Abstract
Character displacement, or a shift in traits where species co-occur, is one of the most common ecological patterns to result from interactions between closely related species. Usually, character displacement is associated to divergence in traits, though, they might be convergent, especially when used for aggressive interference between species. In the context of animal communication, territorial calls are predicted to converge in order to increase context recognition and decrease the costs of ecological interference competition. However, such signals might also be adapted to characteristics of the shared environment. In this study, we used data from 15 groups of two parapatric tamarins, Saguinus midas and S. bicolor, to test for similarities in long calls among sympatric and allopatric groups. We hypothesized that calls would converge in sympatric areas, as it would be mutually beneficial if both species recognize territorial contexts, but that convergence would depend on forest type due to acoustic adaptation. As predicted, long calls converged in sympatry, with S. midas shifting its calls towards S. bicolor’s acoustic pattern. However, this shift only occurred in primary forest. In sympatric areas, S. midas produced sounds with narrower bandwidths in primary than in secondary forest, consistent with optimization of sound propagation while both species produced longer calls in primary forests independently of geographic location (i.e. sympatry and allopatry). Our results suggest that both social and environmental pressures are important in shaping tamarin sounds. As their effects can interact, analyses, which assume that these ecological pressures act independently, are likely to miss important patterns.
Significance statement
Territorial signals between closely related sympatric species are expected to be convergent to increase context recognition and decrease the costs of interference competition. However, such signals might also be adapted to characteristics of the shared environment, such as forest structure characteristics. We analysed vocalisations from two parapatric tamarins and found that, though their long calls asymmetrically converged in sympatry, only red-handed tamarins shifted towards pied tamarin call type, and this only occurred in primary forest. Our results suggest that both social and environmental pressures are important in shaping primate calls. Because their effects can interact, analyses which assume that these pressures act independently, are likely to miss important patterns.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Selection for reducing niche overlap often drives character displacement, i.e. exaggerated differences between species populations in sympatry relative to those in allopatry (Pfennig and Pfennig 2009; Weber and Strauss 2016; Grether et al. 2017; Stuart et al. 2017). Any morphological, ecological, physiological or behavioural trait can follow such a pattern, either through local adaptation or phenotypic plasticity (Brown and Wilson 1956; Grant 1972; Pfennig and Pfennig 2012; Allen et al. 2014; de Moura Bubadué et al. 2016). Traits related to reproductive interference (i.e. sexual interactions between species including courtship and other displays) are more likely to be divergent as pressure for mate recognition and hibridization avoidance is high (West-Eberhard 1983; Okamoto and Grether 2013). Meanwhile, traits related to resource competition and aggressive interference can be convergent or divergent depending on the costs of the interaction between species, their relative abundance, and the evolutionary timeframe of interaction between the species (Grether et al. 2017, 2020; Germain et al. 2018). In fact, when analysing multiple traits, some pairs of species can have both convergent and divergent displacement patterns depending on trait function (Miller et al. 2019; Grether 2020). Regardless, evidence for both divergent and convergent character displacement often requires that traits are displaced in areas where species co-occur, independently of other factors (Schluter 2001; Wilkins et al. 2013).
Many species have calls that are used as a defensive response to territory intrusions from conspecifics and heterospecifics (McGregor 1993; Peiman and Robinson 2010; Christensen and Radford 2018). Such calls may be considered fitness-related traits, since behavioural interference can affect the rate of population growth (Chesson 2000; Grether et al. 2017, 2020). Therefore, selection can favour convergence of such fitness-related traits in sympatry to minimize competitive asymmetries, when competitors engage in direct conflicts (Germain et al. 2018). Accordingly, many bird species have convergently evolved sounds that facilitate the recognition of the competitive context (Laiolo 2012; Tobias et al. 2014; Kirschel et al. 2019). Also, some pairs of frogs species have convergent territory and release calls, which are traits related to aggressive or “neutral” contexts (Leary 2001; Freitas and Toledo 2020; Grether 2020; Grether et al. 2020).
Similar environmental characteristics can also promote trait convergence in ecological communities, independently of species interactions (Cardoso and Price 2010). Animal sounds, for instance, are often well-adapted for propagation in the habitats in which the emitting individuals occur (Bradbury and Vehrencamp 1998). The acoustic adaptation hypothesis posits that in woody forests, high-pitched calls of short duration will degrade quickly, whereas calls that are long, low-pitched, and with narrower bandwidths, will propagate more effectively (Morton 1975; Dabelsteen et al. 1993; Holland et al. 1998). This hypothesis has been tested by numerous studies and corroborated by some (Ey and Fischer 2009), although little or no effect has been found by others (birds: Bicudo et al. 2016; felids: Peters and Peters 2010; marmots: Daniel and Blumstein 1998; howler monkeys: Holzmann and Areta 2020; and gorillas: Hedwig et al. 2015). Many of the studies that did not find support for the acoustic adaptation hypothesis have suggested that social aspects (mainly intraspecific) are also important in shaping acoustic features, although few have tested both factors together (Kirschel et al. 2019, 2020).
The acoustic adaptation hypothesis may be particularly important in understanding the impacts of land use change in human-modified landscapes, as it suggests that habitat modification creates differences in selection pressures for sound propagation optimization. For instance, the significant conversion of tropical forests into secondary forests over the last 50 years (Chazdon 2014; Crist et al. 2017) has increased environmental heterogeneity in these landscapes, with remaining forests varying widely in terms of plant species richness, composition, microclimate and above-ground biomass (Guariguata and Ostertag 2001; Montgomery and Chazdon 2001; Rozendaal et al. 2019). Old-growth (primary or pristine) forests, on the other hand, tend to be more complex in terms of canopy stratification, having larger and taller trunks and more closed canopy (Montgomery and Chazdon 2001), all of which could affect sound propagation.
Neotropical primates are a useful system for investigating the effects of habitat modification on acoustic adaptation and character displacement given that (1) primates are generally thought to produce innate calls, and there is little evidence for plasticity in their vocalisations (Egnor and Hauser 2004; Snowdon 2017a); (2) vocal flexibility may be an important aspect in social signal convergence (Laiolo 2012) and (3) primates have suffered significant and ongoing modification of their natural tropical forests (Estrada et al. 2018). In Central Amazonia, the red-handed tamarin (Saguinus midas) and the pied tamarin (S. bicolor) are two closely related primates with parapatric distributions and a narrow sympatric zone (Röhe 2006; Rylands et al. 2016). While S. midas occurs over much of the northeastern portion of the Amazon region, S. bicolor has one of the smallest ranges of any primate in the world (~ 7500 km2) (Gordo et al. 2017), being restricted to the interfluvial region between the Amazon, Negro, and Urubu rivers, and much of its range lying within the largest urban area of Amazonia — the city of Manaus (Fig. 1; Gordo et al. 2013). S. bicolor is currently classified as Critically Endangered by the International Union for the Conservation of Nature (IUCN) (Gordo et al. 2019a) due to fragmentation and habitat loss, as well as possible competition with S. midas, and is among the 25 most endangered primate species in the world (Gordo et al. 2019b). Additionally, over the last 30 years, the range of S. bicolor has decreased, while that of S. midas has not only increased, but it has encroached upon that of S. bicolor (Röhe 2006; Gordo et al. 2017). Pied and red-handed tamarins are considered potential competitors since both species have very similar body sizes (Hershkovitz 1977), niches (i.e. use of spaces with similar climate, relief, and vegetation types) (Röhe 2006), and they show asymmetrical territorial responses to each other’s calls in sympatric areas (Sobroza et al. 2021).
