Abstract
In most cooperatively breeding species, reproduction is monopolised by a subset of group members. However, in some species most or all individuals breed. The factors that affect reproductive success in such species are vital to understanding why multiple females breed. A key issue is whether or not the presence of other breeders is costly to an individual’s reproductive success. This study examines the factors that affect the post-parturition component of reproductive success in groups of communal-breeding banded mongoose (Mungos mungo), where up to ten females breed together. Per-litter reproductive success was low (only 18% of pups survived from birth to independence). Whilst singular breeding was wholly unsuccessful, there were costs associated with breeding in the presence of increasing numbers of other females and in large groups. Synchronisation of parturition increased litter success, probably because it minimises the opportunity for infanticide or decreases competitive asymmetry between pups born to different females. There was no evidence of inbreeding depression, and reproductive success was generally higher in litters where females only had access to related males within their group. I conclude that communal breeding in female banded mongooses represents a compromise between the benefits of group-living and communal pup care on the one hand, and competition between females to maximise their personal reproductive success on the other. Such conflicts are likely to occur in most communal breeding species. Whilst communal breeding systems are generally considered egalitarian, negative effects of co-breeders on individual reproductive success is still an issue.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Within social groups, the effect of an individual’s reproduction on the fitness of other breeders is a particularly important issue. It has serious implications for the level of reproductive skew. In groups exhibiting high skew (“despotic”), reproduction is monopolised by a small number of individuals relative to the number of non-breeders in the social group. In groups with low skew (‘egalitarian’), reproduction is more evenly distributed, and most individuals breed. Theoretical models of reproductive skew generally assume a cost to the dominant’s fitness when a subordinate breeds (e.g. Vehrencamp 1983; Keller and Reeve 1994; Cant 1998; Reeve et al. 1998). Whilst the factors affecting reproductive success in species with high reproductive skew are well studied, studies of species with low reproductive skew are relatively few (Lewis and Pusey 1997). Understanding the factors that affect reproductive success in species with low reproductive skew, in particular, whether breeding with other females is costly is fundamental to understanding why multiple individuals breed in such species.
In high-skew species, wherein subordinate females sometimes breed, there is often evidence of conflict and direct interference between breeding females (e.g. Koenig et al. 1995; Clutton-Brock et al. 1998). Communal breeding species, exhibiting a form of low skew where more than one female lays eggs or gives birth to young in the same nest or den (“joint-nesting plural breeders”, Brown 1987) are generally assumed to represent a particularly cooperative form of reproduction. However, studies of both the communal breeding pukeko (Porphyrio porphyrio: Jamieson 1997) and groove-billed ani (Crotophaga sulcirostris: Vehrencamp et al. 1988) similarly provide evidence of conflict and direct interference between breeding females. This suggests that within communal breeders, as in species with high reproductive skew, females may compete over reproduction, and that communal breeding is not necessarily adaptive to all reproductive group members. A key contrast in evaluating whether multiple-breeding females are costly or beneficial in communal breeding species is the reproductive success of single-breeding vs multiple-breeding female clutches or litters.
Numerous other variables affect female reproductive success. Ecological factors, especially climate and food availability, are well documented (Clutton-Brock 1988). Whilst the reproductive success of individuals generally increases with food availability, social factors can have positive or negative effects on an individual’s reproductive success. In cooperative breeders (species where some individuals contribute care to the offspring of others) with high reproductive skew, there is a general trend of increasing breeder reproductive success with group size, and this is probably due to increased help, through an increasing helper-to-young ratio (Clutton-Brock et al. 2001b). However, in cooperative breeding species with low reproductive skew, where the number of females that breed generally increases with group size, the availability of help does not necessarily also increase. In communal breeders, the negative effects of competition for food between young may produce a negative correlation between group size and reproductive success as commonly found in non-cooperative social species (e.g. Clutton-Brock et al. 1982).
In social groups where more than one female breeds, the synchrony of parturition can have major implications for the success of the component litters. Selection pressures acting on parturition synchrony include infanticide, interlitter competition, and predation dilution (Bertram 1975; Ims 1990; Mennella et al. 1990). Whether synchronisation benefits all breeders, or particular individuals at a cost to others, determines the distribution of reproductive success within communal litters.
Reproductive success can be affected, not only by extrinsic factors, but also by the genetic quality of offspring. Inbreeding depression occurs where offspring fitness is compromised when close relatives breed. Numerous studies provide evidence for fitness costs to offspring of inbreeding (Jimenez et al. 1994; Brown and Brown 1998; Coulson et al. 1998; Keller 1998; Slate et al. 2000). In contrast, inbreeding without cost has been recorded in the dwarf mongoose Helogale parvula (Keane et al. 1996), and naked mole rat Heterocephalus glaber (Reeve et al. 1990).
