Abstract
Introduction
While uptake of laparoscopic hepatectomy has improved, evidence on laparoscopic re-hepatectomy (LRH) for colorectal liver metastases (CRLMs) is limited and has never been compared to the open approach. We sought to define outcomes of LRH compared to open re-hepatectomy (ORH).
Methods
Patients undergoing re-hepatectomy for CRLM at 39 institutions (2006–2013) were identified. Primary outcomes were 30-day post-operative overall morbidity, mortality, and length of stay. Secondary outcomes were recurrence and survival at latest follow-up. LRHs were matched to ORHs (1:3) using a propensity score created by comparing pre-operative clinicopathologic factors (number and size of liver metastases and major hepatectomy).
Results
Of 376 re-hepatectomies included, 27 were LRH, including 1 (3.7%) conversion. The propensity-matched cohort included 108 patients. Neither median operative time (252 vs. 230 min; p = 0.82) nor overall 30-day morbidity (48.1 vs. 38.3%; p = 0.37) differed. Non-specific morbidity (including cardiac, respiratory, infectious, and renal events) decreased with LRH (11.1 vs. 30.9%, p = 0.04), while surgical-specific morbidity, including liver insufficiency, was higher (44.4 vs. 22.2%, p = 0.03). One ORH and 0 LRH suffered 30-day mortality. Median length of stay (9 vs. 12 days; p = 0.60) was comparable. At latest follow-up, 26 (96.3%) LRH and 67 (82.7%) ORH patients were alive. Eight (29.6%) LRH and 36 (44.4%) ORH patients were alive without disease.
Conclusion
LRH for recurrent CRLM was associated with overall short-term outcomes comparable to ORH, but different morbidity profiles. While it may offer a safe and feasible approach, further insight is necessary to better define patient selection.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Hepatectomy is now established as the treatment of choice for curative-intent treatment of colorectal liver metastases (CRLMs) to provide excellent long-term outcomes with up to 60% 5-year overall survival [1, 2]. However, recurrence following hepatectomy occurs as often as in 60% of cases, most commonly within the liver [3, 4]. Repeat hepatectomy (RH) has been proven to be a safe and effective strategy in the management of recurrent CRLMs. It provides curative-intent therapy with limited morbidity and mortality profile and favorable long-term outcomes [5,6,7,8].
Minimally invasive surgery is increasingly used by a growing number of surgeons in the treatment of benign and malignant diseases [9,10,11,12,13,14,15,16]. It has been repeatedly shown to provide improved outcomes over open techniques in terms of limited blood loss, reduced morbidity and mortality, and faster recovery [9,10,11,12,13,14,15,16]. However, liver surgery presents unique challenges when it comes to laparoscopic surgical approaches, mostly due to the liver’s cephalic location within the abdomen and the variable intra-hepatic anatomy [17, 18]. Thus, the uptake of minimally invasive surgery for hepatectomy has been slower than in other fields, particularly for CRLMs [19, 20]. The evidence supporting the use of minimally invasive hepatectomy remains limited as highlighted by the Second Laparoscopic Liver Resection Consensus Conference held in Morioka in 2014, even more so for CRLMs [19]. Laparoscopic repeat hepatectomy (LRH) for CRLMs is even less commonly reported; data on its feasibility, safety, and efficacy are scarce [21, 22].
In this study, we sought to define the outcomes of LRH compared to open repeat hepatectomy (ORH), using the large contemporary dataset from the Association Française de Chirurgie (French Surgical Association—AFC) CRLM database.
Methods
We conducted a retrospective propensity-matched comparative cohort study using the AFC CRLM database. The study was designed and reported according to the STROBE and RECORD consensus statements [23, 24].
Data sources
The AFC CRLM database is a questionnaire-based survey of patients undergoing hepatectomy for CRLM. Patients undergoing CRLM resection across 32 French institutions over January 1, 2006, to December 31, 2013, are included. The data collection methods as well as abstracted variables details have previously been published [3, 20]. Each institution was responsible for data entry and submitting files to the AFC. Data collection was standardized through institutions. Files were then anonymized and merged into a single database. In case of missing data (>10% per variable), questionnaires were returned to the institutions. Timing and modalities of follow-up after resection were based on institutional practices. Patients lacking long-term follow-up information or with outlying values were excluded. The authors had complete access to the data included in the final dataset.