Species distribution ranges and location of sampling sites. (a) Map of Saguinus midas and S. bicolor ranges and putative sympatric area (based on Röhe 2006). (b) Sampling sites of S. bicolor and S. midas in areas of sympatry and allopatry
Tamarin long calls have an important territorial function (Snowdon 1989, 2017a, b), therefore, we hypothesized that acoustic parameters of pied and red-handed tamarin long calls would converge in sympatry after accounting for potential acoustic adaptation to human-driven habitat heterogeneity (primary or secondary forest). Accordingly, the two species should converge in long call acoustic parameters in sympatry as it would be beneficial if both recognize the territorial context, in order to reduce the probability of interference competition. However, we expected frequency convergence to be stronger (or occur only) in primary forests, assuming that populations occurring in older and/or less disturbed forests are more likely to show acoustic adaptation, since frequency features are more prone to be constrained by morphology and genetics (Riede et al. 2005; Kitchen et al. 2019). In contrast, temporal parameters should converge in both forest types, as these traits tend to be more plastic (Janik and Slater 1997; Kitchen et al. 2019). We also predicted that, independently of geographic condition (i.e. sympatry and allopatry), both species would produce longer and lower-pitched calls in primary forests compared to secondary forests, as predicted by the acoustic adaptation hypothesis.
Material and methods
Study site
We carried out the study in the Brazilian Central Amazon, which included allopatric and sympatric areas in the ranges of S. bicolor and S. midas (Fig. 1). Topography in the region includes an alternation between drainage catchments and plateaus (ca. 60–120 m a.s.l.). Native vegetation cover is tropical humid terra firme forest in both primary and secondary stages of succession, with patches of white sand scrub forest (campina and campinarana) (Oliveira et al. 2008). Within this region, we chose four sites where the two primates were in allopatry, and four sites in sympatry, for a total of eight sites. We determined the geographic condition (sympatry or allopatry) of groups, based on evidence from previous studies (Ayres et al. 1982; Röhe 2006), data from long-term projects (Pied Tamarin Project and Tropical Ecology, Assessment and Monitoring Network), and co-occurrence modelling (Röhe 2006). Simultaneous observations of both species at the same site, mixed groups, or hybrids (at least those physically evident, e.g. Gordo et al. 2017) were never recorded during this study. All sampled areas contained forests with both primary and secondary growth, but forest cover was continuous in all cases. We categorized the location where the groups were detected as primary or secondary forest based on field observation of structural characteristics of the forest, and the presence of trees commonly found in Amazonian secondary forests such as Cecropia spp., Vismia spp. and Bellucia spp. (Mesquita et al. 2001).
Data collection
Fieldwork was carried out over nine field visits from June 2014 to February 2015 and consisted of recording tamarin long calls following standardized playback trials. A total of 275 calls were recorded in a sampling effort that comprised 690 km walked over 97 days distributed across the eight sites, with around nine consecutive sampling days at each site. Data collection was carried out between 06:30 and 16:30, the period when tamarins show the greatest activity (Day and Elwood 1999; Gordo et al. 2017). We used existing trails and roads to survey each area until we found a tamarin group. Once this occurred, we marked the encounter location with a Garmin GPS and characterized the group according to the number of adult individuals. Different sightings at a given site were considered as belonging to the same group when we consistently observed the same number of individuals and the distance between sightings did not exceed 400 m. Although the annual home range size of pied tamarins’ group may reach up to 100 ha (Gordo et al. 2008), at smaller time scales such as 9 days, they rarely exceed 40 ha (Day and Elwood 1999; Monteiro-Neto 2015 TVS and MG pers. obs.). Such small home range sizes are common among callitrichids (Garbino and Martins-Junior 2018), thus supporting the 400 m threshold used here. We considered as independent only those groups separated from each other by at least 1 km.
We recorded tamarin calls for 10 min immediately following playback trials (see details at Sobroza et al. 2021). This duration was chosen because the animals were not habituated to the observers and rarely stayed longer than this at the same place. During these trials, once we encountered a tamarin group, we broadcasted long calls from non-neighbouring pied or red-handed tamarins. The sounds used during the playbacks were composed of long calls from both allopatric and sympatric areas, and we presented these to the species in a balanced way across species and geographic condition (i.e. sympatric and allopatric areas). Individual groups were played recordings of long calls from both species (with a minimum interval of 1 h) and their responses were recorded. We recorded tamarins with.wav files using a HTDZ HT-81 microphone (Frequency response: 1–16.0 kHz) attached to a Zoom H1 recorder (16-bit, 44 kHz). It was not possible to record data blindly because our study involved focal animals in the field.
Acoustics analysis
We analysed pied and red-handed tamarin long call recordings in Raven 1.4 (Cornell University Laboratory of Ornithology 2010). Long calls are either emitted as a territorial signal during intergroup encounters or as part of intragroup communication and cohesion (Sobroza et al. 2017). Since we recorded the calls after broadcasting playbacks, the behavioural context in which they were engaged was predominantly territorial. We defined a long call as a signal composed of variable number of syllables (not more than 8) with ∩ − shaped spectral signature and relatively constant interval between syllables (mean = 0.129 s, SD = 0.045 s). For each call, we analysed nine acoustic parameters (Table 1). We measured temporal parameters from oscillograms and spectral parameters of the first (fundamental) harmonic by analysing power spectra (Sobroza et al. 2017) built with the following configuration: DFT size = 1024 samples, overlap = 80%, window size = 20 ms, window type = Blackmann.