This paper evaluates the effects of social and environmental factors on post-parturition reproductive success in groups of communal-breeding banded mongoose (Mungos mungo). Factors affecting fecundity (whether or not a female gives birth and carries to term, inter-birth interval, and how many foetus she gestates) are dealt with elsewhere (Gilchrist et al. 2004). Banded mongooses are small (<2 kg), insectivorous, group-living mammals that live in equatorial Africa. This species shows extremely low reproductive skew for a cooperatively breeding species, particularly with regard to the other social mongoose species: the meerkat and dwarf mongoose (Cant 2000; De Luca and Ginsberg 2001; Gilchrist et al. 2004). Within groups, females come into behavioural oestrus approximately synchronously and usually copulate with multiple males within their group (Cant 2000). Females breed three to four times a year, and up to ten females give birth approximately synchronously in the same den. If plural breeding is detrimental to individual reproductive success, pup production will be lower when multiple females breed than when a single female breeds, and will decrease with increasing number of breeding females. If synchrony reduces infanticide or competitive asymmetry, litters where all females give birth on the same day will have higher pup production than litters where females give birth on different days. If food availability is an important predictor of reproductive success, pup production will be greater (1) with increasing rainfall (invertebrate prey abundance generally increases with rainfall), and (2) in groups with access to supplemental food. Finally, if inbreeding is detrimental to reproductive success, pup production of litters produced by females breeding in groups with unrelated males will be higher than natal females breeding in groups with only natal males.
Materials and methods
Study area and population
Fieldwork was carried out in an 8-km2 area, on and around Mweya peninsula, Queen Elizabeth National Park, southwest Uganda 0°12′ S and 27°54′ E, from March 1997 to February 2000. The vegetation of the study site was predominantly short and medium fire climax grassland with numerous dense thickets (Lock 1977). The climate was equatorial, with little annual fluctuation in day length or temperature. Daily rainfall data were collected via a rain gauge sited in a central location within the study site.
Banded mongoose groups comprise individuals that forage together as a cohesive unit during the day and sleep communally in a den at night. Groups are stable on a day-to-day basis and are territorial. Data on reproductive success were collected on 11 groups of banded mongoose. Three groups had access to supplemental food from human garbage dumps (Gilchrist and Otali 2002; Otali and Gilchrist 2004). Group size varied from three to 36, and the number of reproductive females within a group varied from one to ten (Gilchrist 2001). Individuals were trapped, anaesthetised, marked and located according to methods outlined elsewhere (Cant 2000; Cant et al. 2001). Procedures were licensed by the Uganda National Council for Science and Technology and the Uganda Wildlife Authority, and followed the guidelines of the Association for the Study of Animal Behaviour (ASAB 1991).
Data collection and analysis
I evaluated mean reproductive success per female per litter using communal pup counts and the number of females that gave birth. It was not possible to ascribe young to specific breeders because females generally mate with multiple males and give birth together underground (Cant 2000). I define a communal litter as the litter born to the group, i.e. the litter produced by all the females that gave birth in a breeding event (one to ten females) within a 30 day period (Gilchrist 2004). Parturition was detected when a pregnant female’s abdomen size changed from swollen to flaccid. Litters where all females gave birth on the same day were classified as synchronous. Litters where females gave birth on different days were classified as asynchronous. It was impossible to directly count communal litter size at birth because pups were usually born in inaccessible subterranean dens. I, therefore, estimated communal litter size at birth by summing the foetus counts made during gestation on all females that subsequently gave birth in a communal litter. By trapping and anaesthetising females 2 to 4 weeks after oestrus (gestation is approximately 8 weeks, Cant 2000), it was possible to detect pregnancy and count the number of foetuses by palpation. There is no evidence that trapping and palpating pregnant females affected the probability that they aborted (χ 2=2.37, df=1, p=0.12, n=264). For females that had given birth but for whom foetal litter size was not counted, foetal litter size was estimated using the predictive equation from a General Linear Model with age as the fixed effect and foetus count as the variate (age being a significant correlate of foetus count, Gilchrist et al. 2004). Results in subsequent analyses were consistent on comparing against analyses restricted to females with foetus counts.
Between birth and emergence, pups spend approximately their first 30 days in the den (Rood 1974; Cant 2003). Pups were defined as ‘emergent’ from the first date that they were observed travelling with a foraging group. The maximum number of emergent pups was counted within a few days of first emergence. After emergence, pups are dependent on older group members for food and protection, and usually form stable associations with ‘escorts’ until approximately 90 days of age (Gilchrist 2004; Gilchrist and Russell, in preparation). Pup number at independence was counted at 90 days after birth.