Selection of participants
We identified all adult patients (≥18 years old) undergoing RH in the AFC CRLM from January 1, 2006, to December 31, 2013. We excluded patients undergoing RH as the second stage of two-stage procedure or deemed unresectable at the time of surgery. Patients were divided into two groups: those who underwent LRH and those who underwent ORH. The choice of surgical approach was at the discretion of the attending surgeon. Patients undergoing LRH converted to an open procedure were included in the LRH group.
Outcomes and data collection
The primary outcomes were 30-day post-operative overall morbidity and mortality. Post-operative morbidity was assessed at 30 days following surgery and subdivided into non-specific morbidity (including cardiac, respiratory, infectious, thromboembolic, and renal events) and specific morbidity (including surgical site infection, liver insufficiency, and biliary leak). Liver insufficiency was defined as persistent rise in bilirubin and INR levels at post-operative day 5 as per the “50/50” criteria [25]. Secondary outcomes were length of stay, recurrence-free survival (RFS—from date of RH to date of recurrence), and vital status at last follow-up (alive without disease, alive with disease, or deceased). Recurrence was defined as intra- or extrahepatic biopsy-proven recurrent adenocarcinoma or a lesion deemed suspicious on cross-sectional imaging, following RH.
We extracted baseline demographics as well as clinical, operative, and post-operative characteristics from the database. Extrahepatic disease was defined as the presence of any non-liver site of colorectal cancer either biopsy proven or identified as a concerning lesion on cross-sectional imaging. Major hepatectomy was considered as the resection of three or more liver segments. Use of systemic therapy was captured and divided into pre-operative (administration of any chemotherapy agent in the 6 months prior to hepatectomy) and post-operative (in the 6 months following hepatectomy).
Statistical analysis
The characteristics of included patients were described stratified by LRH compared to ORH. Categorical data were reported as absolute numbers (n) with proportion (%), and continuous variables as median with interquartile range (IQR). Groups were compared using Chi-square, Fischer exact test, or ANOVA, as appropriate.
The Kaplan–Meier method was used to estimate RFS [26]. Patients were censored at the end of follow-up, defined as date of death or date of last clinical encounter. Differences in RFS between LRH and ORH were examined with the log-rank test [26].
Propensity scoring was used to account for clinical differences between groups. The propensity score was computed using logistic regression modeling with LRH as the dependent variable. Variables included in the propensity score were chosen based on their clinical relevance in the decision to proceed with LRH as opposed to ORH and baseline differences between groups. Age, body mass index category, American Society of Anesthesiology (ASA) classification, solitary liver metastasis, largest liver lesion over 3 cm, and major liver resection were used in the propensity score calculation. The score was then used to match LRH to ORH (1:3) using a nearest-neighbor algorithm, to create comparable cohorts to assess the impact of LRH on outcomes.
Outcomes were compared before (entire cohort) and after matching (matched cohort). Logistic regression models were constructed to assess the association between LRH and primary outcomes. Results were reported as odds ratios (OR) with 95% confidence interval (95%CI). Statistical significance was set at p ≤ 0.05. All analyses were conducted with SPSS 21.0 (IBM Corp., Amon, NY).
Results
Characteristics of the cohort
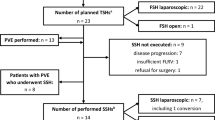
From 3036 entries in the AFC CRLM database, 447 were RH. Of those, 27 were performed with minimally invasive approach and were not missing data for propensity scoring (Fig. 1). LRHs were performed in five institutions. Propensity score matching was used to minimize confounding by indication for surgical approach and thereby create more comparable groups. The matched cohort includes 27 LRH and 82 ORH.
