Abstract
Background
Diffuse sclerosing variant (DSV) of papillary thyroid carcinoma (PTC) is a rare variant more common among younger patients.
Materials and methods
Excluding patients with microcarcinoma, 5848 patients with PTC underwent initial surgery between 1995 and 2011. Twenty-two patients (0.4 %) were histologically diagnosed with DSV, of whom 20 (91 %) were <45 years old. We compared clinicopathologic characteristics and outcomes between patients with DSV and those with classical PTC <45 years old. Univariate analysis by the Kaplan–Meier method in relation to cause-specific survival (CSS) and disease-free survival (DFS) rates was performed with regard to the following variables: sex; anti-thyroglobulin antibody (TgAb) positivity; presence of distant metastasis; pathological lymph node metastasis; extra-thyroidal invasion; and pathological variant (classical vs. DSV).
Results
The 20 patients with DSV <45 years old comprised 18 females and 2 males. Frequencies of TgAb, pN1b, and local recurrence were higher in the DSV group than in the classical PTC group. Ten-year CSS and DFS rates for PTC patients <45 years old were 99.7 and 88.6 % in the classical PTC group and 100 and 60.5 % in the DSV group. CSS rate did not differ between groups, but DFS rate was significantly lower in the DSV group than in the classical PTC group (p < 0.0001, log-rank test). Multivariate analysis identified DSV group and pN1b as prognostic factors for recurrence in young PTC patients.
Conclusions
Most DSV patients were young and had a background of chronic thyroiditis. Outcomes for DSV were very good, but recurrence was more common than in classical PTC.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
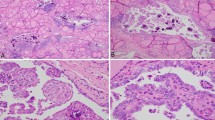
Papillary carcinoma is the most common type of well-differentiated carcinoma of the thyroid. In 1985, Vickery et al. described a new variant of PTC, for which they proposed the term “diffuse sclerosis variant” [1]. According to the World Health Organization (WHO) classification, DSV tumors tend to occur in young patients and are characterized by diffuse involvement of one or both thyroid lobes, usually without formation of a dominant mass, and by extensive squamous metaplasia, numerous psammoma bodies, dense lymphocytic infiltration, and stromal fibrosis. DSV was recognized as a histological variant of PTC in 1988 [2].
Previous studies have demonstrated outcomes for DSV [2–26], with some suggesting high incidences of lymph node metastasis and lung metastasis, and thus a less favorable prognosis compared with classical PTC [3, 4, 8, 21, 23, 27]. Other reports have suggested that the prognosis for DSV is no different from that for classical PTC [6, 9, 12, 15, 19, 20, 22]. In Japan, Fujimoto et al. reported that all 14 of their DSV patients remained alive without distant metastasis as of a mean follow-up period of 16 years [6], while Fukushima et al. reported a better cause-specific survival (CSS) rate was better in DSV patients than in other PTC patients, whereas the disease-free survival (DFS) rate was lower [23]. Pediatric thyroid carcinoma patients show a higher incidence of recurrence and higher prevalence of lymph node metastasis and pulmonary metastasis [27]. To assess the biological aggressiveness of DSV, we retrospectively reviewed the medical records of patients with DSV treated in our hospital. PTC patients in this hospital are staged according to the TNM classification, and separate stage groupings are recommended for patients <45 and ≥45 years old, because age influences the prognosis of PTC. We therefore compared clinicopathologic features and outcomes between DSV patients and classical PTC patients <45 years old.
Materials and methods
Patients
Between 1995 and 2011, a total of 5848 patients underwent initial surgery for PTC, excluding papillary microcarcinoma, at Ito Hospital in Tokyo. Of these, 22 patients (0.4 %) were histologically diagnosed with DSV. All PTC patients underwent ultrasonography (US) and US-guided fine-needle aspiration (FNA) biopsy preoperatively. All 22 DSV patients were diagnosed on the basis of histological examination of surgical specimens, and lesions were classified according to the WHO classification of malignant tumors [2] and staged according to the TNM classification system of the Union for International Cancer Control. We compared clinicopathologic characteristics and prognosis between the 1194 patients who were <45 years old with the presence of lymph node metastasis (1174 classical PTC patients and 20 DSV patients), because most DSV patients (91 %) were <45 years old and all DSV patients showed the presence of lymph node metastasis. To match for N factor between these groups, patients showing positive results for pathological lymph node metastases (pN1) were selected, because all DSV patients showed the presence of pathological lymph node metastasis. Patients underwent central neck lymph node dissection (CND) as routine dissection when diagnosed with PTC preoperatively. Patients diagnosed preoperatively with lateral lymph node metastasis by US-guided FNA underwent lateral compartment neck dissection (level II–V) in addition to routine dissection of the central compartment (level VI). As a result, all DSV patients underwent CND and 17 patients underwent uni- or bilateral modified neck lymph node dissection (MND).