Group size (the number of individuals more than 90 days of age) was counted when a litter was born. All group members more than 6 months are capable of contributing care to the communal litter (Gilchrist 2001; Gilchrist and Russell, in preparation), with males tending to make higher contributions than females through babysitting and escorting (Rood 1974; Cant 2003; Gilchrist 2004; Gilchrist and Russell, in preparation). As an index of care, the number of group members (and males) more than 6 months was counted.
I tested whether reproductive success is correlated with whether females (giving birth) had access to related or unrelated males within the group. In the absence of genetic data, I use group residence status as a surrogate. Group history was known from 1994, and in no case did an individual immigrate into an established group (Cant et al. 2001; Gilchrist 2001). Therefore, females that conceived within their natal group did not have access to ‘unrelated’ males within the group (n=11 litters, two groups), and females that conceived within a group formed by fusion of male and female subgroups from different natal groups had access to ‘unrelated’ males within the group (n=24 litters, six groups). The incidence of extra-group copulations was extremely low (Gilchrist et al. 2004). I restrict analysis only to litters where all females were natal or all females were non-natal.
The selective pressures acting on a female’s post-parturition reproductive success (through pup survival) will be quite different between the two distinct stages of a pup’s life. Before emergence, pups remain in the den and are reliant upon suckling. After emergence, until the pups attain independence, they travel with the group and are fed by group members. Therefore, I independently evaluate the factors that affect reproductive success across these two periods: (1) birth to emergence and (2) emergence to independence. The data on pup counts were non-normally distributed and robust to transformation. To meet the assumptions of parametric statistical analysis, I divided analyses of reproductive success into two subcomponents (Vehrencamp et al. 1988): (a) litter failure/success (all pups died/one or more pups survived), (b) mean number of pups per female in successful litters (for litters where one or more pups survived). In analyses, I excluded four litters that were born during group fission events (Cant et al. 2001), because observations suggested that litter failure was caused by the conflict and confusion of the fission event (three litters failed pre-emergence and one litter produced only one emergent pup).
Statistical analyses were performed using GenStat 6. Normally distributed data were analysed using the General Linear Model (GLM) procedure. Binomial data were analysed using the Generalised Linear Model procedure with logit link function and binomial error distribution. Group identity was fitted as a random term in equivalent Restricted Estimate Maximum Likelihood Models, and Iterated Reweighted Restricted Maximum Likelihood Models but was estimated as a negative or zero component of variance, confirming that litters can be considered statistically independent data points. The statistical independence of communal litters was expected, as group membership and environmental conditions differed between litters within groups. Dispersion was estimated where dispersion >1, and fixed at 1 otherwise. In each analysis, I tested whether reproductive success differed between litters where a single female gave birth (n=8 single-female litters from five groups) and litters where more than one female gave birth (n=54 multiple-female litters from nine groups, with three groups producing single and multiple-female litters during the study). I also fitted social and environmental variables (see Table 1) and relevant two-way interactions as fixed effects. Forward elimination was used to select fixed terms for the minimal model (Sokal and Rohlf 1995), which comprised only those parameters that contributed significantly (p<0.05) to the explanatory power of the model. All means are expressed as ±1 standard error.
For the period between pup birth and emergence, I investigated the factors that affect the probability of a litter emerging by fitting litter survival (0=no pups survived to emergence, 1=one or more pups survived to emergence) as the response variate in a Generalised Linear Model with binomial denominator 1. The factors that affect the mean number of emergent pups per breeding female (excluding litters that failed to emerge) were investigated by fitting mean number of emergent pups per female that gave birth as the response variate in a GLM. These analyses were repeated for the period between pup emergence and independence (excluding litters that failed before emergence and those that failed before independence from the GLM).
Results
Overall litter success: pup independence
Only 54.7% of litters born produced one or more pups at independence (35 litters produced one or more pups at independence, 18 litters failed before emergence and 11 failed between emergence and independence). The mean estimated communal litter size at birth (for the 60 communal litters of which the number of pups was estimated at birth and counted at independence) was 11.70±0.98 pups (total=702 pups), and the mean number of pups to survive to independence was 2.17±0.38 per communal litter (total=130 pups). The mean proportion of pups to survive from birth to independence was 0.18±0.028 per communal litter.
Litter success/failure from birth to emergence
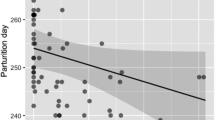
Twenty six percent of communal litters died before emergence (n=17/65 litters). The probability that a litter produced one or more pups at emergence did not differ between single-female and multiple-female litters [F(1,58)=1.22, p=0.12, n=60 litters], but increased with the number of females that gave birth [F(1,58)=4.88, p=0.031, n=60 litters, percentage variance accounted for (r 2)=6.1%; probability of litter success ranged from 0.51±0.13 for one female to 0.98±0.05 for ten females]. Within multiple-female litters, synchronous litters were more likely to produce one or more pups at emergence (1.00±0.00, n=10 litters) than asynchronous litters [0.69±0.09, n=25; F(1,33)=6.28, p=0.012, n=35 litters, r 2=14.2%].