Demographic and clinical characteristics of the entire and matched cohorts are presented in Table 1. Operative characteristics are detailed in Table 2. As expected, there were baseline differences in patients undergoing LRH and ORH. Patients undergoing LRH were operated on more commonly for fewer and larger liver lesions. Larger amount of liver parenchyma was resected with LRH than ORH as evidenced by more common major liver resection (92.6 vs. 24.1%). The LRH group included 25 major liver resections: two left lobectomies, eleven right lobectomies, five right trisegmentetctomies, one left trisegmentectomy, one left lobectomy with wedge resection of segment 8, one right lobectomy with wedge resection of segment 4, and one right lobectomy with wedge resection of segment 2. There was one (3.1%) conversion from LRH to open surgery. No difference was observed in operative time or receipt of blood transfusion between the LRH and ORH groups.
Figure 2a, b presents standardized differences between groups before and after propensity matching and shows adequate balance between groups in the matched cohort for the variables included in the propensity score.
Short-term post-operative outcomes
Figure 3 depicts the 30-day post-operative morbidity and mortality in the entire (Fig. 3a) and the matched (Fig. 3b) cohorts. No post-operative mortality occurred in the LRH group, compared to five (1.4%) events in the entire ORH group and one (1.2%) event in the matched ORH group. Overall morbidity did not differ between groups in the entire and the matched cohorts. Specific morbidity was more frequent in the LRH group in the entire cohort (p < 0.001). This difference was maintained in the matched cohort (44.4 vs. 22.2%; p = 0.026). Analysis of principal causes of specific morbidity revealed higher occurrence of liver insufficiency in the LRH group. There was no difference observed in terms of non-specific morbidity. On regression analysis, LRH was significantly associated with 30-day specific morbidity (OR 2.88; 95%CI: 1.11–7.04), but not with overall morbidity or non-specific morbidity (Table 3). The low number of events in each group did not allow for adjustments for additional covariates.
While median length of stay was shorter for LRH in the entire cohort, with 9 (IQR: 8–18) days compared to 10 (IQR: 8–13) days (p = 0.019), no difference was observed after matching, with 9 (IQR: 8–18) days compared to 12 (IQR: 9–17) days (p = 0.602).
Long-term post-operative outcomes
No statistically significant difference was observed in the ability to achieve R0 resection with LRH and ORH in the entire cohort (84.6 vs. 64.5%; p = 0.233) or the matched cohort (84.6 vs. 72.7%; p = 0.297).
Median follow-up was 20.7 (IQR: 3.3–60.2) months for the LRH group, 15.4 (IQR: 5.7–35.9) months for the entire ORH group, and 23.2 (IQR: 6.4–41.4) months for the matched ORH group. Recurrence did not differ between LRH and ORH in the entire cohort (67.7 vs. 51.9%; p = 0.138). It also did not reach statistical difference in the matched cohort (66.7 vs. 46.9%; p = 0.075). Median time to recurrence did not differ between LRH with 10.8 (IQR: 7.4–17.5) months, and ORH in the entire cohort with 10.2 (IQR: 4.6–17.9) months (p = 0.932), or the matched cohort with 11.0 (IQR: 7.4–17.5) months (p = 0.483).
RFS is presented in Fig. 4. It did not differ, with 5-year RFS of 21.4% for LRH compared to 25.6% for ORH (p = 0.938), in the matched cohort. Due to small number of death events, actuarial overall survival could not be computed. At latest follow-up in the matched cohort, 96.3% of patients undergoing LRH and 82.7% undergoing ORH were alive. Proportion of patients alive without disease at the latest follow-up were 29.6 and 44.4% for LRH and ORH, respectively.
Discussion
In this multi-institutional propensity-matched study, we compared the outcomes of LRH to ORH. While post-operative overall morbidity did not differ between groups, different morbidity profiles were identified. LRH was associated with 2.88 times higher odds of specific morbidity, compared to ORH. No difference was observed in post-operative recovery as assessed by length of stay. Long-term oncologic outcomes assessed by RFS and survival status at latest follow-up did not significantly differ whether patients underwent LRH or ORH.
There is a growing body of literature reporting the effectiveness of RH for the curative-intent management of recurrent CRLMs [5,6,7, 27, 28]. Favorable long-term survival can be achieved with RH and is superior to that obtained with palliative chemotherapy alone [5, 7, 28,29,30]. RH presents specific challenging that makes it theoretically more demanding than the initial hepatectomy. The liver parenchyma is more friable from prior regeneration and potential pre-operative chemotherapy, there is less parenchyma to preserve the future liver remnant function, adhesions render exposure more difficult, and changes in extra- and intra-hepatic anatomy from prior surgery result in more challenging dissection. Nevertheless, RH can be performed with similar morbidity and mortality profiles as the initial hepatectomy [27, 28, 31].