In our institution, routine radioiodine ablation therapy after initial surgery is not performed unless distant metastasis or massive extra-thyroidal extension and lymph node metastasis is clinically apparent at surgery, because the use of radioiodine ablation therapy has been limited in Japan. In the 1194 young patients, CSS rates were calculated and compared according to the following variables: sex; anti-thyroglobulin antibody (TgAb) positivity; presence of distant metastasis; presence of pathological lymph node metastasis (pN1a vs. pN1b); extra-thyroidal invasion; and pathological variant (classical PTC vs. DSV). DFS rate was also calculated for the 1152 young patients (classical PTC, n = 1134; DSV, n = 18) who did not show distant metastasis at presentation and who underwent locally curative surgery.
Statistical analysis
The Kaplan–Meier method was used to calculate cumulative survival rate for patients from the date of diagnosis to the date of death or most recent follow-up examination. The impact of various factors on survival was analyzed using the log-rank test. Multivariate analyses of prognostic factors were based on Cox proportional hazards modeling. Values of p < 0.05 were considered statistically significant. All statistical analyses were performed using JMP version 8.0 software (SAS Institute, Cary, NC, USA).
Results
Clinical features of DSV
Twenty PTC patients <45 years old were diagnosed with DSV, and their clinicopathologic characteristics are shown in Table 1. DSV patients comprised 18 females (90 %) and 2 males (10 %), and median age at diagnosis was 25 years (range 9–35 years). The most common initial manifestations were neck swelling (85 %) and general fatigue (10 %). One DSV patient (5 %) was receiving pharmacotherapy for Graves’ disease. Seventeen DSV patients (85 %) tested positive for TgAb or thyroid peroxidase antibody (TPOAb), and 2 DSV patients (10 %) were receiving pharmacotherapy for Hashimoto’s thyroiditis. Thyroid functions at presentation were euthyroid in 17 patients (85 %), hypothyroid in 2 (10 %), and hyperthyroid in 1 (5 %).
Preoperative US showed that 8 DSV patients (40 %) had thyroid carcinomas in both thyroid lobes, and 12 (60 %) had thyroid carcinoma in only one lobe. All patients showed common US features such as heterogeneous echotexture, solid composition, ill-defined margins, and scattered microcalcifications. The sensitivity of PTC diagnosis by FNA was very high in our series (100 %). All DSV patients underwent initial surgery, comprising total thyroidectomy (TT) in 17 patients and lobectomy (LT) in 3 patients. All DSV patients underwent CND and 17 patients (85 %) underwent uni- or bilateral MND. All 20 DSV patients showed positive results for pathological lymph node metastasis. Radioiodine ablation therapy (RAT) was performed in 12 DSV patients, accounting for 71 % of patients who underwent total thyroidectomy. Two patients with lung metastasis at presentation were treated by total thyroidectomy and RAT (131I, 100 mCi), of whom one achieved complete response. One patient remains alive as of 18 years postoperatively, and the other patient remains alive at 10 years postoperatively.
Outcomes for young patients with DSV and classical PTC
We compared clinicopathologic characteristics between young patients in the DSV group and young patients in the classical PTC group (Table 2). Frequencies of TgAb, pN1b, and local recurrences were higher in patients with DSV than in those with classical PTC. Differences between groups in incidences of distant metastasis and extra-thyroidal extension were not significant.
Prognosis in DSV and classical PTC
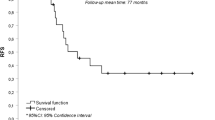
Median overall follow-up after surgery was 9.0 years. None of the DSV patients died of their disease, whereas 3 patients with PTC died due to distant metastases. The overall 10-year CSS rate was 100 % in the DSV group and 99.7 % in the classical PTC group (Fig. 1). The present results showed no significant difference between the DSV and classical PTC groups in terms of CSS rate. In 1152 patients (1134 patients with classical PTC, 18 patient with DSV) who did not show distant metastasis at presentation and who underwent locally curative surgery, the 10-year DFS was 60.5 % in patients with DSV and 88.6 % in those with classical PTC (Fig. 2). The DFS rate was significantly lower in the DSV than in the classical PTC group (p < 0.0001). Seven patients (35 %) were diagnosed with lymph node recurrence, and all patients underwent re-operation (Table 3). The site of lymph node recurrence was the lateral compartment (level III and IV); of those, 5 patients (71 %) showed lymph node recurrences at the operation site. In DSV patients <45 years old, the median duration to recurrence after surgery was 1.4 years. Details of the eight patients who were diagnosed with distant metastasis or lymph node recurrence are shown in Table 3.