Number of pups at emergence
The mean number of emergent pups (excluding failed litters) was 6.13±0.53 per communal litter (n=48 litters), and 1.93±0.18 per female that gave birth (n=43 litters). The number of emergent pups per female was higher in single-female litters (4.00±0.58, n=3 litters) than multiple-female litters [1.75±0.16, n=40; F(1,41)=13.94, p=0.001, n=43 litters, r 2=23.6%). Within multiple-female litters, the number of pups at emergence per female decreased as the number of females that gave birth increased [F(1,38) =7.94, p=0.008, n=40 litters, r 2=15.1%, Fig. 1], and was higher in groups where all females that gave birth were natal (2.67±0.33, n=6 litters) than in groups where all the females that gave birth had formed the group via fusion with unrelated males [1.72±0.24, n=11; F(1,15)=5.48, p=0.033, n=17 litters, r 2=21.9%).
Litter success/failure from emergence to independence
Twenty four percent of emergent litters failed before pup independence (n=11/46 litters). The probability that an emergent litter produced one or more pups at independence was higher for multiple-female litters (0.82±0.06, n=38 litters) than single-female litters (0.00±0.00, n=3 litters; χ 2=9.25, df=1, p=0.002, n=41 litters, r 2=18.3%). Within multiple-female litters, the probability that an emergent litter produced one or more pups at independence increased with the mean daily rainfall in the 90 days post-parturition (χ 2=7.66, df=1, p=0.006, n=38 litters, r 2=18.9%; predicted probability of litter success increasing from 0.43±0.18 for 0 mm rain to 1.00±0.01 for 6 mm rain).
Considering the factors affecting litter success/failure from birth to independence, multi-female litters had a higher probability of producing one or more pups at independence (0.60±0.08, n=52 litters) than single-female litters (0.00±0.00, n=7 litters; F(1,57)=9.33, p=0.003, n=59 litters, r 2=12.6%), and synchronous litters had a higher success probability (0.92±0.10, n=10 litters) than asynchronous litters (0.44±0.12, n=24 litters; F(1,32)=6.19, p=0.018, n=34 litters, r 2=15.4%).
Number of pups at independence
The mean number of pups at independence (excluding litters that failed before independence) was 3.91±0.49 per communal litter (n=35 litters). The mean number of pups at independence per female was 1.09±0.12 per communal litter (n=31 litters). The number of pups at independence per female decreased with increasing number of adults [F(1,29)=5.19, p=0.030, n=31 litters, r 2=12.3%, Fig. 2]. In addition, there was a tendency for the number of pups at independence to increase with the number of pups from the previous cohort still surviving [F(1,28)=3.58, p=0.069, n=31 litters]. The mean number of pups at independence per female tended to be higher in groups with natal-only females (1.63±0.25, n=5 litters) than non-natal female groups (1.05±0.19, n=9 litters; F(1,12)=3.44, p=0.088, n=14 litters, r 2=15.8%).
Discussion
Whereas environmental and social factors have little effect on the fecundity of female banded mongooses (Gilchrist et al. 2004), they have significant effects on post-parturition reproductive success. All litters where only one female gave birth failed, but the number of emergent pups per female decreased as the number of females that gave birth increased. Pup production was higher in litters where all females gave birth on the same day than in asynchronous litters, and was generally higher in litters born to only natal females than in groups where all the females that gave birth were non-natal. In addition, the probability that a litter survived from emergence to independence increased with rainfall, and the number of independent pups per female decreased with increasing numbers of adults. Interpretations of these patterns must be indirect because the cause of death of the majority of pups is unknown, but illness and predation are both implicated (Otali and Gilchrist 2004).
Is it costly to breed with other females?
An assumption of models of reproductive skew is that subordinate reproduction is costly to the dominant individual in the social group (e.g. Vehrencamp 1983; Keller and Reeve 1994). Without such a competitive cost to plural breeding, there would be no motivation for an individual to suppress reproduction of other group members. Do banded mongoose females have higher reproductive success when they breed alone and no other females give birth? No: all communal litters where a single female gave birth failed to produce a single pup at independence. This suggests that monopoly of reproduction by a single female in banded mongoose groups would not be advantageous to that female.
