Abstract
Historic land use changes and subsequent river channelization created deeply incised, unstable stream channels largely devoid of natural cover throughout the Yazoo River basin, Mississippi, USA. Large trash (e.g., televisions, toilets, car parts) dumped in streams provided shelter for some aquatic fauna. To determine whether trash served as a surrogate for natural cover, I examined crayfish use of both cover types. I sampled crayfishes by kick-seining 2 × 1-m plots in three cover classes: trash, natural cover, and no cover. I captured 415 crayfishes from 136 of the 294 plots. Most crayfishes were in natural cover (253), followed by trash (154), and no-cover (8) plots. Trash use varied by crayfish genus and size. Frequencies of all size classes of Procambarus and of the smallest Cambarus were higher in natural cover than trash. Many of the smallest individuals were found in live root mats. As Cambarus and Orconectes grew, they shifted more toward trash, and the largest Orconectes size class was significantly more abundant than expected in trash. Trash served as “artificial reefs,” providing cover for crayfishes and other fauna, but functioned differently than the remaining natural cover. The results confirmed that stream substrate did not provide adequate instream cover for crayfishes in the study area and suggested that high-quality natural cover for large crayfishes was in short supply, at least for some species. Land management that provides for abundant, ongoing input and retention of complex cover, such as trees and live roots, to stream channels should be beneficial for crayfish assemblages.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Most streams in north-central Mississippi are highly degraded and have simplified habitat with minimal instream cover (Shields et al. 1994; Warren et al. 2002). The degraded condition of streams resulted largely from post-European-settlement land use practices applied to highly erodible terrain (Simon and Darby 1997) and subsequent river channelization and dredging that led to headcutting and incision of tributaries (Shields et al. 1994; Shields et al. 2006). Today, even most small streams in the basin are deeply incised with sand/clay beds, little instream cover, and shallow depths at base flow (Warren et al. 2002; Shields et al. 2006). The study area remains predominantly rural, but changes in hydrology and sedimentation are “of the same order of magnitude as those that have been reported to result from urbanization…which is generally considered to represent the most radical change in land use” (Harvey and Watson 1986). Channel incision typically increases peak flows (Gordon et al. 1992), and high energy peak flows regularly either flush out or bury large wood and other forms of cover in the study streams (Warren et al. 2002). However, habitat variation still exists among headwater streams in the basin, with conditions ranging from stream segments with some large wood and mature riparian forest canopy to others with virtually no instream cover and banks covered with invasive kudzu (Pueraria montana) vines (Warren et al. 2002; Warren et al. 2009).
Although channelization, incision, and headcutting are known to influence stream faunal assemblages (Gordon et al. 1992; Shields et al. 1994; Ross et al. 2001), a diverse stream fauna persists in the study area (Warren et al. 2002). More than 35 fish and five crayfish species commonly occur in the streams. A negative species-area relationship exists for crayfishes in the study area (S. Adams, unpublished data), and it is unknown whether this reflects historic patterns or is a result of degraded conditions that provide crayfishes little refuge from predation as fish size increases with stream size.
Superimposed on this landscape is a human propensity for dumping trash, often large items, off of bridges, or steep banks into streams. In some stream reaches, trash appears to provide the most complex and stable cover available. Numerous studies document a variety of reptiles, including snakes, lizards, skinks, and turtles using trash either in summer or in winter as hibernacula, especially where habitat has been disturbed (Neill 1948; Christiansen et al. 1971; Scali and Zuffi 1994). Water snakes (Nerodia sipedon) preferred trash to more natural habitats along one stream despite some survival risks associated with using trash (Pattishall and Cundall 2009). Many studies in marine habitats document examples of large trash functioning as artificial reefs and being used by numerous fish species (Caselle et al. 2002). Nevertheless, trash is seldom considered potentially useful habitat for stream organisms. For example, the US Environmental Protection Agency’s EMAP protocol for stream habitat assessment includes trash as a measure of “anthropogenic alterations and disturbances,” but not as a measure of “habitat complexity and cover for aquatic fauna” (Kaufmann et al. 1999).
