Abstract
Background
Obesity is associated with high morbidity and represents an increasing health care problem worldwide. Laparoscopic sleeve gastrectomy (LSG) has been used effectively for weight loss and co-morbidity remission. In this retrospective study, we evaluated cardiac reverse remodeling at medium-term follow-up by echocardiography, the amount of cardiovascular medications, and the impact of co-morbidities after sleeve gastrectomy.
Methods
Altogether, 16 obese patients (4 men, 12 women; 46.4 ± 10.3 years) underwent complete clinical evaluation, laboratory tests, and color Doppler/tissue Doppler imaging echocardiography preoperatively and 12–20 months after bariatric surgery.
Results
Body weight (mean body mass index) was significantly reduced (from 44.8 ± 8.0 to 31.2 ± 7.8 kg/m2; p = 0.001). Lipid profile significantly improved: total cholesterol and triglycerides decreased (respectively: 215.5 ± 53.8 vs. 205.3 ± 46.6 mg/dl and 184.9 ± 109.3 vs. 116.1 ± 49.9 mg/dl, both p ≤ 0.05), and high-density lipoprotein increased (43.1 ± 10.9 vs. 51.4 ± 12.8 mg/dl, p = 0.005). Systolic blood pressure significantly decreased (from 133.0 ± 17.1 to 120.6 ± 13.7 mmHg; p = 0.04). Diabetes remission was complete in five of six patients (83 %) and sleep apnea in four of five (80 %). Echocardiography showed significantly reduced interventricular septum and posterior wall thickness (11.3 ± 1.8 to 9.4 ± 2.1 mm and 10.4 ± 1.7 to 8.6 ± 1.9 mm, respectively; both p < 0.007) and reduced left ventricular mass (absolute value and indexed by height, respectively: 222.41 ± 78.2 to 172.75 ± 66.3 g (p = 0.003) and 55.9 ± 14.3 to 43.8 ± 17.2 g/m2.7 (p = 0.0004). Antihypertensive drug intake was significantly reduced (p = 0.03), as shown by the 10-year Framingham Risk Score (from 14.2 ± 9.3 to 8.3 ± 9.5 %, p = 0.003).
Conclusions
Sleeve gastrectomy is associated with marked improvement in terms of weight loss, lipid profile, type 2 diabetes, sleep apnea, hypertension, and left ventricular hypertrophy, with a significantly reduced Framingham Risk Score.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Obesity is a systemic disorder associated with multiple co-morbidities, including type 2 diabetes mellitus (T2DM), hypertension, dyslipidemia, metabolic syndrome, nonalcoholic fatty liver disease, nephropathy, left ventricular (LV) hypertrophy, and obstructive sleep apnea syndrome (OSAS). Obesity increases morbidity and mortality risks and represents a major health care problem with epidemic distribution worldwide. Obesity is considered a significant cardiovascular risk factor for premature death and disability from cardiovascular diseases [1, 2]. The secondary cardiac structural and hemodynamic changes that occur when these alterations result in congestive heart failure are overall referred to as “obesity cardiomyopathy” and have been extensively investigated. They include an increase in LV wall thickness, mass, and diameters, with systolic and diastolic dysfunction [3, 4]. Right ventricular dimensions and function may be similarly affected, in relation to pulmonary hypertension, OSAS, and obesity hypoventilation syndrome [5].
Weight loss surgery has been shown to reduce mortality and the incidence of co-morbidities and to ameliorate or remit T2DM [6, 7]. Bariatric surgery seems also able to reverse many of the associated hormonal and hemodynamic changes, reducing the incidence of cardiovascular events [2, 6]. The decrease in LV mass after bariatric surgery, irrespective of the surgical technique, is well documented and may provide beneficial effects on long-term survival in obese patients [8]. Cardiac changes are not pronounced during the first 3 months, when the decrease in glucose, insulin, and cholesterol levels is more significant. LV mass regression continues in a linear fashion and peaks by the ninth postoperative month [8]. In a recent systematic review involving 52 studies and 16,867 patients, a 40 % relative risk reduction for 10-year coronary heart disease, as determined by the Framingham Risk Score, was observed after bariatric surgery [9].