Is there evidence that mean reproductive success decreases as the number of breeding females increases? Yes: the mean number of emergent pups per female that gave birth decreased as the number of females that gave birth increased. In contrast, a previous study of the same population suggested that per capita production of emergent young increased with the number of females that gave birth (Cant 2000, contrast Fig. 3a with Fig. 3b). This difference may be explained by Cant’s singular- and two-female litters (all of which failed) being derived from recently formed groups because such groups tend to experience high rates of litter failure in their initial litters (J.S. Gilchrist, unpublished data).
The mean number of emergent pups per female plotted against the number of females that gave birth: a this study, b Cant (2000). Points joined to the right of a data point indicate the number of litters with the same value for the X and Y axes
The high failure rate of litters with few breeding females may reflect the difficulty females face in combining foraging with suckling young. Females allow allosuckling (Neal 1970; Rood 1975; J. S. Gilchrist, unpublished data), and, therefore, pups may be suckled more often when there is more than one lactating female in the group (Lewis and Pusey 1997). The diminishing reproductive success with increasing number of females that gave birth is more interesting for the lack of evidence of a negative effect of increasing communal litter size, suggesting that competition between pups is not the issue. Whether the detrimental effect of increasing number of breeding females is passive (e.g. scramble competition) or active (e.g. infanticide) is unclear (Gilchrist 2006).
Other habitually communal-breeding (low skew) species exhibit evidence of costs of communal breeding to breeder’s per capita reproductive output. In the pukeko (Craig 1980) and groove-billed ani (Koford et al. 1990), the average reproductive success of joint-nesting individuals is lower than singular breeding females (also see Table 3 in Vehrencamp 2000). Costs to communal breeding through reduced reproductive success (for at least some females) have also been documented in some communal breeding rodents (Marin and Pilastro 1994; Wolff 1994; Gerlach and Bartmann 2002). This suggests that even in communal breeding species, there is commonly a conflict of interest, and that it may not be adaptive to the dominant female to allow subordinate females to breed.
Why then does a female tolerate reproduction by other females when it is costly to her own reproductive success? Low reproductive skew in communal breeding species may represent an inability of individuals to suppress reproduction in other females (Creel and Waser 1997; Packer et al. 2001). The lack of a distinct dominance hierarchy in female banded mongooses is consistent with the suggestion that no female has sufficient leverage to suppress reproduction in subordinates (Gilchrist et al. 2004, but see De Luca and Ginsberg 2001). Alternatively, dominant females will tolerate the costs of communal breeding if the cost of suppressing, or evicting (Johnstone and Cant 1999) subordinates exceeds the cost of the additional female(s) breeding.
Alternatively, a decrease in mean reproductive success per breeding female does not necessarily mean that a dominant female in a group suffers decreased reproductive success. The mean number of emergent pups per female could decrease through increased mortality of subordinate’s pups, with mortality of dominant’s pups unaffected. This could occur through differential infanticide, as in meerkats where most infanticide is conducted by the dominant female on subordinate’s pups (Clutton-Brock et al. 1998, 2001a). Alternatively, subordinate’s pups may be inferior competitors. In the banded mongoose, smaller females gestate smaller foetuses (Gilchrist et al. 2004) and, therefore, probably give birth to smaller pups.
If a dominant female does not incur a reproductive cost from multiple females breeding (Cant 2000), it may be in her interests to allow related females to contribute to the communal litter, as she would gain indirect fitness from relatives’ offspring (Cant and Johnstone 1999). Subordinate reproduction could represent an important component of fitness in the banded mongoose, as indirect fitness to the dominant, and as direct fitness to subordinates, because females within a group are probably always related, and because many communal litters likely contained subordinate pups. Female banded mongooses either breed within their natal group or disperse as a single-sex subgroup, and (within a group) are, therefore, probably always related to one another (Cant et al. 2001; Gilchrist 2001). Sixteen of the 35 litters that survived to independence produced more than three pups (the median foetal litter size per female, Gilchrist et al. 2004), and, therefore, probably contained subordinate pups. The remaining 19 litters, with three or less independent pups, may also have contained subordinate offspring.
Food availability and group size effects on reproductive success
Food resources may be especially important in determining the level of reproductive skew and distribution of reproduction within cooperatively breeding species (Keller and Reeve 1994). I detected a positive correlation between rainfall (commonly a positive correlate of the abundance of invertebrate prey, Rood 1975; Hiscocks and Perrin 1991; Waser et al. 1995; Doolan and Macdonald 1996) and the probability of litter success at independence in banded mongooses. However, groups that had access to a supplemental food source, human food at garbage dumps, did not have higher reproductive success than those groups with access only to natural food sources, even controlling for number of females (also see Otali and Gilchrist 2004). The lack of a supplemental food effect is surprising because adults with access to garbage dumps were heavier (Otali and Gilchrist 2004). This may be expected to have translated into increased pup survival, with females better able to meet the high energetic demands of lactation, and adults provisioning pups with a higher proportion of the food items encountered, as for meerkats (Brotherton et al. 2001). That this was not the case calls into question the nutritional quality of the human food at garbage dumps (Otali and Gilchrist 2004).