Crayfishes are often closely associated with cover and have shown strong avoidance of open sand microhabitats (Parkyn and Collier 2004). Lodge and Hill (1994) found that abundances of crayfishes and shelter were positively correlated in Wisconsin lakes and suggested that the strong interaction between intraspecific competition for habitat (or food) and predation by fishes often limits crayfish populations. Cover is important for foraging, population resilience to flooding, and avoidance of predation and cannibalism (Stein and Magnuson 1976; Blake et al. 1994; Smith et al. 1996; Parkyn and Collier 2004; Adams 2007; Jowett et al. 2008). Tree roots, leaf litter, woody debris, and undercut banks were the cover types most associated with the crayfish Paranephrops planifrons in New Zealand (Jowett et al. 2008). The authors predicted that decreased P. planifrons abundances would result from activities, such as channelization, that increased velocities and removed shallow edge habitats or that lowered water levels so stream edges were separated from streambank cover.
During stream sampling in incised streams of north Mississippi over the previous 8 years, I have often observed crayfishes using trash as cover. Indeed, the largest crayfishes, including females with eggs or young, often seemed to live in large trash items such as old tires or televisions. These observations led me to the following questions: (1) to what extent does instream trash serve as “artificial reefs,” providing cover for crayfishes, and (2) does use of cover types differ among crayfish genera and size classes (e.g., DiStefano et al. 2003)?
In spring 2010, I conducted a study to quantify crayfish use of trash in streams. Prior to beginning, I made the following hypotheses, based on informal observations during previous sampling: (1) crayfish abundance would be higher in plots with trash than with natural or no cover, (2) crayfishes in trash would be larger than those in natural cover, and (3) the use of trash would not vary by species.
Methods
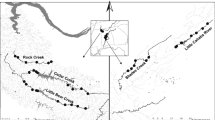
Study sites were in north-central Mississippi within the Northern Hilly Gulf Coastal Plain (65e) and Loess Plains (74b) ecoregions, marked by low to moderate gradient streams with substrates primarily of sand, silt, or clay (Chapman et al. 2004), and occasionally patches of gravel. I selected eight sites (Fig. 1; Table 1) containing garbage small enough to sample effectively. Sites were in eight, wadeable, headwater streams with water velocities slow enough to allow seining. Within each site, I selected an equal number of plots with trash, natural cover, and no cover. I attempted to sample 30 plots per site; however, some sites did not have 10 pieces of garbage that could be sampled, so I sampled additional plots at other sites with abundant garbage (Table 1). Plots were located haphazardly, balancing the total number along each bank and mid-channel.
Crayfishes were sampled by first surrounding the downstream end and sides of the plot with a seine net (3.2-mm mesh). One person then kicked the substrate and disturbed or entirely removed the cover in an area 2-m long by 1-m wide upstream of the seine. Sometimes garbage occurred in aggregate heaps or mixed with large wood. In that case, we only sampled garbage or natural cover that we could isolate with the seine. Crayfishes from each seine sample were held separately. Immediately after sampling ended, crayfishes were identified, sexed, measured with calipers for post-orbital carapace length (POCL; behind eye to the mid-dorsal point on the posterior edge of carapace; POCL equals roughly half of the total body length), then released to the stream. I retained voucher specimens of each species.
The types of cover, if any, and location in the stream (near the stream bank or mid-channel) were recorded. Most plots contained more than one type of cover. One piece of cover frequently served as an anchor to accumulate additional natural or human-made debris. For example, a mattress would accumulate a large amount of leaves in the coils. All types of cover in a plot were recorded.
Comparisons of crayfish frequencies among cover classes and between bank and mid-channel locations were conducted using goodness-of-fit tests (PopTools version 3.2 as an add-in program in Microsoft Excel 14.0; Sokal and Rohlf 1995). Expected frequencies were calculated as equal probabilities among cover classes multiplied by the total number of crayfishes captured across the classes being compared. To create size categories for analyses, I examined length–frequency histograms by genus and identified size class break points that were common to all genera.