The use of minimally invasive surgery has increased significantly over the past decades. Due to reduced morbidity and enhanced recovery, it is now standard of care for a number of procedures to treat benign and malignant diseases [9,10,11,12,13,14,15,16]. The uptake of laparoscopic liver surgery has been slower, with 14–25% of hepatectomies performed laparoscopically in international series [32, 33]. This proportion drops to 6.7% when looking specifically at CRLMs [20]. Nevertheless, when an initial hepatectomy is performed laparoscopically, it is associated with improved short-term outcomes [20, 34, 35].
While the use of LRH has been reported in a limited number of case series, its outcomes have never been compared to those of ORH [21, 22, 36]. This multi-institutional study is the first to compare the results of LRH to ORH. It is an important issue to address. Laparoscopic surgery poses a number of challenges that may be more difficult to overcome for liver resections, including loss of three-dimension vision, hand–eye coordination, access to the liver dome, and visualization and tracking of deep seated lesions [37, 38]. This may be more pronounced when already dealing with the technical difficulties of repeat liver surgery.
This analysis did not identify a significant difference in overall morbidity between LRH and ORH. The selection bias involved in deciding to proceed with LRH or ORH might have contributed to this, as evidenced by different patient characteristics at baseline. Propensity score matching was used to mitigate this [39]. Due to the overall small number of LRHs, it was not possible to assess the impact of interinstitution variability on the reported morbidity. While the current findings seem to oppose reports pertaining to laparoscopic initial hepatectomy, it is important to outline that the morbidity reduction with laparoscopic liver resection is not consistent across studies [35]. This lack of difference may be explained by the special nature of CRLM surgical management. Resection of CRLM is not always ideally suited for laparoscopic approach. In particular, parenchymal sparing principles are key in optimizing post-operative outcomes [40, 41]. This is even more crucial for RH and is more challenging laparoscopically due to the difficult access to some areas of the liver and hampered ability to visualize and track lesions [37, 38]. A high proportion of LRH was major liver resections in this series, which parallels other reports [21, 22]. Resection of larger amounts of parenchyma might thus have been needed to allow for LRH and impacted outcomes. This would explain why the pattern of morbidity varied. Surgery specific morbidity, including liver insufficiency, was more common with LRH. Non-specific morbidity was lower with ORH, mostly due to fewer respiratory events, a known benefit of minimally invasive techniques [42].
It is acknowledged that this study presents limitations, mostly due to its retrospective nature and small sample size. It appraised the results of an overall uncommon procedure in a multi-institutional setting. The decision to proceed with LRH or ORH was left to the surgeons. While propensity score matching was used to account for this selection bias, it cannot correct for all potential confounders. In addition, the dataset used was not created specifically for the purposes of the research question herein addressed. Therefore, some interesting variables were not available. In particular, the details of the initial hepatectomies were not known, including approach (laparoscopic or open), extent of resection, and use of chemotherapy prior to the repeat hepatectomy episode. The impact of the laparoscopic approach on intra-operative blood loss could not be assessed since this difficult to capture variable was lacking from the dataset. This was, however, assessed by examining the use of blood transfusion, which did not differ between LRH and ORH. Finally, LRH was performed by expert laparoscopic liver surgeons, which is critical in achieving good outcomes. For the results reported herein to apply, surgeons have to ensure they received good training in laparoscopic liver resection and completed the learning curve on initial hepatectomies [19, 43, 44].
Nevertheless, this study represents a contemporary multi-institutional assessment of LRH and compared it to ORH for the first time. It provides important insight into the potential benefits and drawbacks of LRH. While good outcomes can be achieved, there is still work to be done before RH can be performed laparoscopically on a routine basis. First, the uptake of laparoscopic liver resection for CRLM should increase such that liver surgeons complete the long but necessary learning curve [19, 43, 44]. Second, development of improved navigation and operative tools would ensure that parenchymal sparing principles can be adhered to [37]. At this moment, LRH appears best to be performed by experts in the fields and in highly selected patients.