Sleeve gastrectomy (SG), originally conceived as the first stage of a biliopancreatic diversion with duodenal switch (BPD-DS), has rapidly gained popularity as a definitive bariatric procedure. SG is associated with a high rate of remission of T2DM and other obesity-associated co-morbidities, such as hypertension, hyperlipidemia, and sleep apnea [10–12]. To our knowledge, no data are available in the literature about specific effects of SG on reverse remodeling of the heart. The aim of the present study was to analyze retrospectively a selected population at mid-term follow-up after SG for: (1) reverse cardiac remodeling, evaluated by transthoracic two-dimensional (2D) Doppler and tissue Doppler imaging (TDI) echocardiography; (2) all co-morbidities that occurred, such as a toxic lipid profile or T2DM; and (3) the amount of cardiovascular medications required.
Materials and methods
Study population
From March 2009 to December 2010, a total of 248 obese patients (mean age 41 ± 10.46 years, range 16–67 years) underwent SG at a single academic medical center. A complete preoperative evaluation by a multidisciplinary team included demographic data, blood tests, and 12-lead electrocardiography. The preoperative workup included anesthesiology and cardiac evaluations. In 17 (6.8 %) of these patients (including only 4 men), whose mean age was 46.4 ± 10.3 years (range 16–58 years), preoperative 2D and Doppler echocardiography was indicated and performed. The indications were LV pressure overload and left atrial (LA) enlargement, as shown by 12-lead electrocardiography (ECG) (11 patients, 69 %), or because of the presence of three or more cardiac risk factors (in the remaining six patients), according to ACC/AHA guidelines [13]. The obese patients in our cohort had multiple cardiac risk factors, but none had a history of ischemic myocardial disease, current instability, or a history of heart failure or cerebrovascular disease. One patient with significant aortic valve stenosis was excluded from the study.
Based on these considerations, routinely after the ECG/cardiac evaluation and the anesthesiologist’s assessment in our center the patients usually were classified as being at low perioperative cardiac risk patients. Only in a very low percentage of these patients is preoperative echocardiography required according to current guidelines, which exclude patients with an absence of an active or highly suspected active cardiac disease (unstable coronary syndromes, decompensated heart failure, significant arrhythmias, and/or severe valvular disease).
Congenital heart diseases, atrial fibrillation, and known coronary artery disease were considered additional exclusion criteria. None was seen in this patient cohort. Preoperative data were retrospectively analyzed.
Sleeve gastrectomy was performed with a technique previously described [11]. The vertical gastric resection started 6 cm from the pylorus up to the angle of His, and it was calibrated along a 48F transoral bougie to obtain a 60–70 ml capacity gastric pouch.
All patients gave written informed consent to follow-up evaluation. The Ethics Committee on Research at our institution approved the study.
Follow-up
Patients were reevaluated postoperatively at a median follow up of 16 months (range 12–20 months), and such follow-up was 100 % complete. Clinical evaluation, changes on lipid profile, systolic and diastolic blood pressures, 12-lead electrocardiography, 2D Doppler/TDI echocardiography, co-morbidities progression, and the use of antihypertensive and antidiabetic medications were recorded. Remission of T2DM was defined as fasting glucose levels <100 mg/dl and hemoglobin A1c (HbA1c) <6.0 % without the use of oral hypoglycemic drugs or insulin. To estimate the 10-year cardiovascular risk, the Framingham Risk Score was calculated, as previously described [14, 15].