Several habitat and water physicochemical parameters were measured to characterize streams. In each site, we chose one location with relatively uniform flow to measure stream width and discharge and calculated the latter using the transect method (Harrelson et al. 1994) with a Marsh-McBirney Flowmate 2000 and a topsetting rod. Water quality parameters, including dissolved oxygen (mg/l and % saturation), conductivity (μS/cm), pH, and temperature (oC) were measured at the time of crayfish sampling using a Hydrolab Quanta. The Hydrolab was calibrated daily for dissolved oxygen and weekly for other measures. Water quality measurements were not taken at Beartail Creek because of an equipment malfunction. Channel substrate size distribution was not characterized because of the apparent uniformity in substrate among sites.
Results
Streams differed from one another primarily in size, with discharge varying two orders of magnitude among sites (Table 1). Specific conductivity was uniformly low (28–70 μS/cm), pH ranged from 5.3 to 6.1, and dissolved oxygen levels ranged from 7.5 to 10.2 mg/l (87–105 % saturation) (Table 1).
I sampled 294 plots, evenly divided among the three cover classes, and collected 413 crayfishes of seven species. Data from three Procambarus too small to identify to species were discarded. Procambarus, represented by four species, was the most abundant genus in the samples, but Orconectes etnieri (sensu Taylor et al. 2013) was the most abundant species, followed by Cambarus striatus. Several individuals were not measured (because of very recent molting or escape after identification), so sample sizes varied slightly among analyses.
In the garbage and natural cover categories, most plots contained more than one type of cover. For garbage plots, I summarized plots by the dominant item that was anchoring other material. Large appliances and house components (e.g., washing machines, televisions, toilets, roofing tin) characterized the greatest number of trash plots (Fig. 2). The most common type of natural cover was small wood (<10 cm diameter or <1.5 m long), followed by leaf packs and large wood (>10 cm diameter or >1.5 m long; Table 2), with all three types often co-occurring.
Crayfishes were not uniformly distributed among cover classes. Across all sites combined, the percentage of plots containing crayfishes was similar between garbage and natural cover plots but significantly lower in no-cover plots (Fig. 3). In three sites, the percentages of plots containing crayfishes were nearly identical between garbage and natural cover, but in the most unbalanced site, Duncans Creek, 86 % of garbage plots had crayfishes compared to 43 % of natural cover plots. In four sites, we found no crayfish in no-cover plots. Although fewer than 10 % of the plots had more than four individuals per plot, more natural cover plots than trash plots contained five or more individuals (Fig. 4).
Use of natural cover versus trash differed by both genus and size class. Goodness-of-fit tests indicated that use of the three cover types was nonuniform for all three genera (all sizes combined; Table 3), due most obviously to the paucity of crayfishes in no-cover plots (Figs. 5, 6). However, within genera, use patterns were consistent among species (all sizes combined; Fig. 5). Procambarus was the only genus in which frequencies were greater in natural cover than trash for all size classes (Table 3; Fig. 6). The smallest size class of Cambarus (<13 mm POCL) used natural cover more than trash, but use of natural cover versus trash did not differ significantly in the larger two size classes (Table 3; Fig. 6). In Orconectes, distributions of the smallest two size classes did not differ from random in trash versus natural cover, but the largest size class used trash more frequently than natural cover (Table 3; Fig. 6). Note that for the larger size classes, sample sizes were smaller and, therefore, statistical power was lower.
Frequency distribution showing number of crayfishes by cover class and genus for individuals <13.0 mm post-orbital carapace length (POCL) (a), 13.0-22.9 mm POCL (b), and >22.9 mm POCL (c). For each genus, goodness-of-fit tests including all cover classes were highly significant. Significant differences between frequencies in trash versus natural cover are indicated by asterisks (**P < 0.001; *P < 0.05; see Table 3 for details)
Crayfish size in relation to cover use can be viewed in a number of other ways as well. Both median crayfish sizes (median test, P = 0.017) and size distributions (Kruskal–Wallis test, P = 0.002) differed among the three cover classes (all species combined; Fig. 7). The no-cover plots had a larger median crayfish size than either trash (P = 0.029) or natural (P = 0.031) cover plots due to the lack of small individuals (Fig. 7). Because the no-cover plots had very few crayfishes (n = 8), they were not analyzed further with respect to crayfish size. Between trash and natural cover plots, crayfish median sizes were similar (Median test, P = 0.053), but size distributions differed (Mann–Whitney U test, P = 0.004), with distributions in the natural cover plots skewed more toward smaller crayfishes (Fig. 7b).