Conclusion
LRH for recurrent CRLM can be safely achieved with overall short-term outcomes comparable to ORH. However, morbidity profiles differed; LRH was associated with more common specific morbidity such as liver insufficiency, but lower non-specific morbidity such as respiratory events. While LRH offers a feasible approach in selected patients and by expert hands, further insight and development of laparoscopic liver techniques appear necessary prior to widespread use.
References
Choti MA, Sitzmann JV, Tiburi MF et al (2002) Trends in long-term survival following liver resection for hepatic colorectal metastases. Ann Surg 235:759–766
Abdalla EK, Vauthey J-N, Ellis LM et al (2004) Recurrence and outcomes following hepatic resection, radiofrequency ablation, and combined resection/ablation for colorectal liver metastases. Ann Surg 239:818–825
Hallet J, Sa Cunha A, Adam R et al (2016) Factors influencing recurrence following initial hepatectomy for colorectal liver metastases. Br J Surg. doi:10.1002/bjs.10191
de Jong MC, Pulitano C, Ribero D et al (2009) Rates and patterns of recurrence following curative intent surgery for colorectal liver metastasis: an international multi-institutional analysis of 1669 patients. Ann Surg 250:440–448
Adam R, Bismuth H, Castaing D et al (1997) Repeat hepatectomy for colorectal liver metastases. Ann Surg 225:51–60
Adam R, Pascal G, Azoulay D et al (2003) Liver resection for colorectal metastases: the third hepatectomy. Ann Surg 238:871–883 (discussion 883–4)
Petrowsky H, Gonen M, Jarnagin W et al (2002) Second liver resections are safe and effective treatment for recurrent hepatic metastases from colorectal cancer: a bi-institutional analysis. Ann Surg 235:863–871
Shaw IM, Rees M, Welsh FKS et al (2006) Repeat hepatic resection for recurrent colorectal liver metastases is associated with favourable long-term survival. Br J Surg 93:457–464
Schlachta CM, Mamazza J, Grégoire R et al (2003) Could laparoscopic colon and rectal surgery become the standard of care? A review and experience with 750 procedures. Can J Surg 46:432–440
Smith CD, Weber CJ, Amerson JR (1999) Laparoscopic adrenalectomy: new gold standard. World J Surg 23:389–396. doi:10.1007/PL00012314
Glasgow RE, Yee LF, Mulvihill SJ (1997) Laparoscopic splenectomy: the emerging standard. Surg Endosc 11:108–112
Keus F, de Jong JAF, Gooszen HG, van Laarhoven CJHM (2006) Laparoscopic versus open cholecystectomy for patients with symptomatic cholecystolithiasis. Cochrane Database Syst Rev. doi:10.1002/14651858.CD006231
Kuo PC, Johnson LB, Sitzmann JV (2000) Laparoscopic donor nephrectomy with a 23-hour stay: a new standard for transplantation surgery. Ann Surg 231:772–779
Lujan J, Valero G, Biondo S et al (2013) Laparoscopic versus open surgery for rectal cancer: results of a prospective multicentre analysis of 4,970 patients. Surg Endosc 27:295–302
Jayne DG, Guillou PJ, Thorpe H et al (2007) Randomized trial of laparoscopic-assisted resection of colorectal carcinoma: 3-year results of the UK MRC CLASICC Trial Group. J Clin Oncol 25:3061–3068
Hallet J, Labidi S, Bouchard-Fortier A et al (2013) Oncologic specimen from laparoscopic assisted gastrectomy for gastric adenocarcinoma is comparable to D1-open surgery: the experience of a Canadian centre. Can J Surg 56:249–255
Massimino KP, Han E, Pommier SEJ, Pommier RF (2012) Laparoscopic surgical exploration is an effective strategy for locating occult primary neuroendocrine tumors. Am J Surg 203:628–631
Ban D, Tanabe M, Ito H et al (2014) A novel difficulty scoring system for laparoscopic liver resection. J Hepato Biliary Pancreat Sci 21(6):745–753