Echocardiography
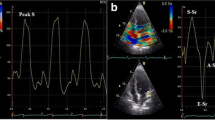
Echocardiographic follow-up studies were performed in left lateral decubitus by an experienced echocardiologist (EC) using commercially available ultrasonography equipment (CX-50; Philips, Andover, MA, USA) with a 1–5 MHz phased array transducer (S5-1; Philips). The images were acquired from parasternal and apical windows and were digitally recorded according to the American Society of Echocardiography (ASE) standards for offline analysis. M-mode, bidimensional echocardiographic data, and pulsed, continuous, color Doppler, and TDI flow mapping were recorded. Two expert ECs independently and blinded from the patients’ clinical records, reviewed each study and measured each referred quantitative parameter in three consecutive cycles. The parameters of LV geometry were measured offline in M-mode, including the interventricular septum (IVS), posterior wall thickness, and LV internal diameters, measured at the end of diastole (end-diastolic diameter) and at the end of systole (end-systolic diameter). When M-mode measurements could not be obtained optimally in three patients (19 %), LV internal dimensions and wall thickness measurements were made using the leading edge convention, as described by the ASE [16]. LV end-diastolic and end-systolic volumes were calculated using the Teichholz method [16]. Relative wall thickness was calculated as follows:
where RWT is the relative wall thickness, LVPW is the left ventricular posterior wall thickness at end-diastole, and EDD is the left ventricular mid-cavity dimension at end-diastole. LV mass was calculated using the corrected AES convention [16] proposed by Devereux:
For more appropriate normalization of LV mass in obese patients, the LV mass indexed by height was calculated by dividing the LV mass by height in meters raised to the 2.7th power [16]. Indexation of LV mass for the allometric power of its relation to body height (height2.7) facilitates identification of LV hypertrophy among obese subjects because height2.7 parallels ideal body size for body height, in contrast to actual body weight [17]. Different methods of indexation of LV mass, as for body surface area (BSA), resulted in considerably different prevalence of LV hypertrophy, which is why indexation for body mass index (BMI) or BSA has been avoided. LV geometry was considered normal if the relative wall thickness (RWT) was <0.45 and the LV mass index (LVMI) was <51 g/m2.7. A normal LVMI with increased RWT (≥0.45) was considered concentric remodeling. A hypertrophic LV (LVMI > 51 g/m2.7) was considered eccentric when the RWT was <0.45 and concentric when the RWT was ≥0.45. Using pulsed-wave Doppler and TDI, early and late diastolic mitral inflow and early diastolic mitral annular velocity, obtained from the average of measurements of the septal and lateral annulus, were recorded to assess diastolic function. The patients were accordingly classified into three groups: normal filling, altered relaxation and pseudonormal filling dynamics. Interobserver agreement was 0.9.
Statistical analysis
Continuous variables are reported as the mean ± SD and categorical variables as the number and percent. Continuous variables were analyzed with Student’s t test for paired data for bivariate analyses and with a generalized linear model for multivariate analyses. Categoric variables were analyzed with χ2 or McNemar tests, when appropriate. Statistical significance was considered for values of p ≤ 0.05. Computations were performed with SPSS 19 (IBM, Armonk, NY, USA).
Results
Sleeve gastrectomy was carried out in all patients without intraoperative complications. Significant postoperative bleeding was registered in one patient (6 %) and required nonoperative management (2-unit blood transfusion). The mean postoperative hospital stay was 4.3 days (range 3–7 days). There were no operative deaths. At the time of surgery, the mean BMI was 44.8 ± 8 kg/m2. Co-morbidities were present as follows: hypertension 10 patients (62.5 %), DM 6 patients (37 %), OSAS 5 patients (31 %) (Table 1). Before the operation, patients with hypertension took 2.7 ± 1.2 different antihypertensive pharmacological class medications daily. At follow-up, the number of medications had decreased to 1.4 ± 1.4 agents (p = 0.03). In four patients, the therapy was completely stopped (Fig. 1). The values of preoperative and postoperative blood tests are reported in Table 2.
Body weight was significantly reduced, with a mean BMI decrease from 44.8 ± 8.0 to 31.2 ± 7.8 kg/m2 (p = 0.001). The lipid profile was significantly improved in terms of lowering of total cholesterol and triglycerides (215.5 ± 53.8 to 205.3 ± 46.6 mg/dl and 184.9 ± 109.3 to 116.1 ± 49.9 mg/dl, p = 0.05 and 0.02, respectively), with an increase in high density lipoprotein (HDL) cholesterol (43.1 ± 10.9 to 51.4 ± 12.8 mg/dl, p = 0.005). A significant reduction in systolic blood pressure was recorded (from 133 ± 17.1 to 120.6 ± 13.7 mmHg, p = 0.04), without a significant variation in diastolic blood pressure. Diabetes remission was complete in five of six patients (83 %). In the patient without remission, the diabetes had been present for more than 10 years. However, glycemic control improved with significant reduction of oral antidiabetic drug use. Sleep apnea was cured in four of five patients (80 %) and was ameliorated in one patient. The mean Framingham Risk Score decreased from 14.2 ± 0.3 % preoperatively to 8.3 ± 0.5 % at follow-up (p = 0.0026) owing to an improved lipid profile, diabetes remission, and less antihypertensive drug need (Table 2).