Several other measures were consistent with size-differentiated tendencies of crayfishes to use different cover types. The largest individuals used trash most frequently. Seven of the 10 largest crayfishes captured were in trash, and the largest individual of five of the seven species was in trash. Also, the largest individual (representing four species) in six of the eight sites was in trash. Finally, 66 % (10 of 15) of crayfishes >30 mm POCL were in trash. Conversely, the smallest crayfish tended to use natural cover most frequently. Seven of the 10 smallest crayfish were in natural cover, and 72 % (36 of 50) of juvenile crayfish <10 mm POCL were in natural cover. The smallest individual of five species was in natural cover. Only two juveniles were in no-cover plots.
Several site characteristics were qualitatively associated with size extremes. Each of the two natural plots that harbored the largest crayfish from a site contained a root wad. The six trash plots that had the largest crayfish from a site always contained trash the size of a car wheel (about 40-cm diameter) or larger. Most plots with the largest crayfish contained a mixture of large and small cover types (e.g., a root wad and leaves or a tire and small woody debris). During sampling, I observed that relatively large groups of the smallest crayfishes often occurred in fine, live roots of woody plants extending into the water from the stream banks.
Distributions of plots within stream channels differed by cover class, but crayfish plot use differed by location only in natural cover. Natural cover plots were more frequent near the sides than in the middle of the channel, because most of the natural cover originated from the stream bank or was in the form of undercut banks (Table 4). Trash plots were distributed about equally between the sides and middle, and no-cover plots were more frequent mid-channel (Table 4). In the trash and no-cover classes, crayfish use of plots was independent of the plot location in a stream; however, in natural cover plots, crayfishes were slightly more abundant than expected in plots near the sides of a stream (Table 4).
Discussion
Nearly all crayfishes in the study were associated with cover, but the nature of the cover used varied by crayfish size and genus. The smallest crayfishes tended to prefer natural cover over trash, and as they grew, became more likely to use trash. These results are consistent with observations I made during previous sampling for other studies. However, the findings that Procambarus spp. of all sizes preferred natural cover to trash and that the preferences for trash versus natural cover varied among genera were contrary to my expectations.
Crayfish size is typically the strongest determinant of success in obtaining possession of desired shelter in both intraspecific (Rabeni 1985; Ranta and Lindström 1993; Figler et al. 1999) and interspecific (Rabeni 1985; Nakata and Goshima 2003) interactions. Therefore, in streams, the largest individuals are expected to occupy the most desirable shelters. Because the largest individuals in most sites chose trash, I inferred that trash frequently provided the best shelter in the site for large adults. Trash was used significantly more than natural cover by the largest size class of Orconectes, indicating that trash provided necessary habitat for large Orconectes in the simplified study streams. Crayfish use of brush bundles added to streams in the region also suggested a lack of quality cover in the incised streams (Warren et al. 2009).