Data analysis
The analysed data consisted of acoustic parameters (Table 1) of individual long calls (S. midas, N = 151; S. bicolor, N = 124) clustered by tamarin group (N = 15) and sampling site (N = 8) (one to three groups per site). Since most acoustic parameters were correlated, data were ordinated using Principal Components Analysis (PCA). We applied the broken stick test (Peres-Neto et al. 2005) to separate meaningful information and choose how many PC axes to use in additional analysis. The test identified the first two axes as significantly informative, but we added the third axis, which contained most of the information on the temporal aspects of sound. The first axis captured 31.7% of the data variability, and the second and third 27.9 and 14.59%, respectively. Therefore, we used PC1, PC2 and PC3 to summarize tamarin sounds and analysed each axis separately as a response variable in Generalized Linear Mixed-effects Models (GLMMs) with Gaussian distribution (Zuur et al. 2009). In all models, we tested for the three-way interaction between predictors that were species (two levels: S. midas or S. bicolor), geographic condition (two levels: sympatry or allopatry) and forest type (two levels: primary and secondary forests). Group and site identities were used as random factors to account for potential autocorrelation among calls from the same group and groups from the same site. All analyses were performed in R software version 4.0 (R Development Core Team 2020), using packages nlme, vegan and vireg (Dixon 2003; Breheny and Burchett 2017; Pinheiro et al. 2018).
Results
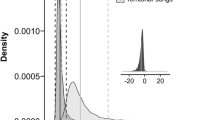
According to the PCA, the red-handed tamarin had a relatively wider acoustic niche, while the pied tamarin appeared to form a nested subgroup within the red-handed tamarin acoustic niche (Fig. 2, Tables 2, 3). We found that bandwidth mainly contributed to PC1, frequency features to PC2, and duration and entropy features to PC3 (Table 2). We found that allopatric populations of S. midas and S. bicolor in primary forests were statistically different in their PC1 scores, with sounds of S. midas in allopatric/primary forested areas differing markedly from S. midas in sympatric/primary forested areas, and from S. bicolor in any condition (Table 4, Fig. 3). In sympatric areas, S. midas sounds were more similar to those of S. bicolor sounds. Thus, S. midas displaced towards convergence to S. bicolor’s acoustic niche, with higher PC1 scores (i.e. narrower bandwidths), but only in primary forest. In contrast, S. bicolor did not show such a pattern (Fig. 3, Table 4). Furthermore, forest type was the only significant predictor of PC2 and PC3 scores (Table 4). PC2 scores were slightly higher in secondary forests than in primary forests (i.e. lower dominant and higher frequencies), while PC3 scores were higher in primary than secondary forests (i.e. longer durations) (Fig. 4).
Acoustic niche of pied and red-handed tamarins. The first two axes of a Principal Component Analysis (PCA) were used to represent long calls based on nine acoustic parameters. Each point in the graph represents a long call of pied tamarins (black symbols) (n = 124) and red-handed tamarins (red symbols) (n = 151) in sympatry (triangles) and allopatry (circles)
Differences of acoustic features of pied and red-handed tamarins relative to interactive effects of habitat type (red = primary forest and blue = secondary forest), and geographic condition (allopatry and sympatry), based on the Generalized Linear Mixed Model (GLMM). Each point represents the PC1 score of a long call (n = 275); lines represent predicted means for each group of observations. Plots use partial residuals of the response variable and thus show the effect of a given interaction while controlling for the effects of remaining predictors
Differences of acoustic features of pied and red-handed tamarins relative to effects of habitat type (red = primary forest and blue = secondary forest) based on the Generalized Linear Mixed Model (GLMM). Each point represents (a) PC 2 and (b) PC 3 scores of long calls (n = 275); lines represent predicted means for each group of observations. Plots use partial residuals of the response variable and thus show the effect of a given variable while controlling for the effects of remaining predictors
Discussion
In this study, we tested whether selection would favour acoustic convergence of territorial calls between sympatric tamarin species, assuming that this would facilitate context recognition when the risk of conflict is imminent (Laiolo 2012). Our results support this hypothesis, but only for one of the two species (S. midas) which showed convergent sounds in areas of sympatry. To the best of our knowledge, this is the first record of asymmetric convergent character displacement in primate social signals. Convergence in competitor recognition characters is expected when levels of interspecific aggressions are intermediate (Grether et al. 2020), whereas asymmetry in character displacement can be a result of differential success in resource holding between species (Grether et al. 2020). Indeed, S. midas appears to be vocally more territorial than S. bicolor during territory intrusion experiments (Sobroza et al. 2021). Nevertheless, the fact that the shift we observed in S. midas calls only occurred in primary forests suggests that multiple and interacting selective forces might be shaping tamarin sounds, as observed in birds and rodents (Campbell et al. 2010; Tobias et al. 2014). Secondary forests contain more food resources for tamarins (Gordo 2012), therefore, competition would be reduced and character displacement less likely.
Many neotropical primates display long calls, which are sounds with multiple functions (Snowdon et al. 1983; Oliveira and Ades 2004). Both pied and red-handed tamarins make long calls that are used for intra- and intergroup communication to aid in group cohesion, movement coordination, and spacing during territorial contexts (Day and Elwood 1999; Snowdon 2017b; Sobroza et al. 2017). In other primates, long calls may also function in mate defence (Snowdon 1989; Wich and Nunn 2002), but we have no evidence supporting such hypothesis for the pied and red-handed tamarins, and only weak evidence for callitrichids, in general. For instance, if long calls also have a reproductive function, we would expect an asymmetric sex ratio during call emission and responses. In captive colonies of tamarins (Saguinus oedipus), results are inconsistent: sometimes males and sometimes females respond more to the opposite sex (McConnell and Snowdon 1986; Scott et al. 2006), while in the field, there are recordings of both males and females of Saguinus spp. emitting long calls during group encounters (Garber et al. 1993; Garber 1997). Therefore, while the territorial function of long calls is relatively well accepted and widespread among callitrichids, the hypothesis that it has a possible reproductive function (e.g. mate attraction or mate-guarding) remains poorly supported. When characters have multiple functions, directions of displacement may be less obvious (Grether et al. 2009), though, our results are under what is expected for territorial signals (Drury et al. 2020).
