Abstract
The cribriform-morular variant (CMV) of papillary thyroid carcinoma (PTC) is a rare histologic subtype of PTC that shows a combination of growth patterns including cribriform and spindle cell areas. The thyroid cancer with this unique histology was originally reported in patients with familial adenomatous polyposis (FAP), although it was later found in patients without polyposis as well. Because of its rarity, its clinical features are not clear. We reviewed seven patients with CMV-PTC who were found among 4194 patients with PTC in our pathology files between June 1991 and March 2003. The prevalence of CMV was 0.16% among all PTCs. We invited these patients to our hospital so we could obtain a detailed family history and recommend colonoscopic examination and germline APC gene analysis. Two patients without subjective symptoms had polyposis of the colon and colon cancers. Germline APC gene mutations were found in both patients. The father of a patient who refused the invitation was revealed to have undergone surgery for colon polyposis. In the remaining four patients, neither polyposis nor APC gene mutation was found. Common clinical features included a young age (mean 25 years), predominance of females, circumscribed tumors, negative node metastasis, and no recurrence of the thyroid cancer after surgery. Two of the three patients with colon polyposis had bilateral multiple thyroid tumors, whereas the remaining four (without polyposis) had a solitary tumor. The histopathology of CMV in patients with PTC should arouse a suspicion of FAP, especially if there are multiple tumors. This finding can lead to early detection of colon cancer.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The cribriform-morular variant (CMV) of papillary thyroid carcinoma (PTC) is a rare, peculiar subtype of PTC. This papillary histotype is present frequently in familial adenomatous polyposis (FAP)-associated thyroid cancers [1, 2]. In 1968, Camiel et al. was the first to suggest the relation of FAP with thyroid carcinoma [3]. In 1994, Harach et al. reported that the thyroid carcinoma of patients with FAP had a distinctive histologic feature different from those of common papillary carcinoma [4]. These atypical histologic features included an intricate blending of cribriform, trabecular, solid, follicular, and papillary patterns of growth, with scattered morular areas. They also suggested that if a physician sees such a patient with thyroid carcinoma of the CMV, the possible presence of FAP should be recognized [4]. Although this relatively new knowledge can be found in a current endocrine pathology textbook [5], not all practicing pathologists and endocrine surgeons are familiar with this potentially important PTC variant.
Familial adenomatous polyposis is a major hereditary colorectal cancer syndrome linked to mutations in the APC gene located in the 5q21 region. Individuals with an inherited APC mutation have almost 100% risk of developing colorectal cancer at a significantly young age [6]. The relative risk for thyroid carcinoma in FAP patients under 35 years of age is 160 times that of the general population. More importantly, thyroid carcinoma may be the first symptom in patients with FAP, and 30% of thyroid carcinomas are diagnosed 4 to 12 years before the development of polyposis [7]. Cameselle-Teijeiro later described sporadic CMV-PTC, which shared morphologic features of FAP-associated thyroid carcinoma in patients but with no family history or clinical manifestations of FAP [8].
The objective of the present study was to clarify the clinical features of patients with CMV-PTC with special attention to a possible association with FAP or colon cancer.
Materials and Methods
Seven patients with CMV were found among 4194 patients with PTC in the pathology files of Kuma Hospital between June 1991 and March 2003. The prevalence of CMV among all PTCs was 1.6/1000. All of the patients were females aged 16 to 32 years (mean 25 years). Clinical features and pathologic findings were reviewed. We invited these patients to our hospital and obtained detailed family histories. We also recommended that they be examined by colonoscopy and APC gene analysis. APC molecular screening was carried out using the protein truncation test followed by the direct sequencing method described elsewhere [9, 10].
Results
The clinical and pathologic findings for the seven cases of CMV are summarized in Table 1.
Clinical Features
Three patients (cases 2, 5, 6) had hard tumors by physical examination, so on first impression the physician suspected that these thyroid tumors were malignant. The tumors of the other patients did not have the characteristics of a malignant thyroid nodule on palpation. Serum levels of free thyroxine, free triiodothyronine, and thyroid-stimulating hormone were within the normal ranges in all patients. Serum thyroglobulin levels were within the normal range except in two patients who had mildly elevated levels. On ultrasonography, all tumors showed a circumscribed capsular pattern. Fine-needle aspiration biopsy showed an unusual cytologic pattern: clusters of spindle form or elongated cells with moderate atypical nuclei suggesting medullary thyroid carcinoma or poorly differentiated papillary carcinoma. Serum calcitonin levels in the six patients examined were normal. One patient was diagnosed as having CMV by FNAB because peculiar nuclear clearing, a characteristic feature of CMV, was seen.