Although small crayfishes also used trash, they were more abundant in natural cover. Crayfishes often exhibit ontogenetic shifts in habitat use, in many cases apparently to adjust to size-dependent changes in predation risks and cover suitabilities. As crayfishes grow, the effectiveness of protection offered by various cover types changes, and predation risks may depend on crayfish size, cover characteristics, and predator size and behavior. For example, small crayfishes often move to stream margins to avoid fish predation, whereas large crayfishes that are less susceptible to fish predation may move to deeper water to avoid terrestrial predators (Garvey et al. 2003). Smaller crayfishes may also use fine roots along stream margins to avoid larger individuals. Habitat shifts by juvenile crayfishes in the presence of adults were demonstrated in Missouri rivers (e.g., Rabeni 1985). In addition, trash, like natural cover laying on the streambed, was highly susceptible to becoming buried or embedded in sand. Often the interstices of fine structures within trash were filled with sand. Conversely, live roots that grew into the water from the stream banks were at least partially suspended in the water column and, therefore, were not as susceptible to becoming embedded. Numerous studies on various crayfish genera have shown that juvenile crayfishes often prefer stream margins characterized by shallower water, generally lower water velocities, finer substrate, and sometimes more macrophytes than mid-channel locations (Rabeni 1985; DiStefano et al. 2003; Jowett et al. 2008). The presence of riparian shrubs and trees and the extension of their roots into the stream were a major influence on the abundance of the crayfish Austropotamobius pallipes in an English drainage, and the stream margins were especially important to juveniles (Smith et al. 1996). Similarly, crayfish selected large, as well as small fibrous tree roots in New Zealand streams (Parkyn and Collier 2004). Smith et al. (1996) postulated that the roots served multiple functions of providing shelter, trapping food, and creating undercut banks, all beneficial to the crayfish. Tree root mats in Missouri streams harbored more insects than did pool or riffle habitats and also contained a unique community, possibly due to the structural complexity the roots created (Wood and Sites 2002).
In addition to crayfishes, we found many other species using trash, suggesting that it provides useful cover for a variety of organisms in the stream. A variety of small-bodied fishes, especially Noturus spp. and Ameiurus spp., commonly used trash. This is consistent with other research that has indicated cover limitations for these and other fishes in the region (Shields et al. 1994; Warren et al. 2002; Warren et al. 2009). We also observed water snakes (Nerodia spp.), cottonmouths (Agkistrodon piscivorus), snapping turtles (Chelydra serpentina), and a wide variety of small invertebrates in trash. Trash apparently provided shelter for potential predators, as well as prey, of crayfishes, but the presence of potential predators does not necessarily preclude use by crayfishes. Several microhabitat-scale studies showed no interactions between abundances of crayfishes and predatory fishes in cover (Bishop et al. 2008; Jowett et al. 2008; Warren et al. 2009).
In natural cover plots, crayfishes were more abundant than expected near stream banks compared to mid-channel. Several plausible explanations exist. First, as noted above, juvenile crayfishes in many systems prefer stream margins, particularly in the presence of fishes or large crayfishes (Rabeni 1985; Englund and Krupa 2000), and juveniles constituted the majority of my catches. Second, tree roots appeared to be a preferred cover type for juveniles, and root mats occurred only along stream banks; however, crayfishes also were more abundant in constructed habitat bundles placed near banks than mid-channel in a nearby stream (Warren et al. 2009), suggesting that the result was not due solely to the presence of tree roots. Third, although not quantified, natural cover near banks seemed to be more complex than mid-channel natural cover, which was often a single log or a small leaf pile. Finally, natural cover plots along stream banks were more likely to be close to other cover, including burrows in banks, than were mid-channel plots, which may make them more suitable or more likely to be encountered and colonized.
Procambarus showed stronger preference for natural cover over trash than did the other genera. Reasons for the difference in preference are not clear, but may relate to general habitat types in which each genus evolved. All three genera are widespread in eastern North America; however, the center of distribution, and probably evolution, for Procambarus is along the Coastal Plain of the lower southeastern United States (Hobbs 1984), whereas centers of distribution in Orconectes and Cambarus are farther north (Guiaşu 2002; Hamr 2002) where rockier habitats prevail. So although all three genera occupy a wide range of habitats, grossly generalized habitat preferences for finer versus coarser sediments and for soft versus hard forms of cover may be reflected in the habitat preferences exhibited by the genera in relation to natural cover versus trash.
As with any sampling method, my approach had its limitations, including the possibility that thorough sampling of crayfishes was least efficient around some of the most complex and stable cover. Large, live roots, (e.g., as of cypress trees Taxodium distichum) seemed particularly difficult to sample effectively. Our results may be somewhat confounded if our sampling was least efficient in the best cover, but regardless, extensive use of trash, especially by large individuals, was clearly documented.