As expected, frequency shifts (see PC1 and PC2, Table 2) occurred mainly in primary forests, and were accompanied by shifts in temporal features (PC3, Table 2). Such acoustic changes are only partially following the acoustic adaptation hypothesis, which predicts that in areas with woody vegetation (e.g. primary forest), signals that are longer, lower-pitched and with a narrow bandwidth would be selected (Morton 1975). We found that S. midas presented narrower bandwidths in primary forests, in accordance with the acoustic adaptation hypothesis, but mainly in sympatric areas. Sounds with narrower bandwidths propagate better in closed habitats because the intensity is concentrated in specific frequency bands (Morton 1975). One possible explanation for the asymmetry in the displacement is that in areas where a possible competitor is present, S. midas emits sounds with narrower bandwidths, and higher amplitude to achieve longer distances, which could also imply an advantage during territorial defence (Ritschard et al. 2012). Even if the effect was not strong, both species produced slightly higher dominant frequencies in primary forests independently of geographic condition. Higher frequencies tend to degrade quickly in closed habitats. So, other strategies that enhance sound propagation may be used by tamarins such as site-selected perching, as occurs in other callithrichids (Sabatini and Ruiz-Miranda 2010). As predicted, both species presented longer calls in primary than in secondary forests, and our findings agree with the idea that frequency features are less plastic than temporal features (Janik and Slater 1997); the strongest effect on frequency features was more evident in primary forests. Amplitude and temporal features are more likely to be modulated in a shorter period through phenotypic plasticity, due to simpler mechanisms of production (Janik and Slater 1997). Plasticity provides an important mechanism that facilitates resource partitioning (Pfennig et al. 2006), but the extent to which the observed acoustic differences reflect plasticity vs. local adaptation is unclear.
Whatever mechanism is behind the acoustic variation that we observed, tamarin calls did not differ significantly between species in secondary forests. If long calls also play a role in sex recognition in pied and red-handed tamarins as is suggested for other callitrichids (Weiss et al. 2001; Miller et al. 2004), this may be related to recognition errors that could result in maladaptive mating and hybridization. During the last three decades, the city of Manaus has been growing rapidly, and new roads have been created in areas where the pied and the red-handed tamarins co-occur, thereby increasing the likelihood of hybridization (McFarlane and Pemberton 2019). Hybridization often causes convergence in physical and behavioural aspects of parapatric species (Brockelman and Schilling 1984; Long et al. 1998; Secondi et al. 2003; Kitchen et al. 2019). As the physical appearance of S. midas and S. bicolor hybrids can be similar to parental species, or a mix of characteristics of parental species (Gordo et al. 2017), it is not possible to rule out the possibility that we recorded individuals that are phenotypically similar to one of the parental species but are hybrids. Many bird species tend to show convergent traits and intense interspecific territoriality when hybridization occurs in sympatric areas (Drury et al. 2020; Cowen et al. 2020), which may also be the case for pied and red-handed tamarins. There is no data available on the actual magnitude of gene flow between the red-handed and the pied tamarin hybrids. If the behavioural dominance of S. midas (Sobroza et al. 2021) also implies differences of gene flow, this could cause the absence of differentiation between species calls and asymmetrical character displacement (Yikweon 2008). Sound signal similarities can be the cause and consequence of hybridization, and the origin or extent to which both processes are occurring in this system remains to be tested.
Sobroza et al. (2021) found that red-handed tamarins responded with more territorial calls when listening to pied tamarin calls than to its own species in sympatric areas; while pied tamarins did not respond differently when listening to its species or the congener. Hence, despite the major pattern of convergence in acoustic features recorded here, such sounds must not be functionally interpreted equally by both species. Not only signal production can be susceptible to character displacement, but also its perception (Seddon and Tobias 2010), if pied and red-handed tamarins use different acoustic features as cues for species recognition or are different in terms of tuning capacity (e.g. Curé et al. 2012; Pasch et al. 2017), this may also be related to the asymmetries found in responses to the playback. Even closely related species can have asymmetrical cognitive and recognition capacities, and some species are capable of detecting specific signals with minimal variance (Seddon and Tobias 2010; Grether 2011; Pasch et al. 2017). It is worth mentioning that in the previous playback experiments, Sobroza et al. (2021) did not present congeneric sounds from all possible combinations (i.e. sympatric\primary forest; sympatric\secondary forest; allopatric\primary forest, and allopatric\ secondary areas), which may be a non-biological explanation related to their findings of asymmetrical responses.
Generally, character displacement and competitor recognition are phenomena involving many signal components, sensory modalities and genes (Ortiz-Barrientos et al. 2004; Grether 2011; Pureswaran et al. 2016). Therefore, even if acoustic signals are convergent and can be influenced by hybridization, species discrimination can still be possible if this function is retained in at least one signal. Many animals are flexible enough to quickly shift communication modalities when the signal transmission is impaired (sensu Partan 2017). Secondary forests usually have a more open canopy, fewer obstacles such as large trunks and lower relative humidity, features that might favour visual and chemical long-distance communication (Müller-Schwarze 2006; Sicsú et al. 2013). Thus, we cannot underestimate the recognition capacity of pied and red-handed tamarins in sympatric areas based only on our acoustic results.
It is instructive to consider the extent to which other factors not linked to forest structure could affect our results. For instance, some gregarious species have sounds that are group-specific (Crockford et al. 2004; Zaccaroni et al. 2012; Mumm and Knörnschild 2017). If tamarins also have sounds that act as group identifiers and the density of tamarins in secondary forest is higher, then acoustic diversity related to group identity may be increased. Indeed, red-handed tamarins are known to occur at higher densities in secondary forests than in primary forests (Lenz et al. 2014). Our statistical analysis included the identity of the groups as a random factor — therefore our results are unlikely to be related to group identity. Primary and secondary forests will also have a distinct composition of vocally-active species other than tamarins (e.g. birds, anurans and insects), such as, creating a local soundscape that might also drive acoustic divergence and sound diversification (Naugler and Ratcliffe 1994; Snowdon and de la Torre 2002; Gasc et al. 2013; Bicudo et al. 2016). Finally, secondary forests themselves can be highly variable in terms of acoustically-relevant structural properties (Wiley and Richards 1978; Montgomery and Chazdon 2001), though, it does not explain the observed displacement among sympatric and allopatric areas. Accounting for all these potential effects is logistically challenging, but our findings provide a template upon which more detailed hypotheses can be elaborated.
In summary, S. midas seems to have a wider acoustic niche and greater vocal flexibility than S. bicolor, responding both to the presence of the congener and forest type. In allopatric areas and primary forest, both species have a unique sound structure. Yet, this structure is consistent with the acoustic adaptation hypothesis, suggesting that it optimizes sound propagation in these habitats. In contrast, in sympatric areas, the major pattern of variation in the acoustic features converges, consistent with selection for enhanced context recognition and, thus, reduces interference competition. Our results suggest that both social and physical constraints interact asymmetrically to shape the sound produced by parapatric tamarins. Thus, analyses that account for these pressures independently are likely to miss important patterns in ecological systems of co-occurring species.
Data availability
Data are available as supplementary material.