In cooperatively breeding species, the reproductive success of breeders commonly increases with group size (for a review, see Emlen 1991). This is generally interpreted as evidence of the positive effect of helpers, e.g. as in meerkats where helpers feed emergent pups (Clutton-Brock et al. 2001b). However, in the banded mongoose, there is evidence of reduced reproductive success in large groups, and, therefore, costs to individual reproductive success at large group size that counteract the benefits of communal breeding. This may explain why eviction events where individuals are forced out of their natal group have only been recorded in the largest groups (Cant et al. 2001; Gilchrist 2006). In the majority of evictions, it is the females that are forcibly expelled (Gilchrist 2001). Eviction may be the only effective way to minimise female intra-group reproductive competition (Johnstone and Cant 1999). These large groups may surpass the threshold cost that can be tolerated by ‘dominant’ reproductive individuals.
Synchrony of parturition
Banded mongooses show a high degree of synchronisation of parturition relative to other mammalian species (Gilchrist 2006). Synchronised litters were more successful than asynchronous litters. There are several possible selective pressures favouring synchronisation of parturition. By giving birth synchronously, a female may limit the ability of other group members to differentiate between their own and others offspring and to kill pups that would compete with their own (e.g. Manning et al. 1995). Alternatively, synchronous parturition may reduce competitive asymmetry between pups of different litters (Menella et al. 1990). Reproductive synchrony could also dilute predation risk of young (Ims 1990). However, in the banded mongoose, economising and coordinating care of young may be a major selective advantage to synchronisation (Cant 2003; Gilchrist 2004). The higher success of synchronous litters raises the question of why more communal litters were not synchronised to same-day parturition. Such instances may highlight a breakdown in coordination, or conflict between females.
Is reproductive success reduced by inbreeding depression?
In many species with high reproductive skew, non-breeders are related to the opposite-sex breeder, and it is proposed that the reason these individuals do not breed is avoidance of inbreeding and the associated costs to offspring produced (Faulkes and Bennett 2001). Gilchrist et al. (2004) found no evidence that banded mongoose females avoid breeding in the presence of only related males within their group. If females regularly breed with related males, are there associated costs to reproductive success? Surprisingly, in this study, litters where females had access to only related males within their group had higher reproductive success than litters where all females had access to unrelated males. Perhaps potential conflicts within and between sexes are reduced when females and males are related, and indirect benefits favour truly communal care (Hamilton 1964). Alternatively, there may be benefits to familiarity in well-established groups that improve coordination of pup care.
Conclusions
This paper demonstrates that whilst singular breeding is unproductive for females of the low-skew banded mongoose, reproductive success declines with increasing numbers of co-breeding females, suggesting costs to multiple-breeding females. In addition, despite their apparently maladaptive fitness returns, singular breeding and asynchronous parturition occur, suggesting conflicts of interest between females. Low reproductive skew could, therefore, represent a lack of dominance control. Alternatively, communal breeding may maximise an individual’s inclusive fitness. A female may allow related females to add young because pup mortality is high and beyond her control. She would then at least gain indirect fitness benefits if her own pups die.