A caveat to this study is that I conducted no toxicity testing of the trash. I witnessed paint cans with paint flowing down the stream, auto parts releasing oil, antifreeze containers, and many other potentially toxic items. A crayfish survey of 28 river and stream sites in Kansas found that two of the four sites lacking crayfishes were full of garbage and oil and another had an oil slick (Durbian et al. 1994), suggesting that toxins associated with the garbage may have eliminated crayfishes. In addition, several of my sites were organically enriched by carcasses (deer, large fishes, etc.) dumped in the stream. We did not sample carcasses directly, but they undoubtedly influenced the water quality of the streams. Presumably, such organic enrichment would benefit some species at the expense of others. At one site (Jones Creek) that always contained an abundant supply of carcasses, the density of O. etnieri during a different study was as high as I have encountered in any north Mississippi stream.
The results may appear to present a management dilemma: to remove or not to remove trash from the stream. Certainly potential toxicity and health risks need to be included in such decision making. From the standpoint of instream cover, large trash appears to be functioning as artificial reefs, providing useful cover to crayfishes and other fauna in these simplified, degraded streams. In the marine literature, the question of whether artificial reefs merely attract fish or actually increase production is unresolved (Carr and Hixon 1997). In the study streams, it appears that trash probably is not an adequate surrogate for natural cover, especially with respect to providing rearing habitat for small crayfishes. However, the largest females have the highest reproductive value, so a stable or increasing population may depend on suitable habitat for large individuals (Rabeni 1985); therefore, artificial cover may benefit crayfish production by increasing survival of large females and, thus, increasing reproductive output.
Taking a broader view, it is difficult to encourage a sense of public stewardship and appreciation of streams that are full of trash. Indeed, watershed planning groups have identified “improving aesthetics” as a desirable goal with progress toward the goal measured by the change in the percent of stream with trash (Merrick and Garcia 2004). Aesthetics (i.e., lack of trash) were assigned economic value in both residential and commercial/industrial metrics (Merrick and Garcia 2004). Therefore, in the interest of avoiding toxic effects and increasing a sense of public “ownership” of stream resources, maintaining trash-free streams remains an appropriate goal.
Ultimately, the conclusions extend beyond whether or not trash is beneficial as cover and serve to illustrate that crayfishes in the region depend heavily on instream cover other than substrate. A further research step would be to determine whether cover is actually limiting to crayfish populations in the streams and whether addition of cover increases population sizes. Long-term conservation of stream crayfishes, as well as fishes and other fauna, will benefit from land management that maintains and restores processes providing ongoing supply and retention of cover, particularly stable, complex cover, in stream channels. Furthermore, to maximize crayfish habitat, stream flows and stream channel morphology need to be such that water remains in contact with stream banks and their associated cover during low flows (Jowett et al. 2008). Although deeply incised alluvial river systems cannot be fully restored, they can reach a new equilibrium in which secondary floodplains develop within the incised channels (Harvey and Watson 1986; Simon 1989). Establishment of new channel equilibria takes time and requires that streambed elevations are not further disturbed and that land use does not change in ways that dramatically alter hydrology (Harvey and Watson 1986). Development of new floodplains should help restore cover to stream channels and facilitate stream water remaining in contact with stream banks and their associated cover.
