authors have not investigated the width of the diverticulum base as a risk factor or authors do not report that diverticulum width was tested as a risk factor [1,2,3, 5,6,7,8, 10, 14, 15, 17, 18, 20].
According to our study outcomes, the width of Meckel’s diverticulum ≥ 1.5 cm is associated with significantly increased risk of gastric heterotopia presence. No correlation between diverticulum width and child’s age was found. Therefore, resection of asymptomatic MD with a base ≥ 1.5 cm in children should be recommended.
There is no clear consensus regarding the optimal type of MD resection in children. From the technical point of view, there are two types of MD resection—diverticulectomy-only and segmental ileal resection (and anastomosis). Whenever there is a macroscopic involvement of the diverticulum base in patients with symptomatic MD, segmental ileal resection is the only possible solution. In other cases, diverticulectomy-only seems to be an acceptable technique, even if there is a small risk of leaving residual heterotopic tissue in situ [4, 5, 10, 15].
It has been reported that an adequate treatment of a bleeding MD can be accomplished through complete resection of the heterotopic gastric mucosa [5, 21]. In the absence of gastric heterotopia, any residual mucosal ulceration should heal due to an elimination of the acidic environment [22]. Robinson et al. measured the distance between gastric oxyntic cells and resection margins in children presenting with a bleeding MD and demonstrated that diverticulectomy-only completely resects gastric heterotopia without increased risk of continued bleeding or other complications (resection margins of all study patients were clear of gastric heterotopia). In addition, children treated with diverticulectomy-only had shorter operative times and shorter hospital stays than children who underwent segmental ileal resection [5]. The adequacy and effectiveness of diverticulectomy-only in the treatment of a bleeding MD has also been demonstrated by several other authors [10, 11, 21].
One of the principal advantages of diverticulectomy-only technique is that it can be accomplished through laparoscopy without a need for an enterectomy using endostapling devices [20, 22, 23]. Several authors demonstrated the safety feasibility of minimally invasive MD resection. Laparoscopic resection is associated with a shorter operative time, reduced patients’ recovery time and shortened duration of hospital stay compared to segmental ileal resection [10, 12, 20, 21]. Chan et al. pointed out that laparoscopic MD resection in children is underused and may significantly improve outcomes and reduce costs. Pediatric surgeons with appropriate skills should therefore consider laparoscopic approach as a suitable option when dealing with symptomatic/asymptomatic MD [21].
During the past 10 years, more than 700 articles have been published on Meckel Diverticulum (according to PubMed search for “Meckel Diverticulum” from 2007 to 2017). Because of the rare incidence of MD, most publications have either been small series or case reports. Various conclusions (such as the age–sex distribution, clinical presentation and factors associated with symptomatic MD, percentage of ectopic tissue in a symptomatic MD, optimal type of surgical treatment, etc.) have been drawn from these limited series. Our objective was to investigate which demographic/clinical parameters predict the presence of gastric heterotopia in MD. To the best of our knowledge, there is no other study in the available literature focused on risk factors associated with gastric heterotopia presence in MD.
In conclusion, asymptomatic MD harboring ectopic mucosa carries a higher risk of development of complications in the future. Therefore, when the presence of gastric heterotopia in MD is suspected, the diverticulum should be resected. Based on our study outcomes, the width of the diverticulum base seems to be a significant predictive factor associated with the presence of gastric heterotopia in MD. Laparoscopic diverticulectomy presents a safe and feasible surgical approach when dealing with this condition in children.