Total thyroidectomy was performed in three patients because their carcinomas were multiple and were located in both lobes or because associated benign nodules were located in the other lobe. Unilateral lobectomy was performed in four patients. Central lymph node dissection alone was performed in three patients, and the other four patients underwent dissection of the lateral compartment as well.
Pathologic Findings
Formalin-fixed and paraffin-embedded sections were stained with hematoxylin and eosin. The maximum diameter of the tumors ranged from 1.3 to 3.7 cm (mean 2.6 cm). Two (cases 1 and 2) of the three patients who had colon polyposis had bilateral multiple thyroid tumors, whereas the remaining four without polyposis had a solitary tumor. All of the nodules showed essentially similar histologic features. They showed an intricate blending of cribriform, follicular, trabecular, solid, papillary, and morular patterns of growth (Fig. 1). The tumors were encapsulated, and colloids were scant. No regional lymph nodal metastasis was found, and vascular and capsular invasion was not noted.
We performed an immunohistochemical study for a primary monoclonal antibody, β-catenin (14; Transduction Laboratories, Lexington, KY, USA), at a dilution of 1:200. The sections were followed by signal detection using the streptavidin-biotin kit (Dako, Carpinteria, CA, USA). For a negative control, the primary monoclonal antibody was replaced with phosphate-buffered saline in duplicate sections. In all cases, β-catenin was expressed strongly in the cytoplasm and nuclei in the morular and cribriform areas of the tumors (Fig. 2); it was expressed only on the cell membrane in the follicular cells of the peritumor thyroid tissue.
Relation to FAP and Family History
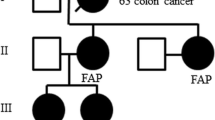
The FAP-related features of the present seven patients are summarized in Table 2. Two patients had a family history of colon polyposis and colorectal cancer. One of them refused the present invitation and examinations. Three patients had relatives with cancer of the digestive organs but not typical colon polyposis; and the remaining patient did not have a family history of malignant tumors. Two patients (cases 1 and 3) were found to have colon polyps by colonoscopy and underwent total or subtotal colectomy. One of them had a small cancer in a large polyp, and the other had small but invasive rectal cancer.
A germline APC gene mutation was found in the patients with colon polyposis. In case 1, a protein truncation test covering exons 9 to 15 detected an abnormal band. The direct sequencing method using polymerase chain reaction (PCR) products of the region revealed a deletion of thymidine in codon 512 resulting in the appearance of a stop codon (GTA GTA GCC to GAG TAG, stop) [10]. In case 3, a protein truncation test covering exons 1 to 10 detected a signal of decreased intensity of wild-type APC protein and a signal of increased intensity of a smaller product, which was the result of an abnormal splicing in exon 9 [9].
The mean follow-up period after thyroid surgery was 142 months (range 7 months to 30 years). No patient developed distant metastases or regional recurrent disease. Four patients with sporadic CMV-PTC have had no evidence of FAP after a follow-up of 1 to 10 years.
Discussion
If we find a young woman to have CMV-PTC, should we recommend that she be screened for FAP? The answer is “yes” because three of the seven patients we evaluated with this histology had associated FAP, and two of them already had colon cancer in the present study. FAP is diagnosed by flexible colonoscopy and an APC gene mutation. The patient’s family history is also useful. APC gene mutations are inherited in an autosomal dominant pattern, but 20% to 30% of FAP cases are caused by de novo mutations [11]. If the screening can detect FAP, it is possible to reduce the individual’s risk of developing colorectal cancer.
The CMV is seen in both FAP-associated PTC and FAP-nonassociated PTC. Cameselle-Teijeiro and Chan proposed the term “cribriform-morular variant of papillary carcinoma” as a variant of PTC and first reported FAP-nonassociated CMV [8]. Past reports stated that FAP-associated CMV is typically characterized by female predominance, an age at tumor diagnosis of less than 30 years in about 90% of cases, and multifocal development [1, 8, 12, 13, 14]. In the present study, all seven patients were female, and their mean age was 25 years (range 16–32 years). Two of the three patients with colon polyposis had bilateral multiple thyroid tumors, whereas the remaining four without polyposis had a single tumor. The other clinicopathologic features were similar.