Prognostic factor for recurrence
In the 1152 young patients with PTC including DSV (classical PTC, n = 1134; DSV, n = 18) who did not show distant metastasis at presentation and who underwent locally curative surgery, we examined prognostic factors associated with recurrence, either of lymph node recurrence or distant metastases. Univariate analysis identified male sex, extra-thyroidal invasion (pEx2), lateral lymph node metastasis (pN1b), and histological type (DSV) as significant prognostic factors for recurrence (Fig. 3), whereas multivariate analysis only identified lateral lymph node metastasis (pN1b) and DSV as independent prognostic factors for recurrence (Table 4).
a Comparison of DFS rates between female and male. b Comparison of DFS rates between patients with pEx 0 or 1 and pEx2. c Comparison of DFS rates between patients with pN1a and pN1b. d Comparison of DFS rates between patients with central compartment dissection, and those with both central and lateral compartment dissection
Discussion
We examined a large series of 22 patients diagnosed with DSV over 16 years in our institution. Previous studies examining larger series of PTC patients have reported prevalence of DSV ranging from 0.74 to 5.3 % of PTC patients as a whole [19]. In our hospital, DSV accounted for 0.4 % of PTCs excluding microcarcinoma. The prevalence of DSV was lower in our series than in other previous studies. Previous reports of DSV exclusive of case reports are summarized in Table 5. Koo et al. reported DSV as the most prevalent subtype of pediatric papillary carcinoma PTC in their series [25]. The mean age of DSV patients in our study was 27.6 years (range 9–59 years), similar to that of DSV patients in previous studies. We therefore studied outcomes in subgroups of young patients <45 years old, because age is thought to influence the prognosis of DSV.
Preoperative neck US may be helpful in detecting intra-thyroidal extension and lymph node metastases in DSV patients. The US features of DSV were heterogeneous echotexture, solid composition, ill-defined margins, scattered microcalcifications with a “snowstorm” appearance, and various echogenicities [28]. In this study, all patients showed the US features of scattered microcalcifications. Eight (40 %) patients showed intra-thyroidal extension of bilateral lobes. Sixteen (80 %) patients showed positive findings for lymph node metastases on preoperative US. The sensitivity of PTC diagnosis by FNA was very high in our series (100 %). DSV can be diagnosed preoperatively based on a combination of typical imaging findings and cytological examination. The sensitivity of PTC diagnosis by FNA was very high in our series (100 %). DSV can be diagnosed preoperatively based on a combination of typical imaging findings and careful cytological examination.
In our series, 91 % of DSV patients complained of neck swelling, and 85 % tested positive for TgAb or TPOAb and showed a background of chronic lymphocytic thyroiditis. In one previous study, approximately 30 % of DSV patients tested positive for TgAb [20], while in another study, a higher percentage of DSV patients were TgAb-positive (DSV 72 vs. PTC 42 %) [23]. In the present series, TgAb-positive results were seen in a higher proportion of DSV patients (85 %) than among patients with classical PTC (30 %). Clinically, DSV is sometimes misdiagnosed as Hashimoto’s thyroiditis due to the diffuse enlargement of the thyroid lobes. Early correct diagnosis and surgical treatment of DSV is required.
In our institution, the standard operation for patients with PTC was hemi-thyroidectomy or total thyroidectomy and CND, even for N0 patients. The indications for total thyroidectomy are intra-thyroidal metastasis, extra-thyroidal invasion, maximum tumor diameter more than 4 cm, presence of distant metastasis, and presence of lymph node metastasis on preoperative US-guided FNA. Patients diagnosed with DSV preoperatively were recommended total thyroidectomy. Patients diagnosed preoperatively with lymph node metastasis on US-guided FNA underwent additional uni- or bi-lateral MND. In this series, total thyroidectomy was performed in 85 % of DSV patients. Lobectomy was performed in a few DSV patients when the primary tumor was located in only one lobe. All DSV patients underwent CND, and 86 % also underwent MND. During follow-up (median, 9 years), lymph node recurrence was diagnosed in 40 % of young DSV patients, and re-operation was performed in all such cases.