Echocardiographic data showed a significant reduction in IVS and posterior wall thickness (PWT) [11.3 ± 1.8 to 9.4 ± 2.1 mm (IVS) and 10.4 ± 1.7 to 8.6 ± 1.9 mm (PWT), p = 0.007 and 0.005, respectively), in LV mass (222.41 ± 78.2 to 172.75 ± 66.3 g, p = 0.003), with a high statistically significance in LV mass indexed by height (55.9 ± 14.3 to 43.8 ± 17.2 g/m2.7, p = 0.0004) (Fig. 2). LV dimensions were not altered, demonstrated by the fact that both LV diameters—end-diastolic and end-systolic—were not significantly modified. RWT was significantly reduced (0.4 ± 0.07 to 0.35 ± 0.07, p = 0.03) with a nonsignificant improvement in ejection fraction (p = 0.11) and in fractional shortening (Table 3). LV geometry tended toward normalization, although not significantly, considering the improvement in the LVM and RTW (Fig. 3). Diastolic function improvement in the degree of LV relaxation was detected in three patients (19 %) (Fig. 4). Moreover, left atrium (LA) enlargement, reflecting chronic LV filling pressures, significantly diminished. The multivariate test for repeated measures showed that the changes in LV mass and the LVMI were significantly correlated witih changes in systolic blood pressure (p < 0.001), RTW (p < 0.003), and BMI (p < 0.001). In a sensitivity analysis, the main computations were repeated using baseline medical therapy as the factor. The obtained results, though, were similar to the main results in terms of direction and magnitude of statistical significance.
Discussion
Obesity is a global public health problem, and its prevalence has dramatically increased over the last few decades [18]. Although diet and physical exercise are the most important modifiable determinants of body weight, many complex factors, including genetic, hormonal, behavioral, psychological, social, and environmental factors, interact to determine a long-term imbalance between energy intake and expenditure [19].
Sleeve gastrectomy is a safe procedure that has quickly gained popularity worldwide in obese patients. Born as a first stage of biliopancreatic diversion with duodenal switch, today SG is considered a sole bariatric operation, and it has rapidly attracted worldwide interest. The Third International Summit on the current status of SG (December 2010) reported the results of 19,605 SGs performed within 3.6 ± 1.5 years [20]. Because of the increasing interest, three consecutive position statements on the use of SG as a sole bariatric procedure were issued from 2007 to 2011 by the American Society for Metabolic and Bariatric Surgery (ASMBS) [12, 21, 22].
Several studies [3, 4] have reported the beneficial effects of bariatric surgery on cardiovascular risk and on cardiac adaptation to long-term sustained weight loss with an adjustment of LV mass to body size. In published studies, the results are from different techniques of bariatric surgical procedures (Roux-en-Y gastric bypass, SG, gastric banding [2, 8, 9]), but to our knowledge no significant data on cardiac remodeling after SG alone are currently available. Our study reports the impact of SG on co-morbidities and cardiac function, as evaluated by color Doppler and TDI echocardiography in a homogeneous group of patients. The results of the present study indicate that LV hypertrophy, a marker of increased cardiovascular risk, correlates with abnormal hemodynamic, hormonal, and metabolic factors. It improves rapidly after SG in obese adults.