We examined β-catenin expression in CMV to investigate whether there is a difference between FAP-associated CMV and sporadic CMV. β-Catenin is localized on the cell membrane, and cytoplasmic β-catenin levels are extremely low in normal cells. Defects in the adenomatous polyposis coli protein (APC) that result in its inability to interact functionally with the β-catenin destruction complex increase free β-catenin and Wnt activation [15]. β-Catenin is diverted to the nucleus, where it stimulates expression of specific target genes [16, 17, 18]. These mechanisms of β-catenin dysregulation have been shown to play an important role in the human tumorigenesis of colonic adenocarcinoma, where they are present in approximately half of the tumors with APC [16]. Kurihara et al. reported that FAP-associated CMV showed cytoplasmic and nuclear immunolocalization of β-catenin. In contrast, the follicular epithelial cells of normal thyroid tissue and common-type PTC displayed strong membrane immunoreactivity without cytoplasmic or nuclear localization [19]. In this study, all cases with CMV showed cytoplasmic and nuclear immunolocalization of β-catenin; no difference in expression or localization of β-catenin was found between the three FAP-associated CMV cases and the four sporadic CMV cases. Currently, no clear-cut difference between FAP-associated CMV and sporadic CMV is known other than the number of tumors. The prognosis for patients with CMV is good [11]. In a past report, the 5- and 20-year survivals of FAP-associated CMV were 90% and 77%, and the long-term prognosis was excellent if the patients underwent therapy for FAP [20]. In our study, the mean follow-up was 142 months. To date, no patient has developed distant metastases or recurrent regional disease. Despite seemingly aggressive pathology, patients with CMV follow a highly favorable long-term course, as do most patients with common PTC.
Conclusions
This unique histology warrants screening for FAP, particularly if the patient has multiple tumors. Screening can detect colon cancer at an early stage when the patients have no subjective symptoms.
Résumé
La variante cribiforme-morulaire (CM) du cancer papillaire de la thyroïde (CPT) est rare: elle combine deux types de croissance cellulaire: cribiforme et fusiforme. Ce type histologique unique de cancer de la thyroïde a été rapporté pour la première fois chez des patients porteurs de polypose adénomateuse familiale (PAF), bien que plus tard, elle a été décrite chez le patient sans polypose. En raison de sa rareté, ses caractéristiques cliniques ne sont pas claires. Nous avons analysé les dossiers de sept patients porteurs de variante CM du CPT retrouvés parmi les 4194 patients porteurs de CPT dans nos archives d’anatomo-pathologie entre juin 1991 et mars 2003. Parmi les CPT, la prévalence de la variante CM a été de 0.16%. Nous avons demandé à ces patients de nous fournir une histoire familiale détaillée et avons recommandé un examen colonoscopique et une analyse « germ-line » de leurs gènes APC. Deux sujets sans symptômes subjectifs avaient à la fois une polypose et un cancer colique. Les mutations génétiques germ-line APC ont été retrouvées chez les deux patients. On a su que le père d’un patient qui avait refusé de coopérer avait eu une intervention chirurgicale pour polypose colique. Parmi les quatre autres patients, on n’a retrouvé ni polypose ni mutation APC. Les données cliniques retrouvées en commun comprenaient: femme jeune (âge moyen de 25 ans), tumeurs circonscrites, absence de métastases ganglionnaires et absence de cancer de la thyroïde après chirurgie. Deux des trois patients porteurs de polypose colique avaient des tumeurs multiples bilatérales de la thyroïde, alors que les quatre autres patients sans polypose avaient une tumeur solitaire. L’histopathologie de tumeurs CM chez les patients porteurs de CPT doit soulever la suspicion de PAF, surtout si les tumeurs sont multiples. La présence de ce type de cancer de la thyroïde pourrait amener à découvrir un cancer colique à un stade précoce.