References
Allen WL, Stevens M, Higham JP (2014) Character displacement of Cercopithecini primate visual signals. Nat Commun 5:4266. https://doi.org/10.1038/ncomms5266
Ayres JM, Mittermeier RA, Constable ID (1982) Brazilian tamarins on the way to extinction? Oryx 16:329–333. https://doi.org/10.1017/S0030605300017786
Bicudo T, Anciães M, Benchimol M, Peres CA, Simões PI (2016) Insularization effects on acoustic signals of 2 suboscine Amazonian birds. Behav Ecol 27:1480–1490. https://doi.org/10.1093/beheco/arw070
Bradbury JW, Vehrencamp SL (1998) Principles of animal communication. Oxford University Press, Oxford
Breheny P, Burchett W (2017) Visualization of regression models using visreg. R J 9:56–71. https://doi.org/10.32614/rj-2017-046
Brockelman WY, Schilling D (1984) Inheritance of stereotyped gibbon calls. Nature 312:634–636. https://doi.org/10.1038/312634a0
Brown WL, Wilson EO (1956) Character displacement. Syst Zool 5:49–64. https://doi.org/10.2307/2411924
Campbell P, Pasch B, Pino JL, Crino OL, Phillips M, Phelps SM (2010) Geographic variation in the songs of neotropical singing mice: testing the relative importance of drift and local adaptation. Evolution 64:1955–1972. https://doi.org/10.1111/j.1558-5646.2010.00962.x
Cardoso GC, Price TD (2010) Community convergence in bird song. Evol Ecol 24:447–461. https://doi.org/10.1007/s10682-009-9317-1
Chazdon R (2014) Second growth: the promise of tropical forest regeneration in an age of deforestation. Chicago Press, Chicago
Chesson P (2000) Mechanisms of maintenance of species diversity. Annu Rev Ecol Syst 31:343–366. https://doi.org/10.1146/annurev.ecolsys.31.1.343
Christensen C, Radford AN (2018) Dear enemies or nasty neighbors? Causes and consequences of variation in the responses of group-living species to territorial intrusions. Behav Ecol 29:1004–1013. https://doi.org/10.1093/beheco/ary010
Cody ML (1969) Convergent characteristics in sympatric species: a possible relation to interspecific competition and aggression. Condor 71:223–239. https://doi.org/10.2307/1366300
Cowen MC, Drury JP, Grether GF (2020) Multiple routes to interspecific territoriality in sister species of North American perching birds. Evolution 74:2134–2148. https://doi.org/10.1111/evo.14068
Crist E, Mora C, Engelman R (2017) The interaction of human population, food production, and biodiversity protection. Science 356:260–264. https://doi.org/10.1126/science.aal2011
Crockford C, Herbinger I, Vigilant L, Boesch C (2004) Wild chimpanzees produce group-specific calls: a case for vocal learning? Ethology 110:221–243. https://doi.org/10.1111/j.1439-0310.2004.00968.x
Curé C, Mathevon N, Mundry R, Aubin T (2012) Acoustic cues used for species recognition can differ between sexes and sibling species: evidence in shearwaters. Anim Behav 84:239–250
Dabelsteen T, Larsen ON, Pedersen SB (1993) Habitat-induced degradation of sound signals: quantifying the effects of communication sounds and bird location on blur ratio, excess attenuation, and signal-to-noise ratio in blackbird song. J Acoust Soc Am 93:2206. https://doi.org/10.1121/1.406682
Daniel JC, Blumstein DT (1998) A test of the acoustic adaptation hypothesis in four species of marmots. Anim Behav 56:1517–1528. https://doi.org/10.1006/anbe.1998.0929
Day RT, Elwood RW (1999) Sleeping site selection by the golden-handed tamarin Saguinus midas midas: the role of predation risk, proximity to feeding sites, and territorial defence. Ethology 105:1035–1051. https://doi.org/10.1046/j.1439-0310.1999.10512492.x
de Moura Bubadué J, Cáceres N, dos Santos CR, Sponchiado J, Passaro F, Saggese F, Mondanaro A, Raia P, Carotenuto F (2016) Character displacement under influence of bergmann’s rule in Cerdocyon thous (Mammalia: Canidae). Hystrix 27:83–90. https://doi.org/10.4404/hystrix-27.2-11433
Dixon P (2003) VEGAN, a package of R functions for community ecology. J Veg Sci 14:927–930. https://doi.org/10.1111/j.1654-1103.2003.tb02228.x
Drury JP, Cowen MC, Grether GF (2020) Competition and hybridization drive interspecific territoriality in birds. P Natl Acad Sci USA 117:12923–12930. https://doi.org/10.1073/pnas.1921380117
Egnor SER, Hauser MD (2004) A paradox in the evolution of primate vocal learning. Trends Neurosci 27:649–654. https://doi.org/10.1016/j.tins.2004.08.009
Estrada A, Garber PA, Mittermeier RA et al (2018) Primates in peril: the significance of Brazil, Madagascar, Indonesia and the Democratic Republic of the Congo for global primate conservation. PeerJ 6:e4869. https://doi.org/10.7717/peerj.4869
Ey E, Fischer J (2009) The “acoustic adaptation hypothesis”—a review of the evidence from birds, anurans and mammals. Bioacoustics 19:21–48. https://doi.org/10.1080/09524622.2009.9753613
Freitas M de O, Toledo LF (2020) Treefrogs with distinct advertisement calls produce similar territorial signals. Bioacoustics (published online, https://doi.org/10.1080/09524622.2020.1791733)
Garber PA (1997) One for all and breeding for one: Cooperation and competition as a tamarin reproductive strategy. Evol Anthropol 5:187–199. https://doi.org/10.1002/(sici)1520-6505(1997)5:6%3c187::aid-evan1%3e3.0.co;2-a
Garber PA, Pruetz JD, Isaacson J (1993) Patterns of range use, range defense, and intergroup spacing in moustached tamarin monkeys (Saguinus mystax). Primates 34:11–25. https://doi.org/10.1007/BF02381276
Garbino GST, Martins-Junior AMG (2018) Phenotypic evolution in marmoset and tamarin monkeys (Cebidae, Callitrichinae) and a revised genus-level classification. Mol Phylogenet Evol 118:156–171. https://doi.org/10.1016/j.ympev.2017.10.002
Gasc A, Sueur J, Jiguet F, Devictor V, Grandcolas P, Burrow C, Depraete M, Pavoine S (2013) Assessing biodiversity with sound: do acoustic diversity indices reflect phylogenetic and functional diversities of bird communities? Ecol Indic 25:279–287. https://doi.org/10.1016/j.ecolind.2012.10.009
Germain RM, Williams JL, Schluter D, Angert AL (2018) Moving character displacement beyond characters using contemporary coexistence theory. Trends Ecol Evol 33:74–84. https://doi.org/10.1016/j.tree.2017.11.002