Many factors account for the reverse LV remodeling. Postoperative arterial pressure reduction seems to be the main mechanism. However, the reduction in LV mass in obese subjects, regardless of whether the subjects have normal or high blood pressure, has been documented [23]. In fact, LV concentric hypertrophy is the subtype more frequently found in obese patients, and it has been shown to carry a higher than the eccentric type [24], being a compensatory process to volume overload. In the present study, concentric and eccentric hypertrophy and concentric remodeling, which is represented by an altered RWT in the presence of a normal LV mass, were recorded in a similar percentage of cases during the preoperative evaluation. After LV mass regression, LV geometry improved, with a homogeneous decrease in the two types of hypertrophy and in the case of cardiac remodeling, in favor of a normal LV structure. The latter plays an important role in lowering cardiac risk stratification.
In the clinical scenario there is a need for a reproducible and reliable index to follow changes in LV shape over time, related to pressure and/or volume overload, in the context of normal or impaired LV function [25]. In line with most other studies [8, 26, 27], in the present series LV systolic function and fractional shortening were well preserved, without significant changes at follow-up. Wong et al. [4] detected a subclinical depression of LV function, probably correlated with the duration of obesity. The impact on LV function may be subtle during the early stage of obesity, and the relatively young age of our patients may justify its absence. In the meanwhile, the global improvement in LV geometry might reverse the subtle and possibly preclinical LV dysfunction, thereby avoiding subsequent deterioration, possibly related to cardiac fibrosis. There is also an improvement in LV relaxation, with a reduction in LV filling pressures, the latter being related to volume expansion in obese patients. This is reflected in a positive correlation with reduced LA dimensions. Further studies are needed to investigate the mechanism by which diastolic function improved.
The LV mass, systolic blood pressure, BMI, and RWT were analyzed with a model for repeated measures, considering not only the difference between pre and post, as in Pearson’s correlation, but also the absolute value. In contrast to previous studies [8, 27], a significant correlation was found between LV mass regression and systolic blood pressure regression as well as BMI and RWT regression. Interestingly, hypertensive patients experienced a significant drop in systolic blood pressure compared to that of normotensive patients (mean delta 11.0 ± 0.8 vs. 8.0 ± 11.3 mmHg, p < 0.005). Based on our results, w believe that LV reverse remodeling is dependent on various parameters combined to effect global cardiac improvement. It is influenced not only by the decreased blood pressure but also by the BMI, volume overload, and T2DM remission—all factors interconnected in the metabolic syndrome.
Algahim et al. [8] hypothesized that the improvement in the sleep apnea syndrome may have neurohumoral effects, influencing a positive cardiac remodeling. Metabolic changes, such as the normalization of the lipid pattern, remission of T2DM, and euglycemia restoration, can be important promoters of cardiac improvements. In other series, T2DM remission after SG was reported with a rate similar to that obtained after gastric bypass [28, 29]. The remission occurs early in the postoperative period and independently from body weight loss: A hormonal mechanism seems to be involved in inducing changes in insulin resistance and glucose homeostasis [30, 31]. In the present study, complete remission of T2DM occurred in five of six patients (83 %) with clinical improvement in the sixth patient. A similar remission occurred for OSAS. In hypertensive patients, the remission rate was 40 %, and clinical improvement occurred in 30 %. As a consequence, pharmacologic therapy was significantly reduced. The lipid profile also ameliorated, with a significant reduction of total cholesterol and triglycerides and an increase in HDL cholesterol levels. It is an interesting finding that the drop in the cholesterol level, although significant, is not as low as one would expect. This is in line with our previous results [32] and with outcomes reported in the literature [33, 34]. In those studies, a significant improvement in HDL cholesterol and triglycerides levels after laparoscopic sleeve gastrectomy (LSG) is observed although LDL cholesterol and total cholesterol levels were unchanged. Benaiges et al. [34] hypothesized that the drop of ghrelin can play a role in this specific effect of LSG on HDL. These data, together with the global improvement of the metabolic syndrome, may be considered the basis for the observed cardiac risk reduction, as evidenced by the significant reduction of the Framingham Risk Score (p = 0.0026).
Limitations
The main limitation of this retrospective study is that the preoperative echocardiographic evaluations were not performed prior to the patients’ inclusion in the study and not in consecutive patients but in a selected population. This weakens our results. On the other hand, the differences between the preoperative and postoperative data on LV geometry and mass are highly statistically significant, suggesting that the changes in this unique population are of relevance. Prospective data are needed.