Resumen
La variante morular-cribriforme (CMV) del carcinoma papilar de tiroides (PTC) constituye un raro subtipo histológico de PTC, cuyo patrón histológico incluye áreas de células cribriformes y aracniformes. Inicialmente se pensó que este cáncer tiroideo, con estas características histológicas únicas, aparecía en pacientes con poliposis adenomatosa familiar (FAP) aunque más tarde se constató la existencia de CMV en pacientes sin poliposis. Debido a su rareza sus características clínicas son poco conocidas. Entre Junio de 1991 y marzo de 2003 se recopilaron 4194 especímenes de PTC encontrándose 7 casos de CMV-PTC. La prevalencia de los CMV fue 0.16% PTC. Invitamos a estos pacientes a volver a nuestro hospital para realizar una detallada historia familiar, recomendándoles estudio colonoscópico y análisis de la línea germinal del gen APC. Dos pacientes asintomáticos eran portadores de poliposis y cáncer de colon; en los dos casos encontramos mutaciones de la línea germinal del gen APC. El padre de un paciente que reusó acudir a esta revisión fue intervenido quirúrgicamente por poliposis de colon. En los 3 pacientes restantes no encontramos ni poliposis ni mutaciones en el gen APC. Los hallazgos clínicos más frecuentes fueron: tumores circunscritos con adenopatías no metastásicas y sin recidiva tras cirugía del cáncer de tiroides, siendo los pacientes jóvenes del sexo femenino (edad media 25 años). Dos de los 3 pacientes con poliposis de colon presentaron tumores múltiples bilaterales de tiroides, mientras que en los otros 4, sin poliposis, el tumor era único. El hallazgo histopatológico de CMV en pacientes con PTC especialmente cuando se trata de tumores múltiples debe hacer sospechar la posibilidad de una poliposis adenomatosa familiar (FAP) propiciando así el diagnóstico precoz del cáncer de colon.
References
F Cetta MR Pelizzo MC Curia et al. (1999) ArticleTitleGenetics and clinicopathological findings in thyroid carcinomas associated with familial adenomatous polyposis Am. J. Pathol. 155 7–9 Occurrence Handle1:STN:280:DyaK1MzisVaruw%3D%3D Occurrence Handle10393829
B Bell EL Mazzaferri (1993) ArticleTitleFamilial adenomatous polyposis (Gardner’s syndrome) and thyroid carcinoma: a case report and review of the literature Dig. Dis. Sci. 38 185–190 Occurrence Handle1:STN:280:ByyC3s%2FntVI%3D Occurrence Handle8420756
MR Camiel JE Mule LL Alexander et al. (1968) ArticleTitleAssociation of thyroid carcinoma with Gardner’s syndrome in siblings N. Engl. J. Med. 278 1056–1058 Occurrence Handle1:STN:280:CCeC1MvptVM%3D Occurrence Handle5644968
HR Harach GT Williams ED Williams (1994) ArticleTitleFamilial adenomatous polyposis associated thyroid carcinoma: a distinct type of follicular cell neoplasm Histopathology 25 549–561 Occurrence Handle1:STN:280:ByqB3M3pvFw%3D Occurrence Handle7698732
Baloch ZW, Lovolsi VA. Pathology of the Thyroid Gland: Endocrine Pathology, New York, Churchill Livingstone, 2002;61–88
KP Nugent AD Spigelman RK Philips (1996) ArticleTitleRisk of extracolonic cancer in familial adenomatous polyposis Br. J. Surg. 83 1121–1122 Occurrence Handle1:STN:280:ByiD383ps1Y%3D Occurrence Handle8869322
RO Plail HJ Bussey G Glazer et al. (1987) ArticleTitleAdenomatous polyposis: an association with carcinoma of the thyroid Br. J. Surg. 74 377–380 Occurrence Handle1:STN:280:BiiB2czitVc%3D Occurrence Handle3036290
J Cameselle-Teijeiro JK Chan (1999) ArticleTitleCribiriform-morular variant of papillary carcinoma: a distinctive variant representing the sporadic counterpart of familial adenomatous polyposis-associated thyroid carcinoma? Mod. Pathol. 12 400–411 Occurrence Handle1:STN:280:DyaK1M3ksVGisw%3D%3D Occurrence Handle10229505