Gordo M (2012) Ecologia e conservação do sauim-de-coleira, Saguinus bicolor (Primates; Callitrichidae). PhD thesis, Museu Paraense Emílio Goeldii
Gordo M, Calleia FO, Vasconcelos SA et al (2013) The challenges of survival in a concrete jungle: conservation of the pied tamarin (Saguinus bicolor) in the urban landscape of manaus, Brazil. In: Marsh L, Chapman CA (eds) Primates in Fragments: Complexity and Resilience. Springer, New York, pp 357–370
Gordo M, Jerusalinsky L, Mittermeier RA, Röhe F, Boubli JP, Subirá RJ, Vidal MD (2019a) Saguinus bicolor. IUCN Red List Threat Species. https://doi.org/10.2305/IUCN.UK.2019-3.RLTS.T40644A17931870.en
Gordo M, Lagroteria D, Röhe F, Jerusalinsky J, Azevedo RB, Vidal MD, Hrbek T, Farias I, Rylands AB (2019b) Pied tamarin: Saguinus bicolor Spix, 1823 Brazil. In: Schwitzer C, Mittermeier RA, Rylands AB (eds) Primates in Peril: the world’s 25 most endangered primates 2018–2020. International Primatological Society, Global Wildlife Conservation and Bristol Zoological Society, Washington DC, IUCN SSC Primate Specialist Group, pp 82–85
Gordo M, Rodrigues LF, Vidal MD, Spironello WR (2008) Primatas. In: Oliveira M, Baccaro FB, Braga-Neto R, Magnusson WE (eds) Reserva Ducke–a Biodiversidade amazônica através de uma Grade. Átema design editorial, Manaus, pp 39–49
Gordo M, Subirá RJ, Vidal MD, Röhe F, Spironello WR, Valente LM, Oliveira JB, Pissinatti A, Wormell D (2017) Contextualização do Sauim-de-coleira. In: Jerusalinsky L, Azevedo R, Gordo M (eds) Plano de ação nacional para a conservação do sauim-de-coleira. Instituto Chico Mendes de Conservação da Biodiversidade, Brasília, pp 25–44
Grant PR (1972) Convergent and divergent character displacement. Biol J Linn Soc 4:39–68. https://doi.org/10.1111/j.1095-8312.1972.tb00690.x
Grether GF (2011) The neuroecology of competitor recognition. Integr Comp Biol 51:807–818. https://doi.org/10.1093/icb/icr060
Grether GF (2020) Convergent and divergent selection drive plumage evolution in woodpeckers. Nat Commun 11:144. https://doi.org/10.1038/s41467-019-14006-3
Grether GF, Drury JP, Okamoto KW, McEachin S, Anderson CN (2020) Predicting evolutionary responses to interspecific interference in the wild. Ecol Lett 23:221–230. https://doi.org/10.1111/ele.13395
Grether GF, Losin N, Anderson CN, Okamoto KW (2009) The role of interspecific interference competition in character displacement and the evolution of competitor recognition. Biol Rev 84:617–635
Grether GF, Peiman KS, Tobias JA, Robinson BW (2017) Causes and consequences of behavioral interference between species. Trends Ecol Evol 32:760–772. https://doi.org/10.1016/j.tree.2017.07.004
Guariguata MR, Ostertag R (2001) Neotropical secondary forest succession: changes in structural and functional characteristics. For Ecol Manage 148:185–206. https://doi.org/10.1016/S0378-1127(00)00535-1
Hedwig D, Mundry R, Robbins MM, Boesch C (2015) Audience effects, but not environmental influences, explain variation in gorilla close distance vocalizations—a test of the acoustic adaptation hypothesis. Am J Primatol 77:1239–1252. https://doi.org/10.1002/ajp.22462
Hershkovitz P (1977) Living New World Monkeys (Platyrrhini). Primates, vol. 1. Chicago University Press, Chicago
Holland J, Dabelsteen T, Pedersen SB, Larsen ON (1998) Degradation of wren Troglodytes troglodytes song: implications for information transfer and ranging. J Acoust Soc Am 103:2154–2166. https://doi.org/10.1121/1.421361
Holzmann I, Areta JI (2020) Reduced geographic variation in roars in different habitats rejects the acoustic adaptation hypothesis in the black-and-gold howler monkey (Alouatta caraya). Ethology 126:76–87. https://doi.org/10.1111/eth.12962
Janik VM, Slater PJB (1997) Vocal learning in mammals. Adv Stud Behav 26:59–99
Kirschel ANG, Nwankwo EC, Seal N, Grether GF (2020) Time spent together and time spent apart affect song, feather colour and range overlap in tinkerbirds. Biol J Linn Soc 129:439–458. https://doi.org/10.1093/biolinnean/blz191
Kirschel ANG, Seddon N, Tobias JA (2019) Range-wide spatial mapping reveals convergent character displacement of bird song. Proc R Soc B 286:20190443. https://doi.org/10.1098/rspb.2019.0443
Kitchen DM, Bergman TJ, Dias PAD, Ho L, Canales-Espinoza D, Cortés-Otiz L (2019) Temporal but not acoustic plasticity in hybrid howler monkey (Alouatta palliata × A. pigra) loud calls. Int J Primatol 40:132–152. https://doi.org/10.1007/s10764-017-0004-8
Laiolo P (2012) Interspecific interactions drive cultural co-evolution and acoustic convergence in syntopic species. J Anim Ecol 81:594–604. https://doi.org/10.1111/j.1365-2656.2011.01946.x
Leary CJ (2001) Evidence of convergent character displacement in release vocalizations of Bufo fowleri and Bufo terrestris (Anura; Bufonidae). Anim Behav 61:431–438. https://doi.org/10.1006/anbe.2000.1597
Lenz BB, Jack KM, Spironello WR (2014) Edge effects in the primate community of the biological dynamics of forest fragments project, Amazonas, Brazil. Am J Phys Anthropol 155:436–446. https://doi.org/10.1002/ajpa.22590
Long AM, Moore NP, Hayden TJ (1998) Vocalizations in red deer (Cervus elaphus), sika deer (Cervus nippon), and red x sika hybrids. J Zool 244:123–134. https://doi.org/10.1017/S0952836998001149
McConnell P, Snowdon CT (1986) Vocal interactions between unfamiliar groups of captive cotton-top tamarins. Behaviour 97:273–296. https://doi.org/10.1163/156853986X00649
McFarlane SE, Pemberton JM (2019) Detecting the true extent of introgression during anthropogenic hybridization. Trends Ecol Evol 34:315–326
McGregor PK (1993) Signalling in territorial systems: a context for individual identification, ranging and eavesdropping. Phil Trans R Soc B 340:237–244. https://doi.org/10.1098/rstb.1993.0063
Mesquita RCG, Ickes K, Ganade G, Bruce Williamson G (2001) Alternative successional pathways in the Amazon Basin. J Ecol 89:528–537. https://doi.org/10.1046/j.1365-2745.2001.00583.x