References
Adams SB (2007) Direct and indirect effects of channel catfish (Ictalurus punctatus) on native crayfishes (Cambaridae) in experimental tanks. Am Midl Nat 158:85–96
Bishop C, Begley B, Nicholas C, Rader J, Reed E, Sykes K, Williams T, Young E, Mullen D (2008) Shelter-use and interactions between banded sculpin (Cottus carolinae) and bigclaw crayfish (Orconectes placidus) in stream-pool habitats. Southeast Nat 7(1):81–90
Blake M, Nyström P, Hart P (1994) The effect of weed cover on juvenile signal crayfish (Pacifastacus leniusculus Dana) exposed to adult crayfish and non-predatory fish. Ann Zool Fenn 31:297–306
Carr MH, Hixon MA (1997) Artificial reefs: the importance of comparisons with natural reefs. Fish 22(4):28–33
Caselle JE, Love MS, Fusaro C, Schroeder D (2002) Trash or habitat? Fish assemblages on offshore oilfield seafloor debris in the Santa Barbara Channel, California. ICES J Mar Sci 59(suppl):S258–S265. doi:10.1006/jmsc.2002.1264
Chapman SS, Griffith GE, Omernik JM, Comstock JA, Beiser MC, Johnson D (2004) Ecoregions of Mississippi (color poster with map, descriptive text, summary tables, and photographs; map scale 1:1,000,000). U.S. Geological Survey, Reston, Virginia
Christiansen JL, Degenhardt WG, White JE (1971) Habitat preferences of Cnemidophorus inornatus and C. neomexicanus with reference to conditions contributing to their hybridization. Copeia 2:357–359. doi:10.2307/1442858
DiStefano RJ, Decoske JJ, Vangilder TM, Barnes LS (2003) Macrohabitat partitioning among three crayfish species in two Missouri streams USA. Crustaceana 76(3):343–362
Durbian FE III, Frey BJ, Moore DW (1994) Crayfish species from creeks and rivers of Cherokee County, Kansas. Kans Acad Sci 97(1/2):13–17. doi:10.2307/3628248
Englund G, Krupa JJ (2000) Habitat use by crayfish in stream pools: influence of predators, depth and body size. Freshw Biol 43(1):75–83
Figler MH, Cheverton HM, Blank GS (1999) Shelter competition in juvenile red swamp crayfish (Procambarus clarkii): the influences of sex differences, relative size, and prior residence. Aquaculture 178(1):63–75
Garvey JE, Rettig JE, Stein RA, Lodge DM, Klosiewski SP (2003) Scale-dependent associations among fish predation, littoral habitat, and distributions of crayfish species. Ecology 84(12):3339–3348
Gordon ND, McMahon TA, Finlayson BL (1992) Stream hydrology: an introduction for ecologists. Wiley, New York
Guiaşu RC (2002) Cambarus. In: Holdich DM (ed) Biology of freshwater crayfish. Blackwell Science, Ames, pp 609–634
Hamr P (2002) Orconectes. In: Holdich DM (ed) Biology of freshwater crayfish. Blackwell Science, Ames, pp 585–608
Harrelson CC, Rawlins CL, Potyondy JP (1994) Stream channel reference sites: an illustrated guide to field technique. U.S. Forest Service General Technical Report RM-245, Fort Collins, Colorado
Harvey MD, Watson CC (1986) Fluvial processes and morphological thresholds in incised channel restoration. Water Resour Bull 22:359–367
Hobbs HH Jr (1984) On the distribution of the crayfish genus Procambarus (Decapoda: Cambaridae). J Crustac Biol 4(1):12–24. doi:10.2307/1547892
Jowett IG, Parkyn SM, Richardson J (2008) Habitat characteristics of crayfish (Paranephrops planifrons) in New Zealand streams using generalised additive models (GAMs). Hydrobiologia 596(1):353–365
Kaufmann PR, Levine P, Robison EG, Seeliger C, Peck DV (1999) Quantifying physical habitat in wadeable streams. Report number EPA/620/R-99/003. Environmental Protection Agency
Lodge DM, Hill AM (1994) Factors governing species composition, population size, and productivity of cool-water crayfishes. Nord J Freshw Res 69:111–136
Merrick JRW, Garcia MW (2004) Using value-focused thinking to improve watersheds. J Am Plan Assoc 70(3):313–327
Nakata K, Goshima S (2003) Competition for shelter of preferred sizes between the native crayfish species Cambaroides japonicus and the alien crayfish species Pacifastacus leniusculus in Japan in relation to prior residence, sex difference, and body size. J Crustac Biol 23(4):897–907