References
Francis A, Kantarovich D, Khoshnam N et al (2016) Pediatric Meckel’s diverticulum: report of 208 cases and review of the literature. Fetal Pediatr Pathol 35(3):199–206
Çelebi S (2017) Male predominance in Meckel’s diverticulum: a hyperacidity hypotheses. Med Hypotheses 104:54–57
Cserni G (1996) Gastric pathology in Meckel’s diverticulum. Review of cases resected between 1965 and 1995. Am J Clin Pathol 106(6):782–785
Sagar J, Kumar V, Shah DK (2006) Meckel’s diverticulum: a systematic review. J R Soc Med 99:501–505
Robinson JR, Correa H, Brinkman AS et al (2017) Optimizing surgical resection of the bleeding Meckel diverticulum in children. J Pediatr Surg 52(10):1610–1615
Stone PA, Hofeldt MJ, Campbell JE et al (2004) Meckel diverticulum: ten-year experience in adults. South Med J 97(11):1038–1041
Ergün O, Celik A, Akarca US et al (2002) Does colonization of Helicobacter pylori in the heterotopic gastric mucosa play a role in bleeding of Meckel’s diverticulum? J Ped Surg 37(11):1540–1542
Groebli Y, Berlin D, Morel P (2001) Meckel’s diverticulum in adults: retrospective analysis of 119 cases and historical review. Eur J Surg 167:518–524
Park JJ, Wolff BG, Tollefson MK et al (2005) Meckel diverticulum: the Mayo clinic experience with 1476 patients (1950–2002). Ann Surg 241:529–533
Ruscher KA, Fisher JN, Hughes CD et al (2011) National trends in the surgical management of Meckel’s diverticulum. J Pediatr Surg 46:893–896
Zani A, Eaton S, Rees CM et al (2008) Incidentally detected Meckel diverticulum: to resect or not to resect? Ann Surg 247:276–281
Lequet J, Menahem B, Alves A et al (2017) Meckel’s diverticulum in the adult. J Visc Surg 154(4):253–259
Meckel JF (1809) Über die Divertikel am Darmkanal. Archiv für die Physiologie (Halle) 9:421–453
Dumper J, Mackenzie S, Mitchell P et al (2006) Complications of Meckel’s diverticula in adults. Can J Surg 49:353–357
Cullen JJ, Kelly KA, Moir CR et al (1994) Surgical management of Meckel’s diverticulum. An epidemiologic, population-based study. Ann Surg 220:564–568
Bani-Hani KE, Shatnawi NJ (2004) Meckel’s diverticulum: comparison of incidental and symptomatic cases. World J Surg 28:917–920. https://doi.org/10.1007/s00268-004-7512-3
Soltero MA, Bill AH (1976) The natural history of Meckel’s diverticulum and its relation to incidental removal. Am J Surg 132:168–173
Mackey WC, Dineen P (1983) A fifty year experience with Meckel’s diverticulum. Surg Gynecol Obstet 156:56–64
Blaser MJ (1987) Gastric campylobacter-like organisms, gastritis and peptic ulcer disease. Gastroenterology 93:371–383
Lee KH, Yeung CK, Tam YH et al (2000) Laparoscopy for definitive diagnosis and treatment of gastrointestinal bleeding of obscure origin in children. J Pediatric Surg 35(9):1291–1293
Chan KW, Lee KH, Mou JW et al (2008) Laparoscopic management of complicated Meckel’s diverticulum in children: a 10-year review. Surg Endosc 22(6):1509–1512
Mukai M, Takamatsu H, Noguchi H et al (2002) Does the external appearance of a Meckel’s diverticulum assist in choice of the laparoscopic procedure? Pediatr Surg Int 18(4):231–233
Palanivelu C, Rangarajan M, Senthilkumar R et al (2008) Laparoscopic management of symptomatic Meckel’s diverticulum. JSLS 12(1):66–70
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
All authors declare that they have no conflict of interest.
Ethical Approval
All procedures performed in the study were in accordance with the ethical standards of the University hospital and Faculty of Medicine, University of Ostrava, Czech Republic, in accordance with the ethical standards of the Helsinki Declaration of 1975, as revised in 2000. For this type of study formal consent is not required.
Rights and permissions
About this article
Cite this article
Slívová, I., Vávrová, Z., Tomášková, H. et al. Meckel’s Diverticulum in Children—Parameters Predicting the Presence of Gastric Heterotopia. World J Surg 42, 3779–3784 (2018). https://doi.org/10.1007/s00268-018-4664-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-018-4664-0