Miller CT, Scarl J, Hauser MD (2004) Sensory biases underlie sex differences in tamarin long call structure. Anim Behav 68:713–720. https://doi.org/10.1016/j.anbehav.2003.10.028
Miller ET, Leighton GM, Freeman BG, Lees AC, Ligon RA (2019) Ecological and geographical overlap drive plumage evolution and mimicry in woodpeckers. Nat Commun 10:1602. https://doi.org/10.1038/s41467-019-09721-w
Monteiro-Neto BD (2015) Área e uso do espaço de Saguinus bicolor (Primates: Callitrichidae). Master Thesis, Universidade Federal do Amazonas
Montgomery RA, Chazdon RL (2001) Forest structure, canopy architecture, and light transmittance in tropical wet forests. Ecology 82:2707–2718. https://doi.org/10.1890/0012-9658(2001)082[2707:FSCAAL]2.0.CO;2
Morton ES (1975) Ecological sources of selection on avian sounds. Am Nat 109:17–34. https://doi.org/10.1086/282971
Müller-Schwarze D (2006) Chemical ecology of vertebrates. Cambridge University Press, Cambridge
Mumm CAS, Knörnschild M (2017) Territorial choruses of giant otter groups (Pteronura brasiliensis) encode information on group identity. PLoS ONE 12:e0185733. https://doi.org/10.1371/journal.pone.0185733
Naugler CT, Ratcliffe L (1994) Character release in bird song: a test of the acoustic competition hypothesis using american tree sparrows Spizella arborea. J Avian Biol 25:142. https://doi.org/10.2307/3677033
Okamoto KW, Grether GF (2013) The evolution of species recognition in competitive and mating contexts: the relative efficacy of alternative mechanisms of character displacement. Ecol Lett 16:670–678. https://doi.org/10.1111/ele.12100
Oliveira DAG, Ades C (2004) Long-distance calls in neotropical primates. An Acad Bras Cienc 76:393–398. https://doi.org/10.1590/S0001-37652004000200031
Oliveira ML, Baccaro FB, Braga-neto R, Magnusson WE (2008) Reserva Ducke: A Biodiversidade Amazônica través de uma grade. Editora INPA, Manaus
Ortiz-Barrientos D, Counterman BA, Noor MAF (2004) The genetics of speciation by reinforcement. PLoS Biol 2:e416. https://doi.org/10.1371/journal.pbio.0020416
Partan SR (2017) Multimodal shifts in noise: switching channels to communicate through rapid environmental change. Anim Behav 124:325–337. https://doi.org/10.1016/j.anbehav.2016.08.003
Pasch B, Sanford R, Phelps SM (2017) Agonistic character displacement in social cognition of advertisement signals. Anim Cogn 20:267–273. https://doi.org/10.1007/s10071-016-1046-6
Peiman KS, Robinson BW (2010) Ecology and evolution of resource-related heterospecific aggression. Q Rev Biol 85:133–158. https://doi.org/10.1086/652374
Peres-Neto PR, Jackson DA, Somers KM (2005) How many principal components? Stopping rules for determining the number of non-trivial axes revisited. Comput Stat Data Anal 49:974–997. https://doi.org/10.1016/j.csda.2004.06.015
Peters G, Peters MK (2010) Long-distance call evolution in the Felidae: effects of body weight, habitat, and phylogeny. Biol J Linn Soc 101:487–500. https://doi.org/10.1111/j.1095-8312.2010.01520.x
Pfennig DW, Pfennig KS (2012) Development and evolution of character displacement. Ann NY Acad Sci 1256:89–107. https://doi.org/10.1111/j.1749-6632.2011.06381.x
Pfennig DW, Rice AM, Martin RA (2006) Ecological opportunity and phenotypic plasticity interact to promote character displacement and species coexistence. Ecology 87:769–779. https://doi.org/10.1890/05-0787
Pfennig KS, Pfennig DW (2009) Character displacement: ecological and reproductive responses to a common evolutionary problem. Q Rev Biol 84:253–276. https://doi.org/10.1086/605079
Pinheiro J, Bates D, DebRoy S, Sarkar D, R Core Team (2018) Package “nlme”: linear and nonlinear mixed effects models, https://CRAN.R-project.org/package=nlme. Accessed 29 June 2020
Pureswaran DS, Hofstetter RW, Sullivan BT, Potter KA (2016) The role of multimodal signals in species recognition between tree-killing bark beetles in a narrow sympatric zone. Environ Entomol 45:582–591. https://doi.org/10.1093/ee/nvw022
R Development Core Team (2011) R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria, http://www.R-project.org
Riede T, Bronson E, Hatzikirou H, Zuberbühler K (2005) Vocal production mechanisms in a non-human primate: morphological data and a model. J Hum Evol 48:85–96. https://doi.org/10.1016/j.jhevol.2004.10.002
Ritschard M, van Oers K, Naguib M, Brumm H (2012) Song amplitude of rival males modulates the territorial behaviour of great tits during the fertile period of their mates. Ethology 118:197–202. https://doi.org/10.1111/j.1439-0310.2011.01999.x
Röhe F (2006) Área de contato entre as distribuições geográficas de Saguinus midas e Saguinus bicolor (Callitrichidae-Primates): a importância de interações e fatores ecológicos. Master Thesis, Instituto Nacional de Pesquisas da Amazônia
Rozendaal DMA, Bongers F, Aide TM et al (2019) Biodiversity recovery of Neotropical secondary forests. Sci Adv 5:eaau3114. https://doi.org/10.1126/sciadv.aau3114
Rylands AB, Heymann EW, Lynch Alfaro J, Buckner JC, Roos C, Boubli JP, Sampaio R, Mittermeier RA (2016) Taxonomic review of the New World tamarins (Primates: Callitrichidae). Zool J Linn Soc 177:1003–1028. https://doi.org/10.1111/zoj.12386
Sabatini V, Ruiz-miranda CR (2010) Does the golden lion tamarin, Leontopithecus rosalia ( Primates : Callitrichidae ), select a location whithin the forest strata for long distance communication ? Zoologia 27:179–183. https://doi.org/10.1590/S1984-46702010000200004
Schluter D (2001) Ecological character displacement. In: Fox CW, Roff DA, Fairbairn DJ (eds) Evolutionary ecology: concepts and case studies. Oxford University Press, New York, pp 265–276
Scott JJ, Carlson KL, Snowdon CT (2006) Labile sex differences in long calling in cotton-top tamarins. Am J Primatol 68:153–160. https://doi.org/10.1002/ajp.20213
Secondi J, Bretagnolle V, Compagnon C, Faivre B (2003) Species-specific song convergence in a moving hybrid zone between two passerines. Biol J Linn Soc 80:507–517. https://doi.org/10.1046/j.1095-8312.2003.00248.x
