Abstract
Objectives
To evaluate the HER2 status in patients with Siewert type II esophagogastric junction carcinoma.
Background
Trastuzumab is now approved for use in the treatment of human epidermal growth factor receptor 2 (HER2)-positive unresectable metastatic gastric or esophagogastric junction (EGJ) carcinoma. Several studies have evaluated HER2 status in EGJ carcinoma, but none has addressed the implication of HER2 positivity in patients with Siewert type II EGJ carcinoma.
Methods
We retrospectively evaluated the frequency of HER2 positivity in a large single-center cohort of 208 patients with Siewert type II tumors. The relations between HER2 expression and the outcomes and other clinicopathologic features were examined.
Results
Overall, 18.2 % (38/208) of patients in our cohort had HER2-positive tumors. HER2 positivity was associated only with differentiated carcinomas. The 5-year overall survival (OS) rate was 58.7 %. The 5-year OS rates in the patient groups with HER2-negative and HER2-positive tumors were 61.2 and 48.5 %, respectively. There was no significant difference between the groups. Recurrence in the liver was observed in 23.7 % patients of the HER2-positive group and 7.6 % patients of the HER2-negative group. Multivariate analysis to identify the risk factors for liver recurrence revealed only HER2 positivity (p = 0.0155) as an independent predictive factor.
Conclusions
HER2 positivity is a powerful predictor of liver recurrence in patients with Siewert type II EGJ carcinoma. Use of trastuzumab in combination with chemotherapy in an adjuvant setting can be a potentially useful therapeutic strategy to prevent hepatic recurrence in patients with resectable EGJ adenocarcinoma showing HER2 overexpression.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Based on recent evidence provided by the Trastuzumab for Gastric Cancer (ToGA) study, trastuzumab is now approved for use in treating human epidermal growth factor receptor 2 (HER2)-positive unresectable metastatic gastric or esophagogastric junction (EGJ) carcinoma [1]. EGJ carcinoma—which involves the anatomic junction between the esophagus and the stomach—has attracted considerable attention recently because of a marked increase in its incidence [2, 3]. EGJ tumors have been classified based on their epicenters according to the Siewert system [4]. Siewert type II cancer is a true carcinoma of the cardia.
Several studies have evaluated the HER2 status in EGJ carcinomas. They reported that the HER2 positivity rates were higher in the EGJ carcinomas than in gastric carcinomas [5–8]. However, the role of HER2 as a prognostic factor in patients with this cancer remains controversial [5, 7, 9, 10] as none of the reports specifically addressed the implication of HER2 positivity in Siewert type II EGJ carcinoma.
The purpose of this study was to evaluate the frequency of HER2 positivity in Siewert type II cancers. The relations between HER2 expression and the prognosis and other clinicopathologic features were examined.
Patients and methods
Patient population and staging
Between January 1980 and January 2010, a total of 11,647 patients with gastric carcinoma underwent surgery at the Gastric Surgery Division, National Cancer Center Hospital, Tokyo. Trastuzumab was not administered to patients during this period. Of these patients, 299 were diagnosed as having Siewert type II EGJ carcinoma. We defined Siewert type II EGJ carcinoma based on a definition of this type of carcinoma described previously [3]. In brief, the epicenter of the tumor was taken as the point midway between the proximal and distal extent of the gross tumor mapped on a photograph of the resected specimen. A lesion was considered a Siewert type II EGJ carcinoma if its epicenter was within 1 cm proximal and 2 cm distal to the anatomic EGJ. It has been routine clinical practice in this hospital since 1962 to obtain detailed clinical photographs of all postoperative specimens. Each specimen is sectioned at 5-mm intervals, and the location of each cut and the tumor are mapped onto these photographs by the pathologist. Because these pathology reports with photographs were made precisely, the tumors are classified precisely.
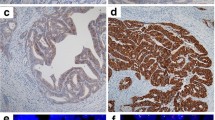
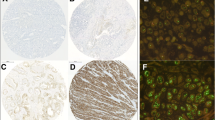
Of the 229 patients, 225 underwent an R0/R1 operation. Of these 225 patients, 208 in whom testing for HER2 was successfully conducted by immunohistochemistry (IHC) and fluorescence in situ hybridization (FISH) were enrolled in this study. All of the specimens were routinely fixed in 10 % formalin and were embedded in paraffin. Using a representative whole section, tumors were tested for the HER2 status with IHC (HercepTest; Dako, Copenhagen, Denmark) and FISH (PathVysion; Abbott, Abbott Park IL, USA) (HER2/CEP17 ratio was ≥2). Tumor samples were scored as IHC 3+ or 2+ if ≥10 % cells anywhere in the tumor tissue showed strong or equivocal membrane staining, respectively. Samples with <10 % of cells showing strong membrane staining were scored as IHC 2+. Cases with IHC 3+ and IHC2+/FISH-positive were judged as being HER2-positve [11].