B Xu A Miyauiti S Uchino et al. (2003) ArticleTitleA predominant increase in the APC gene isoform with exon 9a in a case of attenuated familial adenomatous polyposis Clin. Genet. 63 71–72 Occurrence Handle10.1034/j.1399-0004.2003.630111.x Occurrence Handle1:STN:280:DC%2BD3s%2FivFemtg%3D%3D Occurrence Handle12519375
B Xu K Yoshimoto A Miyauchi et al. (2003) ArticleTitleCribriform-morular variant of papillary thyroid carcinoma: a pathological and molecular genetic study with evidence of frequent somatic mutataion in exon 3 of the β-catenin gene J. Pathol. 199 58–67 Occurrence Handle10.1002/path.1225 Occurrence Handle12474227
S Bulow T Faurschou Nielson C Bulow (1996) ArticleTitleThe incidence rate of familial adenomatous polyposis: result from Danish polyposis register Int. J. Colorectal Dis. 11 88–91 Occurrence Handle10.1007/s003840050027 Occurrence Handle1:STN:280:BymA3M%2Fht1c%3D Occurrence Handle8739833
F Cetta S Olschwang M Petracci et al. (1998) ArticleTitleGenetic alterations in thyroid carcinoma associated with familial adenomatous polyposis: clinical implications and suggestions for early detection World J. Surg. 22 1231–1236 Occurrence Handle10.1007/s002689900550 Occurrence Handle1:STN:280:DyaK1M%2FmsF2msQ%3D%3D Occurrence Handle9841749
SB Ng K Sittampalam YH Goh et al. (2003) ArticleTitleCribriform-morular variant of papillary carcinoma: the sporadic counterpart of familial adenomatous polyposis associated thyroid carcinoma: a case report with clinical and molecular genetic correlation Pathology (Phila.) 35 42–46 Occurrence Handle1:CAS:528:DC%2BD3sXivVWlu74%3D
F Cetta G Montalto M Gori et al. (2000) ArticleTitleGermline mutations of the APC gene in patients with familial adenomatous polyposis-associated thyroid carcinoma: results of a European co-operative study J. Clin. Endocrinol. Metab. 85 286–292 Occurrence Handle10.1210/jc.85.1.286 Occurrence Handle1:CAS:528:DC%2BD3cXjtl2msQ%3D%3D Occurrence Handle10634400
PJ Morin AB Sparks V Korinek et al. (1997) ArticleTitleActivation of beta-catenin-Tcf signaling in colon cancer by mutations in beta-catenin or APC Science 275 1790–1792 Occurrence Handle10.1126/science.275.5307.1790 Occurrence Handle1:CAS:528:DyaK2sXitVGrtbY%3D Occurrence Handle9065403
H Dierick A Bejsovec (1999) ArticleTitleCellular mechanisms of wingless/Wnt signal transduction Curr. Top. Dev. Biol. 43 153–190 Occurrence Handle1:CAS:528:DyaK1MXpsFOm Occurrence Handle9891886
M Peifer P Polakis (2000) ArticleTitleWnt signaling in oncogenesis and embryogenesis: a look outside the nucleus Science 287 1606–1609
JR Miller AM Hocking JD Brown et al. (1999) ArticleTitleMechanism and function of signal transduction by the Wnt/β-catenin and Wnt/Ca pathways Oncogene 18 7860–7872 Occurrence Handle10.1038/sj.onc.1203245 Occurrence Handle1:CAS:528:DC%2BD3cXks1Kmtg%3D%3D Occurrence Handle10630639
K Kurihara S Shimizu J Chong et al. (2000) ArticleTitleNuclear localization of immunoreactive β-catenin is specific to familial adenomatous polyposis in papillary thyroid carcinoma Jpn. J. Cancer Res. 91 1100–1102 Occurrence Handle1:CAS:528:DC%2BD3cXosVGrs7c%3D Occurrence Handle11092973
N Perrier JA Heerden Particlevan JR Goellner et al. (1998) ArticleTitleThyroid cancer in patients with familial adenomatous polyposis World J. Surg. 22 738–743 Occurrence Handle10.1007/s002689900462 Occurrence Handle1:STN:280:DyaK1c3ntV2rsA%3D%3D Occurrence Handle9606291
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tomoda, C., Miyauchi, A., Uruno, T. et al. Cribriform-morular variant of Papillary Thyroid Carcinoma: Clue to Early Detection of Familial Adenomatous Polyposis-associated Colon Cancer. World J. Surg. 28, 886–889 (2004). https://doi.org/10.1007/s00268-004-7475-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-004-7475-4