Neill WT (1948) Hibernation of amphibians and reptiles in Richmond County, Georgia. Herpetologica 4(3):107–114
Parkyn SM, Collier KJ (2004) Interaction of press and pulse disturbance on crayfish populations: flood impacts in pasture and forest streams. Hydrobiologia 527:113–124
Pattishall A, Cundall D (2009) Habitat use by synurbic watersnakes (Nerodia sipedon). Herpetologica 65(2):183–198
Rabeni CF (1985) Resource partitioning by stream-dwelling crayfish: the influence of body size. Am Midl Nat 113(1):20–29
Ranta E, Lindström K (1993) Body size and shelter possession in mature signal crayfish, Pacifastacus leniusculus. Ann Zool Fenn 30:125–132
Ross ST, O’Connell MT, Patrick DM, Latorre CA, Slack WT, Knight JG, Wilkins SD, Montgomery WL (2001) Stream erosion and densities of Etheostoma rubrum (Percidae) and associated riffle-inhabiting fishes: biotic stability in a variable habitat. Copeia 2001(4):916–927. doi:10.1643/0045-8511(2001)001[0916:SEADOE]2.0.CO;2
Scali S, Zuffi M (1994) Preliminary report on a reptile community ecology in a suburban habitat of northern Italy. Ital J Zool 61(1):73–76
Shields FD Jr, Knight SS, Cooper CM (1994) Effects of channel incision on base flow stream habitats and fishes. Environ Manag 18:43–57
Shields FD Jr, Knight SS, Stofleth JM (2006) Large wood addition for aquatic habitat rehabilitation in an incised, sand-bed stream, Little Topashaw Creek, Mississippi. River Res Appl 22:803–817
Simon A (1989) The discharge of sediment in channelized alluvial streams. J Am Water Resour Assoc 25(6):1177–1188
Simon A, Darby SE (1997) Disturbance, channel evolution and erosion rates: Hotophia Creek, Mississippi. In: Wang SSY, Langendoen EJ, Shields FD Jr (eds) Management of landscapes disturbed by channel incision: stabilization, rehabilitation, restoration. University of Mississippi Press, Oxford, pp 476–481
Smith G, Learner M, Slater F, Foster J (1996) Habitat features important for the conservation of the native crayfish Austropotamobius pallipes in Britain. Biol Conserv 75(3):239–246
Sokal RR, Rohlf FJ (1995) Biometry, 3rd edn. W. H. Freeman and Company, New York
Stein RA, Magnuson JJ (1976) Behavioral response of crayfish to a fish predator. Ecology 57:751–761
Taylor CA, Adams SB, Schuster GA (2013) Systematics and biogeography of Orconectes, subgenus Trisellescens, in the southeastern United States, a test of morphology-based classification. J Crustacean Biol. doi:10.1163/1937240X-00002196
Warren ML, Jr., Haag WR, Adams SB (2002) Forest linkages to diversity and abundance in lowland stream fish communities. In: Holland MM, Warren ML, Jr., Stanturf JA (eds) Proceedings of a conference on sustainability of wetlands and water resources: how well can riverine wetlands continue to support society into the 21st century? Gen. Tech. Rep. SRS-50. U.S. Department of Agriculture, Forest Service, Southern Research Station. Asheville, pp 168–182
Warren ML, Jr., Sheldon AL, Haag WR (2009) Constructed microhabitat bundles for sampling fishes and crayfishes in Coastal Plain streams. N Am J Fish Manag 29:330–342
Wood DL, Sites RW (2002) Submerged rootmats: a mesohabitat harboring a distinct insect community in Ozark streams. J Freshw Ecol 17(3):431–440. doi:10.1080/02705060.2002.9663917
Acknowledgments
I thank the FS technicians, M. Bland, A. Commens-Carson, and G. McWhirter, and FS volunteer, J. Ryndock, who went beyond the call of duty in this sampling effort. The research was supported by the USDA Forest Service, Southern Research Station.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Adams, S.B. Crayfish Use of Trash Versus Natural Cover in Incised, Sand-Bed Streams. Environmental Management 53, 382–392 (2014). https://doi.org/10.1007/s00267-013-0197-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00267-013-0197-3