Conclusions
Sleeve gastrectomy is a safe, effective procedure that allows a significant change in LV shape in terms of mass, geometry, and diastolic function. These changes seem to be related to SG-induced weight loss and to the metabolic syndrome improvement. Particularly important, it seems, is the reduced systolic blood pressure, resulting in a marked reduction in the need for antihypertensive therapy. The changes are also related to the remission of T2DM and OSAS and to amelioration of the lipid pattern. Prospective controlled studies are needed to confirm these data and to clarify the possible molecular and/or neurohormonal mechanisms determining these effects.
References
Guh DP, Zhang W, Bansback N et al (2009) The incidence of co-morbidities related to obesity and overweight: a systematic review and meta-analysis. BMC Public Health 9:88
Sjöström L, Peltonen M, Jacobson P et al (2012) Bariatric surgery and long-term cardiovascular events. JAMA 307:56–65
Pascual M, Pascual DA, Soria F et al (2003) Effects of isolated obesity on systolic and diastolic left ventricular function. Heart 89:1152–1156
Wong CY, O’Moore-Sullivan T, Leano R et al (2004) Alterations of left ventricular myocardial characteristics associated with obesity. Circulation 110:3081–3087
Alpert MA (2001) Obesity cardiomyopathy: pathophysiology and evolution of the clinical syndrome. Am J Med Sci 321:225–236
Adams TD, Gress RE, Smith SC et al (2007) Long-term mortality after gastric bypass surgery. N Engl J Med 357:753–761
Buchwald H, Estok R, Fahrbach K et al (2009) Weight and type 2 diabetes after bariatric surgery: systematic review and meta-analysis. Am J Med 122:248–256
Algahim MF, Lux TR, Leichman JG et al (2010) Progressive regression of left ventricular hypertrophy two years after bariatric surgery. Am J Med 123:549–555
Heneghan HM, Meron-Eldar S, Brethauer SA et al (2011) Effect of bariatric surgery on cardiovascular risk profile. Am J Cardiol 108:1499–1507
Gill RS, Birch DW, Shi X et al (2010) Sleeve gastrectomy and type 2 diabetes mellitus: a systematic review. Surg Obes Relat Dis 6:707–713
Basso N, Casella G, Rizzello M et al (2011) Laparoscopic sleeve gastrectomy as first stage or definitive intent in 300 consecutive cases. Surg Endosc 25:444–449
Clinical Issues Committee of the American Society for Metabolic and Bariatric Surgery (2010) Updated position statement on sleeve gastrectomy as a bariatric procedure. Surg Obes Relat Dis 6:1–5
Fleisher LA, Beckman JA, Brown KA et al (2007) ACC/AHA 2007 guidelines on perioperative cardiovascular evaluation and care for noncardiac surgery: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 2002 Guidelines on Perioperative Cardiovascular Evaluation for Noncardiac Surgery) developed in collaboration with the American Society of Echocardiography, American Society of Nuclear Cardiology, Heart Rhythm Society, Society of Cardiovascular Anesthesiologists, Society for Cardiovascular Angiography and Interventions, Society for Vascular Medicine and Biology, and Society for Vascular Surgery. J Am Coll Cardiol 50:e159–e241
Wilson PW, D’Agostino RB, Levy D et al (1998) Prediction of coronary heart disease using risk factor categories. Circulation 97:1837–1847
D’Agostino RB Sr, Vasan RS, Pencina MJ et al (2008) General cardiovascular risk profile for use in primary care: the Framingham Heart Study. Circulation 117:743–753
Lang RM, Bierig M, Devereux RB et al (2005) Recommendations for chamber quantification: a report from the American Society of Echocardiography’s Guidelines and Standards Committee and the Chamber Quantification Writing Group, developed in conjunction with the European Association of Echocardiography, a branch of the European Society of Cardiology. J Am Soc Echocardiogr 18:1440–1463