We conducted the tumor staging according to the International Union Against Cancer (UICC) TNM staging system for EGJ cancer [12]. Well-differentiated and moderately differentiated tubular adenocarcinoma and papillary adenocarcinoma were classified as differentiated-type carcinomas. Poorly differentiated adenocarcinoma, signet-ring cell carcinoma, and mucinous carcinoma were classified as undifferentiated-type carcinomas. Postoperative information was obtained from the follow-up records of the patients and the city registry office.
Statistical analysis
This retrospective study was designed to evaluate the frequency of HER2 positivity in patients with Siewert type II cancer. The relations between HER2 expression and the outcomes and other clinicopathologic features were examined.
Statistical analysis was carried out using JMP 8.0.1 (SAS Institute, Cary, NC, USA). The overall survival (OS) rate was calculated from the date of the operation to the date of death from any cause. Patient survival data were obtained from the patient medical records and census registries. We constructed Kaplan–Meier survival curves to estimate the impact of HER2 positivity on survival. We used the log-rank test for comparisons.
Fisher’s exact test was applied to assess the univariate relations between clinicopathologic variables and HER2 positivity and to assess the correlation of the HER2 status with the initial site of recurrence. Values of p < 0.05 were considered to indicate statistical significance.
A multivariate logistic regression analysis was performed to identify the independent risk factors associated with liver recurrence among the variables: HER2 status, sex, age, presence/absence of lymphatic invasion and venous invasion, tumor size, tumor histology, pT, and pN.
Results
Patient demographics and baseline characteristics
Overall, 18.2 % (38/208) of patients had HER2-positive tumors in our study cohort. The relations between HER2 positivity and various clinicopathologic variables were investigated (Table 1). The results revealed that HER2 positivity was associated only with a differentiated type of tumor histology.
Survival
The median OS was 9.5 years for the entire cohort (208 patients). The median duration of follow-up of the survivors was 6.4 years (range 0.2–20.7 years). In all, 101 patients died during follow-up: 65 deaths were due to recurrence and the remaining 36 to other causes. The 5-year OS rate of the entire cohort with Siewert type II EGJ carcinoma was 58.7 %. The 5-year OS rates in the patient groups with HER2-negative and HER2-positive tumors were 61.2 and 48.5 %, respectively. There is no significant difference between the groups (Fig. 1).
Mode of recurrence
The initial sites of recurrence are shown in Table 2. The most frequent site of recurrence was the liver in the HER2-positive group (23.7 %), followed by the lymph nodes (13.2 %). In the HER2-negative group, the most frequent initial site of recurrence was the lymph nodes (11.8 %), followed by the liver (7.6 %) and peritoneal cavity (6.5 %). There was a statistically significant difference between the two groups regarding the incidence of the initial recurrence in the liver.
The relevance of HER2 for liver recurrence was assessed by multivariate logistic regression analysis after adjustments for the following factors: HER2 status, sex, age, presence/absence of lymphatic invasion and venous invasion, tumor size, tumor histology, pT, and pN (Table 3). Multivariate analysis to identify the independent risk factors for liver recurrence revealed that only HER2 positivity (p = 0.0155) was an independent predictive factor for liver recurrence.
Discussion
There was an 18.2 % incidence of HER2-positive tumors in our study cohort. Reported rates of HER2 positivity in gastric or EGJ carcinomas from previous series using different HER2 scoring systems varied from 4 to 53 % [13]. The series from Japan and Korea using the same scoring system as in the present study reported an HER2 positivity rate of 8–13 % [6, 14, 15]. The prevalence of HER2 positivity in the present study fell beyond the upper limit of this range.
Several authors have reported a strong correlation between HER2 expression and differentiated tumor histology [1, 5, 7, 9, 14, 16, 17]. This correlation was confirmed for Siewert type II tumors in the current study.
In our previous article [3], a multivariate Cox regression analysis revealed that the patient’s age, pN category (pN0/1/2/3), and residual tumor classification (R0/1) were independently associated with outcome. This study showed that HER2 status was not associated with the OS [14, 17, 18]. The role of HER2 as a prognostic factor in gastric cancer remains controversial [5, 7, 9, 10]. HER2-positive tumors are expected to have more aggressive characteristics than HER2-negative tumors because one of the main functions of HER2 is to promote cell growth, differentiation, and survival [19, 20]. In several series of patients with resected disease [14, 17, 18], however, the HER2 status did not influence the outcomes after operation. Although our patient cohort was small, the present results suggest that HER2 status might not influence the outcomes after gastrectomy.
The analysis using the logistic regression model identified only HER2 positivity as a strong predictor of recurrence in the liver as the initial site of recurrence. Janjigian et al. [5] also found that HER2 positivity was associated with a high risk of liver metastasis in gastric cancer patients. The predominant site of recurrence after gastrectomy for differentiated advanced adenocarcinoma is reported to be the liver [21]. We previously also reported vascular invasion as a risk factor for liver recurrence [22].
It is noteworthy that HER2 positivity was identified as the only predictor of liver recurrence in patients with Siewert type II cancer, although we included both vascular invasion and histology as candidate risk factors for liver recurrence in our model for the multivariate analysis. Therefore, we need to bear in mind that HER2 positivity is a powerful predictor of liver recurrence in patients with Siewert type II esophagogastric carcinoma.