References
ASAB (1991) Guidelines for the use of animals in research. Anim Behav 41:183–186
Bertram BCR (1975) Social factors influencing reproduction in wild lions. J Zool 177:463–482
Brotherton PNM, Clutton-Brock TH, O’Riain MJ, Gaynor D, Sharpe L, Kansky R, McIlrath GM (2001) Offspring food allocation by parents and helpers in a cooperative mammal. Behav Ecol 12:590–599
Brown JL (1987) Helping and communal breeding in birds. Princeton University Press, Princeton, New Jersey
Brown JL, Brown ER (1998) Are inbred offspring less fit? Survival in a natural population of Mexican jays. Behav Ecol 9:60–63
Cant MA (1998) A model for the evolution of reproductive skew without reproductive suppression. Anim Behav 55:163–169
Cant MA (2000) Social control of reproduction in banded mongooses. Anim Behav 59:147–158
Cant MA (2003) Patterns of helping effort in co-operatively breeding banded mongooses (Mungos mungo). J Zool 259:115–121
Cant MA, Johnstone RA (1999) Costly young and reproductive skew in animal societies. Behav Ecol 10:178–184
Cant MA, Otali E, Mwanguhya F (2001) Eviction and dispersal in co-operatively breeding banded mongooses (Mungos mungo). J Zool 254:155–162
Clutton-Brock TH (1988) Reproductive success: studies of individual variation in contrasting breeding systems. Chicago University Press, Chicago
Clutton-Brock TH, Albon SD, Guinness FE (1982) Competition between female relatives in a matrilocal mammal. Nature 300:178–180
Clutton-Brock TH, Brotherton PNM, Smith R, McIlrath GM, Kansky R, Gaynor D, O'Riain MJ, Skinner JD (1998) Infanticide and expulsion of females in a cooperative mammal. Proc R Soc Lond B Biol Sci 265:2291–2295
Clutton-Brock TH, Brotherton PNM, Russell AF, O'Riain MJ, Gaynor D, Kansky R, Griffin A, Manser M, Sharpe L, McIlrath GM, Small T, Moss A, Monfort S (2001a) Cooperation, control, and concession in meerkat groups. Science 291:478–481
Clutton-Brock TH, Russell AF, Sharpe LL, Brotherton PNM, McIlrath GM, White S, Cameron EZ (2001b) Effects of helpers on juvenile development and survival in meerkats. Science 293:2446–2449
Coulson TN, Pemberton JM, Albon SD, Beaumont M, Marshall TC, Slate J, Guinness FE, Clutton-Brock TH (1998) Microsatellites reveal heterosis in red deer. Proc R Soc Lond B Biol Sci 265:489–495
Craig JL (1980) Breeding success of a common gallinule. Behav Ecol Sociobiol 6:289–295
Creel SR, Waser PM (1997) Variation in reproductive suppression among dwarf mongooses: interplay between mechanisms and evolution. In: Solomon NG, French JA (eds) Cooperative breeding in mammals. University Press, Cambridge, pp 150–170
De Luca DW, Ginsberg JR (2001) Dominance, reproduction and survival in banded mongooses: towards an egalitarian social system? Anim Behav 61:17–30
Doolan SP, Macdonald DW (1996) Diet and foraging behaviour of group-living meerkats, Suricata suricatta, in the southern Kalahari. J Zool 239:697–716
Emlen ST (1991) Evolution of cooperative breeding in birds and mammals. In: Krebs JR, Davies NB (eds) Behavioural ecology: an evolutionary approach, 3rd edn. Blackwell Scientific Publications, Oxford, pp 301–337
Faulkes CG, Bennett NC (2001) Family values: group dynamics and social control of reproduction in African mole-rats. Trends Ecol Evol 16:184–190
Gerlach G, Bartmann S (2002) Reproductive skew, costs, and benefits of cooperative breeding in female wood mice (Apodemus sylvaticus). Behav Ecol 13:408–418
Gilchrist JS (2001) Reproduction and pup care in the communal breeding banded mongoose. PhD. Dissertation. University of Cambridge, United Kingdom
Gilchrist JS (2004) Pup escorting in the communal breeding banded mongoose: behavior, benefits, and maintenance. Behav Ecol 15:952–960
Gilchrist JS (2006) Female eviction, abortion, and infanticide in banded mongooses (Mungos mungo): implications for social control of reproduction and synchronized parturition. Behav Ecol 17(4):664–669
Gilchrist JS, Otali E (2002) The effects of refuse-feeding on home-range use, group size, and intergroup encounters in the bounded mongoose. Canadian Journal of Zoology-Revue Canadienne De Zoologie 80:1795–1802
Gilchrist JS, Otali E, Mwanguhya F (2004) Why breed communally? Factors affecting fecundity in a communal breeding mammal: the banded mongoose (Mungos mungo). Behav Ecol Sociobiol 57:119–131
Hamilton WD (1964) The genetical evolution of social behaviour. J Theor Biol 7:1–52
Hiscocks K, Perrin MR (1991) A dietary comparison between two sympatric viverrids, Helogale parvula (Sundevall 1846) and Mungos mungo (Gmelin 1788). J Afr Zool 105:307–312
Ims R (1990) The ecology and evolution of reproductive synchrony. Trends Ecol Evol 5:135–140
Jamieson IG (1997) Testing reproductive skew models in a communally breeding bird, the pukeko, Porphyrio porphyrio. Proc R Soc Lond B Biol Sci 264:335–340