Wakabayashi G, Cherqui D, Geller DA et al (2015) Recommendations for laparoscopic liver resection: a report from the second international consensus conference held in Morioka. Ann Surg 261:619–629
Allard M-A, Sa Cunha A, Gayet B et al (2015) Early and long-term oncological outcomes after laparoscopic resection for colorectal liver metastases. Ann Surg 262:794–802
Shafaee Z, Kazaryan AM, Marvin MR et al (2011) Is laparoscopic repeat hepatectomy feasible? A Tri-institutional analysis. JACS 212:171–179
Shelat VG, Serin K, Samim M et al (2014) Outcomes of repeat laparoscopic liver resection compared to the primary resection. World J Surg 38:3175–3180. doi:10.1007/s00268-014-2728-3
von Elm E, Altman DG, Egger M et al (2007) Strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. BMJ 335:806–808
Benchimol EI, Smeeth L, Guttmann A et al (2015) The reporting of studies conducted using observational routinely-collected health data (RECORD) statement. PLoS Med 12:e1001885
Balzan S, Belghiti J, Farges O et al (2005) The “50-50 criteria” on postoperative day 5. Ann Surg 242:824–829
Kaplan EL, Meier P (1958) Nonparametric estimation from incomplete observations. J Am Stat Assoc 53:457–481
Sa Cunha A (2007) A second liver resection due to recurrent colorectal liver metastases. Arch Surg 142:1144–1149
de Jong MC, Mayo SC, Pulitano C et al (2009) Repeat curative intent liver surgery is safe and effective for recurrent colorectal liver metastasis: results from an international multi-institutional analysis. J Gastrointest Surg 13:2141–2151
Meyerhardt JA, Mayer RJ (2005) Systemic therapy for colorectal cancer. N Engl J Med 352:476–487
Brandi G, Corbelli J, de Rosa F et al (2012) Second surgery or chemotherapy for relapse after radical resection of colorectal cancer metastases. Langenbecks Arch Surg 397:1069–1077
Luo LX, Yu ZY, Huang JW, Wu H (2014) Selecting patients for a second hepatectomy for colorectal metastases: an systemic review and meta-analysis. Eur J Surg Oncol 40:1036–1048
Nguyen KT, Gamblin TC, Geller D (2009) World review of laparoscopic liver resection—2,804 patients. Ann Surg 250:831–841
Goumard C, Farges O, Laurent A et al (2015) An update on laparoscopic liver resection: the French Hepato-Bilio-Pancreatic Surgery Association statement. Journal of Visceral Surgery 152:107–112
Topal B, Fieuws S, Aerts R et al (2008) Laparoscopic versus open liver resection of hepatic neoplasms: comparative analysis of short-term results. Surg Endosc 22:2208–2213
Hallet J, Beyfuss KA, Memeo R et al. (2016) Short and long-term outcomes of laparoscopic compared to open liver resection for colorectal liver metastases. Hepatobiliary Surg Nutr 5:300–310
Nomi T, Fuks D, Ogiso S et al. (2016) Second and third laparoscopic liver resection for patients with recurrent colorectal liver metastases. Ann Surg 263:e68–e72
Hallet J, Gayet B, Tsung A et al (2015) Systematic review of the use of pre-operative simulation and navigation for hepatectomy: current status and future perspectives. J Hepato Biliary Pancreat Sci 22(5):353–362
Ban D, Kudo A, Ito H et al. (2015) The difficulty of laparoscopic liver resection. Updates Surg 67:123–128
Lonjon G, Boutron I, Trinquart L et al (2014) Comparison of treatment effect estimates from prospective nonrandomized studies with propensity score analysis and randomized controlled trials of surgical procedures. Ann Surg 259:18–25
Gold JS, Are C, Kornprat P et al (2008) Increased use of parenchymal-sparing surgery for bilateral liver metastases from colorectal cancer is associated with improved mortality without change in oncologic outcome: trends in treatment over time in 440 patients. Ann Surg 247:109–117