Palmieri V, de Simone G, Arnett DK et al (2001) Relation of various degrees of body mass index in patients with systemic hypertension to left ventricular mass, cardiac output, and peripheral resistance (The Hypertension Genetic Epidemiology Network Study). Am J Cardiol 88:1163–1168
Finnucane MM, Stevens GA, Cowan MJ et al (2011) Global Burden of Metabolic Risk Factors of Chronic Disease Collaborating Group (Body Mass Index): national, regional, and global trends in body-mass index since 1980—systematic analysis of health examination surveys and epidemiological studies with 960 country-years and 9.1 million participants. Lancet 377:557–567
Clarke R (2011) Long-term weight loss and prevention of cardiovascular disease. Circulation 124:2801–2802
Deitel M, Gagner M, Erickson AL et al (2011) Third International Summit: current status of sleeve gastrectomy. Surg Obes Relat Dis 7:749–759
Clinical Issues Committee of American Society for Metabolic and Bariatric Surgery (2007) Sleeve gastrectomy as a bariatric procedure. Surg Obes Relat Dis 3:573–576
Anonymous (2011) Updated position statement on sleeve gastrectomy as a bariatric procedure. http://asmbs.org/guidelines-statements/. Accessed 28 Oct 2011
Himeno E, Nishino K, Nakashima Y et al (1996) Weight reduction regresses left ventricular mass regardless of blood pressure level in obese subjects. Am Heart J 131:313–319
Kardassis D, Bech-Hanssen O, Schönander M et al (2012) The influence of body composition, fat distribution and sustained weight loss on left ventricular mass and geometry in obesity. Obesity 20:605–611
De Castro S, Caselli S, Maron M et al (2007) Left ventricular remodelling index (LVRI) in various pathophysiological conditions: a real-time three-dimensional echocardiographic study. Heart 93:205–209
Hsuan CF, Huang CK, Lin JW et al (2010) The effect of surgical weight reduction on left ventricular structure and function in severe obesity. Obesity 18:1188–1193
Syed M, Torosoff M, Rosati C et al (2010) Effect of comorbidities and medications on left ventricular mass regression after bariatric surgery. J Clin Hypertens 12:223–227
Abbatini F, Rizzello M, Casella G et al (2010) Long-term effects of laparoscopic sleeve gastrectomy, gastric bypass, and adjustable gastric banding on type 2 diabetes. Surg Endosc 24:1005–1010
Vidal J, Ibarzabal A, Romero F et al (2008) Type 2 diabetes mellitus and the metabolic syndrome following sleeve gastrectomy in severely obese subjects. Obes Surg 18:1077–1082
Basso N, Capoccia D, Rizzello M et al (2011) First-phase insulin secretion, insulin sensitivity, ghrelin, GLP-1, and PYY changes 72 h after sleeve gastrectomy in obese diabetic patients: the gastric hypothesis. Surg Endosc 25:3540–3550
Rizzello M, Abbatini F, Casella G et al (2010) Early postoperative insulin-resistance changes after sleeve gastrectomy. Obes Surg 20:50–55
Leonetti F, Capoccia D, Coccia F et al (2012) Obesity, type 2 diabetes mellitus, and other comorbidities: a prospective cohort study of laparoscopic sleeve gastrectomy vs medical treatment. Arch Surg 147:694–700
Zhang F, Strain GW, Lei W et al (2011) Changes in lipid profiles in morbidly obese patients after laparoscopic sleeve gastrectomy (LSG). Obes Surg 21:305–309
Benaiges D, Flores-Le-Roux JA, Pedro-Botet J et al (2012) Impact of restrictive (sleeve gastrectomy) vs hybrid bariatric surgery (roux-en-Y gastric bypass) on lipid profile. Obes Surg 22:1268–1275
Acknowledgments
This study was supported by Sapienza University of Rome Research Grant and grants from Fondazione Roma. We are grateful to Dr. Isotta Chimenti for her critical reading.
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Elena Cavarretta and Giovanni Casella contributed equally to this work.
Rights and permissions
About this article
Cite this article
Cavarretta, E., Casella, G., Calì, B. et al. Cardiac Remodeling in Obese Patients After Laparoscopic Sleeve Gastrectomy. World J Surg 37, 565–572 (2013). https://doi.org/10.1007/s00268-012-1874-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-012-1874-8