The lung has been the most common site of distant metastasis in reports of DSV in the literature, and the mean incidence of distant metastasis reported in DSV is 14.9 % [19]. In our own study, 2 DSV patients (10 %) showed lung metastasis at presentation. Both M1 patients were treated by total thyroidectomy as the initial surgical procedure, followed by RAT. Both patients showed positive 131I uptake into lung metastases and one achieved complete remission with 5 administrations of RAT. We recommend initial surgery and subsequent RAT.
Previous studies comparing DSV with classical PTC have reported a similar predilection of DSV for females, younger age at tumor presentation, and higher incidences of cervical lymph node involvement and lung metastasis [3, 4, 6, 19, 20, 22, 23]. Regalbuto et al. reported finding that DSV patients have a poorer prognosis than low-risk PTC patients and can be assimilated into patients with high-risk PTC [26]. The present study found that DSV has a higher incidence of lymph node metastasis and extra-thyroidal extension, but the frequency of distant metastasis did not differ from that in other PTCs. We found that the DFS rate of DSV patients was significantly lower than that of classical PTC patients. We also performed multivariate analysis to identify prognostic factors for recurrence, showing the histological type of DSV as an independent prognostic factor. Patients with DSV are classified as a high-risk group in PTC. Initial surgical treatment with total thyroidectomy and subsequent RAT are recommended and long-term follow-up is required for this group of patients.
Various limitations must be considered in interpreting the results of this retrospective study. First, surgical strategies for the extent of thyroidectomy in PTC patients remain controversial in Japan. In 2010, a guideline for thyroid tumor was created by the Japan Association of Endocrine Surgeons and the Japanese Society of Thyroid Surgery [29]. As we have mentioned before, the indications for total thyroidectomy in our institution are a little different from them. In this study, all DSV patients were diagnosed with PTC by fine-needle aspiration and 17 DSV patients (85 %) underwent total thyroidectomy because DSV was suspected preoperatively. Only 3 patients underwent lobectomy, because the primary tumors were located in only one lobe. The present study showed no remnant thyroidal recurrence in DSV patients.
This was attributed to strong selection bias in the selection of total thyroidectomy or lobectomy. Further investigation is needed to appropriately determine indications for the extent of thyroidectomy.
Second, the validity of the radioiodine ablation therapy after initial surgery cannot be compared between classical PTC patients and DSV patients, because the indications for RAT in Japan are limited to patients with the presence of extra-thyroidal invasion, massive lymph node metastasis, or distant metastasis. Follow-up of patients with DSV for a long time is necessary to investigate the biological characteristics of this pathology.
Conclusions
DSV was more common in younger PTC patients. A high proportion of DSV patients tested positive for TgAb and showed a background of chronic lymphocytic thyroiditis. Outcomes for DSV patients were very good, similar to those for classical PTC, but lymph node recurrence was more common than with classical PTC.
References
Vickery AL Jr, Carcangiu ML, Johannessen JV et al (1985) Papillary carcinoma. Sem Diagn Pathol 2:90–100
Hedinger C, Williams ED, Sobin LH (1989) The WHO histological classification of thyroid tumors: a commentary on the second edition. Cancer 63:908–911
Carcangiu ML, Bianchi S (1989) Diffuse sclerosing variant of papillary thyroid carcinoma. Clinicopathologic study of 15 cases. Am J Surg Pathol 13:1041–1049
Soares J, Limbert E, Sobrinho-Simoes M (1989) Diffuse sclerosing variant of papillary thyroid carcinoma. A clinicopathologic study of 10 cases. Pathol Res Pract 185:200–206
Wu PS, Leslie PJ, McLaren KM et al (1989) Diffuse sclerosing papillary carcinoma of thyroid: a wolf in sheep’s clothing. Clin Endocrinol (Oxf) 31:535–544
Fujimoto Y, Obara T, Ito Y et al (1990) Diffuse sclerosing variant of papillary carcinoma of the thyroid. Clinical importance, surgical treatment, and follow-up study. Cancer 66:2306–2312
Hayashi Y, Sasao T, Takeichi N et al (1990) Diffuse sclerosing variant of papillary carcinoma of the thyroid. A histopathological study of four cases. Acta Pathol Jpn 40:193–198