Seddon N, Tobias JA (2010) Character displacement from the receiver’s perspective: species and mate recognition despite convergent signals in suboscine birds. Proc R Soc Lond B 277:2475–2483. https://doi.org/10.1098/rspb.2010.0210
Sicsú P, Manica LT, Maia R, Macedo RH (2013) Here comes the sun: multimodal displays are associated with sunlight incidence. Behav Ecol Sociobiol 67:1633–1642. https://doi.org/10.1007/s00265-013-1574-x
Snowdon CT (1989) Vocal communication in New World monkeys. J Hum Evol 18:611–633. https://doi.org/10.1016/0047-2484(89)90097-3
Snowdon CT (2017a) Learning from monkey “talk.” Science 355:1120–1122. https://doi.org/10.1126/science.aam7443
Snowdon CT (2017b) Vocal communication in family-living and pair-bonded primates. In: Quam RM, Ramsier MA, Fay RR, Popper AN (eds) Primate Hearing and Communication. Springer, Cham, pp 141–174
Snowdon CT, Cleveland J, French JA (1983) Responses to context- and individual-specific cues in cotton-top tamarin long calls. Anim Behav 31:92–101. https://doi.org/10.1016/S0003-3472(83)80177-8
Snowdon CT, de la Torre S (2002) Multiple environmental contexts and communication in pygmy marmosets (Cebuella pygmaea). J Comp Psychol 116:182–188. https://doi.org/10.1037/0735-7036.116.2.182
Sobroza TV, Cerqueda LS, Simões PI, Gordo M (2017) Vocal repertoire and its behavioral contexts in the pied tamarin, Saguinus bicolor. Int J Primatol 38:642–655. https://doi.org/10.1007/s10764-017-9971-z
Sobroza TV, Gordo M, Barnett APA, Boubli JP, Spironello WR (2021) Parapatric pied and red-handed tamarin responses to congeneric and conspecific calls. Acta Oecol 110:103688. https://doi.org/10.1016/j.actao.2020.103688
Stuart YE, Inkpen SA, Hopkins R, Bolnick DI (2017) Character displacement is a pattern: so, what causes it? Biol J Linn Soc 121:711–715. https://doi.org/10.1093/biolinnean/blx013
Tobias JA, Planqué R, Cram DL, Seddon N (2014) Species interactions and the structure of complex communication networks. P Natl Acad Sci USA 111:1020–1025. https://doi.org/10.1073/pnas.1314337111
Weber MG, Strauss SY (2016) Coexistence in close relatives: beyond competition and reproductive isolation in sister taxa. Annu Rev Ecol Evol S 47:359–381. https://doi.org/10.1146/annurev-ecolsys-112414-054048
Weiss DJ, Garibaldi BT, Hauser MD (2001) The production and perception of long calls by cotton-top tamarins (Saguinus oedipus): acoustic analyses and playback experiments. J Comp Psychol 115:258–271. https://doi.org/10.1037//0735-7036.115.3.258
West-Eberhard MJ (1983) Sexual selection, social competition, and speciation. Q Rev Biol 58:155–183. https://doi.org/10.2307/2828804
Wich SA, Nunn CL (2002) Do male “long-distance calls” function in mate defense? A comparative study of long-distance calls in primates. Behav Ecol Sociobiol 52:474–484. https://doi.org/10.1007/s00265-002-0541-8
Wiley RH, Richards DG (1978) Physical constraints on acoustic communication in the atmosphere: implications for the evolution of animal vocalizations. Behav Ecol Sociobiol 3:69–94. https://doi.org/10.1007/BF00300047
Wilkins MR, Seddon N, Safran RJ (2013) Evolutionary divergence in acoustic signals: causes and consequences. Trends Ecol Evol 28:156–166. https://doi.org/10.1016/j.tree.2012.10.002
Yikweon J (2008) Asymmetry in reproductive character displacement. J Ecol Environ 31:255–260. https://doi.org/10.5141/jefb.2008.31.4.255
Zaccaroni M, Passilongo D, Buccianti A, Dessí-Fulgheri F, Facchini C, Gazzola A, Maggini I, Apollonio M (2012) Group specific vocal signature in free-ranging wolf packs. Ethol Ecol Evol 24:322–331. https://doi.org/10.1080/03949370.2012.664569
Zuur AF, Ieno EN, Walker N, Saveliev AA, Smith GM (2009) Mixed effects models and extensions in ecology with R. Springer, New York
Acknowledgements
We thank Cristiane Rangel, Benedito Monteiro, Laynara Santos, Viviane Costa, Dayane Seyfarth, Guilherme Christo, Karina Solis, Arthur Citó, Gueviston Lima, and Gilberto for fieldwork help. We are also grateful to the Projeto Sauim-de-Coleira, UFAM, PARNA Viruá, LBA, BDFFP, Balbina Dam, Gonçalo Ferraz and the owners of private proprieties (especially Sra Ana and Sr. Dalton Valle) for field facilitation. We thank the editors and two anonymous reviewers for their helpful feedback, as well as Dr. Charles Snowdon for sharing his knowledge and impressions about the function of long calls in tamarin species. This is contribution 811 of the BDFFP Technical Series and 52 from the Amazonian Mammals Research group.
Funding
During the study TVS and received a scholarship from Fundação de Amparo à Pesquisa do Estado do Amazonas (FAPEAM) (062.01758/2018). RMR received a scholarship from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (142352/2017–9). The study is part of a project supported by the "Primate Action Fund" (PAF 14–15; CI 1000796), Idea Wild and the Pignose Company.
Author information
Authors and Affiliations
Contributions
TVS originally formulated the research topic, collected data, and wrote the original draft. TVS, PACLP, JCD and RMR performed exploratory and current statistical analyses. AAPB, MG, and JCD supervised. TVS and WRS raised funds. AAPB and JCD checked the English. All authors contributed to the writing and reviewing.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All applicable international, national, and/or institutional guidelines for the use of animals were followed. The study was non-invasive and complied with Brazilian law (permit for Viruá National Park access 47755–1; and for S. midas and S. bicolor research 10286 – 3 issued by SISBIO/Brazilian Ministry of Environment).
Conflicts of interest
The authors declare no competing interests.
Additional information
Communicated by M. A van Noordwijk.
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sobroza, T.V., Gordo, M., Pequeno, P.A.C.L. et al. Convergent character displacement in sympatric tamarin calls (Saguinus spp.). Behav Ecol Sociobiol 75, 88 (2021). https://doi.org/10.1007/s00265-021-03028-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-021-03028-x