From this point of view, although HER2 may not be a negative prognostic factor for survival in Siewert type II EGJ carcinoma after gastrectomy with curative intent, it does not negate its value as a predictive marker. Although control of liver recurrence is beyond the hands of the surgeon, the ToGA study demonstrated the usefulness of trastuzumab administered in combination with chemotherapy for HER2-positive advanced gastric or EGJ cancer. Use of trastuzumab in combination with chemotherapy in an adjuvant setting can be a potentially useful therapeutic strategy to prevent hepatic recurrence in patients with resectable EGJ adenocarcinoma displaying HER2 overexpression.
References
Bang YJ, Van Cutsem E, Feyereislova A et al (2010) Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet 376:687–697
Kusano C, Gotoda T, Khor CJ et al (2008) Changing trends in the proportion of adenocarcinoma of the esophagogastric junction in a large tertiary referral center in Japan. J Gastroenterol Hepatol 23:1662–1665
Yamashita H, Katai H, Morita S et al (2011) Optimal extent of lymph node dissection for Siewert type II esophagogastric junction carcinoma. Ann Surg 254:274–280
Siewert JR, Stein HJ (1996) Carcinoma of the gastroesophageal junction: classification, pathology and extent of resection. Dis Esophagus 9:173–182
Janjigian YY, Werner D, Pauligk C et al (2012) Prognosis of metastatic gastric and gastroesophageal junction cancer by HER2 status: a European and USA international collaborative analysis. Ann Oncol 23:2656–2662
Kataoka Y, Okabe H, Yoshizawa A et al (2013) HER2 expression and its clinicopathological features in resectable gastric cancer. Gastric Cancer 16:84–93
Gravalos C, Jimeno A (2008) HER2 in gastric cancer: a new prognostic factor and a novel therapeutic target. Ann Oncol 19:1523–1529
Bang Y, Chung J, Xu J, et al (2009) Pathological features of advanced gastric cancer (GC): relationship to human epidermal growth factor receptor 2 (HER2) positivity in the global screening programme of the ToGA trial. J Clin Oncol (Meeting Abstracts) 27(Suppl 15):abstract 4556
Begnami MD, Fukuda E, Fregnani JH et al (2011) Prognostic implications of altered human epidermal growth factor receptors (HERs) in gastric carcinomas: HER2 and HER3 are predictors of poor outcome. J Clin Oncol 29:3030–3036
Kim KC, Koh YW, Chang HM et al (2011) Evaluation of HER2 protein expression in gastric carcinomas: comparative analysis of 1,414 cases of whole-tissue sections and 595 cases of tissue microarrays. Ann Surg Oncol 18:2833–2840
Ruschoff J, Hanna W, Bilous M et al (2012) HER2 testing in gastric cancer: a practical approach. Mod Pathol 25:637–650
Sobin LH, Gospodarowicz MK, Wittekind C (eds) (2009) TNM classification of malignant tumours, 7th edn. Wiley-Blackwell, Hoboken
Chua TC, Merrett ND (2012) Clinicopathologic factors associated with HER2-positive gastric cancer and its impact on survival outcomes: a systematic review. Int J Cancer 130:2845–2856
Terashima M, Kitada K, Ochiai A et al (2012) Impact of expression of human epidermal growth factor receptors EGFR and ERBB2 on survival in stage II/III gastric cancer. Clin Cancer Res 18:5992–6000
Park YS, Ryu M, Park HJ, et al (2011) HER2 status as an independent prognostic marker in patients with advanced gastric cancer receiving adjuvant chemotherapy after curative gastrectomy. J Clin Oncol (Meeting Abstracts) 29(Suppl 15):abstract 4084
Park DI, Yun JW, Park JH et al (2006) HER-2/neu amplification is an independent prognostic factor in gastric cancer. Dig Dis Sci 51:1371–1379
Grabsch H, Sivakumar S, Gray S et al (2010) HER2 expression in gastric cancer: rare, heterogeneous and of no prognostic value—conclusions from 924 cases of two independent series. Cell Oncol 32:57–65
Sasano H, Date F, Imatani A et al (1993) Double immunostaining for c-erbB-2 and p53 in human stomach cancer cells. Hum Pathol 24:584–589
Slamon DJ, Clark GM, Wong SG et al (1987) Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science 235:177–182
Slamon DJ, Godolphin W, Jones LA et al (1989) Studies of the HER-2/neu proto-oncogene in human breast and ovarian cancer. Science 244:707–712
Katai H, Maruyama K, Sasako M et al (1994) Mode of recurrence after gastric cancer surgery. Dig Surg 11:99–103
Ishida M, Morita S, Saka M et al (2012) Metachronous liver metastasis from early gastric cancer. J Gastrointest Surg 16:837–841
Conflict of interest
The authors have no conflicts of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Katai, H., Ishida, M., Yamashita, H. et al. HER2 Expression in Carcinomas of the True Cardia (Siewert Type II Esophagogastric Junction Carcinoma). World J Surg 38, 426–430 (2014). https://doi.org/10.1007/s00268-013-2256-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-013-2256-6