Mizukami Y, Nonomura A, Michigishi T et al (1990) Diffuse sclerosing variant of papillary carcinoma of the thyroid. Report of three cases. Acta Pathol Jpn 40:676–682
Schroder S, Bay V, Dumke K et al (1990) Diffuse sclerosing variant of papillary thyroid carcinoma. S-100 protein immunocytochemistry and prognosis. Virchows Arch A Pathol Anat Histopathol 416:367–371
Gomez-Morales M, Alvaro T, Munoz M et al (1991) Diffuse sclerosing papillary carcinoma of the thyroid gland: immunohistochemical analysis of the local host immune response. Histopathology 18:427–433
Gomez-Morales M, Munoz M, Bustos M et al (1991) Diffuse sclerosing papillary carcinoma of the thyroid. Clin Endocrinol (Oxf) 34:432
Macak J, Michal M (1993) Diffuse sclerosing variant of papillary thyroid carcinoma. Ceskoslov Patol 29:6–8
Moreno EA, Rodriguez GJM, Sola PJ et al (1994) Clinicopathological study of the diffuse sclerosing variety of papillary cancer of the thyroid. Presentation of 4 new cases and review of the literature. Eur J Surg Oncol 20:7–11
Caplan RH, Wester S (1997) Kisken AW Diffuse sclerosing variant of papillary thyroid carcinoma: case report and review of the literature. Endocr Pract 3:287–292
Albareda M, Puig-Domingo M, Wengrowicz S et al (1998) Clinical forms of presentation and evolution of diffuse sclerosing variant of papillary carcinoma and insular variant of follicular carcinoma of the thyroid. Thyroid 8:385–391
Khan AR, Abu-Eshy SA (1998) Variants of papillary carcinoma of the thyroid: experience at Asir Central Hospital. J R Coll Surg Edinb 43:20–25
Imamura Y, Kasahara Y, Fukuda M (2000) Multiple brain metastases from a diffuse sclerosing variant of papillary carcinoma of the thyroid. Endocr Pathol 11:97–108
Kebapci N, Efe B, Kabukcuoglu S et al (2002) Diffuse sclerosing variant of papillary thyroid carcinoma with primary squamous cell carcinoma. J Endocrinol Invest 25:730–734
Chow SM, Chan JK, Law SC et al (2003) Diffuse sclerosing variant of papillary thyroid carcinoma–clinical features and outcome. Eur J Surg Oncol 29:446–449
Thompson LD, Wieneke JA (2005) Heffess CS Diffuse sclerosing variant of papillary thyroid carcinoma: a clinicopathologic and immunophenotypic analysis of 22 cases. Endocr Pathol 16:331–348
Falvo L, Giacomelli L, D’Andrea V et al (2006) Prognostic importance of sclerosing variant in papillary thyroid carcinoma. Am Surg 72:438–444
Lam AK, Lo CY (2006) Diffuse sclerosing variant of papillary carcinoma of the thyroid: a 35-year comparative study at a single institution. Ann Surg Oncol 13:176–181
Fukushima M, Ito Y, Hirokawa M et al (2009) Clinicopathologic characteristics and prognosis of diffuse sclerosing variant of papillary thyroid carcinoma in Japan: an 18-year experience at a single institution. World J Surg 33:958–962. doi:10.1007/s00268-009-9940-6
Koo JS, Hong S (2009) Park CS Diffuse sclerosing variant is a major subtype of papillary thyroid carcinoma in the young. Thyroid 19:1225–1231
Koo JS, Shin E, Hong SW (2010) Immunohistochemical characteristics of diffuse sclerosing variant of papillary carcinoma: comparison with conventional papillary carcinoma. APMIS 118:744–752
Regalbuto C, Malandrino P, Tumminia A et al (2011) A diffuse sclerosing variant of papillary thyroid carcinoma: clinical and pathologic features and outcomes of 34 consecutive cases. Thyroid 21:383–389
Zimmerman D, Hay ID, Gough IR et al (1988) Papillary thyroid carcinoma in children and adults: long-term follow-up of 1039 patients conservatively treated at one institution during three decades. Surgery 104:1157–1166
Lee JY, Shin JH, Han BK et al (2007) Diffuse sclerosing variant of papillary carcinoma of the thyroid: imaging and cytologic findings. Thyroid 17:567–573
Takami H, Ito Y, Okamoto T et al (2014) Revisiting the guidelines issued by the Japanese Society of thyroid surgeons and Japan Association of Endocrine Surgeons: a gradual move towards consensus between japanese and western practice in the management of thyroid carcinoma. World J Surg 38:2002–2010. doi:10.1007/s00268-014-2498-y
Conflict of interest
The authors have no conflict of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Akaishi, J., Sugino, K., Kameyama, K. et al. Clinicopathologic Features and Outcomes in Patients with Diffuse Sclerosing Variant of Papillary Thyroid Carcinoma. World J Surg 39, 1728–1735 (2015). https://doi.org/10.1007/s00268-015-3021-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-015-3021-9