Kingham TP, Correa-Gallego C, D’Angelica MI, et al. (2015) hepatic parenchymal preservation surgery: decreasing morbidity and mortality rates in 4,152 resections for malignancy. J Am Coll Surg 220:471–479
Fuks D, Cauchy F, Ftériche S et al (2016) Laparoscopy decreases pulmonary complications in patients undergoing major liver resection: a propensity score analysis. Ann Surg 263:353–361. doi:10.1097/SLA.0000000000001140
Viganò L, Laurent A, Tayar C et al (2009) The learning curve in laparoscopic liver resection: improved feasibility and reproducibility. Ann Surg 250:772–782
Nomi T, Fuks D, Kawaguchi Y et al (2015) Learning curve for laparoscopic major hepatectomy. Br J Surg 102:796–804
Collaborators of the French Colorectal Liver Metastases Working Group, Association Française de Chirurgie
Amiens, CHU Amiens Picardie: Cyril Cosse, Delphine Lignier, Jean Marc Regimbeau; Angers, CHU Angers: Julien Barbieux, Emilie Lermite, Antoine Hamy; Beauvais, CH Beauvais: François Mauvais; Bordeaux, Groupe Hospitalier Saint André: Christophe Laurent; Chambery, CH Chambery: Irchid Al Naasan; Créteil, CHU Henri Mondor: Alexis Laurent, Daniel Azoulay, Philippe Compagnon, Chetana Lim; Eaubonne, Hôpital Simone Veil: Mohammed Sbai Idrissi; Epinal, Polyclinique de la Ligne Bleue: Fréderic Martin; Gap, CH des Alpes du Sud: Jerôme Atger; Lyon, Hôpital de la Croix Rousse: Jacques Baulieux, Benjamin Darnis, Jean Yves; Mabrut; Lyon, Hôpital Edouart-Herriot: Vahan Kepenekian, Julie Perinel, Mustapha Adham; Lyon, CH Lyon Sud: Olivier Glehen; Lyon, Centre Léon Bérard: Michel Rivoire; Marseille, Hôpital de la Conception: Jean Hardwigsen, Anais Palen, Emilie Grégoire, Yves Patrice LeTreut; Marseille, Institut Paoli-Calmettes: Jean Robert Delpero, Olivier Turrini; Montpellier, Hôpital Saint Éloi: Astrid Herrero, Francis Navarro, Fabrizio Panaro; Nancy, CHU Brabois: Ahmet Ayav, Laurent Bresler; Nancy, Institut de Cancérologie de Lorraine Alexis-Vautrin: Philippe Rauch, François; Guillemin, Fréderic Marchal; Nice, Hôpital de l’Archet: Jean Gugenheim, Antonio Iannelli; Kremlin Bicêtre, CHU Kremlin Bicêtre: Stephane Benoist, Antoine Brouquet; Paris, Hôpital Lariboisière: Marc Pocard, Réa Lo Dico; Paris, Institut Mutualiste Montsouris: Brice Gayet, David Fuks; Paris, Hôpital Saint Antoine: Olivier. Scatton, Olivier Soubrane; Paris, Hôpital de la Pitié Salpetrière: Jean-Christophe Vaillant; Reims, Hôpital Robert Debré: Tullio Piardi, Daniel Sommacale, Reza Kianmanesh; La Roche-sur-Yon, Centre départemental de Vendée: Michel Comy; Strasbourg, Hôpital Hautepierre: Philippe Bachellier, Elie Oussoultzoglou, Pietro Addeo; Strasbourg, Nouvel Hôpital Civil: Dimitrios Ntourakis, Patrick Pessaux, Didier Mutter, Jacques Marescaux; Toulouse, Hôpital Rangueil: Loïc Raoux, Bertrand Suc, Fabrice Muscari; Troyes, Hôpital des Hauts-Clos: Georges ELHOMSY; Villejuif, Hôpital Paul Brousse: Maximiliano Gelli, Antonio Sa Cunha, René Adam, Denis Castaing, Daniel Cherqui; Gabriella PIttau, Oriana Ciacio, Eric Vibert; Villejuif, Gustave Roussy: Dominique Elias, Diane Goéré, Fabrizio Vittadello.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Hallet, J., Sa Cunha, A., Cherqui, D. et al. Laparoscopic Compared to Open Repeat Hepatectomy for Colorectal Liver Metastases: a Multi-institutional Propensity-Matched Analysis of Short- and Long-Term Outcomes. World J Surg 41, 3189–3198 (2017). https://doi.org/10.1007/s00268-017-4119-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-017-4119-z