Jimenez JA, Hughes KA, Alaks G, Graham L, Lacy RC (1994) An experimental study of inbreeding depression in a natural habitat. Science 266:271–273
Johnstone RA, Cant MA (1999) Reproductive skew and the threat of eviction: a new perspective. Proc R Soc Lond B 266:275–279
Keane B, Creel SR, Waser PM (1996) No evidence of inbreeding avoidance or inbreeding depression in a social carnivore. Behav Ecol 7:480–489
Keller L, Reeve HK (1994) Partitioning of reproduction in animal societies. Trends Ecol Evol 9:98–103
Keller LF (1998) Inbreeding and its fitness effects in an insular population of song sparrows (Melospiza melodia). Evolution 52:240–250
Koenig WD, Mumme RL, Stanback MT, Pitelka FA (1995) Patterns and consequences of egg destruction among joint-nesting acorn woodpeckers. Anim Behav 50:607–621
Koford RR, Bowen BS, Vehrencamp SL (1990) Groove-billed anis: joint-nesting in a tropical cuckoo. In: Stacey PB, Koenig WD (eds) Cooperative breeding in birds: long-term studies of ecology and behaviour. Cambridge University Press, Cambridge, pp 333–356
Lewis SE, Pusey AE (1997) Factors influencing the occurrence of communal care in plural breeding mammals. In: Solomon NG, French JA (eds) Cooperative breeding in mammals. University Press, Cambridge, pp 335–363
Lock JM (1977) The vegetation of Rwenzori National Park, Uganda. Botanische Jahrbücher für Systematik, Pflanzengeschichte und Pflanzengeographie 98:372–448
Manning CJ, Dewsbury DA, Wakeland EK, Potts WK (1995) Communal nesting and communal nursing in house mice (Mus musculus domesticus). Anim Behav 50:741–751
Marin G, Pilastro A (1994) Communally breeding dormice, glis-glis, are close kin. Anim Behav 47:1485–1487
Mennella JA, Blumberg MS, McClintock MK, Moltz H (1990) Interlitter competition and communal nursing among Norway rats—advantages of birth synchrony. Behav Ecol Sociobiol 27:183–190
Neal E (1970) The banded mongoose, Mungos mungo Gmelin. East Afr Wildl J 8:63–71
Otali E, Gilchrist JS (2004) Effects of refuse feeding on body condition, reproduction and survival of banded mongooses. J Mammal 85:491–497
Packer C, Pusey AE, Eberly LE (2001) Egalitarianism in female African lions. Science 293:690–693
Reeve HK, Westneat DF, Noon WA, Sherman PW, Aquadro CF (1990) DNA Fingerprinting reveals high-levels of inbreeding in colonies of the eusocial naked mole-rat. Proc Natl Acad Sci USA 87:2496–2500
Reeve HK, Emlen ST, Keller L (1998) Reproductive sharing in animal societies: reproductive incentives or incomplete control by dominant breeders? Behav Ecol 9:267–278
Rood JP (1974) Banded mongoose males guard young. Nature 248:176–177
Rood JP (1975) Population dynamics and food habitats of the banded mongoose. East Afr Wildl J 13:89–111
Slate J, Kruuk LEB, Marshall TC, Pemberton JM, Clutton-Brock TH (2000) Inbreeding depression influences lifetime breeding success in a wild population of red deer (Cervus elaphus). Proc R Soc Lond B Biol Sci 267:1657–1662
Sokal RR, Rohlf FJ (1995) Biometry: the principles and practice of statistics in biological research, 3rd edn. W. H. Freeman, New York
Vehrencamp SL (1983) A model for the evolution of despotic versus egalitarian societies. Anim Behav 31:667–682
Vehrencamp SL, Koford RR, Bowen BS (1988) The effect of breeding-unit size on fitness components in groove-billed anis. In: Clutton-Brock TH (ed) Reproductive success: studies of individual variation in contrasting breeding systems. University of Chicago Press, Chicago, pp 291–304
Waser PM, Elliott LF, Creel NM, Creel SR (1995) Habitat variation and mongoose demography. In: Sinclair ARE, Arcese P (eds) Serengeti II: dynamics, management and conservation of an ecosystem. University Press, Chicago, pp 421–448
Wolff JO (1994) Reproductive success of solitarily and communally nesting white-footed mice and deer mice. Behav Ecol 5:206–209
Acknowledgements
I thank Emily Otali and Francis Mwanguhya for their invaluable assistance with data collection, Tim Clutton-Brock and Tim Coulson for advice, and Andy Russell, Mike Cant, Giacomo Tavecchia and the reviewers for comments on the manuscript. I am grateful to the Uganda Wildlife Authority for allowing me to conduct this research in Queen Elizabeth National Park. For financial support, I thank the Biotechnology and Biological Sciences Research Council, the Ian Karten Charitable Trust, and in Cambridge: Magdalene College, the Board of Graduate Studies, the Cambridge Philosophical Society, and the Department of Zoology.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by E. Korpimäki
Rights and permissions
About this article
Cite this article
Gilchrist, J.S. Reproductive success in a low skew, communal breeding mammal: the banded mongoose, Mungos mungo . Behav Ecol Sociobiol 60, 854–863 (2006). https://doi.org/10.1007/s00265-006-0229-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00265-006-0229-6